Effect of Iron Limitation, Elevated Temperature, and Florfenicol on the Proteome and Vesiculation of the Fish Pathogen Aeromonas salmonicida
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strain, Cultivation, Stress Conditions, and Harvest
2.2. Preparation of Cytosolic, Inner Membrane, and Outer Membrane Protein Fractions
2.3. Preparation of OMV Protein Fraction
2.4. OMV Nanoparticle-Tracking Analysis
2.5. S-Trap Protein Digestion and Peptide Fractionation
2.6. Preparation of the Extracellular Protein Fraction and In-Gel Digestion
2.7. Bioinformatic Tools
2.7.1. PSORTb
2.7.2. SignalP
2.7.3. eggNOG
2.8. Mass Spectrometry Data Acquisition and Analysis
2.9. Transmission Electron Microscopy
2.10. Field Emission Scanning Electron Microscopy
3. Results
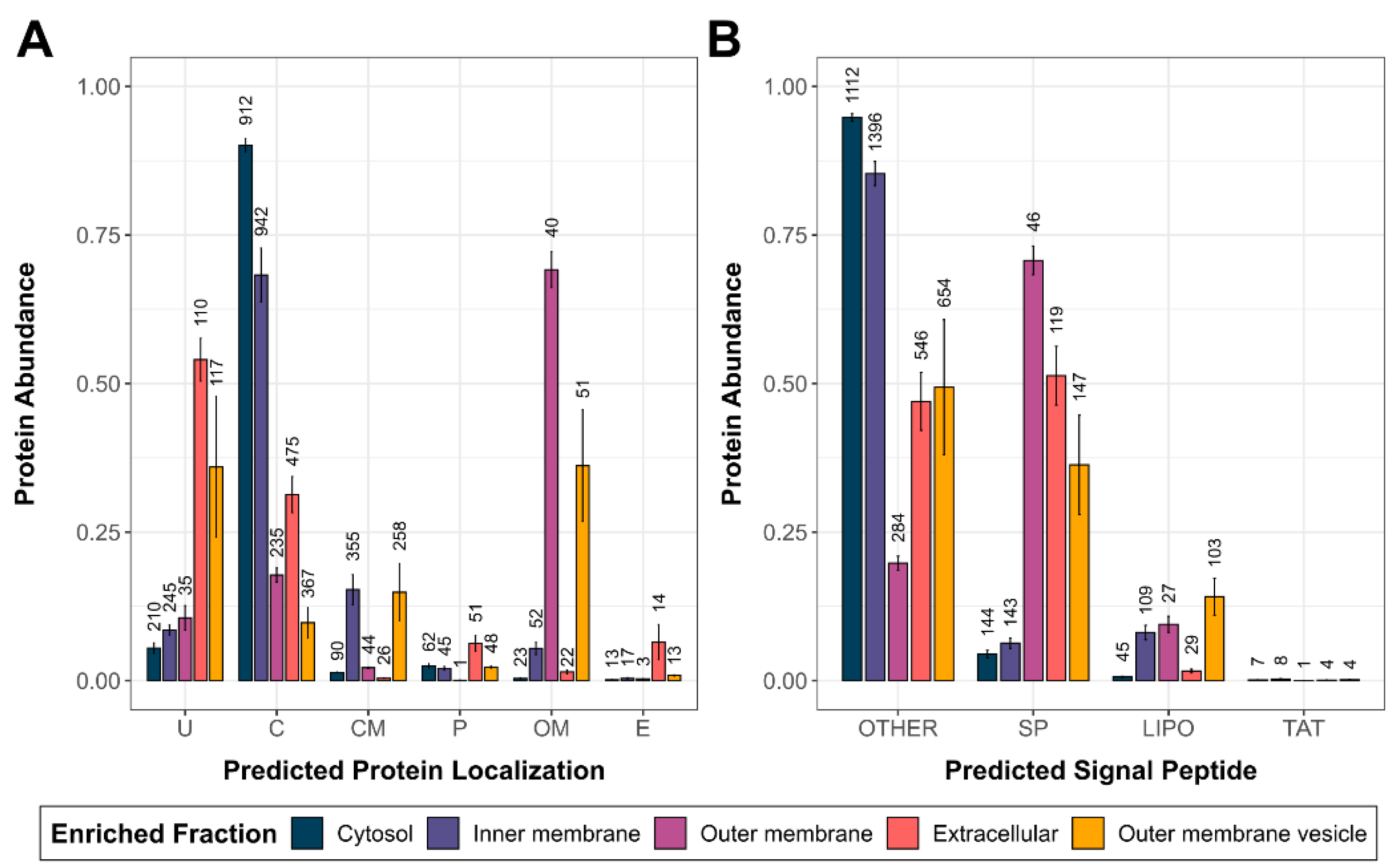
3.1. Evaluation of the Subcellular Fractionation
3.2. Influence of the Stress Conditions on the Composition of the Subproteomes of A. salmonicida
3.3. The Outer Membrane Vesicles of A. salmonicida
4. Discussion
4.1. Subcellular Fractionation
4.2. Response to the Iron Limitation
4.3. Response to an Elevated Incubation Temperature
4.4. Response to Florfenicol
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Garduno, R.A.; Moore, A.R.; Olivier, G.; Lizama, A.L.; Garduno, E.; Kay, W.W. Host Cell Invasion and Intracellular Residence by Aeromonas salmonicida: Role of the S-Layer. Can. J. Microbiol. 2000, 46, 660–668. [Google Scholar] [CrossRef] [PubMed]
- Najimi, M.; Lemos, M.L.; Osorio, C.R. Identification of Siderophore Biosynthesis Genes Essential for Growth of Aeromonas salmonicida under Iron Limitation Conditions. Appl. Environ. Microbiol. 2008, 74, 2341–2348. [Google Scholar] [CrossRef] [PubMed]
- Najimi, M.; Lemos, M.L.; Osorio, C.R. Identification of Iron Regulated Genes in the Fish Pathogen Aeromonas salmonicida Subsp. Salmonicida: Genetic Diversity and Evidence of Conserved Iron Uptake Systems. Vet. Microbiol. 2009, 133, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Najimi, M.; Lemos, M.L.; Osorio, C.R. Identification of Heme Uptake Genes in the Fish Pathogen Aeromonas salmonicida Subsp. Salmonicida. Arch. Microbiol. 2008, 190, 439–449. [Google Scholar] [CrossRef]
- Lemos, M.L.; Balado, M. Iron Uptake Mechanisms as Key Virulence Factors in Bacterial Fish Pathogens. J. Appl. Microbiol. 2020, 129, 104–115. [Google Scholar] [CrossRef]
- Chacón, M.R.; Figueras, M.J.; Castro-Escarpulli, G.; Soler, L.; Guarro, J. Distribution of Virulence Genes in Clinical and Environmental Isolates of Aeromonas Spp. Antonie Van Leeuwenhoek 2003, 84, 269–278. [Google Scholar] [CrossRef]
- Lee, K.K.; Ellis, A.E. Glycerophospholipid:Cholesterol Acyltransferase Complexed with Lipopolysaccharide (LPS) Is a Major Lethal Exotoxin and Cytolysin of Aeromonas salmonicida: LPS Stabilizes and Enhances Toxicity of the Enzyme. J. Bacteriol. 1990, 172, 5382–5393. [Google Scholar] [CrossRef] [PubMed]
