PvMSP8 as a Novel Plasmodium vivax Malaria Sero-Marker for the Peruvian Amazon
Abstract
:1. Introduction
2. Results
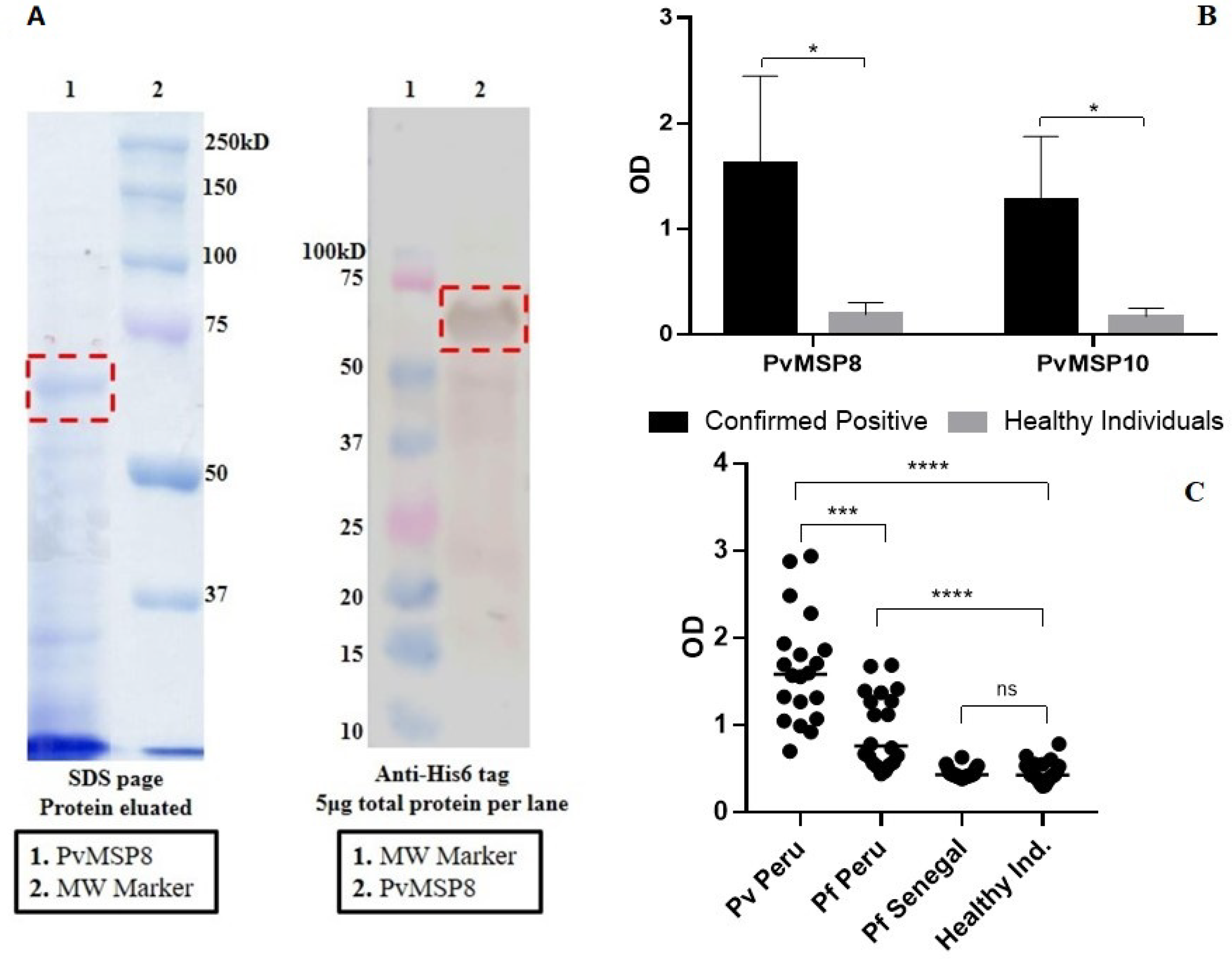
2.1. Preliminary Assessment after PvMSP8 Protein Production
2.2. Evaluation Using Samples of Individuals with Known History of P. vivax Exposure (Cohort Study)
2.3. Evaluation Using Samples Collected in a Population-Based Cross-Sectional Survey
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Study Design
5.2. Samples of Individuals with Known History of P. vivax Exposure (Cohort Study)
5.3. Samples Collected in a Population-Based Cross-Sectional Survey
5.4. Laboratory Procedures
5.4.1. Microscopy
5.4.2. Real-Time Quantitative PCR (qPCR)
5.4.3. Recombinant Proteins
5.4.4. Luminex
5.4.5. ELISA
5.4.6. Data Management and Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Appendix A
- The post-signal amino acid sequences of PvMSP8 (PVX_097625) were synthesized in Sf9 insect cell-optimized codons and cloned into the pFastBacDual expression plasmid. Signal peptides were not included in the recombinant protein, which was engineered to have a His-Tag for purification.
