1. Introduction
Implementing the idea of a circular economy and the change in energy production methods associated with the green transformation present considerable challenges to the construction sector. These challenges include, among other things, reducing the energy and carbon intensity of construction processes by changing the organization of work and reducing the carbon footprint of construction materials.
Reducing the carbon footprint of construction materials can be achieved through changes in manufacturing processes related to developing innovative technologies, improving the efficiency of existing ones, introducing CCS (carbon capture and storage) technologies, using green energy and reducing waste generation, as well as using waste materials from other processes in their composition.
Using waste materials from other processes (often different types of ash) is desirable from the point of view of a circular economy and combating climate change. It brings challenges related to, among other things, obtaining and maintaining adequate properties of construction materials and eliminating risks to people and the environment. One such risk is the possibility of releasing heavy metals.
Table 1 shows the illustrative contents of selected heavy metals in cement, several types of ashes and tires (they are often used as an alternative fuel in cement plants).
The changes above are pushing manufacturers of construction materials, especially binders and concrete, to use new waste materials in their products. Such a material is fly ash from municipal sewage sludge incineration (SSA). Sewage sludge is formed during the biological wastewater treatment phase. It is by far the largest in volume among the by-products of wastewater treatments [
12]. Incomplete statistics indicate that between 2010 and 2021, the current EU-27 countries produced at least between 2.1 and nearly 7 million tons of municipal sewage sludge dry matter yearly [
13]. Despite the goals of the Sewage Sludge Directive [
14] related to increasing the amount of sewage sludge used in agriculture, much of the material is subject to incineration. It was 23.6% of the total production in 2011 [
15]. SSA is the most significant share of the by-products of municipal sewage sludge incineration.
Research into the use of SSA in the following areas is ongoing: for the production of phosphorous fertilizers [
16,
17,
18,
19], for wastewater treatment (after pre-processing) [
20], as a component in mineral binders [
21], in concrete [
22,
23,
24], in ceramic materials [
25,
26] and as a lightweight aggregate [
27]. Some of these applications require a change in the properties or pretreatment of the ash which unfortunately increases the cost and/or carbon footprint of the material. One potential use of SSA in its raw form is in self-hardening slurries (SHSs).
An SHS is a mixture of water, binder and clay material with the optional addition of other components. A detailed description of this type of building material can be found, for example, in the following articles [
28,
29]. A SHS is used in cut-off walls. Cut-off walls are vertical barriers built in the ground in order to limit the horizontal groundwater filtration flow, including that of contaminated water [
30]. They are used in dams, levees and landfills.
This paper focuses on determining the immobilization and release mechanism of heavy metals from SHSs with the addition of SSA destined for cut-off walls, considering the character of their operation. The study is important from the point of view of the following:
- -
By-product utilization. It draws attention to the need for prudent use of waste in construction materials.
- -
Waste management. It shows the possibility of using the waste produced in massive quantities.
- -
Environmental protection. It points out the safe possibility of using SSA in the building material from the point of view of groundwater heavy metal contamination.
2. Leaching of Heavy Metals from Cement-Based Materials
Not all the elemental content in the material is available for leaching. In the case of heavy metals, their release mainly depends on the strength of the bond, the chemical mechanisms of leaching, the physical factors of the environment and the form of the compound in which they are present. There are three chemical mechanisms that control the leaching of substances [
31]:
- -
Solubility control, the process which involves a disintegration of ionic lattices due to a solvent (e.g., ZnO + 2H+ ⇔ Zn2+ + H2O).
- -
Adsorption processes, in which a layer of adsorbate (metal ions) is formed on the surface of adsorbents and forms a molecular or atomic film [
32]. Adsorption is mainly classified into two types: physical adsorption and chemisorption [
33]. For example, the major adsorption scheme of Cu
2+ on the surface of alumina may be written as Cu
2+ + AlOH ⇔ AlOCu
+ + H
+ [
34].
- -
Ion exchange (availability control, sometimes called wash-out), the process which may be defined as the exchange of ions between the substrate and surrounding medium [
35]. Ion exchange can occur in the case of very easily soluble salts (such as NaCl), the release of which is not determined by the process of mineral solubility and adsorption [
36]. As a result of contact with water, they are rapidly released in massive quantities.
There are three basic processes of physical mass transport that determine the phenomenon of leaching of heavy metals [
31]:
- -
Advection process, in which dissolved or precipitated chemicals are dynamically transported by a flowing liquid. The release of heavy metals can be a long-term process due to the material’s exposure to fresh portions of liquid. A special case of advection is percolation, which is when a fluid flows through a porous material. This process mainly applies to materials in a fragmented form. For example, groundwater flows through a porous soil medium.
