Abstract
Cutaneous leishmaniasis (CL) due to a New World species of Leishmania is increasingly seen among returning international travelers, and most cases arise from travel to Mexico, Central and South America. We described a case of CL in a women presenting a nonhealing ulceration under her right ear with slight increase of size of the left parotid gland under the skin lesion, evolving for 4 months. In her history of travel, she reported a ten-day stay in Mexico during the Christmas vacation in the Yucatan region with only half a day walking in the tropical forest. Diagnosis of CL due to Leishmania mexicana was done via PCR detection and sequencing from swab sampling of the lesion. The patient recovered without antiparasitic treatment. Clinicians should consider diagnosing Chiclero’s ulcer in patients returning from endemic areas such as Central America and Texas who present with chronic ulceration. A noninvasive sampling is sufficient for the PCR-based diagnosis of this disease.
1. Introduction
Cutaneous leishmaniasis (CL) due to Leishmania species of the New World is increasingly seen among returning international travelers, and most cases arise from travel to Mexico, Central and/or South America [1]. In Mexico and Central America, Leishmania mexicana is endemic and extends north into central Texas [2]. The vectors of this infection are Lutzomyia sand flies that transmit Leishmania among different mammalian reservoirs (rodents, opossums, armadillos, cats and dogs) [2]. The typical clinical presentation of L. mexicana infection is called the “Chiclero’s ulcer”, because it was first described in Mexican “Chicleros”: forest workers of the Yucatan peninsula who collected the gum of chicozapote tropical trees that was used in the confection of chewing gum (“Chiclets” in Spanish) [3]. The clinical presentation is characterized by a single ulceration classically associated with involvement of the ear [3]. We report a case of Chiclero’s ulcer in a traveler returning from Mexico. A review of the literature about CL due to this species in travelers was conducted.
