Pseudomonas aeruginosa PAO1 Is Attracted to Bovine Bile in a Novel, Cystic Fibrosis-Derived Bronchial Epithelial Cell Model
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Growth Conditions
2.2. CFBE-Cell Preparation
2.3. Chemotaxis Assays
2.3.1. Chemoattractant Preparation
2.3.2. Qualitative Capillary Assay
2.3.3. Quantitative Capillary Assay
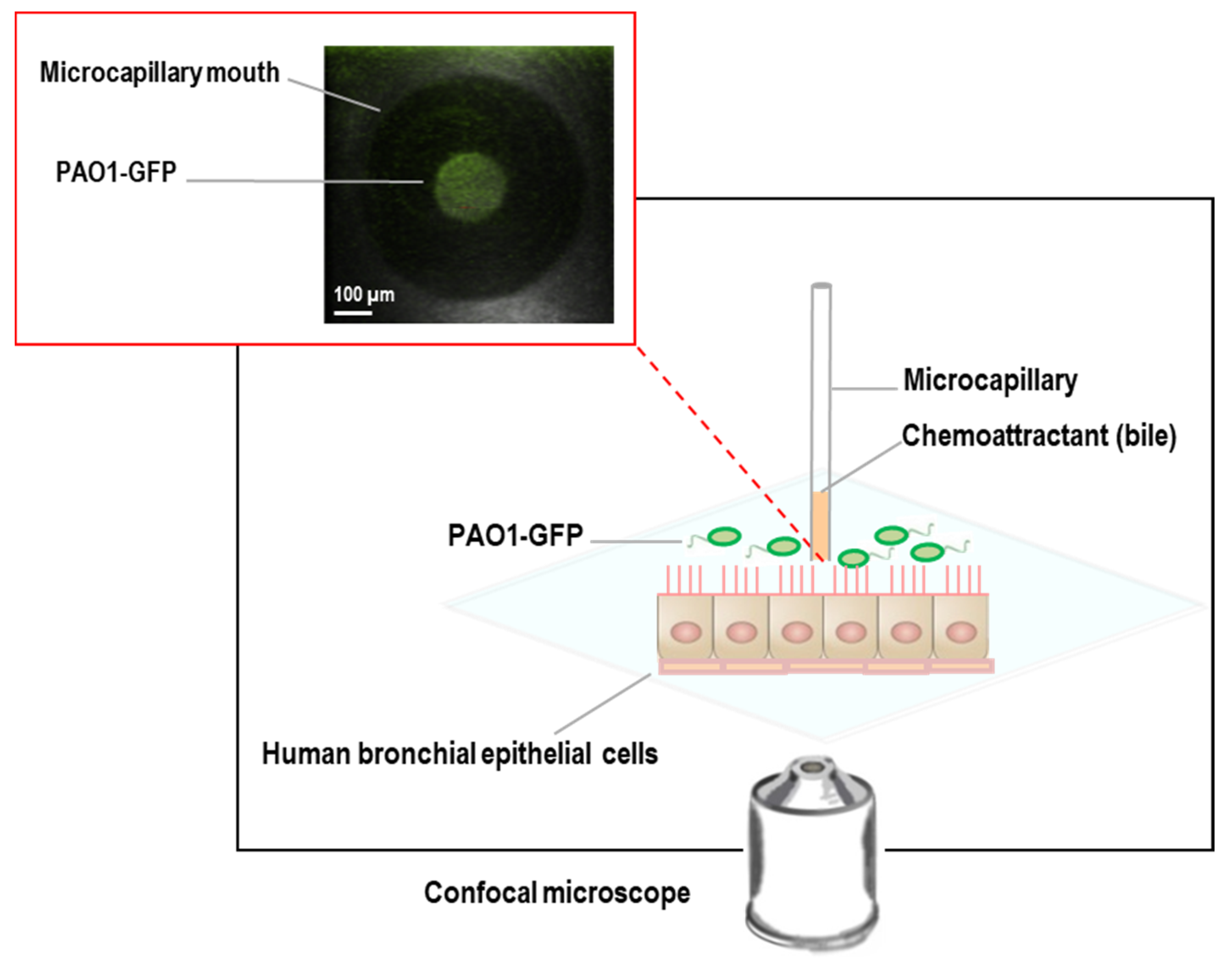
2.3.4. CFBE Cell-Bacteria Chemotaxis Assay
2.3.5. Replicates
2.4. Microscopy
3. Results and Discussion
3.1. P. aeruginosa PAO1 Is Attracted toward Bovine Bile and Correlates with Bile Concentration
3.2. PAO1 Is Attracted to Bovine Bile in the Novel CFBE Experimental System
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Adler, J. Chemotaxis in bacteria. Science 1966, 153, 708–716. [Google Scholar] [CrossRef]
- Webre, D.J.; Wolanin, P.M.; Stock, J.B. Bacterial chemotaxis. Cur. Biol. 2003, 13, R47–R49. [Google Scholar] [CrossRef] [Green Version]
- Wadhams, G.H.; Armitage, J.P. Making sense of it all: Bacterial chemotaxis. Nat. Rev. Mol. Cell. Biol. 2004, 5, 1024–1237. [Google Scholar] [CrossRef]
- Matilla, M.A.; Krell, T. The effect of bacterial chemotaxis on host infection and pathogenicity. FEMS Microbiol. Rev. 2018, 42, fux052. [Google Scholar] [CrossRef] [Green Version]
- Sampedro, I.; Parales, R.E.; Krell, T.; Hill, J.E. Pseudomonas chemotaxis. FEMS Microbiol. Rev. 2015, 39, 17–46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goncz, K.K.; Kunzelmann, K.; Xu, Z.; Gruenert, D.C. Targeted replacement of normal and mutant CFTR sequences in human airway epithelial cells using DNA fragments. Hum. Mol. Genet. 1998, 7, 1913–1919. [Google Scholar] [CrossRef] [Green Version]
- Ehrhardt, C.; Collnot, E.M.; Baldes, C.; Becker, U.; Laue, M.; Kim, K.J.; Lehr, C.M. Towards an in vitro model of cystic fibrosis small airway epithelium: Characterisation of the human bronchial epithelial cell line CFBE41o-. Cell Tissue Res. 2006, 323, 405–415. [Google Scholar] [CrossRef]
- Gottschalk, L.B.; Vecchio-Pagan, B.; Sharma, N.; Han, S.T.; Franca, A.; Wohler, E.S.; Batista, D.A.; Goff, L.A.; Cutting, G.R. Creation and characterization of an airway epithelial cell line for stable expression of CFTR variants. J. Cyst. Fibros. 2016, 15, 285–294. [Google Scholar] [CrossRef] [Green Version]
- Ratjen, F.; Bell, S.C.; Rowe, S.M.; Goss, C.H.; Quittner, A.L.; Bush, A. Cystic fibrosis. Nat. Rev. Dis. Primers 2015, 1, 15010. [Google Scholar] [CrossRef] [PubMed]
