Quantitative G6PD Deficiency Screening in Routine Malaria Diagnostic Units in the Brazilian Amazon (SAFEPRIM): An Operational Mixed-Methods Study
Abstract
:1. Introduction
2. Methods
2.1. Study Design
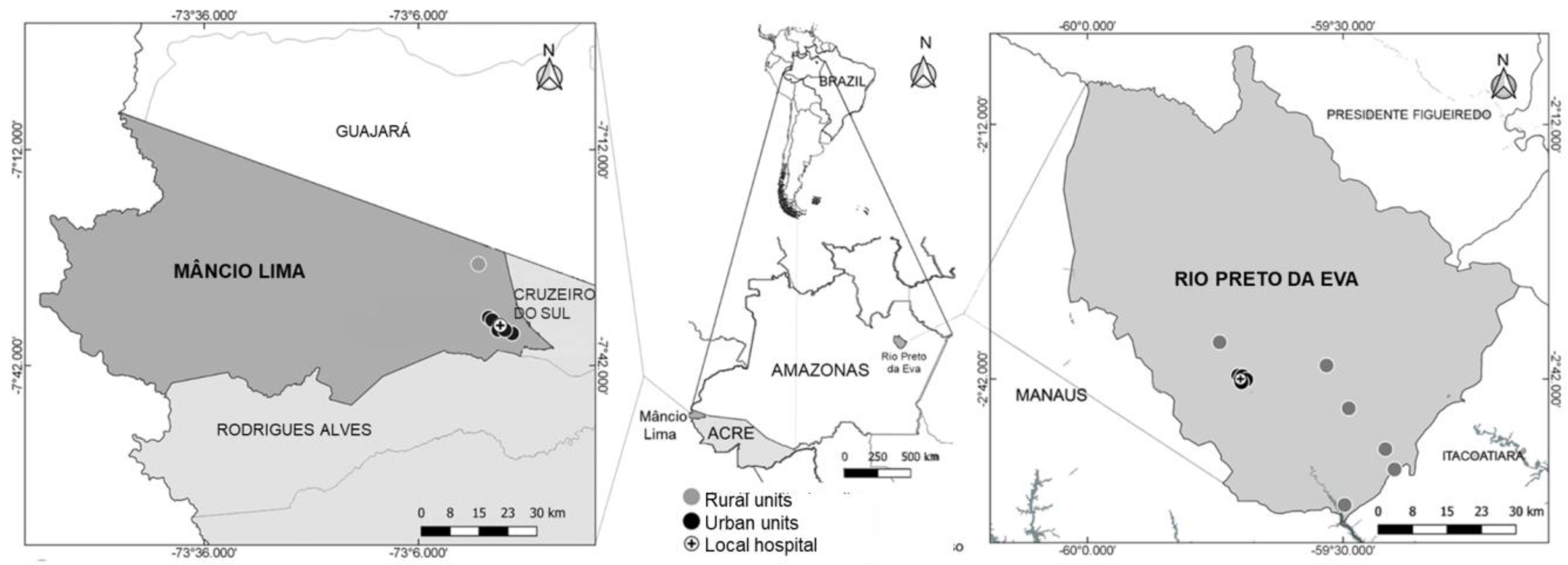
2.2. Study Sites
2.3. HCPs Training and Proficiency Assessment
2.4. G6PD Testing, Results Recording and Pharmacovigilance of Hemolysis
2.5. Genotyping of A Subset of Samples
2.6. Quality Assurance and Oversight
2.7. Perceptions of HCPs and Patients about Implementation
2.8. Data Collection
2.9. Qualitative Data Analysis
2.10. Statistical Analysis
2.11. Ethical Clearance
3. Results
3.1. Study Population, G6PD Distribution and Treatment
3.2. Confirmatory G6PD Genotyping
3.3. Training and Reliability of Interpretation
“The training was very good, we practiced too. What stuck most in my mind was the treatment, because many people were in doubt about which medication would be used if the person could not take the medications daily. There were many people with doubts when the G6PD value was below four…”(HCP001_081720)
3.4. Perceptions about Test Usage
“I don’t understand why this test is done. It was exactly with this doubt that I left the training…”(HCP014_082020)
“On the day of training I had no difficulty handling it, but I’ll be honest, if you asked me to do it now I wouldn’t know how, because I did the training but I didn’t practice in the field. So for me it’s difficult.”(HCP001_081720)
“The test is for finding people who have a reaction with chloroquine, not primaquine.”(HCP012_082020)
3.5. Malaria Card and G6PD Result Recording
“It’s important because it’s like proof that I had malaria, I can get to someone and show that I had malaria, it’s like a document I think. That’s why I consider it important because even for me to travel, if I arrive at a place and there is a police barrier, I can show that I already had (malaria) and had the treatment and I can travel. My card is kept in my wallet, I always carry it, when I travel I like to carry my documents. I think it’s necessary to always show the card to prove that it has already happened. If I lose my card, it’s difficult for me to prove that I had malaria.”(patient09_082020)
3.6. Perceptions about Acute Hemolytic Anemia
“Anemia is when the person has weakness in the legs, lack of blood. That’s anemia. I don’t think it’s related to primaquine… but I can’t explain why not.”(HCP014_082020)
“Hemolytic anemia, I think it’s a serious anemia, I think it’s due to having several malarias, and not because of the medicine.”(HCP016_082020)
“With the first doses patients always feel very ill, with dizziness, weakness, fainting. I’ve seen reports of yellowish eyes, dark urine, but that was with the first three doses of the medicine, they got better afterwards.”(HCP013_082020)
