1. Introduction
Many consumers favor fried food because of its crisp taste, quick preparation, and convenience. To reduce costs, reusing fried oil has become a common practice in many fast-food restaurants. Cooking oil is easily oxidized by frequent heating to high temperatures, which not only reduces its nutritional value but also can form toxic chemicals, including benzopyrene and heterocyclic amines, that can leach into food. Eating oil-fried food can lead to obesity, metabolic disorders, systemic low-grade inflammation, and other diseases [
1,
2].
The consumption of fried food commonly induces systemic low-grade inflammation. This state is non-specific, persistent, and mild, as evidenced by a 2-4-fold increase in plasma inflammatory factors such as TNF-α [
3], a cytokine considered to be a key factor in age-related diseases, autoimmune disorders, cancer, cardiovascular diseases, and diabetes [
4]. Therefore, it is important to investigate the low-grade inflammation mechanism caused by the repeated use of oil for frying to understand the mechanistic targets and their regulation.
Some studies suggest that the damage caused by fried oil to the body systems is a result of oxidative stress [
5]. However, there is now strong evidence that the gut microbiota and its metabolic disturbances are also responsible for the induction of systemic low-grade inflammation, especially by changes in the composition and levels of short-chain fatty acids (SCFAs), which are mostly the metabolic end products of the gut microbiota [
6]. They regulate body inflammatory responses by inducing or inhibiting cytokine production [
7]. However, the changes in the microbiota and associated metabolite composition caused by different inducers and biomarkers of systemic low-grade inflammation are different. Therefore, clarifying the effects of fried oil on gut microbiota structure and SCFA levels is important for identifying the mechanisms by which fried oil induces systemic low-grade inflammation. Based on this, we hypothesized that a short-term consumption of repeatedly fried soybean oil would induce systemic low-grade inflammation by altering the structure of the gut microbiota community and the production of SCFAs, thus providing a basis for exploring targeted interventions to mitigate the health risks associated with a frequent consumption of fried foods.
The current literature on the harm caused by fried oil is mostly based on a long-term (≥30 days) and excessive consumption of fried oil [
8,
9], and there is a lack of research on the effects of short-term (one week) intake of fried oil. The short-term consumption of fried oil aligns with the frequency of the individual consumption of fried foods in daily life. Investigating the mechanism of induction of systemic low-grade inflammation following short-term exposure may offer more precise health recommendations.
This study explored the mechanism of low-grade inflammation induced by the short-term dietary intake of fried oil to provide theoretical support for exploring specific control targets.
2. Materials and Methods
2.1. Preparation of Fried Oil
Chicken fillets, French fries, and dough were fried in fresh soybean oil for 10 min each hour for 8 h (from 9 a.m. to 5 p.m.) for 4 consecutive days, for a total of 32 times. Four sets of soybean-fried oils were prepared based on the percentage of heated soybean oil in the mix. We used unheated soybean oil (C), 25% fried soybean oil +75% unheated soybean oil (low dose: LD), 50% fried soybean oil +50% unheated soybean oil (medium dose: MD), and 100% fried soybean oil (high dose: HD).
2.2. Animal Study
Six-week-old male Kunming mice (30 ± 2 g) were purchased from Changsha Tianqin Biotechnology Co., Ltd., Changsha, China (SPF, SCXK [Xiang] 2019-0014). The experimental animals were maintained at room temperature (RT: 25 ± 2 °C), a humidity of 40–70%, and a 12 h light/dark cycle. The experimental animals were approved by the Animal Ethics Committee of Guangdong Ocean University (GDOU-LAE-2020-018) and raised in strict accordance with the operating standards of the Experimental Animal Center of Guangdong Ocean University (SYXK2014-0053). The standard polypropylene cages used in the experiments were 53 cm in length, 39 cm in width, and 20 cm in height, which provided ample living space for the mice and complied with animal welfare and ethical standards. Water bottles, cages, and bedding materials were sterilized using high-pressure steam, and the water bottles and bedding materials were changed three times a week. After one week of adaptive feeding, 20 mice were randomly divided into 4 groups with 5 animals each: C, LD, MD, and HD. All mice received an intragastric administration of either unheated soybean oil (C) or fried oil (LD, MD, HD) at 4.4 g/kg, respectively, once daily for 6 d. The body weights of the mice were measured daily during the experimental period.
