Effect of Aging Temperature on Microstructure, Mechanical, and Wear Properties of 18Ni-300 Maraging Steel Produced by Powder Bed Fusion
Abstract
1. Introduction
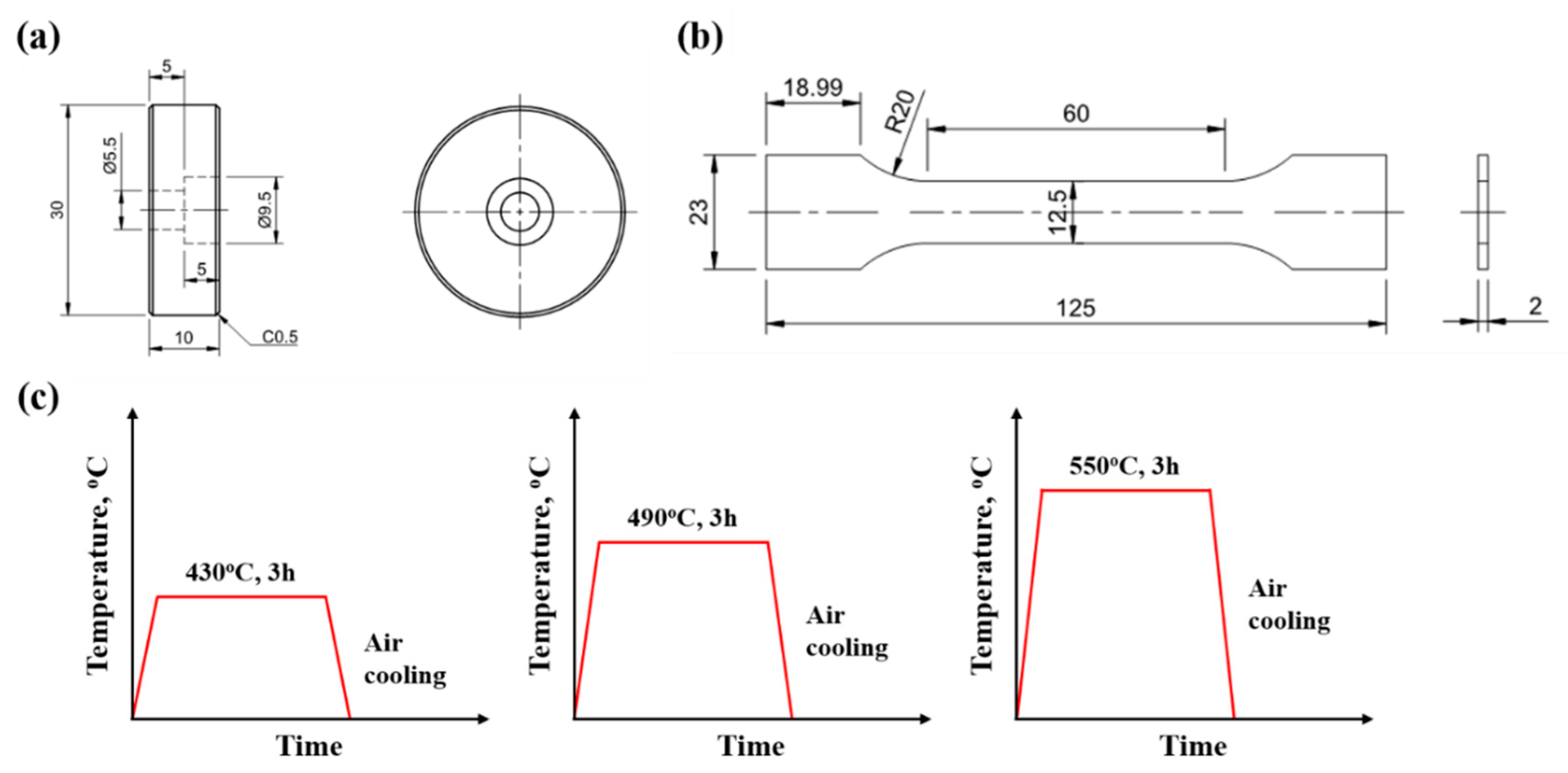
2. Experimental Procedure
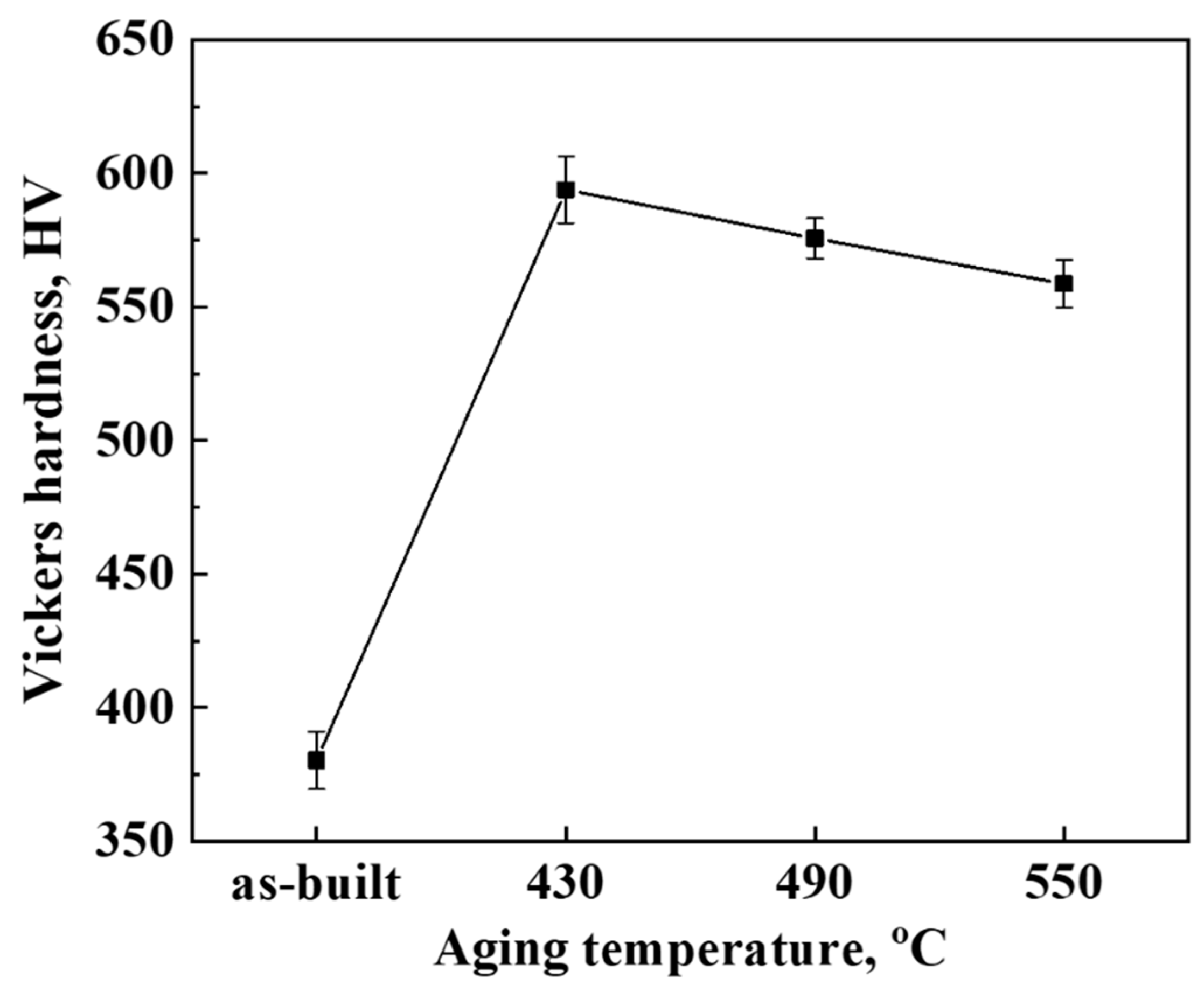
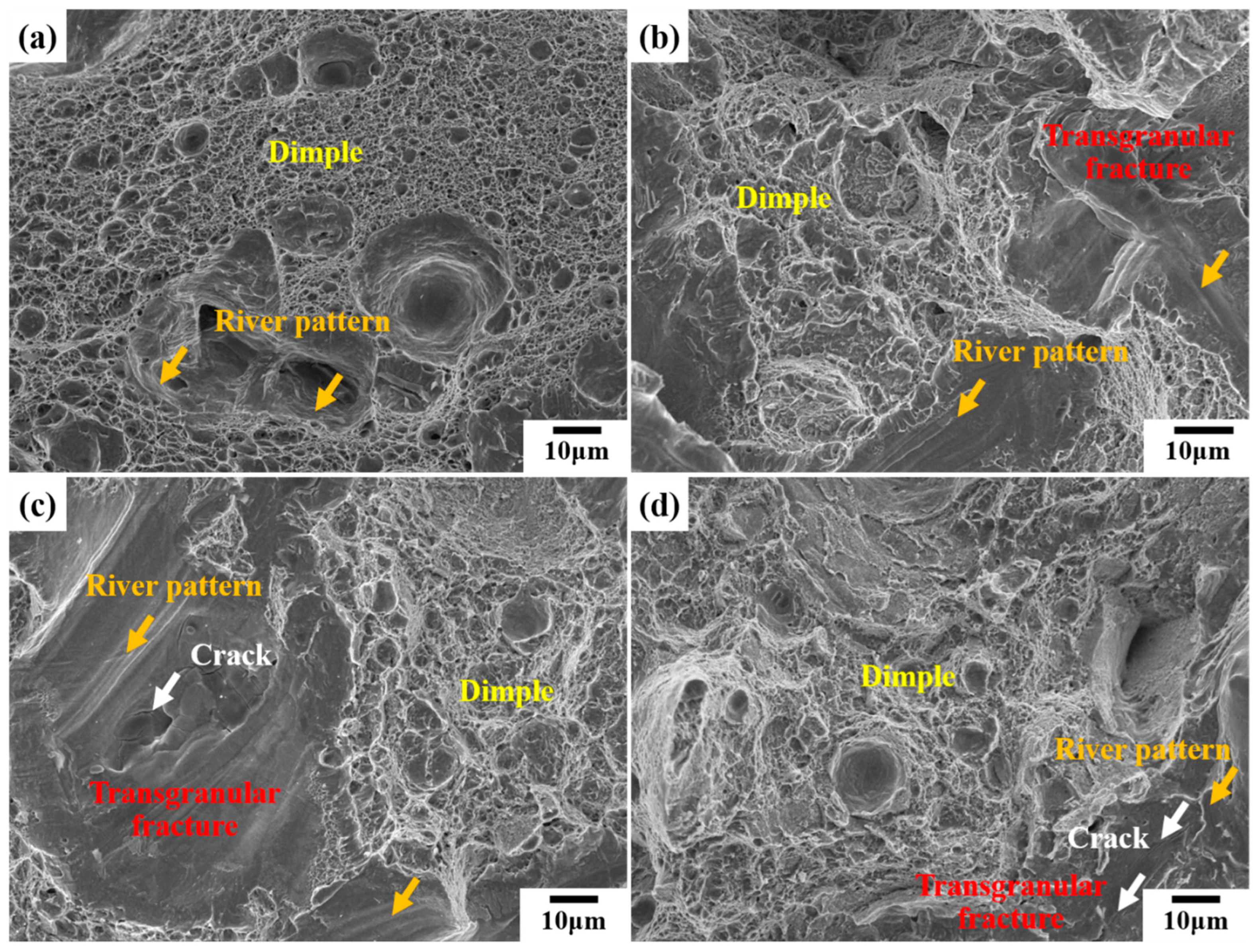
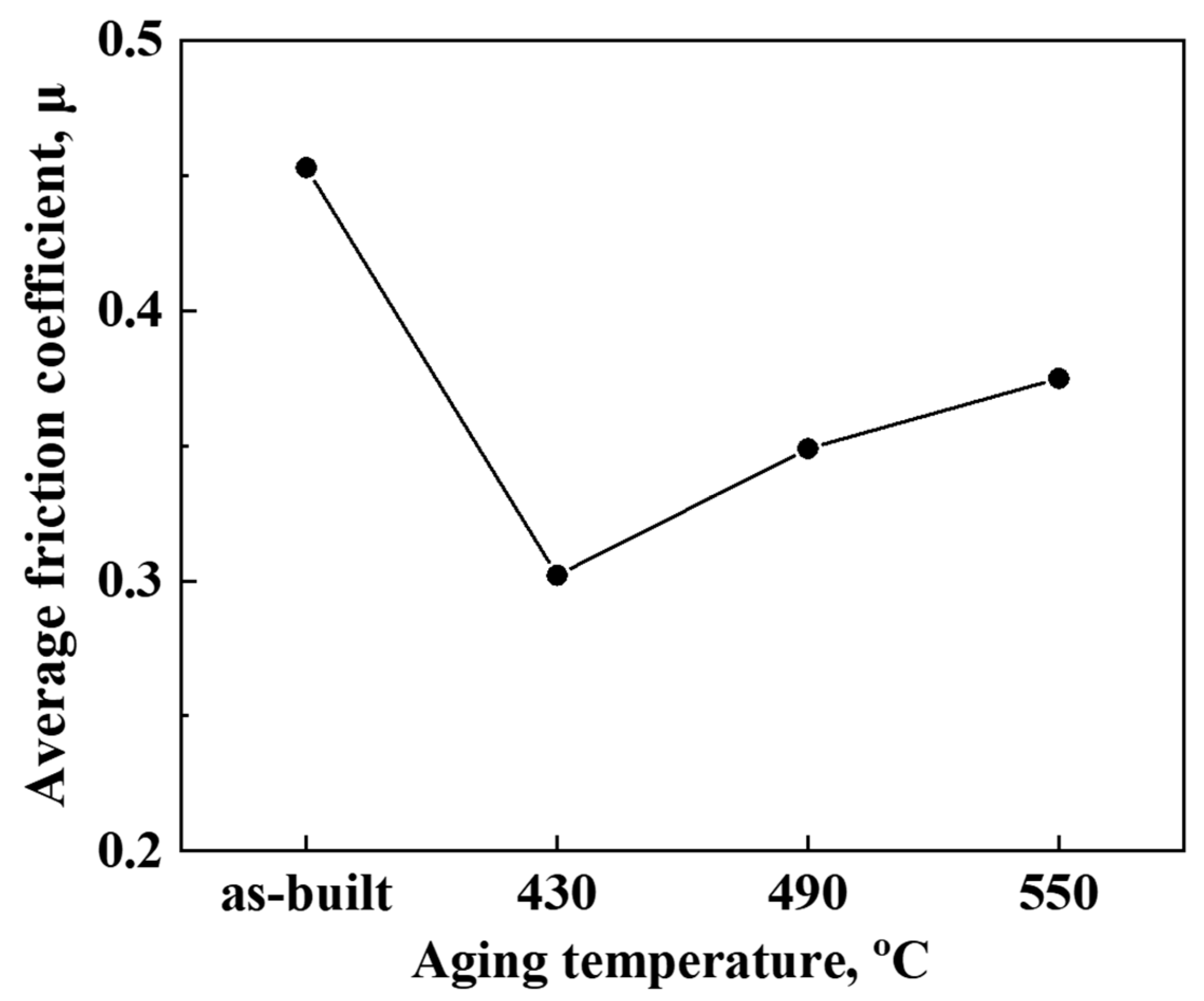
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Floreen, S. The Physical Metallurgy of Maraging Steels. Metall. Rev. 1968, 13, 115–128. [Google Scholar] [CrossRef]