- PLAS PVX_097625 PvMSP8 amino acid sequence (highlighted in yellow), including His-tag (underlined)
- MEGNVSPPNFNDNRVNGNNGNKGNGNDNDVPSFIGGNNNNVNGNNDDNIFNKNGKDVTRNDGDAKDGENRNNKKNENGSGSNENNSIANADNGSGKSDANANQIDEDGNKMDEASLKKILKIVDEMENIQGLLDGDYSILDKYSVKLVDEDDGETNKRKIIGEYDLKMLKNILLFREKISRVCENKYNKNLPVLLKKCSNVDDPKLSKSREKIKKGLAKNNMSIEDFVVGLLEDLFEKINEHFIKDDSFDLSDYLADFELINYIIMHETSELIDELLNIIESMNFRLESGSLEKMVKSAESGMNLNCKMKEDIIHLLKKSSAKFFKIEIDRKTKMIYPVQATHKGANMKQLALSFLQKNNVCEHKKCPLNSNCYVINGEEVCRCLPGFSDVKIDNVMNCVRDDTLDCSNNNGGCDVNATCTLIDKKIVCECKDNFEGDGIYCGHHHHHH
- PVX_097625 PvMSP8 DNA sequence (highlighted in yellow), including His-tag (underlined) and EcoRI (in red) and HindIII (in green) restriction sites for cloning in to pFastBac expression vector.
- GAATTCGGCATGGAAGGAAACGTTAGCCCACCCAACTTTAATGACAACAGGGTAAACGGCAACAATGGAAATAAAGGCAACGGAAATGACAACGACGTGCCGTCGTTCATTGGAGGAAACAATAATAACGTGAACGGCAATAATGATGATAACATTTTTAATAAAAATGGAAAGGATGTCACCCGAAATGATGGCGATGCAAAGGATGGAGAAAATCGAAATAACAAGAAAAACGAAAATGGCAGTGGCTCCAATGAGAATAACTCCATTGCAAATGCGGACAATGGTAGCGGCAAATCTGATGCGAATGCCAACCAAATTGATGAGGATGGAAATAAAATGGATGAAGCATCTTTAAAGAAAATCCTCAAAATTGTAGACGAAATGGAAAATATTCAAGGACTGCTCGATGGAGATTACAGCATTTTGGATAAGTACAGTGTCAAATTAGTTGATGAAGATGATGGAGAAACGAATAAAAGAAAAATCATTGGAGAATATGATTTGAAAATGTTAAAAAATATTTTATTGTTCAGAGAAAAAATTTCCCGAGTTTGTGAAAATAAATACAATAAAAATTTACCCGTCTTGTTAAAAAAATGCTCAAATGTGGATGACCCCAAATTGAGTAAATCCAGGGAAAAAATTAAAAAAGGATTAGCAAAAAATAATATGAGCATTGAAGATTTTGTGGTAGGTTTGTTGGAAGATTTATTTGAGAAAATTAATGAACATTTTATTAAAGACGATTCATTTGATTTGAGTGACTATTTAGCCGATTTCGAGCTCATCAATTATATAATTATGCACGAAACGTCCGAATTGATCGATGAGCTTTTGAACATAATAGAGTCCATGAATTTCAGATTGGAATCCGGATCTTTGGAGAAAATGGTTAAATCTGCAGAATCAGGAATGAACTTAAATTGCAAAATGAAGGAAGACATAATTCACTTACTTAAGAAATCCTCCGCCAAATTTTTTAAAATCGAAATTGACAGAAAGACCAAGATGATATACCCAGTGCAGGCTACACACAAAGGTGCCAACATGAAACAACTCGCCCTGAGCTTCCTCCAGAAGAACAATGTATGTGAACATAAAAAGTGCCCATTGAACTCCAACTGCTATGTTATAAATGGAGAGGAGGTCTGCAGATGTCTACCCGGATTTAGCGACGTCAAAATTGATAACGTGATGAACTGCGTTAGGGATGATACCCTTGACTGTAGCAACAACAACGGTGGCTGTGATGTGAACGCAACGTGTACCCTTATAGACAAAAAAATTGTGTGTGAATGCAAGGACAACTTTGAGGGAGACGGAATATACTGCGGCCATCATCACCATCACCACTAGAAGCTT

References
- Tizifa, T.A.; Kabaghe, A.N.; McCann, R.S.; van den Berg, H.; Van Vugt, M.; Phiri, K.S. Prevention Efforts for Malaria. Curr. Trop. Med. Rep. 2018, 5, 41–50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosas-Aguirre, A.; Gamboa, D.; Manrique, P.; Conn, J.E.; Moreno, M.; Lescano, A.G.; Sanchez, J.F.; Rodriguez, H.; Silva, H.; Llanos-Cuentas, A.; et al. Epidemiology of Plasmodium Vivax Malaria in Peru. Am. J. Trop. Med. Hyg. 2016, 95, 133–144. [Google Scholar] [CrossRef] [Green Version]
- Ministerio de Salud del Peru (MINSA). Documento Técnico Plan Malaria Cero Período 2017–2021—RESOLUCION MINISTERIAL-No 244-2017MINSA. PDF 2017. Available online: https://cdn.www.gob.pe/uploads/document/file/190350/189846_RM_244-2017-MINSA.PDF20180823-24725-1yk3i0y.PDF (accessed on 21 February 2021).