2.3. Serum TNF-α
After 6 days of gavage, blood was collected from the eyeballs of mice on day 7, and centrifuged at 4 °C, 8300 r/min, for 5min in a cryo-centrifuge (ST16R, Thermo Fisher Technology [China] Co., Ltd., Shanghai, China). The supernatant was aspirated and placed in a −80 °C ultra-low-temperature refrigerator (JW-86-158-LA). Serum TNF-α concentration was measured using a TNF-α ELISA kit (Shenzhen Xinbosheng Biotechnology Co., Ltd., Shenzhen, China) procedure.
2.4. Gut Microbiota
On day 7, the fecal samples of mice were collected into 1.5 mL sterile EP tubes and stored in an ultra-low-temperature refrigerator at −80 °C. Fecal samples were sent to Hangzhou GH Information Technology Co., Ltd., Hangzhou, China. for 16S rRNA gene sequencing and detection using an Illumina HiSeq4000 pair-end 2 × 150 bp sequencing platform. Gut microbiota strain categories were identified by database comparisons.
2.5. Fecal SCFAs
Fecal SCFA levels were detected using GC-MS (GCMS-TQ8050 NX, Shimadzu Co., Ltd., Kyoto City, Japan). Fecal samples (50 mg) were added to 100 μL of 15% phosphoric acid, followed by 100 μL of a 125 μg/mL isocaproic acid solution and 900 μL diethyl ether, and then homogenized and centrifuged for 10 min. The mixed solution was filtered using a 0.22 μm organic microporous membrane. The GC conditions were as follows: a VF-17MS capillary column; inlet temperature of 250 °C; ion source temperature of 230 °C; transmission line temperature of 250 °C; and four-bar temperature of 150 °C. MS conditions were the following: an electron bombardment ionization source, single-ion scanning mode, and electron energy of 70 eV.
2.6. Statistical Analysis
The experimental data were statistically analyzed using SPSS27.0 software. Pearson’s method was used to analyze data correlation. Data are expressed as the “mean ± SD”, and a one-way analysis of variance was used to compare the means of each group, with p < 0.05 indicating a statistically significant difference.
