1. Introduction
Since the discovery of penicillin by Alexander Fleming in 1929, various antimicrobials have been designed and translated into clinical practice. Along with their active use in human and veterinary medicine for infection treatment, they have been intensively used in livestock and poultry as animal growth enhancers and diseases prevention tools since at least the 1940s [
1]. As a result, antibiotics continuously accumulate in soils and water, thereby causing either resistance development or the acquisition of resistance genes by bacteria [
2,
3]. The growing spread of bacterial resistance to antimicrobials threatens the health of both humans and animals; this is emerging as a significant global health challenge. Despite legal restrictions on antibiotic use in some countries, their consumption is expected to increase by more than 60 percent by 2030 [
4,
5,
6], challenging the development of their environmentally friendly and natural alternatives.
Various food supplements have been suggested as substitutes for antibiotics. In some studies, organic acids (formic, acetic or propionic) were added to drinking water or animal feed, leading to decreased number of bacteria (
Salmonella and
Campylobacter) in the intestines of poultry [
7,
8,
9]. To suppress bacterial growth (such as
Salmonella,
Clostridium and
Escherichia coli), various plant extracts and essential oils (for example, thyme, cinnamon, black pepper, and many others) have also been investigated [
10,
11,
12,
13]. The composition of these plant extracts may include carotenoids, phenols, alkaloids, terpenes, peptides and many others [
14,
15,
16]. Antimicrobial peptides can be isolated not only from plants, but also from completely different origins: animals, insects or created using genetic engineering methods [
17].
Probiotic bacteria with antagonistic activities against pathogens are often considered as promising substitutes to antibiotics [
18,
19,
20]. The Food and Agriculture Organization of the United Nations (FAO) and the World Health Organization (WHO) defined probiotics as “live microorganisms which when administered in adequate amounts confer a health benefit on the host” [
21]. The development of probiotic drugs and attendant scientific and clinical research continues to develop at pace [
22]. In animal husbandry, poultry and aquaculture, probiotics are used as growth stimulants as well as for the prevention and treatment of diseases [
23,
24]. Among several prominent examples,
Lactococci,
Lactobacilli,
Enterococci and
Bacilli are commonly used [
25,
26].
Although the intestinal microbiota represents a huge complex community of various bacteria, many species of the genera
Lactobacillus and
Bifidobacterium have been shown to offer protection against enteric infections. Several species of the genus
Lactococci are intentionally introduced in the food chain, being involved in a range of food and feed fermentations, and are applied as probiotics in humans and animals [
27,
28]. The use of viable spores of
Bacillus as a probiotic supplement raised a number of questions, including their safety; several
Bacillus species used as animal feed supplements, probiotics, plant protection products or seed coating agents are also known as agents of food poisoning. However, they are widely used in animal husbandry, in particular as a probiotic supplement for the swine livestock [
29,
30].
Lactic acid bacteria (LAB) typically exhibit strong antagonism against other bacteria by using various tools, including synthesis of organic acids (mainly lactic acid) and acidification of the broth [
31], hydrogen peroxide production [
32] and secretion of antimicrobial peptides as well as bacteriocins [
33,
34]. For example, in vaginal secretions, probiotic
Lactobacilli exhibit an acidifying ability, synthesize hydrogen peroxide and form co-aggregates with pathogenic strains of
Streptococcus agalactiae [
35]. The probiotic strain
L. diolivorans 1Z contributed to animal survival after infection by
Salmonella enterica (
Typhimurium serotype) due to colonization resistance and immunomodulation of the host intestine [
36]. Oral probiotics have been evaluated for their possible application in the prevention and treatment of caries in an in vivo rat model, indicating that lactobacillus strains exhibit strong colonization resistance and are capable of destroying biofilms of caries-inducing
Streptococcus mutans [
37]. Moreover, probiotic
Lactobacilli can stimulate the growth and maturation of animals, improving both the innate and the acquired immune mechanisms of the host [
38,
39]. They produce lactic acid as the major metabolic end product of carbohydrate fermentation while also supporting food digestion via production of hydrogen peroxide and other substances, thus creating an unfavorable acidic environment for harmful or pathogenic organisms [
40,
41,
42]. Thus, direct activity of
Lactobacilli on other microorganisms prevents the development of infections and serves as an effective tool to control and restore the microbial equilibrium in the gut [
43,
44]. Furthermore, various bacterial genera possess genotoxins, for example, the cytolethal distending toxin (CDT), which causes DNA double-strand breaks (DSBs) [
45]. Others alter the host DNA damage response, potentially resulting in mutations and cancer (reviewed in [
46]). A deleterious action on host DNA integrity has been described for Gram-negative bacteria such as
Helicobacter sp.,
Chlamydia sp.,
Salmonella sp. or
Escherichia coli, demonstrating that these mechanisms may lead to genomic alterations and transformations associated with cancer development [
46].