- Ministerio de Salud del Peru (MINSA). Fuente: Centro Nacional de Epidemiologia, Prevención y Control de Enfermedades MINSA. Sala de situación de salud Perú a la SE 52-2018. Available online: http://www.dge.gob.pe/portal/docs/vigilancia/sala/2018/salaSE52.zip (accessed on 28 February 2021).
- Ministerio de Salud del Peru (MINSA). Centro Nacional de Epidemiologia, Prevención y Control de Enfermedades—MINSA. Sala de situación de salud Perú a la SE 52-2019. Available online: http://www.dge.gob.pe/portal/docs/vigilancia/sala/2019/salaSE52.zip (accessed on 28 February 2021).
- Adams, J.H.; Mueller, I. The Biology of Plasmodium Vivax. Cold Spring Harb. Perspect. Med. 2017, 7, a025585. [Google Scholar] [CrossRef] [Green Version]
- Greenhouse, B.; Daily, J.; Guinovart, C.; Goncalves, B.; Beeson, J.; Bell, D.; Chang, M.A.; Cohen, J.M.; Ding, X.; Domingo, G.; et al. Priority Use Cases for Antibody-Detecting Assays of Recent Malaria Exposure as Tools to Achieve and Sustain Malaria Elimination. Gates Open Res. 2019, 3, 131. [Google Scholar] [CrossRef] [PubMed]
- Longley, R.J.; White, M.T.; Takashima, E.; Brewster, J.; Morita, M.; Harbers, M.; Obadia, T.; Robinson, L.J.; Matsuura, F.; Liu, Z.S.J.; et al. Development and Validation of Serological Markers for Detecting Recent Plasmodium Vivax Infection. Nat. Med. 2020, 26, 741–749. [Google Scholar] [CrossRef]
- Cunha, M.G.; Silva, E.S.; Sepúlveda, N.; Costa, S.P.T.; Saboia, T.C.; Guerreiro, J.F.; Póvoa, M.M.; Corran, P.H.; Riley, E.; Drakeley, C.J. Serologically Defined Variations in Malaria Endemicity in Pará State, Brazil. PLoS ONE 2014, 9, e113357. [Google Scholar] [CrossRef] [Green Version]
- Rosas-Aguirre, A.; Llanos-Cuentas, A.; Speybroeck, N.; Cook, J.; Contreras-Mancilla, J.; Soto, V.; Gamboa, D.; Pozo, E.; Ponce, O.J.; Pereira, M.O.; et al. Assessing Malaria Transmission in a Low Endemicity Area of North-Western Peru. Malar. J. 2013, 12, 339. [Google Scholar] [CrossRef] [PubMed]
- Cook, J.; Reid, H.; Iavro, J.; Kuwahata, M.; Taleo, G.; Clements, A.; McCarthy, J.; Vallely, A.; Drakeley, C. Using Serological Measures to Monitor Changes in Malaria Transmission in Vanuatu. Malar. J. 2010, 9, 169. [Google Scholar] [CrossRef] [Green Version]
- Cook, J.; Speybroeck, N.; Sochanta, T.; Somony, H.; Sokny, M.; Claes, F.; Lemmens, K.; Theisen, M.; Soares, I.S.; D’Alessandro, U.; et al. Sero-Epidemiological Evaluation of Changes in Plasmodium Falciparum and Plasmodium Vivax Transmission Patterns over the Rainy Season in Cambodia. Malar. J. 2012, 11, 86. [Google Scholar] [CrossRef] [PubMed]
- Rosas-Aguirre, A.; Speybroeck, N.; Llanos-Cuentas, A.; Rosanas-Urgell, A.; Carrasco-Escobar, G.; Rodriguez, H.; Gamboa, D.; Contreras-Mancilla, J.; Alava, F.; Soares, I.S.; et al. Hotspots of Malaria Transmission in the Peruvian Amazon: Rapid Assessment through a Parasitological and Serological Survey. PLoS ONE 2015, 10, e0137458. [Google Scholar] [CrossRef] [PubMed]