3. Results
3.1. Effects of Fried Oil on Body Weights
As shown in
Figure 1, the change in body weight in the LD, MD, and HD groups was significantly higher than that in group C, with increases of 86.3% (
p < 0.05), 137%, and 168% (
p < 0.01), respectively.
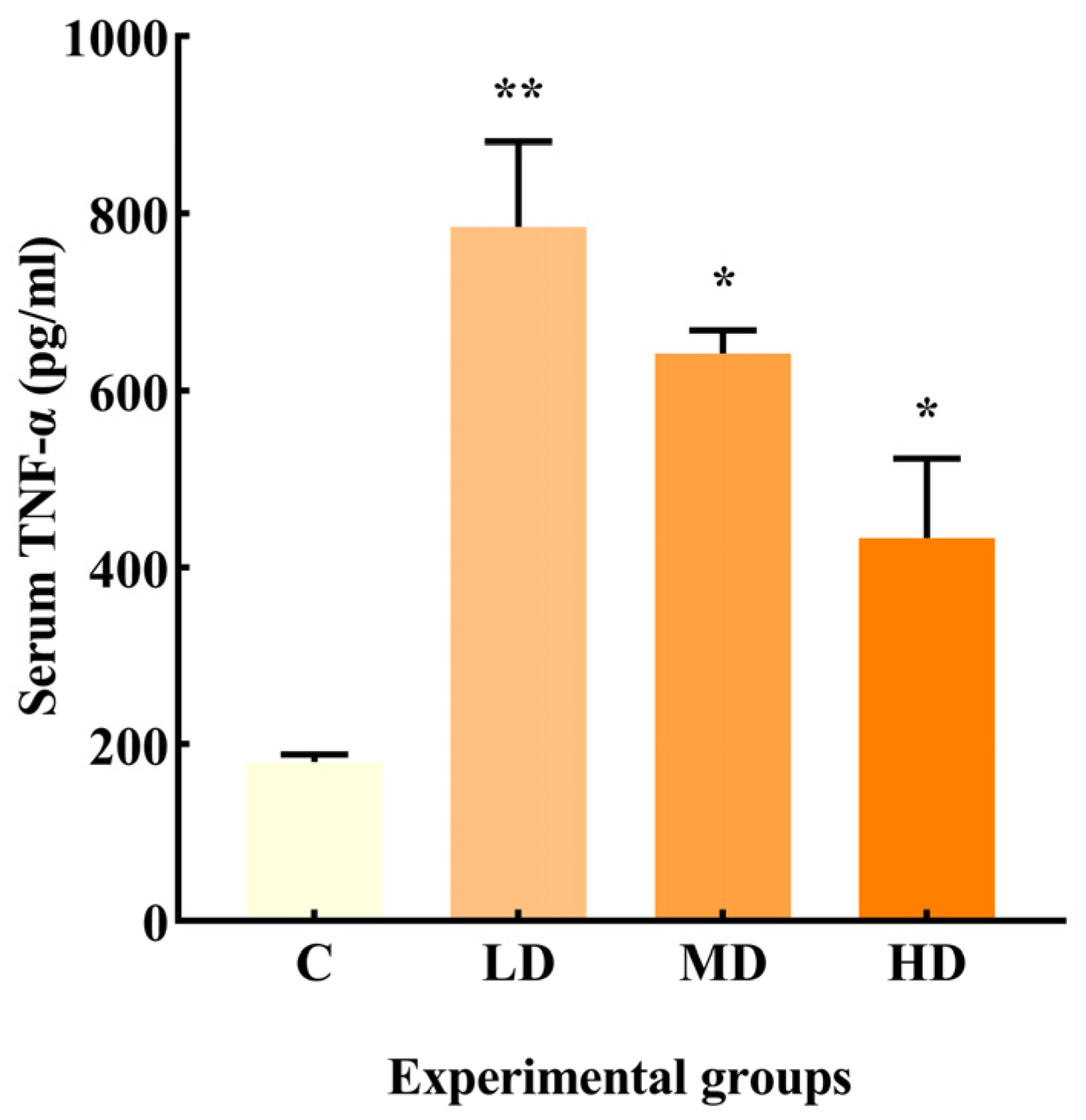
3.2. Effects of Fried Oil on Serum TNF-α
As illustrated in
Figure 2, the LD, MD, and HD groups exhibited a significant increase in TNF-α production by 335% (
p < 0.01) and 256% and 140% (
p < 0.05), respectively, compared to the C group.
3.3. Effects of Fried Oil on the Gut Microbiota Diversity
As shown in
Figure 3A, 3209, 3186, 3125, and 2997 OTUs were obtained in the C, LD, MD, and HD groups, respectively. With an increase in the fried oil dose, the OTU values of the gut microbiota in mice gradually decreased. These results indicate that a certain dose of fried oil can reduce the species diversity of the gut microbiota in mice in the short term. The total OUT counts in the LD, MD, HD, and C groups were 2845, 2771, and 2689, respectively. As the dosage of fried oil increased, the population of OTUs shared in the gut microbiota of mice treated with fried oil and non-fried oil gradually declined, indicating an increasing divergence in the gut microbiota between the two groups. A principal coordinate analysis using UniFrac indicated differences in the gut microbiota between samples and revealed different clusters of microbiota composition in each group. As illustrated in
Figure 3B, the differences in the gut microbiota between samples were insignificant, indicating that the structural characteristics of the gut microbiota were analogous in all groups. In
Figure 3C, the Shannon index of the MD and HD groups was significantly lower than that of the C group (
p < 0.05).