“In the case of patients who use the weekly treatment, they always report that they understand now why the urine was dark before, but now there are some patients who do not accept it, they say it will not cure, they think it is too much medicine and it has already happened that they abandoned treatment.”(HCP010_081820)
“Patients prefer the seven-day treatment, especially those who like alcoholic drinks. Sometimes, mothers of young children don’t like the weekly treatment.”(FGD001_081720)
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Howes, R.E.; Dewi, M.; Piel, F.B.; Monteiro, W.M.; Battle, K.E.; Messina, J.P.; Sakuntabhai, A.; Satyagraha, A.W.; Williams, T.N.; Baird, J.K.; et al. Spatial distribution of G6PD deficiency variants across malaria-endemic regions. Malar. J. 2013, 12, 418. [Google Scholar] [CrossRef] [PubMed]
- Cappellini, M.D.; Fiorelli, G. Glucose-6-phosphate dehydrogenase deficiency. Lancet 2008, 371, 64–74. [Google Scholar] [CrossRef]
- Carson, P.E.; Flanagan, C.L.; Ickes, C.E.; Alving, A.S. Enzymatic Deficiency in Primaquine-Sensitive Erythrocytes. Science 1956, 124, 484–485. [Google Scholar] [CrossRef]
- Monteiro, W.M.; Franca, G.P.; Melo, G.C.; Queiroz, A.L.; Brito, M.; Peixoto, H.M.; Oliveira, M.R.F.; Romero, G.A.; Bassat, Q.; Lacerda, M.V. Clinical complications of G6PD deficiency in Latin American and Caribbean populations: Systematic review and implications for malaria elimination programmes. Malar. J. 2014, 13, 70. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brito-Sousa, J.D.; Santos, T.C.; Avalos, S.; Fontecha, G.; Melo, G.C.; Val, F.; Siqueira, A.M.; Alecrim, G.C.; Bassat, Q.; Lacerda, M.V.; et al. Clinical Spectrum of Primaquine-induced Hemolysis in Glucose-6-Phosphate Dehydrogenase Deficiency: A 9-Year Hospitalization-based Study from the Brazilian Amazon. Clin. Infect. Dis. 2019, 69, 1440–1442. [Google Scholar] [CrossRef]
- Nascimento, J.R.; Brito-Sousa, J.D.; Almeida, A.C.G.; Melo, M.M.; Costa, M.R.F.; Barbosa, L.R.A.; Ramos, R.N.; Silva-Neto, A.V.; da Silva Balieiro, P.C.; Figueiredo, E.F.G.; et al. Prevalence of glucose 6-phosphate dehydrogenase deficiency in highly malaria-endemic municipalities in the Brazilian Amazon: A region-wide screening study. Lancet Reg. Health Am. 2022, 12, 100273. [Google Scholar] [CrossRef]
- Recht, J.; Ashley, E.A.; White, N.J. Use of primaquine and glucose-6-phosphate dehydrogenase deficiency testing: Divergent policies and practices in malaria endemic countries. PLoS Negl. Trop. Dis. 2018, 12, e0006230. [Google Scholar] [CrossRef] [Green Version]