- Shteinberg, M.; Haq, I.J.; Polineni, D.; Davies, J.C. Cystic fibrosis. Lancet 2021, 397, 2195–2211. [Google Scholar] [CrossRef]
- Goetz, D.; Ren, C.L. Review of Cystic Fibrosis. Pediatr. Ann. 2019, 48, e154–e161. [Google Scholar] [CrossRef]
- Rossi, E.; La Rosa, R.; Bartell, J.A.; Marvig, R.L.; Haagensen, J.A.J.; Sommer, L.M.; Molin, S.; Johansen, H.K. Pseudomonas aeruginosa adaptation and evolution in patients with cystic fibrosis. Nat. Rev. Microbiol. 2021, 19, 331–342. [Google Scholar] [CrossRef]
- Moradali, M.F.; Ghods, S.; Rehm, B.H. Pseudomonas aeruginosa lifestyle: A paradigm for adaptation, survival, and persistence. Front. Cell Infect. Microbiol. 2017, 7, 39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Faure, E.; Kwong, K.; Nguyen, D. Pseudomonas aeruginosa in Chronic Lung Infections: How to Adapt within the Host? Front. Immunol. 2018, 9, 2416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bongiovanni, A.; Manti, S.; Parisi, G.F.; Papale, M.; Mulè, E.; Rotolo, N.; Leonardi, S. Focus on gastroesophageal reflux disease in patients with cystic fibrosis. World J. Gastroenterol. 2020, 26, 6322–6334. [Google Scholar] [CrossRef] [PubMed]
- Robinson, N.B.; DiMango, E. Prevalence of gastroesophageal reflux in cystic fibrosis and implications for lung disease. Ann. Am. Thorac. Soc. 2014, 11, 964–968. [Google Scholar] [CrossRef]
- Feigelson, J.; Sauvegrain, J. Refux gastro-oesophagien dans la mucoviscidose [Letter: Gastro-esophageal reflux in mucoviscidosis]. Nouv Presse Med. 1975, 4, 2729–2730. (In French) [Google Scholar]
- Reen, F.J.; Woods, D.F.; Mooij, M.J.; Chróinín, M.N.; Mullane, D.; Zhou, L.; Quille, J.; Fitzpatrick, D.; Glennon, J.D.; McGlacken, G.P.; et al. Aspirated bile: A major host trigger modulating respiratory pathogen colonisation in cystic fibrosis patients. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 1763–1771. [Google Scholar] [CrossRef] [Green Version]
- Hay, A.J.; Zhu, J. In Sickness and in Health: The Relationships between Bacteria and Bile in the Human Gut. Adv. Appl. Microbiol. 2016, 96, 43–64. [Google Scholar] [CrossRef] [PubMed]
- Begley, M.; Gahan, C.G.; Hill, C. The interaction between bacteria and bile. FEMS Microbiol. Rev. 2005, 29, 625–651. [Google Scholar] [CrossRef] [Green Version]
- Gipson, K.S.; Nickerson, K.P.; Drenkard, E.; Llanos-Chea, A.; Dogiparthi, S.K.; Lanter, B.B.; Hibbler, R.M.; Yonker, L.M.; Hurley, B.P.; Faherty, C.S. The Great ESKAPE: Exploring the Crossroads of Bile and Antibiotic Resistance in Bacterial Pathogens. Infect. Immun. 2020, 88, e00865-19. [Google Scholar] [CrossRef]