In conclusion, this indicates that in terms of alpha diversity, intake of medium and high doses of fried oil can reduce the richness and diversity of the gut microbiota.
3.4. Effects of Fried Oil on the Abundance of Gut Microbiota at the Phylum Level
The gut microbiota constitutive abundance of the mice in each group at the phylum level is shown in
Figure 4. The relative abundances of gut microbiota Firmicutes and Desulfobacterota in the LD group increased by 25% and 19%, respectively, compared to the C group. The relative abundance of Deferribacterota and Campilobacterota decreased by 31% and 37%, respectively.
The relative abundance of Proteobacteria in the gut microbiota of mice in the MD group increased by 122% (p < 0.05), and that of Deferribacterota increased by 27%. The relative abundance of Firmicutes and Campilobacterota decreased by 46% and 60%, respectively (p < 0.05), and Desulfobacterota decreased by 53%.
The relative abundance of Proteobacteria in the HD group was significantly increased by 156% (p < 0.05), whereas the relative abundance of Desulfobacterota, Firmicutes, and Campilobacterota was significantly decreased by 64%, 58%, and 85%, respectively (p < 0.05).
3.5. Effects of Fried Oil on Gut Microbiota Abundance at Genus Level
The gut microbiota of the mice in each group at the genus level are shown in
Figure 5. The relative abundance of
Muribaculaceae and
Alistipes in the gut microbiota of mice in the LD group was reduced by 37% and 60%, respectively, compared with the C group.
Alloprevotella significantly decreased by 76% (
p < 0.05). The relative abundance of
Lachnospiraceae_NK4A136_group,
Helicobacter, and
Roseburia increased by 96%, 170%, and 123%, respectively. Notably, the abundance of Lactobacillus significantly increased by 385% (
p < 0.05).
The relative abundance of Escherichia–Shigella in the MD group increased 8-fold, and Bacteroides, Roseburia, Lactobacillus, and Muribaculaceae by 62%, 95%, 207%, and 6%, respectively, whereas the relative abundance of Lachnospiraceae_NK4A136_group, Alloprevotella, Parabacteroides, and Alistipes decreased by 52%, 58%, 56%, and 42%, respectively.
The relative abundance of Escherichia–Shigella increased by 39-fold in the HD group. Parabacteroides, Muribaculaceae, and Lactobacillus increased by 45%, 37%, and 174%, respectively, while the abundance of Bacteroides, Helicobacter, Alloprevotella, and Alistipes decreased by 3%, 29%, 37%, and 13%, respectively.
3.6. Effects of Fried Oil on Fecal SCFAs
Figure 6 reveals that there was no significant change in the levels of acetic acid, propionic acid, butyric acid, isobutyric acid, isovaleric acid, and caproic acid in the feces of mice in the fried oil group compared with the C group, but the levels of valeric acid were significantly increased by 153% (
p < 0.01), 89%, and 78%, respectively (
p < 0.05).
3.7. Correlation Analysis of Gut Microbiota, SCFA Concentration, and TNF-α Levels
As shown in
Figure 7,
Alloprevotella abundance was significantly negatively correlated with TNF-α levels (
p < 0.01).
Lactobacillus abundance was significantly positively correlated with TNF-α levels (
p < 0.05). Valeric acid concentration was significantly positively correlated with TNF-α levels (
p < 0.05) and
Lactobacillus abundance (
p < 0.01), and significantly negatively correlated with
Alloprevotella abundance (
p < 0.05).
Lactobacillus abundance was significantly negatively correlated with the abundance of
Alloprevotella (
p < 0.05).
4. Discussion
Previous research indicates that obese individuals with lower bacterial richness gain more weight over time [
10]. Additionally, a prolonged consumption of high-fat diets can also alter the gut microbiota composition, leading to an increased inflammatory state and obesity [
11]. In the current study, a significant increase in body weight was observed in mice in the LD, MD, and HD groups compared to the C group, suggesting that even a short-term intake of a certain dose of fried oil can significantly increase body weight, while systemic low-grade inflammation can contribute to obesity.