- -
Diffusion process, in which mass flow is caused by a concentration gradient generated in an isothermal single-phase system. Dissolved substances in a solvent spontaneously move from areas of higher concentration to regions of lower concentration, striving to reach equilibrium in the lack of fluid flow. The molecular cause of the diffusion phenomenon is the thermal movement of molecules. In this case, the form of the material does not matter.
- -
Surface wash-off process, in which the release of heavy metals occurs at the initial stage of leaching when the material is exposed to a fresh portion of the liquid. The process applies to materials in a monolithic form. It occurs only when unbound substances are available on the surface and often results in high concentrations of heavy metals in the early fractions of the eluate. Once the surface is depleted, further leaching from the monolithic forms takes place by diffusion.
The leaching of components from building materials is complex, and several of the listed earlier processes can co-occur, depending on the circumstances.
When considering the phenomenon of the leaching of heavy metals from construction materials, the entire life cycle of the material should be considered. Not only how and under what conditions it is used but also what happens after its end-of-life stage. The way it is re-used or landfilled and the form in which the material is found (monolithic or fragmented) also has a particular impact on the leachability of heavy metals [
32,
37]. It is, therefore, essential to adapt the testing procedure to the appropriate material use scenario [
31,
37,
38] and form [
5,
39].
In the case of monolithic forms (e.g., large concrete elements working in the ground or landfilled), it is believed that the release of metals is determined by diffusion and surface wash-off. With fragmented forms (e.g., concrete debris), the advection process plays a significant role. Conductivity (hydraulic permeability) determines how easily water can filter through a material and hence how quickly its components will be flushed out. In the case of fragmented materials with a lower conductivity coefficient, the liquid washes over the grains rather than flowing through them. Therefore, leaching can be controlled by diffusion or percolation (depending on the filtration rate). Due to their low permeability, some fragmented materials behave as monolithic forms.
In the literature, much attention has been paid to the effect of the soil-water environment’s pH and the material’s pore solution on the degree of heavy metal release [
31,
37,
40,
41,
42,
43]. The intensity of heavy metal leaching from cement-based materials depends on the pH of the environment in which the composite is located. Most cations form readily soluble compounds in acidic environments but hardly in alkaline ones. For anions, for some elements, the trend is the opposite.
Leaching is also affected by the age of the material [
41]. A mature material, especially cement-based, is likely to contain more stable phases that resist leaching [
40].
Composites based on mineral binders are used not only for construction purposes but also for environmental purposes. Among other things, cementitious binders are used to produce matrices that solidify waste containing heavy metals [
43]. Immobilization can occur in three ways: by sorption, the chemical incorporation of ions (e.g., by precipitation or exchange) and occlusion [
44] (mechanical retention of foreign ions or molecules inside crystals during precipitation [
45]). The most significant contribution to heavy metal immobilization processes is attributed to the components of the cement matrix, hydrated calcium silicates—the C-S-H phase (C stands for CaO, S stands for SiO
2, and H stands for H
2O, according to cement chemist notation) [
10]—the final product of the hydration of alite and belite. Hydration is a complex process of reacting compounds in cement with water; a detailed description of this process can be found in [
46].
The efficiency of heavy metal immobilization is closely related to the microstructure of the hardened binder [
43]. Adsorption properties characterize the C-S-H phase due to its highly developed specific surface area, amounting to (depending on the test method) 25 to 700 m
2/g [
47]. The gel molecules, connected by van der Waals forces, form pores between themselves, which are involved in the adsorption of heavy metal ions, mainly on the outer surfaces of the phase layers [
48]. In addition, the immobilization potential of C-S-H is influenced by their ability to retain and exchange (incorporate) foreign ions in their structure, the low permeability of the C-S-H gel, hindering the migration of absorbed substances [
10], and the high pH of the concrete pore solution, resulting in the formation of poorly soluble heavy metal compounds.
3. Materials
Table 2 shows the composition of the tested SHS and the content of selected heavy metals in it and its components.
Table 3 shows the properties of the tested SHS and the reference to the test method.