- Frederiksen, R.F.; Paspaliari, D.K.; Larsen, T.; Storgaard, B.G.; Larsen, M.H.; Ingmer, H.; Palcic, M.M.; Leisner, J.J. Bacterial Chitinases and Chitin-Binding Proteins as Virulence Factors. Microbiology 2013, 159, 833–847. [Google Scholar] [CrossRef]
- Dallaire-Dufresne, S.; Tanaka, K.H.; Trudel, M.V.; Lafaille, A.; Charette, S.J. Virulence, Genomic Features, and Plasticity of Aeromonas salmonicida Subsp. Salmonicida, the Causative Agent of Fish Furunculosis. Vet. Microbiol. 2014, 169, 1–7. [Google Scholar] [CrossRef]
- Reith, M.E.; Singh, R.K.; Curtis, B.; Boyd, J.M.; Bouevitch, A.; Kimball, J.; Munholland, J.; Murphy, C.; Sarty, D.; Williams, J.; et al. The Genome of Aeromonas salmonicida Subsp. Salmonicida A449: Insights into the Evolution of a Fish Pathogen. BMC Genom. 2008, 9, 1–15. [Google Scholar] [CrossRef]
- Burr, S.E.; Stuber, K.; Wahli, T.; Frey, J. Evidence for a Type III Secretion System in Aeromonas salmonicida Subsp. Salmonicida. J. Bacteriol. 2002, 184, 5966–5970. [Google Scholar] [CrossRef] [PubMed]
- Burr, S.E.; Wahli, T.; Segner, H.; Pugovkin, D.; Frey, J. Association of Type III Secretion Genes with Virulence of Aeromonas salmonicida Subsp. Salmonicida. Dis. Aquat. Org. 2003, 57, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Vanden Bergh, P.; Frey, J. Aeromonas salmonicida Subsp. Salmonicida in the Light of Its Type-Three Secretion System. Microb. Biotechnol. 2014, 7, 381–400. [Google Scholar] [CrossRef] [PubMed]
- Origgi, F.C.; Benedicenti, O.; Segner, H.; Sattler, U.; Wahli, T.; Frey, J. Aeromonas salmonicida Type III Secretion System-Effectors-Mediated Immune Suppression in Rainbow Trout (Oncorhynchus mykiss). Fish Shellfish Immunol. 2017, 60, 334–345. [Google Scholar] [CrossRef]
- Matys, J.; Turska-Szewczuk, A.; Sroka-Bartnicka, A. Role of Bacterial Secretion Systems and Effector Proteins -Insights into Aeromonas Pathogenicity Mechanisms. Acta Biochim. Pol. 2020, 67, 283–293. [Google Scholar] [CrossRef]
- Ebanks, R.O.; Knickle, L.C.; Goguen, M.; Boyd, J.M.; Pinto, D.M.; Reith, M.; Ross, N.W. Expression of and Secretion through the Aeromonas salmonicida Type III Secretion System. Microbiology 2006, 152, 1275–1286. [Google Scholar] [CrossRef]
- Stuber, K.; Burr, S.E.; Braun, M.; Wahli, T.; Frey, J. Type III Secretion Genes in Aeromonas salmonicida Subsp. Salmonicida Are Located on a Large Thermolabile Virulence Plasmid. J. Clin. Microbiol. 2003, 41, 3854–3856. [Google Scholar] [CrossRef]
- Vincent, A.T.; Hosseini, N.; Charette, S.J. The Aeromonas salmonicida Plasmidome: A Model of Modular Evolution and Genetic Diversity. Ann. N. Y. Acad. Sci. 2021, 1488, 16–32. [Google Scholar] [CrossRef]
- Tanaka, K.H.; Dallaire-Dufresne, S.; Daher, R.K.; Frenette, M.; Charette, S.J. An Insertion Sequence-Dependent Plasmid Rearrangement in Aeromonas salmonicida Causes the Loss of the Type Three Secretion System. PLoS ONE 2012, 7, e33725. [Google Scholar] [CrossRef]