S. aureus also was shown to trigger ROS-mediated DNA damage, thus affecting their genomic integrity [
47]. Thus, the decrease of pathogenic bacteria in the gut by their substitution by probiotic bacteria would explain, at least partially, their antimutagenic activity (reviewed in [
48,
49]).
In previous studies, several strains of lactic acid bacteria with probiotic properties were isolated from clover silage [
50,
51,
52]. Among them,
L. plantarum AG10 demonstrated the most promising probiotic features, as well as antimicrobial activity against pathogens in both liquid medium and biofilm. Furthermore, during milk fermented by
L. plantarum AG10, the dry matter content and product density were slightly higher than in the milk fermented by conventional industrial strains [
53]. At the same time, it produces less lactic acid during milk fermentation and, as a result, has a lower acidification rate. On the other hand, the amount of EPS in fermented milk
L. plantarum AG10 is higher when compared to a control industrial strain [
52]. As a consequence of the larger production of expanded polystyrene, fat-free samples obtained from
L. plantarum AG10 fermented milk demonstrated significantly higher viscosity, product adhesion and higher resistance to destruction [
53].
Drosophila melanogaster is a common and convenient model organism widely used to study the effects of the intestinal microbiota and the effects of probiotics on the host organism. Flies have several features that make them suitable model organisms [
54]. Their intestines have structural and functional similarities with the intestines of mammals [
55]. Flies and mammals have similar signaling pathways, such as Toll and Toll-like receptors, as well as similar protective immune mechanisms [
56]. Genes involved in immune responses, such as signaling, gene expression, metabolism, immune system communication with other organs and systems, tissue homeostasis, intestinal physiology, development and metabolism, are preserved in both
Drosophila and vertebrates [
57].
The fruit fly
Drosophila is widely used as an in vivo model to study bacterial [
58], viral [
59] and even fungal infections [
60]. The insect is used as a model in the study of host-microorganism relationships, especially in relation to residential microflora, such as
L. planatrum, which are nowadays commonly used in the food and pharmaceutical industries. The effects of
L. plantarum on the host physiology are relevant for both fundamental and applied investigations aimed at using these bacteria as probiotics. Studies of the interactions between
Lactobacteria and
Drosophila are interesting because physiological, especially neurophysiological, and reproduction processes can be modulated through probiotics [
61,
62]. To date, particular relationship between
D. melanogaster and its symbiote
L. plantarum have revealed various facets of micro-macro-organism interaction [
63,
64].
Lactobacilli have been found to promote larvae [
65,
66] and protein production [
67,
68], regulate host dietary behavior [
69,
70,
71] and induce the formation of active oxygen species (AFC) with NADP oxidase [
72], thereby protecting fruit fly cells and tissues from damaging agents [
73]. Available data on
L. plantarum and
D. melanogaster indicate the complex and ambiguous character of bacterial-host interactions. However, their potential role in protecting the host organism, as well as maintaining its homeostasis while infected with pathogens, could hardly be overstated.