- Chuquiyauri, R.; Brouwer, K.C.; Felgner, P.; Gilman, R.H.; Molina, D.M.; Moss, E.L.; Vinetz, J.M.; Gardner, M.J.; Neafsey, D.E.; Torres, S.; et al. Genome-Scale Protein Microarray Comparison of Human Antibody Responses in Plasmodium Vivax Relapse and Reinfection. Am. J. Trop. Med. Hyg. 2015, 93, 801–809. [Google Scholar] [CrossRef] [Green Version]
- Rosas-Aguirre, A.; Patra, K.P.; Calderón, M.; Torres, K.; Gamboa, D.; Arocutipa, E.; Málaga, E.; Garro, K.; Fernández, C.; Trompeter, G.; et al. Anti–MSP-10 IgG Indicates Recent Exposure to Plasmodium Vivax Infection in the Peruvian Amazon. JCI Insight 2020, 5, e130769. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mandrekar, J.N. Receiver Operating Characteristic Curve in Diagnostic Test Assessment. J. Thorac. Oncol. 2010, 5, 1315–1316. [Google Scholar] [CrossRef] [Green Version]
- Pan, W.; Ravot, E.; Tolle, R.; Frank, R.; Mosbach, R.; Türbachova, I.; Bujard, H. Vaccine Candidate MSP-1 from Plasmodium Falciparum: A Redesigned 4917 Bp Polynucleotide Enables Synthesis and Isolation of Full-Length Protein from Escherichia Coli and Mammalian Cells. Nucleic Acids Res. 1999, 27, 1094–1103. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Black, C.G.; Wang, L.; Wu, T.; Coppel, R.L. Apical Location of a Novel EGF-like Domain-Containing Protein of Plasmodium Falciparum. Mol. Biochem. Parasitol. 2003, 127, 59–68. [Google Scholar] [CrossRef]
- Giraldo, M.A.; Arevalo-Pinzon, G.; Rojas-Caraballo, J.; Mongui, A.; Rodriguez, R.; Patarroyo, M.A. Vaccination with Recombinant Plasmodium Vivax MSP-10 Formulated in Different Adjuvants Induces Strong Immunogenicity but No Protection. Vaccine 2009, 28, 7–13. [Google Scholar] [CrossRef]
- Rodrigues, M.H.C.; Cunha, M.G.; Machado, R.L.; Ferreira, O.C.; Rodrigues, M.M.; Soares, I.S. Serological Detection of Plasmodium Vivax Malaria Using Recombinant Proteins Corresponding to the 19-KDa C-Terminal Region of the Merozoite Surface Protein-1. Malar. J. 2003, 2, 39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, Y.; Wang, B.; Changrob, S.; Han, J.-H.; Sattabongkot, J.; Ha, K.-S.; Chootong, P.; Lu, F.; Cao, J.; Nyunt, M.H.; et al. Naturally Acquired Humoral and Cellular Immune Responses to Plasmodium Vivax Merozoite Surface Protein 8 in Patients with P. Vivax Infection. Malar. J. 2017, 16, 211. [Google Scholar] [CrossRef]
- Chen, J.-H.; Jung, J.-W.; Wang, Y.; Ha, K.-S.; Lu, F.; Lim, C.S.; Takeo, S.; Tsuboi, T.; Han, E.-T. Immunoproteomics Profiling of Blood Stage Plasmodium Vivax Infection by High-Throughput Screening Assays. J. Proteome Res. 2010, 9, 6479–6489. [Google Scholar] [CrossRef]
- Hostetler, J.B.; Sharma, S.; Bartholdson, S.J.; Wright, G.J.; Fairhurst, R.M.; Rayner, J.C. A Library of Plasmodium Vivax Recombinant Merozoite Proteins Reveals New Vaccine Candidates and Protein-Protein Interactions. PLoS Negl. Trop. Dis. 2015, 9, e0004264. [Google Scholar] [CrossRef]