- Daher, R.K.; Filion, G.; Tan, S.G.E.; Dallaire-Dufresne, S.; Paquet, V.E.; Charette, S.J. Alteration of Virulence Factors and Rearrangement of PAsa5 Plasmid Caused by the Growth of Aeromonas salmonicida in Stressful Conditions. Vet. Microbiol. 2011, 152, 353–360. [Google Scholar] [CrossRef]
- Nordmo, R.; Holth Riseth, J.M.; Varma, K.J.; Sutherland, I.H.; Brokken, E.S. Evaluation of Florfenicol in Atlantic Salmon, Salmo Salar L.: Efficacy against Furunculosis Due to Aeromonas salmonicida and Cold Water Vibriosis Due to Vibrio salmonicida. J. Fish Dis. 1998, 21, 289–297. [Google Scholar] [CrossRef]
- Midtlyng, P.J.; Reitan, L.J.; Lillehaug, A.; Ramstad, A. Protection, Immune Responses and Side Effects in Atlantic Salmon (Salmo salar L.) Vaccinated against Furunculosis by Different Procedures. Fish Shellfish Immunol. 1996, 6, 599–613. [Google Scholar] [CrossRef]
- Marana, M.H.; Skov, J.; Chettri, J.K.; Krossøy, B.; Dalsgaard, I.; Kania, P.W.; Buchmann, K. Positive Correlation between Aeromonas salmonicida Vaccine Antigen Concentration and Protection in Vaccinated Rainbow Trout Oncorhynchus mykiss Evaluated by a Tail Fin Infection Model. J. Fish Dis. 2017, 40, 507–516. [Google Scholar] [CrossRef] [PubMed]
- Marana, M.H.; Von Gersdorff Jørgensen, L.; Skov, J.; Chettri, J.K.; Mattsson, A.H.; Dalsgaard, I.; Kania, P.W.; Buchmann, K. Subunit Vaccine Candidates against Aeromonas salmonicida in Rainbow Trout Oncorhynchus mykiss. PLoS ONE 2017, 12, e0171944. [Google Scholar] [CrossRef] [PubMed]
- Chukwu-Osazuwa, J.; Cao, T.; Vasquez, I.; Gnanagobal, H.; Hossain, A.; Machimbirike, V.I.; Santander, J. Comparative Reverse Vaccinology of Piscirickettsia salmonis, Aeromonas salmonicida, Yersinia ruckeri, Vibrio anguillarum and Moritella viscosa, Frequent Pathogens of Atlantic Salmon and Lumpfish Aquaculture. Vaccines 2022, 10, 473. [Google Scholar] [CrossRef] [PubMed]
- Midtlyng, P.J. A Field Study on Intraperitoneal Vaccination of Atlantic Salmon (Salmo salar L.) against Furunculosis. Fish Shellfish Immunol. 1996, 6, 553–565. [Google Scholar] [CrossRef]
- Balhuizen, M.D.; Veldhuizen, E.J.A.; Haagsman, H.P. Outer Membrane Vesicle Induction and Isolation for Vaccine Development. Front. Microbiol. 2021, 12, 79. [Google Scholar] [CrossRef]
- Gilmore, W.J.; Johnston, E.L.; Zavan, L.; Bitto, N.J.; Kaparakis-Liaskos, M. Immunomodulatory Roles and Novel Applications of Bacterial Membrane Vesicles. Mol. Immunol. 2021, 134, 72–85. [Google Scholar] [CrossRef]
- Kroniger, T.; Flender, D.; Schlüter, R.; Köllner, B.; Trautwein-Schult, A.; Becher, D. Proteome Analysis of the Gram-Positive Fish Pathogen Renibacterium salmoninarum Reveals Putative Role of Membrane Vesicles in Virulence. Sci. Rep. 2022, 12, 3003. [Google Scholar] [CrossRef]
- Toyofuku, M.; Nomura, N.; Eberl, L. Types and Origins of Bacterial Membrane Vesicles. Nat. Rev. Microbiol. 2019, 17, 13–24. [Google Scholar] [CrossRef]
- Schwechheimer, C.; Kuehn, M.J. Outer-Membrane Vesicles from Gram-Negative Bacteria: Biogenesis and Functions. Nat. Rev. Microbiol. 2015, 13, 605–619. [Google Scholar] [CrossRef] [PubMed]