There are many studies of the effect of probiotics on the
Drosophila melanogaster model. Thus, the authors of [
74] evaluated the effect of a probiotic drug on the locomotor functions of flies and changes in the composition of their intestinal microbiota. The following studies show the beneficial effect of probiotic drugs against pathogenic microorganisms for the treatment of flies [
75,
76].
Here we show that the L. plantarum AG10 strain exhibits strong antagonistic activity against several opportunistic intestinal microorganisms, and thus is capable of suppressing the growth of S. aureus and E. coli in vitro during milk fermentation; in addition, it can reduce the negative effects of S. aureus and E. coli in vivo in the survival model of Drosophila melanogaster at the embryonic (larval) and pupal stages. The whole genome sequencing allowed the identification, with high probability, the putative antimicrobial peptide responsible for the above activity. The genome contains extremely low amounts of putative virulence factors indicating that L. plantarum AG10 represents a promising probiotic.
2. Materials and Methods
2.1. Strains and Growth Conditions
Lactiplantibacillus (
Lactobacillus)
plantarum AG10 isolated from silage provides a high milk acidification rate and exhibited potential probiotic properties [
50]. As a reference, we used the
Lactiplantibacillus plantarum 8PA3 strain, which is approved as a probiotic strain (Biomed, Russia) and is widely used in various probiotic products, including “Lactobacterin”, “Biovestin-lacto”, “Lactonorm” and “Florin-forte”. It is also known for its effectiveness against diarrhea of various etiologies, dysbiosis, bacterial vaginosis, acute intestinal infection in children, as well as in the complex therapy of diseases associated with
Helicobacter pylori [
77].
LABs were stored in de Man, Rogosa and Sharpe (MRS) broth with 50% glycerol at −80 °C. Bacteria were seeded from the stock into MRS broth with the addition of 2% glucose and grown at 37 °C for 24 h. The obtained culture was added to skimmed milk to receive a starter culture.
Escherichia coli MG1655 (K-12), Staphylococcus aureus subsp. aureus ATCC 29213, Klebsiella pneumonia (clinical isolate), Pseudomonas aeruginosa ATCC 27853, Serratia marcescens (clinical isolate) and Bacillus cereus (clinical isolate) were used in this study as test bacteria for the evaluation of the antibacterial activity of the LABs. Clinical isolates of B. cereus and K. pneumonia were kindly provided by the Kazan Institute of Epidemiology and Microbiology (Kazan, Russia). The S. marcescens strain was kindly provided by the Institute of Medical Microbiology (Giessen, Germany).
E. coli and S. aureus strains carrying pCtuf Amp Gfpr plasmid providing constitutive GFP synthesis were used as markers of the presence of bacteria in the flies’ gut. The pCtuf Amp Gfpr plasmid was kindly provided by Prof. F. Götz, University Tübingen, Germany.
2.2. Antibacterial Activity of LAB Strains (Agar Drop Diffusion Test)
The overnight culture of
Lactobacilli was inoculated as a lawn on MRS agar and incubated for 48 h at 37 °C. The agar blocks with
Lactobacilli colonies were cut out with a sterile cork borer and set in Petri dishes onto a surface of solid LB medium inoculated with an 8- to 10-h culture of test organisms. The antagonistic activity was assessed after 24-h incubation at 37 °C by the diameter of growth inhibition zones of the test microorganisms around the agar blocks with
Lactobacilli [
78].
2.3. Antibacterial Activity of LAB Strains in the Fermented Milk
The antagonistic properties of LAB strains against pathogens during milk fermentation were tested after 1, 3, 7 and 21 days.