- Casalino, G.; Campanelli, S.L.; Contuzzi, N.; Ludovico, A.D. Experimental Investigation and Statistical Optimisation of the Selective Laser Melting Process of a Maraging Steel. Opt. Laser Technol. 2015, 65, 151–158. [Google Scholar] [CrossRef]
- Hermann Becker, T.; Dimitrov, D. The Achievable Mechanical Properties of SLM Produced Maraging Steel 300 Components. Rapid Prototyp. J. 2016, 22, 487–494. [Google Scholar] [CrossRef]
- Zhang, S.; Zhou, N.; Ma, C.; Liu, J.; Liu, S.; Misra, R.D.K. Simultaneous Enhancement of Strength and Hydrogen Embrittlement Resistance of Laser-Powder Bed Fusion Maraging Steel via Long-Term Low-Temperature Aging. Corros. Sci. 2023, 223, 111440. [Google Scholar] [CrossRef]
- Jägle, E.A.; Choi, P.-P.; Humbeeck, J.V.; Raabe, D. Precipitation and austenite reversion behavior of a maraging steel produced by selective laser melting. J. Mater. Res. 2014, 29, 2072–2079. [Google Scholar] [CrossRef]
- Chen, T.; Shea, K. Computational design-to-fabrication using spatial grammars: Automatically generating printable car wheel design variants. Des. Soc. A Worldw. Community 2015, 35–44. Available online: https://www.designsociety.org/publication/37710/COMPUTATIONAL+DESIGN-TO-FABRICATION+USING+SPATIAL+GRAMMARS%3A+AUTOMATICALLY+GENERATING+PRINTABLE+CAR+WHEEL+DESIGN+VARIANTS (accessed on 20 February 2024).