- Bitto, N.J.; Cheng, L.; Johnston, E.L.; Pathirana, R.; Phan, T.K.; Poon, I.K.H.; O’Brien-Simpson, N.M.; Hill, A.F.; Stinear, T.P.; Kaparakis-Liaskos, M. Staphylococcus aureus Membrane Vesicles Contain Immunostimulatory DNA, RNA and Peptidoglycan That Activate Innate Immune Receptors and Induce Autophagy. J. Extracell. Vesicles 2021, 10, e12080. [Google Scholar] [CrossRef] [PubMed]
- Mehanny, M.; Kroniger, T.; Koch, M.; Hoppstädter, J.; Becher, D.; Kiemer, A.K.; Lehr, C.M.; Fuhrmann, G. Yields and Immunomodulatory Effects of Pneumococcal Membrane Vesicles Differ with the Bacterial Growth Phase. Adv. Healthc. Mater. 2022, 11, 2101151. [Google Scholar] [CrossRef] [PubMed]
- Orench-Rivera, N.; Kuehn, M.J. Differential Packaging Into Outer Membrane Vesicles Upon Oxidative Stress Reveals a General Mechanism for Cargo Selectivity. Front. Microbiol. 2021, 12, 1810. [Google Scholar] [CrossRef]
- Keenan, J.I.; Allardyce, R.A. Iron Influences the Expression of Helicobacter Pylori Outer Membrane Vesicle-Associated Virulence Factors. Eur. J. Gastroenterol. Hepatol. 2000, 12, 1267–1273. [Google Scholar] [CrossRef]
- Briaud, P.; Frey, A.; Marino, E.C.; Bastock, R.A.; Zielinski, R.E.; Wiemels, R.E.; Keogh, R.A.; Murphy, E.R.; Shaw, L.N.; Carroll, R.K. Temperature Influences the Composition and Cytotoxicity of Extracellular Vesicles in Staphylococcus aureus. mSphere 2021, 6, e00676-21. [Google Scholar] [CrossRef]
- Prados-Rosales, R.; Weinrick, B.C.; Piqué, D.G.; Jacobs, W.R.; Casadevall, A.; Rodriguez, G.M. Role for Mycobacterium tuberculosis Membrane Vesicles in Iron Acquisition. J. Bacteriol. 2014, 196, 1250–1256. [Google Scholar] [CrossRef]
- McBroom, A.J.; Kuehn, M.J. Release of Outer Membrane Vesicles by Gram-Negative Bacteria Is a Novel Envelope Stress Response. Mol. Microbiol. 2007, 63, 545–558. [Google Scholar] [CrossRef]
- Seike, S.; Kobayashi, H.; Ueda, M.; Takahashi, E.; Okamoto, K.; Yamanaka, H. Outer Membrane Vesicles Released From Aeromonas Strains Are Involved in the Biofilm Formation. Front. Microbiol. 2021, 11, 3374. [Google Scholar] [CrossRef]
- Micoli, F.; MacLennan, C.A. Outer Membrane Vesicle Vaccines. Semin. Immunol. 2020, 50, 101433. [Google Scholar] [CrossRef]
- Narciso, A.R.; Iovino, F.; Thorsdottir, S.; Mellroth, P.; Codemo, M.; Spoerry, C.; Righetti, F.; Muschiol, S.; Normark, S.; Nannapaneni, P.; et al. Membrane Particles Evoke a Serotype-Independent Cross-Protection against Pneumococcal Infection That Is Dependent on the Conserved Lipoproteins MalX and PrsA. Proc. Natl. Acad. Sci. USA 2022, 119, e2122386119. [Google Scholar] [CrossRef] [PubMed]
- Mehanny, M.; Lehr, C.M.; Fuhrmann, G. Extracellular Vesicles as Antigen Carriers for Novel Vaccination Avenues. Adv. Drug Deliv. Rev. 2021, 173, 164–180. [Google Scholar] [CrossRef] [PubMed]
- Lusta, K.A.; Kozlovskii, Y.E. Outer Membrane Nanovesicles of Gram-Negative Bacteria Aeromonas hydrophila and Aeromonas salmonicida. Microbiology 2011, 80, 519–524. [Google Scholar] [CrossRef]