L. plantarum 8PA3 was used as a reference strain. To obtain an LAB starter culture, an overnight culture of
Lactobacilli was added to 5 mL of milk and incubated for 12 h at 37 °C. To 19 mL of milk, 1 mL of starter LAB culture (10
8–10
9 CFU/mL) was added; an overnight culture of
E. coli or
S. aureus were added to the final concentration of 10
6 CFU/mL when desired and incubation was conducted for 8 h at 40 °C. Then, the samples were stored at 4 °C for 21 days. CFUs were counted using the drop-plate assay [
79] with modifications [
80,
81]. Ten-fold dilution series of the fermented milk were prepared and plated drops (5 μL each) on differential media. MRS agar, mannitol-salt agar and Endo agar were used to differentiate LAB,
S. aureus and
E. coli, respectively. CFUs were counted from the two last drops, typically containing 5–15 colonies.
2.4. D. melanogaster Husbandry
All experiments were performed with virgin female D. melanogaster, since non-virgin females would have a chance to produce offspring from undesirable males. In the SMART test, the virgin females should be used to ensure the genotype clearance. Canton-Sflies were used as wild-type strains in all following experiments unless otherwise specified. For the SMART assay, two mutant Drosophila lines were used: mwh, flr3, carriers of the multiple wing hairs (mwh, 3-0.3) and flare3 (flr3, 3–38.8) marker genes. These lines were kept in thermostat stock in ¼ vol flasks containing culture medium for D. melanogaster (1000 mL water, 20 g Saccharomyces cerevisiae yeast, 7 g agar, 35 g sugar, 40 g semolina and 1 mL propionic acid) at 25 °C and 65% relative humidity.
Infection of flies with L. plantarum 8PA3, L. plantarum AG10, E. coli and S. aureus strains, as well as recombinant strains E. coli pCtufAmpGfpr and S. aureus pCtuf-gfpCmr with constitutive GFP synthesis, were performed through a nutrient substrate. For that, synchronous embryo clutches were obtained and transferred to the surface of the Drosophila nutrient medium with the addition of 100 μL of bacterial cell suspension washed in phosphate-buffered saline (107–108 CFU/mL in sterile PBS).
2.5. The Analysis of Flies Viability on Embryonic and Metamorphosis Stages
The frequency of lethal mutations during embryogenesis was used as an indicator of the changes occurring in gametes of imagoes. The frequency of embryo lethality was determined as the fraction of the eggs that stopped their development at a certain stage out of the total number of the eggs laid. To perform the experiment, virgin imagoes from all control and experimental groups were separated according to their sex within the first day after eclosion and kept separately in vials with temporary culture medium until they reached a sexually mature age (three days). Then, males and females were put together for 12 h for mating. Subsequently, inseminated females were placed in Petri dishes (d = 10 cm) with temporary medium (5 individuals per dish) for 12 h to obtain eggs. In the next day, the eggs were counted using a stereoscopic microscope and then placed in a thermostat (t = 24 °C) for 48 h. The sample size was 10 Petri dishes for each experimental group. The embryo lethality level was calculated according to [
82].
Viability at the pupa stage was estimated as a percentage of those that did not hatch at the end of the period of emergence of adults from puparia in the offspring of five parental pairs. At the same time, the preimaginal death was assessed by changes in the morphology of the pupa.
2.6. Fertility Analysis
For the fertility analysis, five newly emerged females and five males were placed in vials with 10 mL of standard medium (control) and medium containing bacteria of all experimental groups for the oviposition period of 7 days. The vials were kept in an incubator until progeny appeared. Then, parental insects were removed from the tube. The number of adult males and females was fixed. For each experimental group, 10 vials were analyzed [
83].
2.7. DNA Comet Assay
To assess DNA damage in the gut of flies, an alkaline variant of the DNA comet assay was used; this allows for the determination of single-strand DNA breaks in cells [
84]. For all experimental groups, the gut of third-in star
Drosophila larvae was isolated and mechanically suspended in the Poel’s salt solution (15 mMNaCl, 6.4 mM NaH
2PO
4, 42 mMKCl, 7.9 mM CaCl
2, 1.8 mM KHCO
3, 20.8 mM MgSO
4; pH 6.95). Enterocytes were embedded in 0.75% agarose on slides. To prepare each single slide, a total of 5 guts were used. The slides were placed in a lysis solution (2.5 M NaCl, 100 mM EDTA, 10 mMTris, 1% Triton X-100; pH 10) for 1 h at 4 °C. Then the slides were incubated in an alkaline electrophoresis buffer (0.3 M NaOH, 1 mM EDTA, pH 13) for 10 min followed by electrophoresis at 15 V/300 mA for 10 min at 4 °C. Slides were washed 3 times in 20 mMTris (pH 7.5) and 3 times in distilled water and then fixed in ethanol for 10 min. Fluorescence microscopy (Carl Zeiss Axio Imager M2, Oberkochen, Germany) was used to visualize and rank the DNA comets.