- Carrasco-Escobar, G.; Gamboa, D.; Castro, M.C.; Bangdiwala, S.I.; Rodriguez, H.; Contreras-Mancilla, J.; Alava, F.; Speybroeck, N.; Lescano, A.G.; Vinetz, J.M.; et al. Micro-Epidemiology and Spatial Heterogeneity of P. Vivax Parasitaemia in Riverine Communities of the Peruvian Amazon: A Multilevel Analysis. Sci. Rep. 2017, 7, 8082. [Google Scholar] [CrossRef] [Green Version]
- Bendezu, J.; Villasis, E.; Morales Ruiz, S.; Garro, K.; Infante, B.; Gutierrez-Loli, R.; Rodríguez, P.; Fernández-Díaz, M.; Gamboa, D.; Torres, K. Evaluation of Plasmodium Falciparum MSP10 and Its Development as a Serological Tool for the Peruvian Amazon Region. Malar. J. 2019, 18, 327. [Google Scholar] [CrossRef] [PubMed]
- Victor, M.E.; Bengtsson, A.; Andersen, G.; Bengtsson, D.; Lusingu, J.P.; Vestergaard, L.S.; Arnot, D.E.; Theander, T.G.; Joergensen, L.; Jensen, A.T.R. Insect Cells Are Superior to Escherichia Coli in Producing Malaria Proteins Inducing IgG Targeting PfEMP1 on Infected Erythrocytes. Malar. J. 2010, 9, 325. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perraut, R.; Joos, C.; Sokhna, C.; Polson, H.E.J.; Trape, J.-F.; Tall, A.; Marrama, L.; Mercereau-Puijalon, O.; Richard, V.; Longacre, S. Association of Antibody Responses to the Conserved Plasmodium Falciparum Merozoite Surface Protein 5 with Protection against Clinical Malaria. PLoS ONE 2014, 9, e101737. [Google Scholar] [CrossRef]
- Perraut, R.; Varela, M.-L.; Joos, C.; Diouf, B.; Sokhna, C.; Mbengue, B.; Tall, A.; Loucoubar, C.; Touré, A.; Mercereau-Puijalon, O. Association of Antibodies to Plasmodium Falciparum Merozoite Surface Protein-4 with Protection against Clinical Malaria. Vaccine 2017, 35, 6720–6726. [Google Scholar] [CrossRef]
- Pizarro, J.C.; Chitarra, V.; Verger, D.; Holm, I.; Pêtres, S.; Dartevelle, S.; Nato, F.; Longacre, S.; Bentley, G.A. Crystal Structure of a Fab Complex Formed with PfMSP1-19, the C-Terminal Fragment of Merozoite Surface Protein 1 from Plasmodium Falciparum: A Malaria Vaccine Candidate. J. Mol. Biol. 2003, 328, 1091–1103. [Google Scholar] [CrossRef]
- Joos, C.; Varela, M.-L.; Mbengue, B.; Mansourou, A.; Marrama, L.; Sokhna, C.; Tall, A.; Trape, J.-F.; Touré, A.; Mercereau-Puijalon, O.; et al. Antibodies to Plasmodium Falciparum Merozoite Surface Protein-1p19 Malaria Vaccine Candidate Induce Antibody-Dependent Respiratory Burst in Human Neutrophils. Malar. J. 2015, 14, 409. [Google Scholar] [CrossRef]
- Jarvis, D.L. Baculovirus-Insect Cell Expression Systems. Meth. Enzymol. 2009, 463, 191–222. [Google Scholar] [CrossRef]
- Mehlin, C.; Boni, E.; Buckner, F.S.; Engel, L.; Feist, T.; Gelb, M.H.; Haji, L.; Kim, D.; Liu, C.; Mueller, N.; et al. Heterologous Expression of Proteins from Plasmodium Falciparum: Results from 1000 Genes. Mol. Biochem. Parasitol. 2006, 148, 144–160. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Jain, A.; Liang, L.; Obiero, J.M.; Hamapumbu, H.; Stevenson, J.C.; Thuma, P.E.; Lupiya, J.; Chaponda, M.; Mulenga, M.; et al. Distinct Antibody Signatures Associated with Different Malaria Transmission Intensities in Zambia and Zimbabwe. mSphere 2019, 4. [Google Scholar] [CrossRef] [Green Version]