- Avila-Calderón, E.D.; Otero-Olarra, J.E.; Flores-Romo, L.; Peralta, H.; Aguilera-Arreola, M.G.; Morales-García, M.R.; Calderón-Amador, J.; Medina-Chávez, O.; Donis-Maturano, L.; Del Socorro Ruiz-Palma, M.; et al. The Outer Membrane Vesicles of Aeromonas hydrophila ATCC 7966: A Proteomic Analysis and Effect on Host Cells. Front. Microbiol. 2018, 9, 2765. [Google Scholar] [CrossRef]
- Thein, M.; Sauer, G.; Paramasivam, N.; Grin, I.; Linke, D. Efficient Subfractionation of Gram-Negative Bacteria for Proteomics Studies. J. Proteome Res. 2010, 9, 6135–6147. [Google Scholar] [CrossRef]
- Malherbe, G.; Humphreys, D.P.; Davé, E. A Robust Fractionation Method for Protein Subcellular Localization Studies in Escherichia coli. Biotechniques 2019, 66, 171–178. [Google Scholar] [CrossRef]
- Voß, F.; Kohler, T.P.; Meyer, T.; Abdullah, M.R.; Van Opzeeland, F.J.; Saleh, M.; Michalik, S.; Van Selm, S.; Schmidt, F.; De Jonge, M.I.; et al. Intranasal Vaccination with Lipoproteins Confers Protection against Pneumococcal Colonisation. Front. Immunol. 2018, 9, 2405. [Google Scholar] [CrossRef]
- Kim, Y.S.; Yoon, N.; Karisa, N.; Seo, S.; Lee, J.; Yoo, S.; Yoon, I.; Kim, Y.; Lee, H.; Ahn, J. Identification of Novel Immunogenic Proteins against Streptococcus Parauberis in a Zebrafish Model by Reverse Vaccinology. Microb. Pathog. 2019, 127, 56–59. [Google Scholar] [CrossRef]
- Wilson, M.M.; Bernstein, H.D. Surface-Exposed Lipoproteins: An Emerging Secretion Phenomenon in Gram-Negative Bacteria. Trends Microbiol. 2016, 24, 198–208. [Google Scholar] [CrossRef]
- Brandt, M.E.; Riley, B.S.; Radolf, J.D.; Norgard, M.V. Immunogenic Integral Membrane Proteins of Borrelia burgdorferi Are Lipoproteins. Infect. Immun. 1990, 58, 983–991. [Google Scholar] [CrossRef] [Green Version]
- Kovacs-Simon, A.; Titball, R.W.; Michell, S.L. Lipoproteins of Bacterial Pathogens. Infect. Immun. 2011, 79, 548–561. [Google Scholar] [CrossRef] [PubMed]
- Braun, M.; Stuber, K.; Schlatter, Y.; Wahli, T.; Kuhnert, P.; Frey, J. Characterization of an ADP-Ribosyltransferase Toxin (AexT) from Aeromonas salmonicida Subsp. Salmonicida. J. Bacteriol. 2002, 184, 1851–1858. [Google Scholar] [CrossRef] [PubMed]
- Hobb, R.I.; Fields, J.A.; Burns, C.M.; Thompson, S.A. Evaluation of Procedures for Outer Membrane Isolation from Campylobacter jejuni. Microbiology 2009, 155, 979–988. [Google Scholar] [CrossRef]
- Bonn, F.; Bartel, J.; Büttner, K.; Hecker, M.; Otto, A.; Becher, D. Picking Vanished Proteins from the Void: How to Collect and Ship/Share Extremely Dilute Proteins in a Reproducible and Highly Efficient Manner. Anal. Chem. 2014, 86, 7421–7427. [Google Scholar] [CrossRef] [PubMed]
- Yu, N.Y.; Wagner, J.R.; Laird, M.R.; Melli, G.; Rey, S.; Lo, R.; Dao, P.; Cenk Sahinalp, S.; Ester, M.; Foster, L.J.; et al. PSORTb 3.0: Improved Protein Subcellular Localization Prediction with Refined Localization Subcategories and Predictive Capabilities for All Prokaryotes. Bioinformatics 2010, 26, 1608–1615. [Google Scholar] [CrossRef]