2. Case Report and Review of the Literature
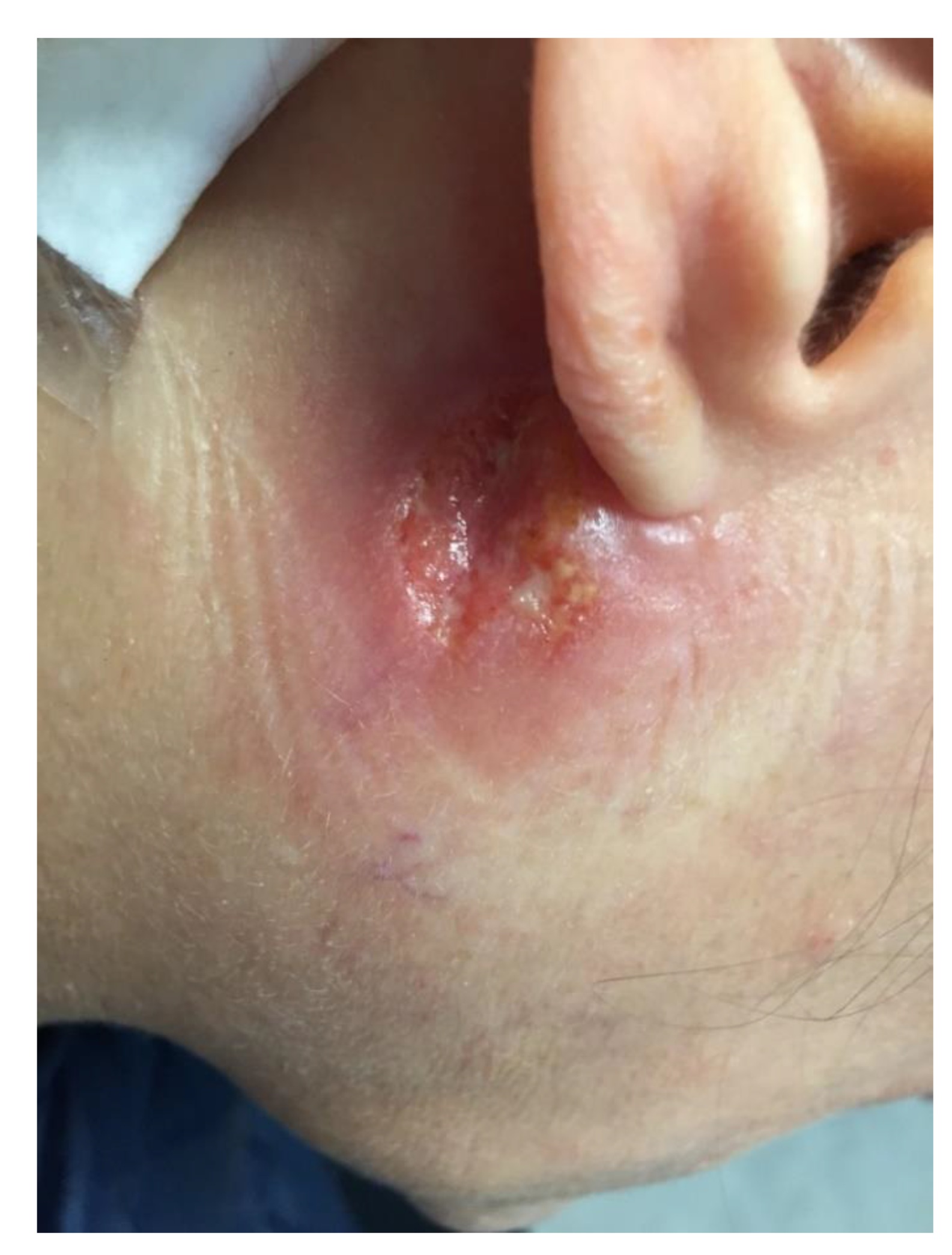
A previously healthy 44-year-old woman presented to the infectious diseases outpatient department of the University Hospital (IHU Méditerranée Infection) of Marseille, France, in May 2019 for a non-healing ulcer located under her right ear slightly extending to the lower part of the ear lobe, which evolved since the end of January (Figure 1).
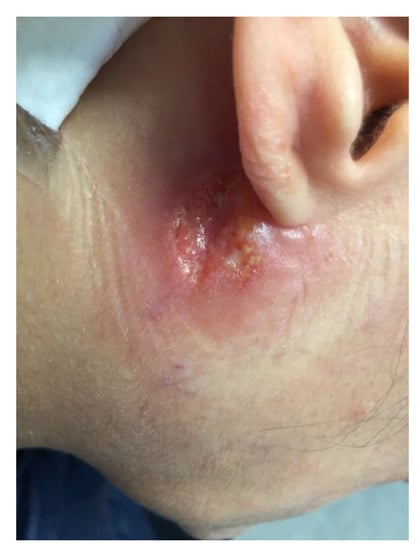
Figure 1.
Nonhealing ulcer due to Leishmania mexicana, localized under the right ear in a patient 4 months after returning from a travel to Mexico.
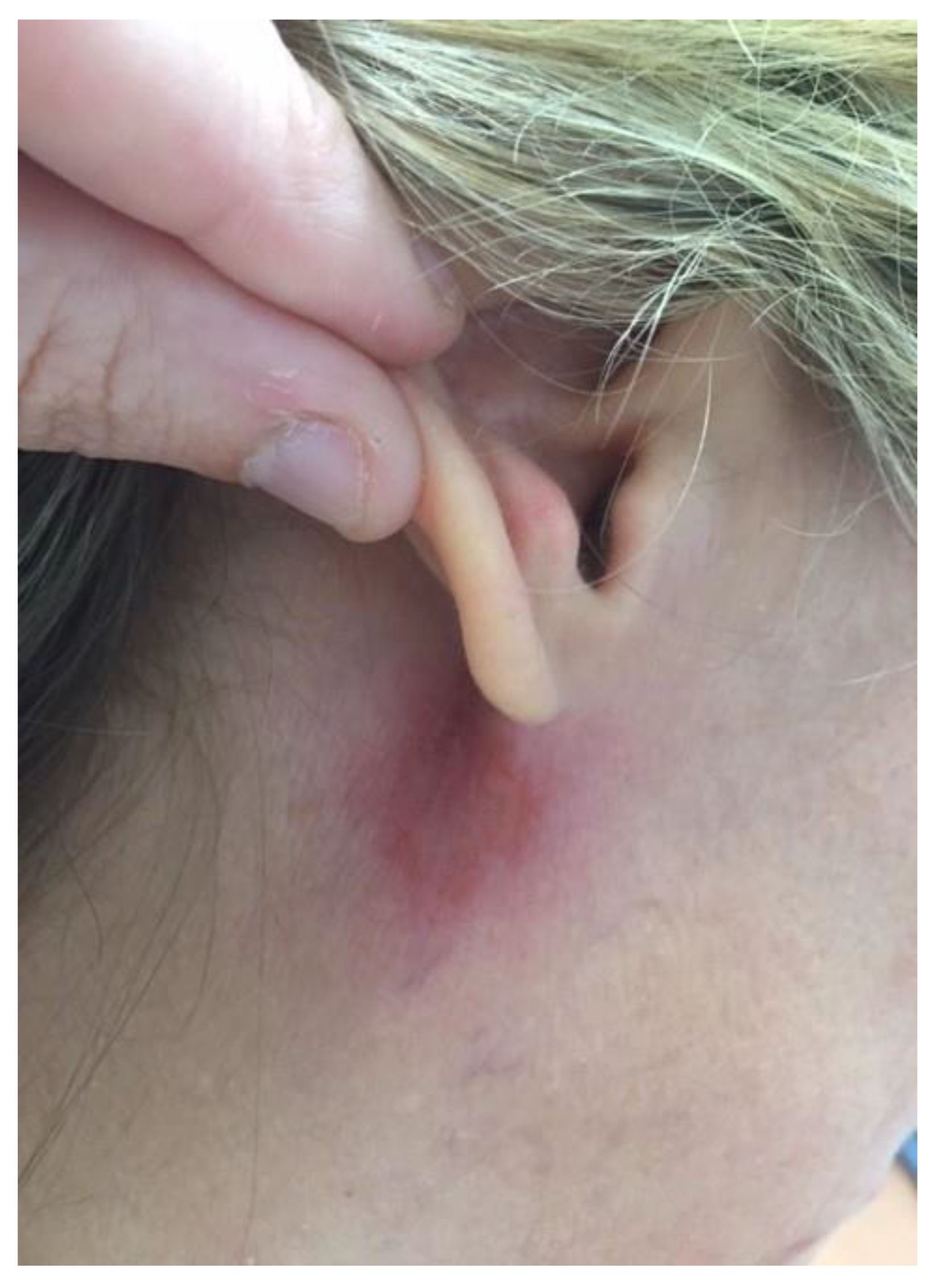
Regarding her travel history, she reported a stay of ten days in Mexico during the Christmas vacation in the Yucatan region with only half a day walking in the tropical forest. She reported paresthesia in the area surrounding the lesion. With a the cervical and thoracic CT-scan, there was no lymphadenitis observed but a slight increase of size of the left parotid gland under the skin lesion. Routine laboratory tests were normal, including C-reactive protein level and blood cell counts. Swab samples of the ulcer were collected for microbiological investigations. Polymerase chain reaction (PCR) and culture aimed at detecting Mycobacterium spp, dermatophytes and pyogenic bacteria were negative. Real-time PCR tests targeting the kinetoplastic minicircle gene successfully detected Leishmania spp. [4]. Subsequent sequencing targeting both the ITS1 and ITS2 region of the rRNA gene performed by the laboratory of the National Reference Center—Laboratory Expert (CNRL) for Leishmania (Montpellier, France) found 