- Dong, Z.; Ouyang, P.; Zhang, S.; Liu, L.; Li, H.; Wu, Y. Effect of Building Direction on Anisotropy of Mechanical Properties of GH4169 Alloy Fabricated by Laser Powder Bed Fusion. Mater. Sci. Eng. A 2023, 862, 144430. [Google Scholar] [CrossRef]
- Shamsdini, S.; Pirgazi, H.; Ghoncheh, M.H.; Sanjari, M.; Amirkhiz, B.S.; Kestens, L.; Mohammadi, M. A Relationship between the Build and Texture Orientation in Tensile Loading of the Additively Manufactured Maraging Steels. Addit. Manuf. 2021, 41, 101954. [Google Scholar] [CrossRef]
- Song, J.; Tang, Q.; Feng, Q.; Ma, S.; Setchi, R.; Liu, Y.; Han, Q.; Fan, X.; Zhang, M. Effect of Heat Treatment on Microstructure and Mechanical Behaviours of 18Ni-300 Maraging Steel Manufactured by Selective Laser Melting. Opt. Laser Technol. 2019, 120, 105725. [Google Scholar] [CrossRef]
- Casati, R.; Lemke, J.N.; Tuissi, A.; Vedani, M. Aging Behaviour and Mechanical Performance of 18-Ni 300 Steel Processed by Selective Laser Melting. Metals 2016, 6, 218. [Google Scholar] [CrossRef]
- Tan, C.; Zhou, K.; Tong, X.; Huang, Y.; Li, J.; Ma, W.; Li, F.; Kuang, T. Microstructure and Mechanical Properties of 18Ni-300 Maraging Steel Fabricated by Selective Laser Melting; Atlantis Press: Amsterdam, The Netherlands, 2017; pp. 404–410. [Google Scholar]
- Kim, D.; Kim, T.; Ha, K.; Oak, J.-J.; Jeon, J.B.; Park, Y.; Lee, W. Effect of Heat Treatment Condition on Microstructural and Mechanical Anisotropies of Selective Laser Melted Maraging 18Ni-300 Steel. Metals 2020, 10, 410. [Google Scholar] [CrossRef]
- Wang, S.; Zhao, T.; Liu, Z.; Du, C.; Li, X. Exploration of the Processing Scheme of a Novel Ni(Fe, Al)-Maraging Steel. J. Mater. Res. Technol. 2021, 10, 225–239. [Google Scholar] [CrossRef]
- Yan, J.; Meng, X.; Ou, B.; Xie, Y.; Cai, B.; Zhang, Y.; Fang, S. Dependence of Microstructure and Properties in Additive Manufactured 18Ni300 on Heat Treatment and Surface Enhancement. J. Mater. Res. Technol. 2024, 29, 969–981. [Google Scholar] [CrossRef]
- Yin, S.; Chen, C.; Yan, X.; Feng, X.; Jenkins, R.; O’Reilly, P.; Liu, M.; Li, H.; Lupoi, R. The Influence of Aging Temperature and Aging Time on the Mechanical and Tribological Properties of Selective Laser Melted Maraging 18Ni-300 Steel. Addit. Manuf. 2018, 22, 592–600. [Google Scholar] [CrossRef]
- Mooney, B.; Kourousis, K.I. A Review of Factors Affecting the Mechanical Properties of Maraging Steel 300 Fabricated via Laser Powder Bed Fusion. Metals 2020, 10, 1273. [Google Scholar] [CrossRef]
- Nouri, N.; Li, Q.; Damon, J.; Mühl, F.; Graf, G.; Dietrich, S.; Schulze, V. Characterization of a Novel Maraging Steel for Laser-Based Powder Bed Fusion: Optimization of Process Parameters and Post Heat Treatments. J. Mater. Res. Technol. 2022, 18, 931–942. [Google Scholar] [CrossRef]