- Almagro Armenteros, J.J.; Tsirigos, K.D.; Sønderby, C.K.; Petersen, T.N.; Winther, O.; Brunak, S.; von Heijne, G.; Nielsen, H. SignalP 5.0 Improves Signal Peptide Predictions Using Deep Neural Networks. Nat. Biotechnol. 2019, 37, 420–423. [Google Scholar] [CrossRef]
- Huerta-Cepas, J.; Szklarczyk, D.; Heller, D.; Hernández-Plaza, A.; Forslund, S.K.; Cook, H.; Mende, D.R.; Letunic, I.; Rattei, T.; Jensen, L.J.; et al. EggNOG 5.0: A Hierarchical, Functionally and Phylogenetically Annotated Orthology Resource Based on 5090 Organisms and 2502 Viruses. Nucleic Acids Res. 2019, 47, D309–D314. [Google Scholar] [CrossRef]
- Tyanova, S.; Temu, T.; Cox, J. The MaxQuant Computational Platform for Mass Spectrometry-Based Shotgun Proteomics. Nat. Protoc. 2016, 11, 2301–2319. [Google Scholar] [CrossRef]
- Tyanova, S.; Temu, T.; Sinitcyn, P.; Carlson, A.; Hein, M.Y.; Geiger, T.; Mann, M.; Cox, J. The Perseus Computational Platform for Comprehensive Analysis of (Prote) Omics Data. Nat. Methods 2016, 13, 731–740. [Google Scholar] [CrossRef]
- Nairz, M.; Schroll, A.; Sonnweber, T.; Weiss, G. The Struggle for Iron—A Metal at the Host-Pathogen Interface. Cell. Microbiol. 2010, 12, 1691–1702. [Google Scholar] [CrossRef]
- Doherty, C.P. Host-Pathogen Interactions: The Role of Iron. J. Nutr. 2007, 137, 1341–1344. [Google Scholar] [CrossRef]
- Boltaña, S.; Rey, S.; Roher, N.; Vargas, R.; Huerta, M.; Huntingford, F.A.; Goetz, F.W.; Moore, J.; Garcia-Valtanen, P.; Estepa, A.; et al. Behavioural Fever Is a Synergic Signal Amplifying the Innate Immune Response. Proc. R. Soc. B Biol. Sci. 2013, 280, 20131381. [Google Scholar] [CrossRef] [PubMed]
- Huntingford, F.; Rey, S.; Quaggiotto, M.M. Behavioural Fever, Fish Welfare and What Farmers and Fishers Know. Appl. Anim. Behav. Sci. 2020, 231, 105090. [Google Scholar] [CrossRef]
- Lulijwa, R.; Rupia, E.J.; Alfaro, A.C. Antibiotic Use in Aquaculture, Policies and Regulation, Health and Environmental Risks: A Review of the Top 15 Major Producers. Rev. Aquac. 2020, 12, 640–663. [Google Scholar] [CrossRef]
- Devos, S.; Stremersch, S.; Raemdonck, K.; Braeckmans, K.; Devreese, B. Intra-and Interspecies Effects of Outer Membrane Vesicles from Stenotrophomonas maltophilia on β-Lactam Resistance. Antimicrob. Agents Chemother. 2016, 60, 2516–2518. [Google Scholar] [CrossRef]
- Bielaszewska, M.; Bauwens, A.; Kunsmann, L.; Karch, H.; Mellmann, A. Antibiotic-Mediated Modulations of Outer Membrane Vesicles in Enterohemorrhagic Escherichia coli O104:H4 and O157:H7. Antimicrob. Agents Chemother. 2017, 61, e00937-17. [Google Scholar] [CrossRef]
- Vanden Bergh, P.; Heller, M.; Braga-Lagache, S.; Frey, J. The Aeromonas salmonicida Subsp. Salmonicida Exoproteome: Global Analysis, Moonlighting Proteins and Putative Antigens for Vaccination against Furunculosis. Proteome Sci. 2013, 11, 44. [Google Scholar] [CrossRef]