- Siqueira, A.M.; Lacerda, M.V.; Magalhães, B.M.; Mourão, M.P.; Melo, G.C.; Alexandre, M.A.; Alecrim, M.G.; Kochar, D.; Kochar, S.; Kochar, A.; et al. Characterization of Plasmodium vivax-associated admissions to reference hospitals in Brazil and India. BMC Med. 2015, 13, 57. [Google Scholar] [CrossRef] [Green Version]
- Brito-Sousa, J.D.; Murta, F.; Vitor-Silva, S.; Sampaio, V.S.; Mendes, M.O.; Brito, M.A.; Batista, T.S.; Santos, A.P.; Marques, L.L.; Barbosa, L.R.; et al. Real-life implementation of a G6PD deficiency screening qualitative test into routine vivax malaria diagnostic units in the Brazilian Amazon (SAFEPRIM study). PLoS Negl. Trop. Dis. 2021, 15, e0009415. [Google Scholar] [CrossRef]
- Bancone, G.; Gornsawun, G.; Chu, C.S.; Porn, P.; Pal, S.; Bansil, P.; Domingo, G.J.; Nosten, F. Validation of the quantitative point-of-care CareStart biosensor for assessment of G6PD activity in venous blood. PLoS ONE 2018, 13, e0196716. [Google Scholar] [CrossRef]
- Pal, S.; Bansil, P.; Bancone, G.; Hrutkay, S.; Kahn, M.; Gornsawun, G.; Penpitchaporn, P.; Chu, C.S.; Nosten, F.; Domingo, G.J. Evaluation of a Novel Quantitative Test for Glucose-6-Phosphate Dehydrogenase Deficiency: Bringing Quantitative Testing for Glucose-6-Phosphate Dehydrogenase Deficiency Closer to the Patient. Am. J. Trop. Med. Hyg. 2019, 100, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Ley, B.; Winasti Satyagraha, A.; Rahmat, H.; von Fricken, M.E.; Douglas, N.M.; Pfeffer, D.A.; Espino, F.; von Seidlein, L.; Henriques, G.; Oo, N.N.; et al. Performance of the Access Bio/CareStart rapid diagnostic test for the detection of glucose-6-phosphate dehydrogenase deficiency: A systematic review and meta-analysis. PLOS Med. 2019, 16, e1002992. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brazilian Ministry of Health. Lista de Municípios Pertencentes às Áreas de Risco ou Endêmicas para Malária. 2020. Available online: https://www.gov.br/saude/pt-br/media/pdf/2021/agosto/19/lista-de-municipios-pertencentes-as-areas-de-risco-ou-endemicas-para-malaria.pdf (accessed on 10 January 2022).
- PATH. G6PD Operational Research Community of Practice. Available online: https://www.path.org/resources/safeprim-study-materials-training-primary-healthcare-workers-sd-biosensor-standard-g6pd-screening-test/ (accessed on 15 December 2021).
- Zobrist, S.; Brito, M.; Garbin, E.; Monteiro, W.M.; Clementino Freitas, S.; Macedo, M.; Soares Moura, A.; Advani, N.; Kahn, M.; Pal, S.; et al. Evaluation of a point-of-care diagnostic to identify glucose-6-phosphate dehydrogenase deficiency in Brazil. PLoS Negl. Trop. Dis. 2021, 15, e0009649. [Google Scholar] [CrossRef] [PubMed]
- Brazilian Ministry of Health. Guia de Tratamento da Malária no Brasil. Brasília Ministério da Saúde. 2020. Available online: https://www.saude.gov.br/images/pdf/2020/marco/17/guia-tratamento-malaria-.pdf (accessed on 19 December 2021).