- Perez-Leal, O.; Sierra, A.Y.; Barrero, C.A.; Moncada, C.; Martinez, P.; Cortes, J.; Lopez, Y.; Torres, E.; Salazar, L.M.; Patarroyo, M.A. Plasmodium Vivax Merozoite Surface Protein 8 Cloning, Expression, and Characterisation. Biochem. Biophys. Res. Commun. 2004, 324, 1393–1399. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Wang, B.; Sattabongkot, J.; Lim, C.S.; Tsuboi, T.; Han, E.-T. Immunogenicity and Antigenicity of Plasmodium Vivax Merozoite Surface Protein 10. Parasitol. Res. 2014, 113, 2559–2568. [Google Scholar] [CrossRef]
- Elshal, M.F.; McCoy, J.P. Multiplex Bead Array Assays: Performance Evaluation and Comparison of Sensitivity to ELISA. Methods 2006, 38, 317–323. [Google Scholar] [CrossRef] [Green Version]
- Kingsmore, S.F. Multiplexed Protein Measurement: Technologies and Applications of Protein and Antibody Arrays. Nat. Rev. Drug. Discov. 2006, 5, 310–320. [Google Scholar] [CrossRef] [Green Version]
- Moncunill, G.; Campo, J.J.; Dobaño, C. Quantification of Multiple Cytokines and Chemokines Using Cytometric Bead Arrays. In Cytokine Bioassays: Methods and Protocols; Vancurova, I., Ed.; Methods in Molecular Biology; Springer: New York, NY, USA, 2014; pp. 65–86. ISBN 978-1-4939-0928-5. [Google Scholar]
- Amanna, I.J.; Slifka, M.K. Mechanisms That Determine Plasma Cell Lifespan and the Duration of Humoral Immunity. Immunol. Rev. 2010, 236, 125–138. [Google Scholar] [CrossRef]
- Carter, M.J.; Mitchell, R.M.; Meyer Sauteur, P.M.; Kelly, D.F.; Trück, J. The Antibody-Secreting Cell Response to Infection: Kinetics and Clinical Applications. Front. Immunol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Ly, A.; Hansen, D.S. Development of B Cell Memory in Malaria. Front. Immunol. 2019, 10, 559. [Google Scholar] [CrossRef] [PubMed]
- Silveira, E.L.V.; Dominguez, M.R.; Soares, I.S. To B or Not to B: Understanding B Cell Responses in the Development of Malaria Infection. Front. Immunol. 2018, 9, 2961. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clark, E.H.; Silva, C.J.; Weiss, G.E.; Li, S.; Padilla, C.; Crompton, P.D.; Hernandez, J.N.; Branch, O.H. Plasmodium Falciparum Malaria in the Peruvian Amazon, a Region of Low Transmission, Is Associated with Immunologic Memory. Infect. Immun. 2012, 80, 1583–1592. [Google Scholar] [CrossRef] [Green Version]
- Elliott, S.R.; Fowkes, F.J.I.; Richards, J.S.; Reiling, L.; Drew, D.R.; Beeson, J.G. Research Priorities for the Development and Implementation of Serological Tools for Malaria Surveillance. F1000Prime Rep. 2014, 6. [Google Scholar] [CrossRef]
- Longley, R.J.; Sattabongkot, J.; Mueller, I. Insights into the Naturally Acquired Immune Response to Plasmodium Vivax Malaria. Parasitology 2016, 143, 154–170. [Google Scholar] [CrossRef]