Among the pro-inflammatory cytokines, TNF-α can mediate macrophages to release cytokines, causing low-grade inflammation and apoptosis [
12]. Long-term intake of fried oil can increase plasma pro-inflammatory cytokines, but short-term intake of fried oil significantly increased by two to four times in body systems is rarely reported [
13]. In this study, serum TNF-α was significantly increased by two to four times in mice administered fried oil within a week compared to that in the C group.
The Shannon index analysis showed that the richness and diversity of gut microbiota in the MD and HD groups were significantly reduced at 6d, which is consistent with the results of a previous study in mice fed fried corn oil and lard for six weeks [
14].
The relative abundance of Proteobacteria is associated with toxins, which contain a variety of pathogenic bacteria (such as
Escherichia,
Shigella, etc.) that mediate cytokine aggregation and the formation and exacerbation of inflammation, which is a potential diagnostic indicator of ecological disorders and disease risk, and an increase in the abundance of Proteobacteria may further facilitate invasion by exogenous pathogens [
15]. In intestinal inflammatory syndromes, such as irritable bowel syndrome (IBS) and metabolic syndrome, an increase in the abundance of Proteobacteria has been evident [
16]. In our study, the relative abundance of Proteobacteria in gut microbiota of mice in the MD and HD groups was significantly increased by 122% and 156%, respectively, with the relative abundance of
Escherichia–Shigella increased by 8 and 39 times, respectively. Furthermore, the abundance of
Lactobacillus in the LD group was significantly increased, whereas that of
Alloprevotella significantly decreased. All these factors point to a disturbance in the gut microbiota upon ingesting fried oil, even in the short term.
Lactic acid bacteria can aid in balancing the gut microbiota and boosting immunity.
Lactobacillus converts lactose into lactic acid, which is an important metabolic intermediate in SCFA metabolism but can also serve as a fuel source for cancer cells, promoting inflammation, angiogenesis, and metastasis [
17]. The immunomodulatory effect of
Lactobacillus can also be achieved through the release of cytokines induced by its metabolites, including SCFAs, tumor necrosis factor, interferon, and transforming growth factor [
18]. A long-term high-fat diet can lead to a reduced number of
Lactobacilli in the gut; conversely,
Lactobacillus can alleviate high-fat diet-induced obesity in mice [
19,
20]. However, in our study, the relative abundance of
Lactobacillus in the LD group significantly increased by up to 385% in the short term. According to the Pearson correlation analysis, the relative abundance of
Lactobacillus was significantly positively correlated with TNF-α levels. This result contradicts the common belief that an increase in
Lactobacillus abundance can reduce the levels of inflammatory factors. It seems that an increase in
Lactobacilli does not necessarily mean better health, and in fact a large increase in
Lactobacillus abundance may promote an increase in inflammatory factor levels [
21,
22].
Alloprevotella can decompose protein- and carbohydrate-containing foods. It can also function as a conditional pathogen that induces intestinal inflammation.
Alloprevotella is more abundant in vegetarians compared with non-vegetarians [
23], and is generally considered to be a bacterium associated with a healthy plant-based diet, acting as a “probiotic” in the human body and therefore any reduction can lead to disease [
24]. Recent human studies have linked increased
Alloprevotella abundance to local and systemic low-grade inflammation [
25]. In our study,
Alloprevotella abundance decreased significantly in the LD group and was significantly negatively correlated with
Lactobacillus abundance and TNF-α levels, suggesting that frying increased soybean oil acidity, making the intestinal environment more favorable for
Lactobacillus growth. The proliferation of
Lactobacillus produces metabolites such as lactic acid and valeric acid, which further increase gut acidity and inhibit the growth, reproduction, and activity of
Alloprevotella, culminating in a reduced abundance [
26]. It has been suggested that the decrease in
Alloprevotella abundance may indirectly contribute to reducing the inflammatory response and promoting gut ecological balance in the context of fried oil intake. In some studies, in rats fed fried soybean oil for six weeks, it was found that gut
Lactobacillus abundance was significantly increased and
Alloprevotella abundance was significantly decreased, which is consistent with our study. It is speculated that the immune system has the capacity to suppress multiple oxidative stress products formed during feeding of fried oil by promoting the proliferation of
Lactobacillus, whereas the decrease in
Alloprevotella abundance is related to the fermentation and utilization of carbohydrates and monosaccharides [
27].