- Monteiro, W.M.; Val, F.F.; Siqueira, A.M.; Franca, G.P.; Sampaio, V.S.; Melo, G.C.; Almeida, A.C.; Brito, M.A.; Peixoto, H.M.; Fuller, D.; et al. G6PD deficiency in Latin America: Systematic review on prevalence and variants. Mem. Inst. Oswaldo Cruz 2014, 109, 553–568. [Google Scholar] [CrossRef] [PubMed]
- Palinkas, L.A.; Horwitz, S.M.; Green, C.A.; Wisdom, J.P.; Duan, N.; Hoagwood, K. Purposeful Sampling for Qualitative Data Collection and Analysis in Mixed Method Implementation Research. Adm. Policy Ment. Health 2015, 42, 533–544. [Google Scholar] [CrossRef] [Green Version]
- Tong, A.; Sainsbury, P.; Craig, J. Consolidated criteria for reporting qualitative research (COREQ): A 32-item checklist for interviews and focus groups. Int. J. Qual. Health Care 2007, 19, 349–357. [Google Scholar] [CrossRef] [Green Version]
- Murta, F.L.G.; Mendes, M.O.; Sampaio, V.S.; Junior, A.S.B.; Díaz-Bermúdez, X.P.; Monteiro, W.M.; Lacerda, M.V.G. Misperceptions of patients and health workers regarding malaria elimination in the Brazilian Amazon: A qualitative study. Malar. J. 2019, 18, 223. [Google Scholar] [CrossRef] [Green Version]
- Ley, B.; Winasti Satyagraha, A.; Kibria, M.G.; Armstrong, J.; Bancone, G.; Bei, A.K.; Bizilj, G.; Brito, M.; Ding, X.C.; Domingo, G.J.; et al. Repeatability and reproducibility of a handheld quantitative G6PD diagnostic. PLoS Negl. Trop. Dis. 2022, 16, e0010174. [Google Scholar] [CrossRef]
- Ley, B.; Alam, M.S.; Kibria, M.G.; Marfurt, J.; Phru, C.S.; Ami, J.Q.; Thriemer, K.; Auburn, S.; Jahan, N.; Johora, F.T.; et al. Glucose-6-phosphate dehydrogenase activity in individuals with and without malaria: Analysis of clinical trial, cross-sectional and case–control data from Bangladesh. PLOS Med. 2021, 18, e1003576. [Google Scholar] [CrossRef]
- Brito-Sousa, J.D.; Peixoto, H.M.; Devine, A.; Silva-Neto, A.V.; Balieiro, P.C.; Sampaio, V.S.; Vitor-Silva, S.; Mendes, M.O.; Souza, B.K.; Lacerda, M.V.; et al. Real-life quantitative G6PD screening in Plasmodium vivax patients in the Brazilian Amazon: A cost-effectiveness analysis. PLoS Negl. Trop. Dis. 2022, 16, e0010325. [Google Scholar] [CrossRef]
- Santana, M.S.; Monteiro, W.M.; Siqueira, A.M.; Costa, M.F.; Sampaio, V.; Lacerda, M.V.; Alecrim, M.G. Glucose-6-phosphate dehydrogenase deficient variants are associated with reduced susceptibility to malaria in the Brazilian Amazon. Trans. R Soc. Trop. Med. Hyg. 2013, 107, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Brito-Sousa, J.D.; Phanor, J.; Balieiro, P.C.D.S.; Silva-Neto, A.V.; Cordeiro, J.S.M.; Vitor-Silva, S.; Mendes, M.; Sampaio, V.S.; Melo, G.C.D.; Lacerda, M.; et al. Effect of weekly versus daily primaquine on Plasmodium vivax malaria recurrences: A real-life cohort study. Rev. Soc. Bras. Med. Trop. 2022, 55. [Google Scholar] [CrossRef] [PubMed]