- Bae, K.; Shin, D.; Kim, J.-H.; Lee, W.; Jo, I.; Lee, J. Influence of Post Heat Treatment Condition on Corrosion Behavior of 18Ni300 Maraging Steel Manufactured by Laser Powder Bed Fusion. Micromachines 2022, 13, 1977. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Sha, W. Quantification of Precipitate Fraction in Maraging Steels by X-ray Diffraction Analysis. Mater. Sci. Technol. Mater. Sci. Technol. 2004, 20, 126–130. [Google Scholar] [CrossRef]
- Calro, M. Microstructural and Mechanical Properties of Maraging Steel Parts Produced by Selective Laser Melting. Master’s Thesis, School of Industrial and Information Engineering, Politecnico di Milano, Milan, Italy, 2015. [Google Scholar]
- Kempen, K.; Yasa, E.; Thijs, L.; Kruth, J.-P.; Van Humbeeck, J. Microstructure and Mechanical Properties of Selective Laser Melted 18Ni-300 Steel. Phys. Procedia 2011, 12, 255–263. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, C.; Zhang, S.L.; Ding, Y.L.; Ge, Q.L.; Su, J. Effect of Aging Temperature on the Precipitation Behavior and Mechanical Properties of Fe–Cr–Ni Maraging Stainless Steel. Mater. Sci. Eng. A 2021, 806, 140763. [Google Scholar] [CrossRef]
- Herzog, D.; Seyda, V.; Wycisk, E.; Emmelmann, C. Additive Manufacturing of Metals. Acta Mater. 2016, 117, 371–392. [Google Scholar] [CrossRef]
- Pardal, J.M.; Tavares, S.S.M.; Cindra Fonseca, M.P.; da Silva, M.R.; Neto, J.M.; Abreu, H.F.G. Influence of Temperature and Aging Time on Hardness and Magnetic Properties of the Maraging Steel Grade 300. J. Mater. Sci. 2007, 42, 2276–2281. [Google Scholar] [CrossRef]
- Bai, Y.; Wang, D.; Yang, Y.; Wang, H. Effect of Heat Treatment on the Microstructure and Mechanical Properties of Maraging Steel by Selective Laser Melting. Mater. Sci. Eng. A 2019, 760, 105–117. [Google Scholar] [CrossRef]
- Rohit, B.; Muktinutalapati, N.R. Austenite Reversion in 18% Ni Maraging Steel and Its Weldments. Mater. Sci. Technol. 2018, 34, 253–260. [Google Scholar] [CrossRef]
- Kong, D.; Dong, C.; Wei, S.; Ni, X.; Zhang, L.; Li, R.; Wang, L.; Man, C.; Li, X. About Metastable Cellular Structure in Additively Manufactured Austenitic Stainless Steels. Addit. Manuf. 2021, 38, 101804. [Google Scholar] [CrossRef]
- Kong, D.; Dong, C.; Ni, X.; Liang, Z.; Man, C.; Li, X. Hetero-Deformation-Induced Stress in Additively Manufactured 316 L Stainless Steel. Mater. Res. Lett. 2020, 8, 390–397. [Google Scholar] [CrossRef]
- Mei, X.; Yan, Y.; Fu, H.; Gao, X.; Huang, S.; Qiao, L. Effect of Aging Temperature on Microstructure Evolution and Strengthening Behavior of L-PBF 18Ni(300) Maraging Steel. Addit. Manuf. 2022, 58, 103071. [Google Scholar] [CrossRef]
- Wang, W.; Chen, Z.; Lu, W.; Meng, F.; Zhao, T. Heat Treatment for Selective Laser Melting of Inconel 718 Alloy with Simultaneously Enhanced Tensile Strength and Fatigue Properties. J. Alloys Compd. 2022, 913, 165171. [Google Scholar] [CrossRef]