2.8. SMART Test
The genotoxicity of
L. plantarum strains and strains of
E. coli and
S. aureus was assessed using standard cross versions of SMART on
D. melanogaster. Females from the
flr3/In(3LR)TM3 line were mated with
mwh/mwh males [
85,
86,
87]. The lines were kindly provided by O. N. Antosyuk, Ural Federal University (UrFU, Ekaterinburg, Russia). The emergent adults were collected and preserved in 70% ethanol. The preserved fly wings were placed under a stereoscopic microscope using entomological pincers and the wing pairs were spread over codified slides. A Faure solution (30 g gum Arabic, 20 mL glycerol, 50 g chloral hydrate, and 50 mL distilled water) was used to fix the wings. After mounting, the slides remained on a warm plate (60 °C) for 1 h. The spots (single or twin) on the wings were counted by using a Carl Zeiss Observer 1.0 microscope (Carl Zeiss, Oberkochen, Germany) at 400× magnification.
2.9. DNA Extraction
A single colony of L. plantarum AG10 was grown in Man–Rogosa–Sharpe (MRS) Broth (SigmaAldrich, St. Louis, MO, USA) under microaerophilic conditions at 37 °C overnight. Then, bacterial cells were harvested by centrifugation at 10,000 rpm for 5 min and the genomic DNA was extracted using GeneJET Genomic DNA Purification Kit (ThermoFisher, Waltham, MA, USA) according to the manufacturer’s protocol. The quality and purity of DNA was checked by using 0.7% agarose gel electrophoresis and OD260/OD280 ratio on Nanodrop2000 systems.
2.10. Genome Sequencing and Assembling
The
L. plantarum AG10 whole genome sequencing was performed using a high-throughput IlluminaMiSeq platform. For IlluminaMiSeq sequencing, DNA was sheared to fragments ranging between 300 and 500 bp using the Covaris S220 (Covaris, Woburn, MA, USA) The fragmented DNA sample was end-paired, dA-tailed and ligated to multiple adapters. The ligated products were purified and further enriched using PCR, and paired-end sequencing was performed by using IlluminaMiseq (Illumina, San Diego, CA, USA). The quality of sequence reads was assessed using FastQC (version 0.11.9) [
88]; the genome was assembled using SPAdes 3.15.3 [
89]. The raw data of complete genome sequence of
L. plantarum AG10 are available in NCBI GenBank database as BioProject PRJNA953814.
2.11. Bioinformatic Analysis
The assembled genome was annotated using Prokka [
90]. Distribution of genes into subsystem categories was performed using the RAST server [
91].
2.12. Statistical Analysis
All experiments were performed in biological triplicates with three repeats in each unless otherwise specifically stated. Statistical data analysis was performed using one-factor analysis of variance (one-way ANOVA) statistical test with Holm–Sidak correction for multiple testing in GraphPad Prism version 6.0 for Windows (GraphPad Software). For each indicator, the arithmetic mean, its estimation error and standard deviation were calculated. For data with non-Gaussian distribution, a non-parametric one-way analysis of variance (Kruskal–Wallis) test has been performed and median values with interquartile ranges were shown.
4. Discussion
The widespread emergence of pathogenic bacteria resistant to conventional antimicrobials represents one of the key health challenges worldwide, and thus active screening of new approaches for the prevention and treatment of diseases is essential. Probiotic bacteria, such as lactic acid bacteria,
Bacilli and their metabolites, have been intensively investigated as promising natural and eco-friendly substitutes to antibiotics [
18,
19,
20,
25,
26].