- Gerth-Guyette, E.; Adissu, W.; Brito, M.; Garbin, E.; Macedo, M.; Sharma, A.; Das, S.; Lacerda, M.V.; Pereira, D.; Talukdar, A.; et al. Usability of a point-of-care diagnostic to identify glucose-6-phosphate dehydrogenase deficiency: A multi-country assessment of test label comprehension and results interpretation. Malar. J. 2021, 20, 307. [Google Scholar] [CrossRef] [PubMed]
- Engel, N.; Ghergu, C.; Matin, M.A.; Kibria, M.G.; Thriemer, K.; Price, R.N.; Ding, X.C.; Howes, R.E.; Ley, B.; Incardona, S.; et al. Implementing radical cure diagnostics for malaria: User perspectives on G6PD testing in Bangladesh. Malar. J. 2021, 20, 217. [Google Scholar] [CrossRef]
- Ley, B.; Thriemer, K.; Jaswal, J.; Poirot, E.; Alam, M.S.; Phru, C.S.; Khan, W.A.; Dysoley, L.; Qi, G.; Kheong, C.C.; et al. Barriers to routine G6PD testing prior to treatment with primaquine. Malar. J. 1981, 16, 329. [Google Scholar] [CrossRef]

| Variable | Rio Preto da Eva n = 413 | Mâncio Lima n = 1241 |
|---|---|---|
| Females, n (%) | 159/413 (38.5%) | 497/1241 (40.0%) |
| Males, n (%) | 254/413 (61.5%) | 744/1241 (60.0%) |
| Median age (SD) | 32.3 (18.7%) | 24.6 (17.2%) |
| G6PD Activity Ranges | ||
| <4.0 IU/gHb (M/F) | 15/8 | 24/16 |
| ≥4.0 and ≤6.0 IU/gHb (M/F) | 16/10 | 63/59 |
| >6.0 (M/F) | 56/36 | 505/298 |
| Radical Cure Treatments | ||
| Study participants prescribed 7-day primaquine | 126/150 (84.0%) | 1104/1208 (91.4%) |
| Study participants prescribed weekly dose primaquine | 22/150 (14.7%) | 34/1208 (2.8%) |
| Other | 2/150 (1.3%) | 70/1208 (5.8%) |
| Males (n = 81) | Females (n = 59) | ||||
| STANDARDTM G6PD Test Result | Hemizygous Deficient | Hemizygous Normal | Homozygous Deficient | Heterozygous | Homozygous Normal |
| <4.0 U/gHb | 18 | 10 | - | 3 | 13 |
| ≥4.0 and ≤6.0 U/gHb | - | 15 | - | - | 8 |
| >6.0 U/gHb | - | 38 | - | - | 35 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brito-Sousa, J.D.; Murta, F.; Vitor-Silva, S.; Sampaio, V.; Mendes, M.; Souza, B.; Batista, T.; Santos, A.; Marques, L.; Barbosa, L.; et al. Quantitative G6PD Deficiency Screening in Routine Malaria Diagnostic Units in the Brazilian Amazon (SAFEPRIM): An Operational Mixed-Methods Study. Pathogens 2022, 11, 1328. https://doi.org/10.3390/pathogens11111328
Brito-Sousa JD, Murta F, Vitor-Silva S, Sampaio V, Mendes M, Souza B, Batista T, Santos A, Marques L, Barbosa L, et al. Quantitative G6PD Deficiency Screening in Routine Malaria Diagnostic Units in the Brazilian Amazon (SAFEPRIM): An Operational Mixed-Methods Study. Pathogens. 2022; 11(11):1328. https://doi.org/10.3390/pathogens11111328
Chicago/Turabian StyleBrito-Sousa, Jose Diego, Felipe Murta, Sheila Vitor-Silva, Vanderson Sampaio, Maxwell Mendes, Brenda Souza, Talita Batista, Alicia Santos, Leonardo Marques, Laila Barbosa, and et al. 2022. "Quantitative G6PD Deficiency Screening in Routine Malaria Diagnostic Units in the Brazilian Amazon (SAFEPRIM): An Operational Mixed-Methods Study" Pathogens 11, no. 11: 1328. https://doi.org/10.3390/pathogens11111328