100% identity with L. mexicana (Genbank FJ948435.1). Serological investigation showed a positive serology for Leishmania spp. only on immunoblotting assay (LD-Bio Diagnostic, Lyons, France). The diagnosis of cutaneous leishmaniasis due to L. mexicana was retained. At a one month of follow-up, the patient had recovered and skin ulceration and inflammation had disappeared (Figure 2).
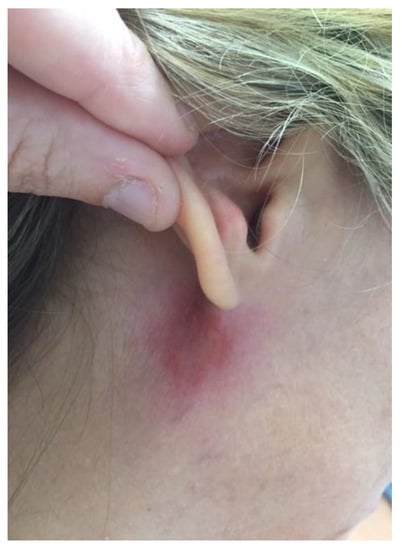
Figure 2.
Evolution of the skin lesion 1 month later without treatment.
We performed a review of the literature in Medline in the English language using the following keywords “Cutaneous leishmaniasis”, “Leishmania mexicana” AND “travel” OR “traveler”. We found 12 references reporting a total of twenty-eight humans cases of CL caused by L. mexicana in travelers (Table 1) [1,3,5,6,7,8,9,10,11,12,13]. Most cases were reported in male travelers returning from Belize, followed by Mexico. Two references reported cases in soldiers who underwent jungle military training in Belize [1,8]. The others reported travels were mainly for touristic reasons. Except for the present case, none involved a French traveler. Regarding clinical features, only three patients presented with the typical localization involving the ear, and three patients had lesions on the face (nose and eyelid). For all patients except our case, the diagnosis was performed on a skin biopsy. The reported treatment strategies were highly heterogeneous. Only two cases, (including ours) received no treatment and healed spontaneously. The other treatments reported in the literature included liposomal amphotericin B, intravenous and intralesional sodium stibogluconate, IV or oral fluconazole, cryotherapy, thermotherapy and topical imiquimod. Two patients had recurrence of their lesions after a first line of treatment and required a second-line treatment for healing.