Most previous studies on the induction mechanism of systemic low-grade inflammation have primarily focused on other signaling pathways such as nuclear factor kappa-B (NF-κB) and mitogen-activated protein kinase (MAPK). However, research has linked gut microbiota metabolites and SCFAs to act as anti-inflammatory agents via G protein-coupled receptors and histone deacetylase [
28,
29]. Recent research has indicated that valeric acid, a branched short-chain fatty acid, is negatively correlated with good health. Excessive blood valerate concentration exacerbates inflammation and damages brain cells and dendritic branches; however, gut microbiota changes and elevated blood valerate levels can be alleviated by appropriate exercise [
30]. The fecal valeric acid concentration in children with autism is higher than that of acetic acid and butyric acids [
31]. A significant increase in fecal valeric acid levels occurs in patients with IBS, and fecal valeric acid levels are significantly reduced following treatment [
32], suggesting that an excessive accumulation of valeric acid in the gut is detrimental to health. The Pearson correlation analysis showed a significant positive correlation between fecal valeric acid and TNF-α levels, which suggests that fried oil consumption affects health, even in the short term. Thus, an increase in fecal valeric acid levels, a metabolic product of the gut microbiota, increases TNF-α levels, and induces low-grade inflammation in the body. This result also provides an important theoretical basis for the search for control targets for systemic low-grade inflammation induced by fried oil.
This study indicated that fried oil, even at a low concentration and for a short duration, can cause gut microbiota changes. Such disturbances can result in the overgrowth of
Lactobacillus and a reduction in the proliferation of
Alloprevotella. Subsequently, there was an increase in the bacterial metabolite valeric acid, contributing to a significant increase in serum TNF-α levels. However, upon exposure to a high concentration of fried oil, TNF-α declined, but the gut microbiota metabolites isovaleric acid, butyric acid, and caproic acid showed an increasing trend with minimal changes in SCFAs. Notably, this phenomenon may stem from the non-linear dose–response relationships that are common in biological systems. Within a certain range, increasing exposure levels may attenuate, rather than enhance, the strength of an organism’s response, reflecting the complex regulatory mechanisms in the body, including adaptive responses and negative feedback inhibitory pathways [
33]. For example, initial exposure to low doses of fried oil may stimulate a rapid TNF-α response; however, the subsequent activation of negative feedback mechanisms may have led to a decrease in its levels at the time point of our observations [
34]. In addition, our study focuses on short-term exposure effects, where organisms may utilize adaptive mechanisms to respond effectively to such inflammatory stimuli, explaining the observation of a short-term decline in TNF-α. However, long-term exposure scenarios, where these adaptive mechanisms may gradually fail and cumulative damage exceeds repair capacity, may eventually manifest as a persistent or exacerbated inflammatory response, leading to increased TNF-α levels, a direction worthy of further exploration in future studies [
35]. Previous reports have shown that an increase in gut butyric acid concentration induces an inhibitory effect on TNF-α levels [
36]. Whether butyric acid influences TNF-α levels can be tested by extending the test period in future studies. A future goal would be to regulate gut microbiota to stimulate the growth of specific microorganisms, thereby increasing the proportion of target gut microbiota or finding a substance that can inhibit the increase in the gut metabolite valeric acid, thereby minimizing inflammation in the body. According to our study, consuming a certain dose of fried oil even for a short period can lead to an increase or decrease in selective gut microbiota, resulting in an imbalance in microbiota homeostasis and an increase in fecal valeric acid and inflammatory factor TNF-α in serum, inducing a low-degree inflammation in the body.