- Lei, S.; Xu, Z.; Peng, L.; Lai, X. Effect of Grain Size on the Ductile-Brittle Fracture Behavior of Commercially Pure Titanium Sheet Metals. Mater. Sci. Eng. A 2021, 822, 141630. [Google Scholar] [CrossRef]
- Yingjie, L.; Xingui, B.; Keqiang, C. A Study on the Formation of Wear Debris during Abrasion. Tribol. Int. 1985, 18, 107–111. [Google Scholar] [CrossRef]
- Mabrouk, A.; Farhat, Z. Novel Ni-P-Tribaloy Composite Protective Coating. Materials 2023, 16, 3949. [Google Scholar] [CrossRef]
- Swain, B.; Bhuyan, S.; Behera, R.; Mohapatra, S.S.; Behera, A. Wear: A Serious Problem in Industry. In Tribology in Materials and Manufacturing—Wear, Friction and Lubrication; IntechOpen: London, UK, 2020; ISBN 978-1-83880-575-3. [Google Scholar]
- Hong, W.; Cai, W.; Wang, S.; Tomovic, M.M. Mechanical Wear Debris Feature, Detection, and Diagnosis: A Review. Chin. J. Aeronaut. 2018, 31, 867–882. [Google Scholar] [CrossRef]
- Wei, M.X.; Chen, K.M.; Wang, S.Q.; Cui, X.H. Analysis for Wear Behaviors of Oxidative Wear. Tribol. Lett. 2011, 42, 1–7. [Google Scholar] [CrossRef]
- Zhang, C. 14—Understanding the Wear and Tribological Properties of Ceramic Matrix Composites. In Advances in Ceramic Matrix Composites; Low, I.M., Ed.; Woodhead Publishing: Cambridge, UK, 2014; pp. 312–339. ISBN 978-0-85709-120-8. [Google Scholar]





| Element | Ni | Co | Mo | Ti | Al | Si | C | P | S | Fe |
|---|---|---|---|---|---|---|---|---|---|---|
| Wt% | 17.70 | 8.66 | 4.58 | 0.68 | 0.089 | 0.01 | 0.0043 | <0.005 | <0.003 | Bal. |
| Martensite (%) | Austenite (%) | |
|---|---|---|
| as-built | 100 | 0 |
| 430 °C | 98.8 | 1.2 |
| 490 °C | 87.1 | 12.9 |
| 550 °C | 78.2 | 21.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwak, N.; Lim, Y.; Heo, S.; Jeon, C.; Jo, I. Effect of Aging Temperature on Microstructure, Mechanical, and Wear Properties of 18Ni-300 Maraging Steel Produced by Powder Bed Fusion. Metals 2024, 14, 375. https://doi.org/10.3390/met14040375
Kwak N, Lim Y, Heo S, Jeon C, Jo I. Effect of Aging Temperature on Microstructure, Mechanical, and Wear Properties of 18Ni-300 Maraging Steel Produced by Powder Bed Fusion. Metals. 2024; 14(4):375. https://doi.org/10.3390/met14040375
Chicago/Turabian StyleKwak, Nawon, Yujin Lim, Seokha Heo, Chami Jeon, and Ilguk Jo. 2024. "Effect of Aging Temperature on Microstructure, Mechanical, and Wear Properties of 18Ni-300 Maraging Steel Produced by Powder Bed Fusion" Metals 14, no. 4: 375. https://doi.org/10.3390/met14040375
APA StyleKwak, N., Lim, Y., Heo, S., Jeon, C., & Jo, I. (2024). Effect of Aging Temperature on Microstructure, Mechanical, and Wear Properties of 18Ni-300 Maraging Steel Produced by Powder Bed Fusion. Metals, 14(4), 375. https://doi.org/10.3390/met14040375