- Bachta, K.E.R.; Allen, J.P.; Cheung, B.H.; Chiu, C.H.; Hauser, A.R. Systemic infection facilitates transmission of Pseudomonas aeruginosa in mice. Nat. Commun. 2020, 11, 543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sistrunk, J.R.; Nickerson, K.P.; Chanin, R.B.; Rasko, D.A.; Faherty, C.S. Survival of the Fittest: How Bacterial Pathogens Utilize Bile to Enhance Infection. Clin. Microbiol. Rev. 2016, 29, 819–836. [Google Scholar] [CrossRef] [Green Version]
- Reen, F.J.; Flynn, S.; Woods, D.; Dunphy, N.; Chróinín, M.N.; Mullane, D.; Stick, S.; Adams, C.; O’Gara, F. Bile signalling promotes chronic respiratory infections and antibiotic tolerance. Sci. Rep. 2016, 6, 29768. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Flynn, S.; Reen, F.J.; O’Gara, F. Exposure to Bile Leads to the Emergence of Adaptive Signaling Variants in the Opportunistic Pathogen Pseudomonas aeruginosa. Front. Microbiol. 2019, 10, 2013. [Google Scholar] [CrossRef] [Green Version]
- Hugdahl, M.B.; Beery, J.T.; Doyle, M.P. Chemotactic behavior of Campylobacter jejuni. Infect. Immun. 1988, 56, 1560–1566. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holloway, B.W. Genetics of Pseudomonas. Bacteriol. Rev. 1969, 33, 419–443. [Google Scholar] [CrossRef]
- Hentchel-Franks, K.; Lozano, D.; Eubanks-Tarn, V.; Cobb, B.; Fan, L.; Oster, R.; Sorscher, E.; Clancy, J.P. Activation of airway cl- secretion in human subjects by adenosine. Am. J. Respir. Cell Mol. Biol. 2004, 31, 140–146. [Google Scholar] [CrossRef]
- Yu, Q.; Griffin, E.F.; Moreau-Marquis, S.; Schwartzman, J.D.; Stanton, B.A.; O’Toole, G.A. In vitro evaluation of tobramycin and aztreonam versus Pseudomonas aeruginosa biofilms on cystic fibrosis-derived human airway epithelial cells. J. Antimicrob. Chemother. 2012, 67, 2673–2681. [Google Scholar] [CrossRef]
- Sampedro, I.; Kato, J.; Hill, J.E. Elastin degradation product isodesmosine is a chemoattractant for Pseudomonas aeruginosa. Microbiology 2015, 161, 1496–1503. [Google Scholar] [CrossRef] [Green Version]
- Reen, F.J.; Woods, D.F.; Mooij, M.J.; Adams, C.; O’Gara, F. Respiratory pathogens adopt a chronic lifestyle in response to bile. PLoS ONE 2012, 7, e45978. [Google Scholar] [CrossRef] [PubMed]