- Devos, S.; Van Putte, W.; Vitse, J.; Van Driessche, G.; Stremersch, S.; Van Den Broek, W.; Raemdonck, K.; Braeckmans, K.; Stahlberg, H.; Kudryashev, M.; et al. Membrane Vesicle Secretion and Prophage Induction in Multidrug-Resistant Stenotrophomonas maltophilia in Response to Ciprofloxacin Stress. Environ. Microbiol. 2017, 19, 3930–3937. [Google Scholar] [CrossRef]
- Pérez-Cruz, C.; Carrión, O.; Delgado, L.; Martinez, G.; López-Iglesias, C.; Mercade, E. New Type of Outer Membrane Vesicle Produced by the Gram-Negative Bacterium Shewanella vesiculosa M7T: Implications for DNA Content. Appl. Environ. Microbiol. 2013, 79, 1874–1881. [Google Scholar] [CrossRef]
- Zingl, F.G.; Kohl, P.; Cakar, F.; Leitner, D.R.; Mitterer, F.; Bonnington, K.E.; Rechberger, G.N.; Kuehn, M.J.; Guan, Z.; Reidl, J.; et al. Outer Membrane Vesiculation Facilitates Surface Exchange and In Vivo Adaptation of Vibrio cholerae. Cell Host Microbe 2020, 27, 225–237.e8. [Google Scholar] [CrossRef] [Green Version]
- Ebanks, R.O.; Dacanay, A.; Goguen, M.; Pinto, D.M.; Ross, N.W. Differential Proteomic Analysis of Aeromonas salmonicida Outer Membrane Proteins in Response to Low Iron and in Vivo Growth Conditions. Proteomics 2004, 4, 1074–1085. [Google Scholar] [CrossRef]
- Menanteau-Ledouble, S.; Kattlun, J.; Nöbauer, K.; El-Matbouli, M. Protein Expression and Transcription Profiles of Three Strains of Aeromonas salmonicida Ssp. Salmonicida under Normal and Iron-Limited Culture Conditions. Proteome Sci. 2014, 12, 29. [Google Scholar] [CrossRef]
- Menanteau-Ledouble, S.; El-Matbouli, M. Antigens of Aeromonas salmonicida Subsp. Salmonicida Specifically Induced In Vivo in Oncorhynchus mykiss. J. Fish Dis. 2016, 39, 1015–1019. [Google Scholar] [CrossRef]
- Barnes, A.C.; Horne, M.T.; Ellis, A.E. Effect of Iron on Expression of Superoxide Dismutase by Aeromonas salmonicida and Associated Resistance to Superoxide Anion. FEMS Microbiol. Lett. 1996, 142, 19–26. [Google Scholar] [CrossRef]
- Lim, C.K.; Hassan, K.A.; Tetu, S.G.; Loper, J.E.; Paulsen, I.T. The Effect of Iron Limitation on the Transcriptome and Proteome of Pseudomonas fluorescens Pf-5. PLoS ONE 2012, 7, e39139. [Google Scholar] [CrossRef]
- Lan, Y.; Zhou, M.; Li, X.; Liu, X.; Li, J.; Liu, W. Preliminary Investigation of Iron Acquisition in Hypervirulent Klebsiella pneumoniae Mediated by Outer Membrane Vesicles. Infect. Drug Resist. 2022, 15, 311–320. [Google Scholar] [CrossRef]
- Groberg, W.J., Jr.; McCoy, R.H.; Pilcher, K.S.; Fryer, J.L. Relation of Water Temperature to Infections of Coho Salmon (Oncorhynchus kisutch), Chinook Salmon (O. tshawytscha), and Steelhead Trout (Salmo gairdneri) with Aeromonas salmonicida and A. hydrophila. J. Fish. Res. Board Can. 1978, 35, 1–7. [Google Scholar] [CrossRef]
- Knøchel, S. Effect of Temperature on Hemolysin Production in Aeromonas Spp. Isolated from Warm and Cold Environments. Int. J. Food Microbiol. 1989, 9, 225–235. [Google Scholar] [CrossRef]