Table 1.
Cases reports of Leishmania mexicana in travelers, review of literature.
3. Discussion
We detected and identified L. mexicana in a French traveler returning from Mexico by using PCR-based methods on a swab sample from the ulcer. All previous cases had been diagnosed on a skin biopsy sample. Swabbing of ulcers or eschars is a noninvasive, highly sensitive, diagnostic method that advantageously avoids performing a skin biopsy [14]. It is noteworthy that antibody detection by immunoblotting detected antibodies directed against the 14 and 16 kDa L. infantum antigens, which are present in the Leishmania species of the Vianna complex [15]. In a recent report of the Geosentinel network, L. mexicana was found in 3.6% of 274 returned travelers presenting with CL, confirming that it is a relatively rare but emerging CL species in travelers [1]. For clinicians, the main issue about CL in travelers returning from Central and South America is to assess the risk of mucocutaneous or mucous leishmaniasis, which are dilapidating and difficult to treat diseases. In the reported cases of CL due to L. mexicana, involvement of the ear or nose may have prompted clinicians to treat it aggressively because of a putative risk of mucocutaneous involvement. However, analyzing the literature on L. mexicana CL is reassuring because neither lymphatic nor mucosal involvement has been reported with this species [3]. The majority of cases heal spontaneously after 3–9 months [3]. Spontaneous healing occurred after 5 months in our patient, and after 11 months in the other untreated case reported in the literature [3]. However, recurrences were observed in two patients after systemic intravenous treatment (liposomal amphotericin B and IV sodium stilbogluconate) [7,10]. IV or oral fluconazole was not recommended, whereas it was administrated in two cases. The efficiency is difficult to evaluate because the therapeutic scheme included two other successive treatments with different molecules [16]. In fact, recent guidelines about the treatment of CL due to L. mexicana in travelers recommend abstention in patients presenting with up to three lesions if there is no aesthetic disfigurement risk [16]. In patients presenting with more than three lesions, local therapy (topic or intralesional) is recommended [16]. In case of recurrence, lesions with sensitive localization, and/or a larger than 30 mm diameter, the first-line systemic treatment recommended is ketoconazole [16]. Yet, we should stress that ketoconazole has been administered to only one of the 28 reported cases. Protection against sandfly bites, including avoidance of outdoor activities from dusk to dawn, appropriate clothes, use of indoor fans, use of DEET (N,N-diethyl-meta-toluamide) repellents or pyrethroid impregnated bednets, remain the only ways to prevent leishmaniosis in endemic countries.
4. Conclusions
Clinicians should consider Chiclero’s ulcer in patients presenting with a chronic ulceration and returning from endemic areas, including Central America and Texas. A noninvasive swab sampling of the ulceration is enough for the PCR-based microbiological diagnosis of CL due to L. mexicana.
Author Contributions
Resources, C.l. and C.E.; Writing-original draft preparation, C.E. and C.l.; writing-review and editing, S.R. and P.G.; supervision, P.P. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the French Government under the Investissements d’avenir (Investments for the Future) program managed by the Agence Nationale de la Recherche (ANR, fr: National Agency for Research) (reference: Méditerranée Infection 10-IAHU-03) and by Région Provence Alpes Côte d’Azur and European funding FEDER PRIMI.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Ethics Committee of the Assistance Publique des Hôpitaux de Marseille (APHM) (protocol code 2019-73 on 29 May 2019).
Informed Consent Statement
The patient gave her oral and written consent for the publication of this clinical case and photos.
Data Availability Statement
All data are available within the article.