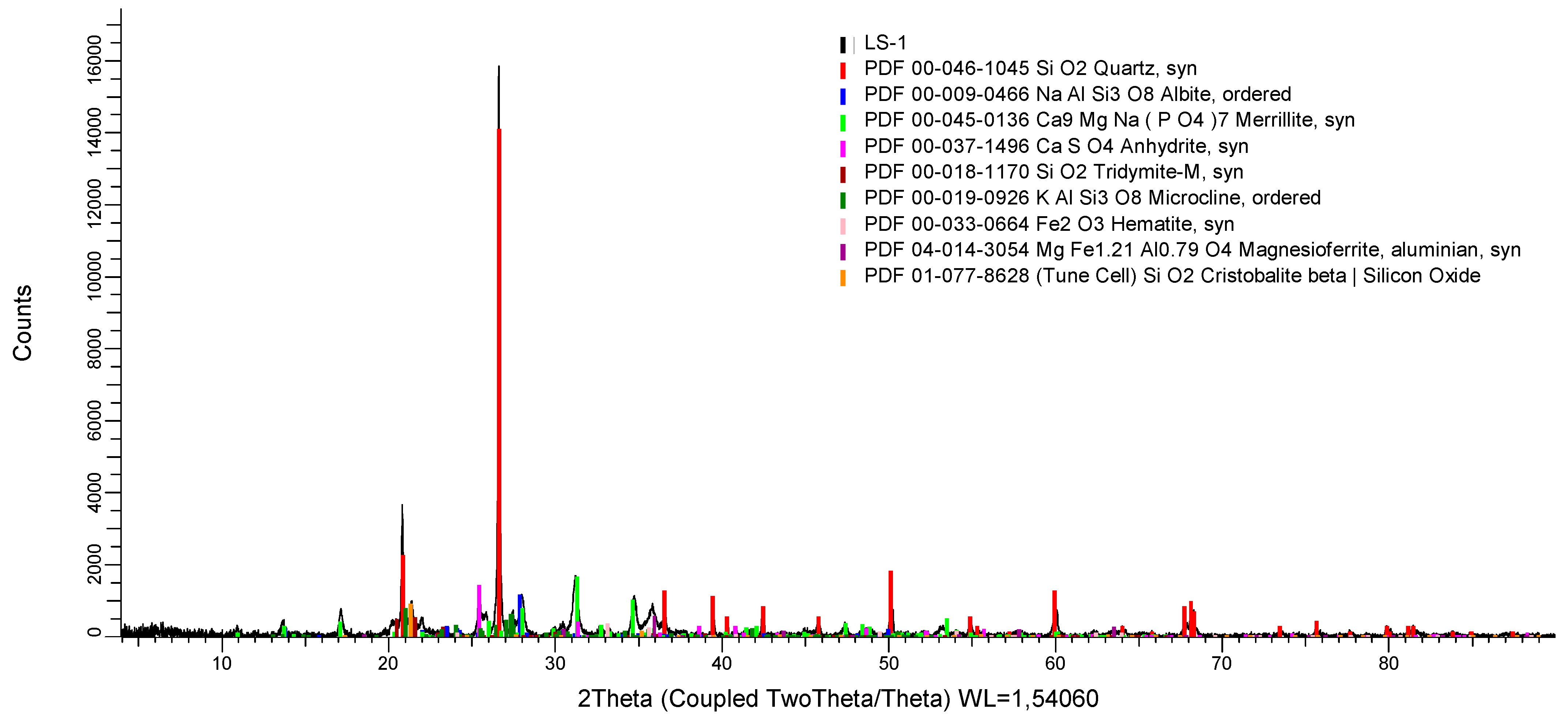
SSA from a municipal sewage treatment plant equipped with a Sewage Sludge Thermal Treatment Station was used in the SHS. SSA was collected in the multicyclone dust separator in the process line responsible for flue gas cleaning. Selected chemical and physical properties of SSA are shown in
Table 4. The phase composition is shown in
Figure 1. The examined SSA exhibits a relatively high content of phosphorus (
Table 4) and heavy metals (
Table 3). Its high water demand and fineness, and low hydraulic and pozzolanic activity (
Table 4), hinder its application within concrete technology.
Considering the size of the reflections (
Figure 1), it can be concluded that quartz was probably the most numerous phase in the studied sample. This is confirmed by an examination of the chemical composition of the ash (
Table 2), in which silicon dioxide is the dominant compound. The background of the sample probably indicates the low amorphousness of the material. The content of anhydrite (derived from the flue gas desulfurization process), as well as calcium oxide, aluminum oxide and reactive silicon dioxide (
Table 2), may indicate the ash’s binding potential, but this is not confirmed by activity index studies (
Table 4). The occurrence of phases that include phosphorus is confirmed by the literature data on the relatively high content of this element in SSA [
55,
56].