- Frias, A.; Manresa, A.; de Oliveira, E.; López-Iglesias, C.; Mercade, E. Membrane Vesicles: A Common Feature in the Extracellular Matter of Cold-Adapted Antarctic Bacteria. Microb. Ecol. 2010, 59, 476–486. [Google Scholar] [CrossRef]
- Vincent, A.T.; Charette, S.J. To Be or Not to Be Mesophilic, That Is the Question for Aeromonas salmonicida. Microorganisms 2022, 10, 240. [Google Scholar] [CrossRef]
- Dowling, P.M. Chloramphenicol, Thiamphenicol, and Florfenicol. In Antimicrobial Therapy in Veterinary Medicine; Wiley: Hoboken, NJ, USA, 2013; pp. 269–277. [Google Scholar]
- Desbois, A.P.; Cook, K.J.; Buba, E. Antibiotics Modulate Biofilm Formation in Fish Pathogenic Isolates of Atypical Aeromonas salmonicida. J. Fish Dis. 2020, 43, 1373–1379. [Google Scholar] [CrossRef] [PubMed]
- Perez-Riverol, Y.; Bai, J.; Bandla, C.; García-Seisdedos, D.; Hewapathirana, S.; Kamatchinathan, S.; Kundu, D.J.; Prakash, A.; Frericks-Zipper, A.; Eisenacher, M.; et al. The PRIDE Database Resources in 2022: A Hub for Mass Spectrometry-Based Proteomics Evidences. Nucleic Acids Res. 2022, 50, D543–D552. [Google Scholar] [CrossRef] [PubMed]






| Iron Limitation | Temperature | Antibiotics | |
|---|---|---|---|
| Excerpt of the proteomics results | Several upregulated IROMPs (TonB-dependent proteins, iron/siderophore transporters, hemolysin, aerolysin) | Upregulated hemolysin, putative hemin receptor, oxidative-stress chaperon, pullulanase, uncharacterized protein | Increased ribosomal proteins in extracellular space and outer membrane vesicles |
| Vesiculation (Mean particle size of control: 172 nm) | Slightly increased vesicle size (Mean particle size: 195 nm) | Slightly decreased vesicle size (Mean particle size: 151 nm) | Decreased vesicle size (Mean particle size: 102 nm) potentially due to prophage induction |
| Vesiculation amount | Comparable vesiculation amount to control condition | Less vesiculation compared to control | Comparable vesiculation amount to control condition |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kroniger, T.; Mehanny, M.; Schlüter, R.; Trautwein-Schult, A.; Köllner, B.; Becher, D. Effect of Iron Limitation, Elevated Temperature, and Florfenicol on the Proteome and Vesiculation of the Fish Pathogen Aeromonas salmonicida. Microorganisms 2022, 10, 1735. https://doi.org/10.3390/microorganisms10091735
Kroniger T, Mehanny M, Schlüter R, Trautwein-Schult A, Köllner B, Becher D. Effect of Iron Limitation, Elevated Temperature, and Florfenicol on the Proteome and Vesiculation of the Fish Pathogen Aeromonas salmonicida. Microorganisms. 2022; 10(9):1735. https://doi.org/10.3390/microorganisms10091735
Chicago/Turabian StyleKroniger, Tobias, Mina Mehanny, Rabea Schlüter, Anke Trautwein-Schult, Bernd Köllner, and Dörte Becher. 2022. "Effect of Iron Limitation, Elevated Temperature, and Florfenicol on the Proteome and Vesiculation of the Fish Pathogen Aeromonas salmonicida" Microorganisms 10, no. 9: 1735. https://doi.org/10.3390/microorganisms10091735