Recent data indicate that the
L. plantarum AG10 strain, isolated previously from clover silage, demonstrates promising antagonistic activity and probiotic properties in vitro [
50,
52]. In this work, we also show that it competes with pathogens in vivo, as well as diminishes their negative effects on the host. Besides the pronounced repression of most foodborne pathogens in the classic agar drop diffusion test (
Table 1), this bacterium was able to suppress the growth of
S. aureus and
E. coli during milk fermentation. Thus, the amount of viable
E. coli cells in the final product decreased from 10
6 to 10
5 after 8 h of milk fermentation by
L. plantarum AG10 (
Figure 1A); in the milk fermented by approved probiotic
L. plantarum 8PA3, the viable
E. coli cells increased until 10
8 (
Figure 1B). Similar suppression has been observed for
S. aureus under the same conditions (Compare
Figure 1C,D), suggesting antimicrobial potential of the strain. In addition, low amounts of putative virulence factors have been detected in silico in the genome of
L. plantarum AG10 by using the VFDB database (in contrast to 30 found in
L. plantarum 8PA3 [
100]), suggesting its prominent safety profile as a potential probiotic.
The ability of
L. plantarum AG10 to compete with pathogenic flora has been further demonstrated in vivo using a
Drosophila melanogaster model. AG10 could diminish the negative effects of both
S. aureus and
E. coli on the survival characteristics of
Drosophila melanogaster at both embryonic (larvae) and pupa stages, while the reference strain was active mainly against
S. aureus only. Thus, the addition of either
L. plantarum AG10 or the reference
L. plantarum 8PA3 strain significantly reduced the mortality of
Drosophila embryo and pupa infected with
S. aureus, while
L. plantarum AG10 could also level out the effects of
E. coli on the survival of insects (
Figure 3A,B). Consequently, in groups infected with pathogens in the presence of
Lactobacilli, the amount of imago was similar to the untreated group or groups treated solely with
Lactobacilli (
Figure 3). Since bacteria added to the flies’ growth media were preliminary washed by PBS, the observed effects are apparently driven by antimicrobial substances produced by
Lactobacilli de novo. Moreover, the experiment with
S. aureus and
E. coli overexpressing GFP confirmed that pathogens are captured by larvae (