4. Methods and Procedures
The main chemical mechanism of leaching is considered to be dissolution, occurring as a result of contact of the substance with water (solvent). Still, the release process tends to be complex, and in addition to chemistry, physical transport processes are also critical. Therefore, it is impossible to cover all essential leaching aspects in a single test method based on current knowledge. The authors of [
57] pointed out the need to match the leaching test to the conditions of the material’s actual impact on the environment. Therefore, it was necessary to develop a method that simulates the actual application of SHS in cut-off walls—called the filtration leaching test.
4.1. Filtration Leaching Test
The filtration leaching test is based on the use scenario of an SHS in cut-off walls, in which the porous material is exposed to penetration (filtration) of a liquid with different degrees of aggressiveness. The test procedure allows for the filtration of a liquid with preset properties through the sample at a constant or variable hydraulic gradient. The filtration speed depends on the material’s properties under test, as well as the gradient (which reflects the material’s behavior in the working environment). A quasi-constant hydraulic gradient was used in the test method, so the filtration velocity fluctuated due to the processes occurring in the test material and could not be controlled.
The proposed eluate collection procedure can be a particular extrapolation of the percolation method [
58] to monolithic materials with a filtration coefficient of k < 10
−8 m/s. Therefore, the eluate was taken in 7 fractions until the ratio of L/S = 10 dm
3/kg d.m. (dry matter) was obtained according to [
58]. The L/S ratio used is in accordance with the provisions of [
59]. The leaching liquid was distilled water.
Reaction and specific conductance were measured immediately after collection in all eluate fractions. The device used was an Elmetron CPC-511 equipped with an ECF-1 conductivity sensor, an IJ-44C compound electrode and a temperature sensor. The obtained eluates were filtered through ethanol-wetted PTFE membrane filters with a pore diameter of ϕ = 0.45 μm. The filtered fraction was acidified by adding about 1 mL of concentrated nitric acid(V) solution per 100 mL of eluate and tested for heavy metal content.
The SHS samples for testing were made in cylindrical, chemically resistant polyvinyl chloride (PVC) molds with dimensions as in
Figure 2. Until hardening, the samples were stored in molds covered with foil to prevent drying of the material. After 2–7 days, the samples in the molds were fully immersed in tap water (at +18 ± 2 °C), where they were matured until testing. The properties were determined after 28 days of curing the samples.
Heavy metal release calculations were analogous to the percolation method [
58]. In all fractions of the eluate, the amounts of heavy metals released were calculated according to Formula (1).
Here,
released quantity of the component in eluate fraction
i, converted to material dry matter [mg/kg d.m.];
eluate
i fraction volume [dm
3];
concentration of the considered component in eluate
i fraction [mg/dm
3];
sample dry matter [kg]; and
eluate fraction index (
i = 1, 2, …, 7) [-].
The cumulative released quantity from the sample was calculated according to Formula (2).
When the concentration of the test component was below the limit of determination, two calculations of
values were conducted. The upper limit of
was obtained by substituting into Formula (1)
equal to the limit of quantification, and the lower limit by substituting
equal to 0 mg/dm
3. Similarly, in the case of the cumulative released quantity of the component, this was performed.
It was impossible to meet the standard’s requirements [
58] for the liquid flow speed through the samples. The flow rate, and thus also the contact time of the leaching medium with the sample and the length of the test period, depended on the properties of the test material and differed for each sample.