- Tagliacozzi, D.; Mozzi, A.F.; Casetta, B.; Bertucci, P.; Bernardini, S.; Di Ilio, C.; Urbani, A.; Federici, G. Quantitative analysis of bile acids in human plasma by liquid chromatography-electrospray tandem mass spectrometry: A simple and rapid one-step method. Clin. Chem. Lab. Med. 2003, 41, 1633–1641. [Google Scholar] [CrossRef] [PubMed]
- Washizu, T.; Tomoda, I.; Kaneko, J.J. Serum bile acid composition of the dog, cow, horse and human. J. Vet. Med. Sci. 1991, 53, 81–86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parales, R.E.; Harwood, C.S. Bacterial chemotaxis to pollutants and plant-derived aromatic molecules. Curr. Opin. Microbiol. 2002, 5, 266–273. [Google Scholar] [CrossRef]
- Bloemberg, G.V.; O’Toole, G.A.; Lugtenberg, B.J.; Kolter, R. Green fluorescent protein as a marker for Pseudomonas spp. Appl. Environ. Microbiol. 1997, 63, 4543–4551. [Google Scholar] [CrossRef] [Green Version]
- Olivares, E.; Badel-Berchoux, S.; Provot, C.; Jaulhac, B.; Prévost, G.; Bernardi, T.; Jehl, F. Tobramycin and Amikacin Delay Adhesion and Microcolony Formation in Pseudomonas aeruginosa Cystic Fibrosis Isolates. Front. Microbiol. 2017, 8, 1289. [Google Scholar] [CrossRef] [PubMed]
- Schwarzer, C.; Fischer, H.; Machen, T.E. Chemotaxis and Binding of Pseudomonas aeruginosa to Scratch-Wounded Human Cystic Fibrosis Airway Epithelial Cells. PLoS ONE 2016, 11, e0150109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farina, A.; Dumonceau, J.M.; Lescuyer, P. Proteomic analysis of human bile and potential applications for cancer diagnosis. Expert. Rev. Proteom. 2009, 6, 285–301. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Behroozian, S.; Sampedro, I.; Dhodary, B.; Her, S.; Yu, Q.; Stanton, B.A.; Hill, J.E. Pseudomonas aeruginosa PAO1 Is Attracted to Bovine Bile in a Novel, Cystic Fibrosis-Derived Bronchial Epithelial Cell Model. Microorganisms 2022, 10, 716. https://doi.org/10.3390/microorganisms10040716
Behroozian S, Sampedro I, Dhodary B, Her S, Yu Q, Stanton BA, Hill JE. Pseudomonas aeruginosa PAO1 Is Attracted to Bovine Bile in a Novel, Cystic Fibrosis-Derived Bronchial Epithelial Cell Model. Microorganisms. 2022; 10(4):716. https://doi.org/10.3390/microorganisms10040716
Chicago/Turabian StyleBehroozian, Shekooh, Inmaculada Sampedro, Basanta Dhodary, Stephanie Her, Qianru Yu, Bruce A. Stanton, and Jane E. Hill. 2022. "Pseudomonas aeruginosa PAO1 Is Attracted to Bovine Bile in a Novel, Cystic Fibrosis-Derived Bronchial Epithelial Cell Model" Microorganisms 10, no. 4: 716. https://doi.org/10.3390/microorganisms10040716