Acknowledgments
The authors gratefully thank the French National Reference Center for Leishmanioses (Montpellier, France) for their technical assistance and Sante publique France for their funding of National Reference Center for Leishmanioses activities. The authors thank Pierre Dudouet for providing informed consent form.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Boggild, A.K.; Caumes, E.; Grobusch, M.P.; Schwartz, E.; A Hynes, N.; Libman, M.; A Connor, B.; Chakrabarti, S.; Parola, P.; Keystone, J.S.; et al. Cutaneous and mucocutaneous leishmaniasis in travellers and migrants: A 20-year GeoSentinel Surveillance Network analysis. J. Travel Med. 2019, 26. [Google Scholar] [CrossRef]
- McIlwee, B.E.; Weis, S.E.; Hosler, G.A. Incidence of Endemic Human Cutaneous Leishmaniasis in the United States. JAMA Dermatol. 2018, 154, 1032–1039. [Google Scholar] [CrossRef] [Green Version]
- Blaylock, J.M.; Wortmann, G.W. A case report and literature review of “Chiclero’s ulcer”. Travel Med. Infect. Dis. 2012, 10, 275–278. [Google Scholar] [CrossRef] [PubMed]
- Mary, C.; Faraut, F.; Lascombe, L.; Dumon, H. Quantification of Leishmania infantum DNA by a Real-Time PCR Assay with High Sensitivity. J. Clin. Microbiol. 2004, 42, 5249–5255. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bailey, M.S.; Langman, G. Misdiagnosis of cutaneous leishmaniasis and recurrence after surgical excision. J. R. Army Med. Corps 2013, 160, 314–316. [Google Scholar] [CrossRef] [PubMed]
- Van Thiel, P.; Zeegelaar, J.; Van Gool, T.; Faber, W.; Kager, P. Cutaneous leishmaniasis in three Dutch military cohorts following jungle training in Belize. Travel Med. Infect. Dis. 2011, 9, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Demers, E.; Forrest, D.M.; Weichert, G.E. Cutaneous leishmaniasis in a returning traveller. Can. Med. Assoc. J. 2013, 185, 681–683. [Google Scholar] [CrossRef] [Green Version]
- Montalvo, A.M.; Fraga, J.; Blanco, O.; González, D.; Monzote, L.; Soong, L.; Capó, V. Imported leishmaniasis cases in Cuba (2006–2016): What have we learned. Trop. Dis. Travel Med. Vaccines 2018, 4, 7. [Google Scholar] [CrossRef]
- Zlotogorski, A.; Gilead, L.; Jonas, F.; Horev, L.; Klaus, S.N. South American cutaneous leishmaniasis: Report of ten cases in Israeli travelers. J. Eur. Acad. Dermatol. Venereol. 1998, 11, 32–36. [Google Scholar] [CrossRef]
- Vinetz, J.M.; Soong, L. Leishmania mexicana infection of the eyelid in a traveler to Belize. Braz. J. Infect. Dis. 2007, 11, 149–152. [Google Scholar] [CrossRef]
- Cohen, J.M.; Saavedra, A.P.; Sax, P.E.; Lipworth, A.D. Pink plaque on the arm of a man after a trip to Mexico: Cutaneous leishmaniasis. Dermatol. Online J. 2015, 21. [Google Scholar] [CrossRef]
- Fuentes-Nava, G.; Tirado-Sánchez, A.; Fernández-Figueroa, E.A.; Sánchez-Montes, S.; Becker, I.; Bonifaz, A. Efficacy of imiquimod 5% cream as first-line management in cutaneous leishmaniasis caused by Leishmania mexicana. Rev. Da Soc. Bras. De Med. Trop. 2021, 54, e0305. [Google Scholar] [CrossRef]
- Tsai, P.-H.; Chen, Y.-T.; Liau, J.-Y.; Huang, M.-H.; Hsu, H.-M.; Yeong, E.-K.; Hung, C.-C. Molecular diagnosis and therapy for cutaneous leishmaniasis of a returned traveler from Mexico. J. Microbiol. Immunol. Infect. 2020. [Google Scholar] [CrossRef] [PubMed]
- Morand, A.; Angelakis, E.; Ben Chaabane, M.; Parola, P.; Raoult, D.; Gautret, P. Seek and Find! PCR analyses of skin infections in West-European travelers returning from abroad with an eschar. Travel Med. Infect. Dis. 2018, 26, 32–36. [Google Scholar] [CrossRef]
- Menu, E.; Blaizot, R.; Mary, C.; Simon, S.; Adenis, A.; Blanchet, D.; L’Ollivier, C.; Ranque, S.; Demar, M. Immunoblot for the Diagnosis of Cutaneous Leishmaniasis in French Guiana. Am. J. Trop. Med. Hyg. 2021, 104, 2091–2096. [Google Scholar] [CrossRef] [PubMed]
- Blum, J.; Buffet, P.; Visser, L.; Harms, G.; Bailey, M.S.; Caumes, E.; Clerinx, J.; van Thiel, P.P.A.M.; Morizot, G.; Hatz, C.; et al. LeishMan recommendations for treatment of cutaneous and mucosal leishmaniasis in travelers, 2014. J. Travel Med. 2014, 21, 116–129. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).