4.2. Release Mechanism
The release mechanism was analyzed according to the method described in the works [
32,
57]. For this purpose, graphs of heavy metal release (component release, cumulative component release and component concentration versus cumulative L/S ratio) were prepared. The graphs obtained were compared with the literature data (
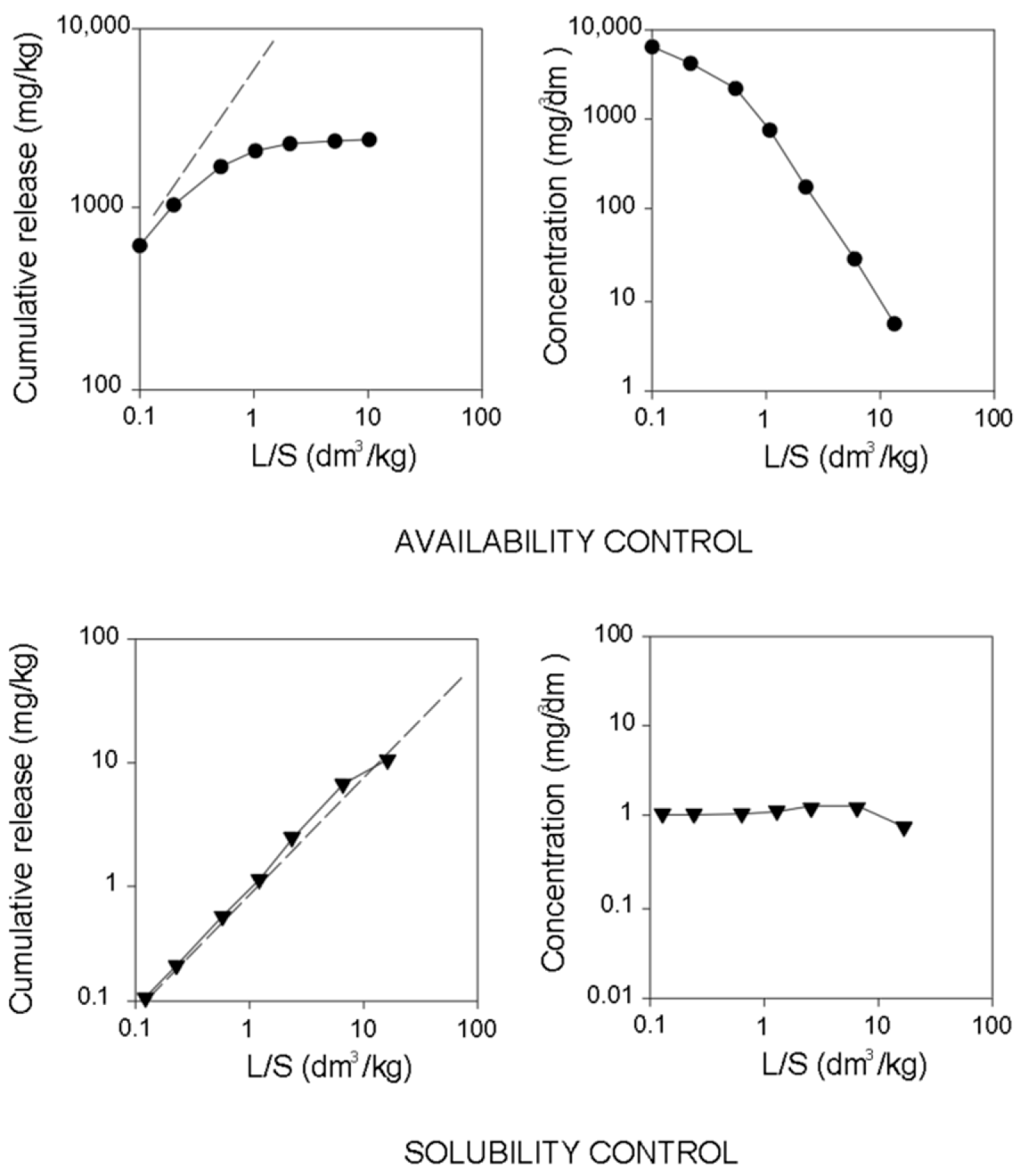
Figure 3). Based on the similarity between the graphs obtained and the theoretical shapes of the curves of leaching and the concentration of the components (
Figure 3), the processes controlling the release of heavy metals from the materials studied were determined.
4.3. Determination of Heavy Metal Concentrations in Eluates
The total content of heavy metals (Cd, Cr, Cu, Pb and Zn) was determined by flame atomization atomic absorption spectrometry (F-AAS) on a PERKIN ELMER AAnalyst 300. The standard curves used for the study were determined from MERC’s standard solutions. A 100 mm torch fed with a stoichiometric air–acetylene mixture was used. The flame reached temperatures in the range of 2100–2300 °C. The radiation source was a cavity cathode lamp made by Philips. The method’s limits of quantification for the elements analyzed are listed in
Table 5.
4.4. Statistical Analysis
To examine a relationship between two variables when the other variables were excluded, the partial correlation coefficients were used. The partial correlation coefficients were used to analyze the relationship between the pH value of the eluates, the test duration or the cumulative L/S ratio and the concentration of individual metals (only fractions of eluates with concentrations above the limit of determination were used for calculations). Due to the abnormal form of the distribution of the obtained results, the number of cases, as well as the often clearly non-linear relationships, Spearman’s rank correlation coefficient ρc, was used. The significance of correlation coefficients was evaluated at the significance level of α = 0.05.
The complete differential method determined uncertainty in the released quantity of the component.
4.5. Immobilization Level
The level of immobilization of heavy metals in the SHS was determined using Formula (3).
Here,
I—immobilization level [%];
mi,e—mass of
i heavy metal in eluate [mg]; and
mi,m—mass of
i heavy metal in the material subjected to leaching [mg].