- Mount, A.M.; Mwapasa, V.; Elliott, S.R.; Beeson, J.G.; Tadesse, E.; Lema, V.M.; Molyneux, M.E.; Meshnick, S.R.; Rogerson, S.J. Impairment of Humoral Immunity to Plasmodium Falciparum Malaria in Pregnancy by HIV Infection. Lancet 2004, 363, 1860–1867. [Google Scholar] [CrossRef]
- Flateau, C.; Le Loup, G.; Pialoux, G. Consequences of HIV Infection on Malaria and Therapeutic Implications: A Systematic Review. Lancet Infect. Dis. 2011, 11, 541–556. [Google Scholar] [CrossRef]
- Fillol, F.; Sarr, J.B.; Boulanger, D.; Cisse, B.; Sokhna, C.; Riveau, G.; Simondon, K.B.; Remoué, F. Impact of Child Malnutrition on the Specific Anti-Plasmodium Falciparum Antibody Response. Malar. J. 2009, 8, 116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Genton, B.; Al-Yaman, F.; Ginny, M.; Taraika, J.; Alpers, M.P. Relation of Anthropometry to Malaria Morbidity and Immunity in Papua New Guinean Children. Am. J. Clin. Nutr. 1998, 68, 734–741. [Google Scholar] [CrossRef] [Green Version]
- Greenhouse, B.; Smith, D.L.; Rodríguez-Barraquer, I.; Mueller, I.; Drakeley, C.J. Taking Sharper Pictures of Malaria with CAMERAs: Combined Antibodies to Measure Exposure Recency Assays. Am. J. Trop. Med. Hyg. 2018, 99, 1120–1127. [Google Scholar] [CrossRef] [Green Version]
- Medzihradsky, O.F.; Kleinschmidt, I.; Mumbengegwi, D.; Roberts, K.W.; McCreesh, P.; Dufour, M.-S.K.; Uusiku, P.; Katokele, S.; Bennett, A.; Smith, J.; et al. Study Protocol for a Cluster Randomised Controlled Factorial Design Trial to Assess the Effectiveness and Feasibility of Reactive Focal Mass Drug Administration and Vector Control to Reduce Malaria Transmission in the Low Endemic Setting of Namibia. BMJ Open 2018, 8, e019294. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Molineaux, L.; Gramiccia, G. The Garki Project: Research on the Epidemiology and Control of Malaria in the Sudan Savanna of West Africa; World Health Organization: Geneva, Switzerland, 1980; ISBN 978-92-4-156061-0. [Google Scholar]
- Stresman, G.; Cameron, A.; Drakeley, C. Freedom from Infection: Confirming Interruption of Malaria Transmission. Trends Parasitol. 2017, 33, 345–352. [Google Scholar] [CrossRef]
- Kochayoo, P.; Kittisenachai, N.; Changrob, S.; Wangriatisak, K.; Muh, F.; Chootong, P.; Han, E.-T. The Acquisition of Long-Lived Memory B Cell Responses to Merozoite Surface Protein-8 in Individuals with Plasmodium Vivax Infection. Malar. J. 2019, 18, 188. [Google Scholar] [CrossRef] [Green Version]
- de Carvalho, M.E.; Glasser, C.M.; de Ciaravolo, R.M.C.; Etzel, A.; dos Santos, L.A.; Ferreira, C.S. Sorologia de malária vivax no foco Aldeia dos Índios, município de Peruíbe, Estado de São Paulo, 1984 a 1986. Cad. Saúde Pública 1988, 4, 276–292. [Google Scholar] [CrossRef] [Green Version]
- Ministerio de Salud del Peru (MINSA). Fuente: Centro Nacional de Epidemiologia, Prevención y Control de Enfermedades MINSA. Sala de situación de salud Perú a la Semana Epidemiologica N°52-2012. Available online: http://www.dge.gob.pe/vigilancia/sala/2012/salaSE52.zip (accessed on 28 February 2021).