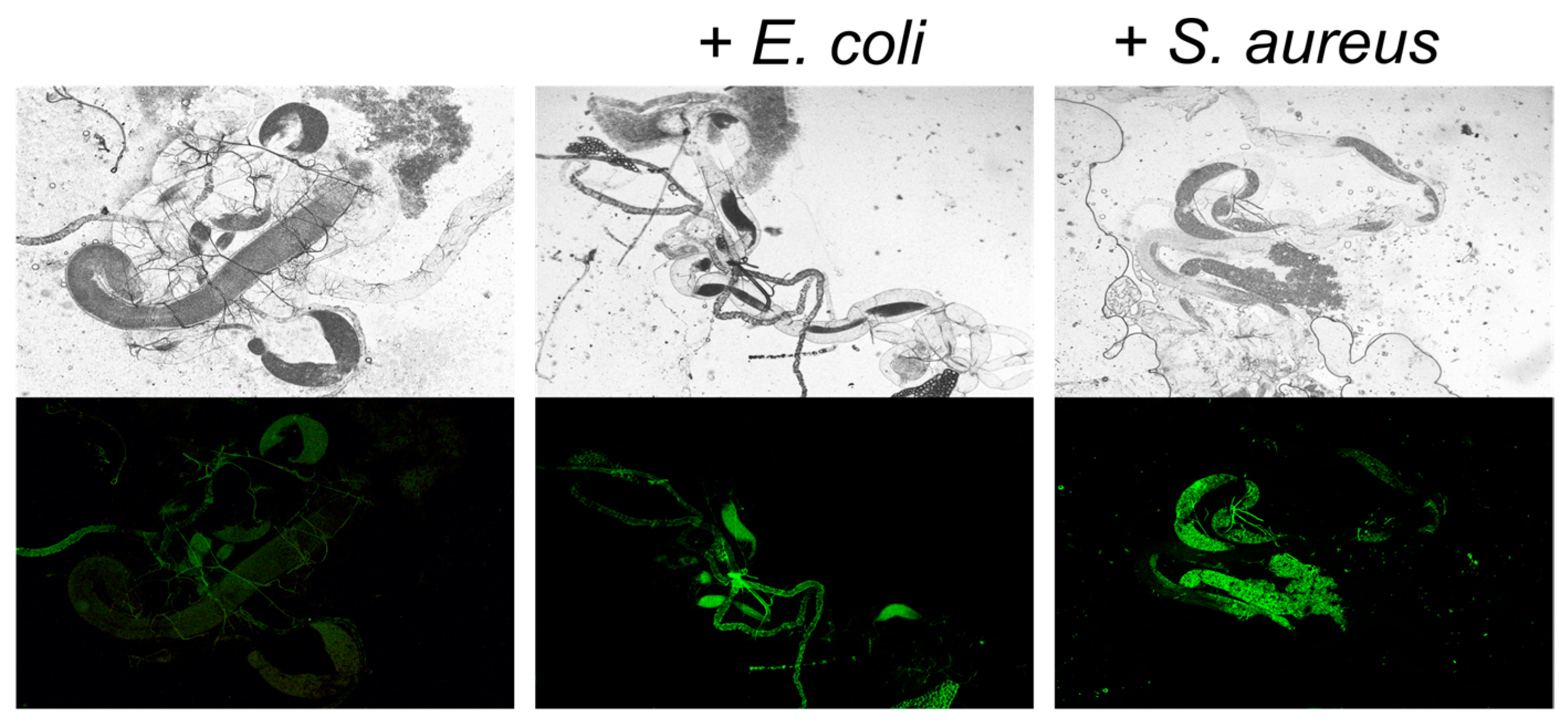
Figure 2), whereas successful introduction of
Lactobacilli to the larvaes’ gut was shown previously [
100], suggesting that the repression of pathogens occurs in vivo, while ex vivo interactions remain also possible.
The exact mechanism of the observed antimicrobial activity remains open to discussion. Lactic acid bacteria (LAB) compete with other bacteria by using various mechanisms such as synthesis of organic acids (mainly lactic acid) and acidification of the broth [
31], hydrogen peroxide production [
32] and secretion of antimicrobial peptides, as well as bacteriocins [
33,
34]. While broth acidification seems to be the main factor of antagonism in both agar drop diffusion and milk fermentation tests, antimicrobial peptides could play the leading role in vivo [
33,
34]. Moreover, it has been reported previously that
L. plantarum AG10 acidifies the milk in a less pronounced manner than the industrial strains [
52,
53], assuming that the production of antimicrobial peptides could be apparently responsible for the observed effects both in vitro and in vivo. Indeed, the screening of putative antimicrobial peptides in the genome of
L. plantarum AG10 in comparison with the dbAMP database allowed identification of three peptides with over 90% homology with peptides characterized by presumable antimicrobial activity (
Table 3). Among them, LLCBMPMO_01967 seems to be active only against
S. aureus, while LLCBMPMO_01968 exhibit antimicrobial properties against both
S. aureus and
E. coli with a high probability (
Table 5). These findings agree with observations of the apparent repression of pathogenic bacteria in
Drosophila (
Figure 3 and
Figure 4). The reference strain
L. plantarum 8PA3 also carries four putative AMPs in the genome (
Table 4) with WP_046947768.1, with predicted activity against both pathogens for the latter (
Table 5). Nevertheless, during in vivo experiments,
L. plantarum 8PA3 did not reduce the mortality in
E. coli infected group (
Figure 3 and
Figure 4), suggesting that this AMP is apparently either inactive or non-expressed under experimental conditions. Thus, despite the relatively high possibility of antimicrobial properties predicted for AMPs in silico (
Table 4), an independent in vitro verification of their activity is required in further investigations.