- Ministerio de Salud del Perú (MINSA). Norma Técnica Para La Atención de La Malaria y Malaria Severa En El Perú. Available online: Http://Bvs.Minsa.Gob.Pe/Local/MINSA/4378.Pdf (accessed on 21 February 2021).
- Velasco, A.; Instituto Nacional de Salud (Perú). Norma Técnica de Salud Para el Control de Calidad del Diagnóstico Microscópico de Malaria; Instituto Nacional de Salud, Ministerio de Salud: Lima, Peru, 2010; p. 41. Available online: http://www.ins.gob.pe/insvirtual/images/otrpubs/pdf/FINAL%20MALARIA%2028.12.10%5B1%5D.pdf (accessed on 21 February 2021).
- Manrique, P.; Miranda-Alban, J.; Alarcon-Baldeon, J.; Ramirez, R.; Carrasco-Escobar, G.; Herrera, H.; Guzman-Guzman, M.; Rosas-Aguirre, A.; Llanos-Cuentas, A.; Vinetz, J.M.; et al. Microsatellite Analysis Reveals Connectivity among Geographically Distant Transmission Zones of Plasmodium Vivax in the Peruvian Amazon: A Critical Barrier to Regional Malaria Elimination. PLoS Negl. Trop. Dis. 2019, 13, e0007876. [Google Scholar] [CrossRef]
- Pacheco, M.A.; Elango, A.P.; Rahman, A.A.; Fisher, D.; Collins, W.E.; Barnwell, J.W.; Escalante, A.A. Evidence of Purifying Selection on Merozoite Surface Protein 8 (MSP8) and 10 (MSP10) in Plasmodium spp. Infect. Genet. Evol. 2012, 12, 978–986. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Longley, R.J.; França, C.T.; White, M.T.; Kumpitak, C.; Sa-Angchai, P.; Gruszczyk, J.; Hostetler, J.B.; Yadava, A.; King, C.L.; Fairhurst, R.M.; et al. Asymptomatic Plasmodium Vivax Infections Induce Robust IgG Responses to Multiple Blood-Stage Proteins in a Low-Transmission Region of Western Thailand. Malar. J. 2017, 16, 178. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- DeLong, E.R.; DeLong, D.M.; Clarke-Pearson, D.L. Comparing the Areas under Two or More Correlated Receiver Operating Characteristic Curves: A Nonparametric Approach. Biometrics 1988, 44, 837–845. [Google Scholar] [CrossRef] [PubMed]






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Villasis, E.; Garro, K.; Rosas-Aguirre, A.; Rodriguez, P.; Rosado, J.; Gave, A.; Guzman-Guzman, M.; Manrique, P.; White, M.; Speybroeck, N.; et al. PvMSP8 as a Novel Plasmodium vivax Malaria Sero-Marker for the Peruvian Amazon. Pathogens 2021, 10, 282. https://doi.org/10.3390/pathogens10030282
Villasis E, Garro K, Rosas-Aguirre A, Rodriguez P, Rosado J, Gave A, Guzman-Guzman M, Manrique P, White M, Speybroeck N, et al. PvMSP8 as a Novel Plasmodium vivax Malaria Sero-Marker for the Peruvian Amazon. Pathogens. 2021; 10(3):282. https://doi.org/10.3390/pathogens10030282
Chicago/Turabian StyleVillasis, Elizabeth, Katherine Garro, Angel Rosas-Aguirre, Pamela Rodriguez, Jason Rosado, Anthony Gave, Mitchel Guzman-Guzman, Paulo Manrique, Michael White, Niko Speybroeck, and et al. 2021. "PvMSP8 as a Novel Plasmodium vivax Malaria Sero-Marker for the Peruvian Amazon" Pathogens 10, no. 3: 282. https://doi.org/10.3390/pathogens10030282
APA StyleVillasis, E., Garro, K., Rosas-Aguirre, A., Rodriguez, P., Rosado, J., Gave, A., Guzman-Guzman, M., Manrique, P., White, M., Speybroeck, N., Vinetz, J. M., Torres, K., & Gamboa, D. (2021). PvMSP8 as a Novel Plasmodium vivax Malaria Sero-Marker for the Peruvian Amazon. Pathogens, 10(3), 282. https://doi.org/10.3390/pathogens10030282





