1. Introduction
Salmonella enterica serovars Enteritidis and Typhimurium have been reported as the most common causes of salmonellosis outbreaks related to food contamination [
1]. The US Centers for Disease Control and Prevention (CDC) reported an estimation of 1 million domestically acquired salmonellosis cases with 19,000 hospitalizations and 380 deaths [
2]. Animal farms are typically associated with the presence of diverse
Salmonella serovars [
3,
4]. The environments around farms such as feedstuff, soil, water, and feces are common sources of
Salmonella [
4]. Distribution of
Salmonella on farms leads to the likelihood of
Salmonella contamination in various food of animal origin and produce.
Salmonella serovars predominantly present in food can be differed, for example, serovars Enteritidis and Typhimurium are common in eggs [
5], serovars Kentucky and Enteritidis are common in poultry [
6], serovars Newport, Infantis and Javiana are common in fruits and vegetables [
7]. The environments of food processing plant have been linked to occurrence of diverse
Salmonella serovars. For example, serovars Weltevreden [
8,
9], Anatum, Enteritidis, Newport, and Typhimurium have been previously detected in wastewater from the plant [
10].
Salmonella serovars predominant in human are also of crucial concern. These include
Salmonella serovars Weltevreden, Enteritidis and Anatum which have been reported as the most common serovars isolated from human in Thailand [
11]. The occurrence of
Salmonella contaminated in poultry meat has been often reported [
12]. Fresh produce such as sprout has also been reported as the high risk food that is commonly been contaminated by
Salmonella [
12,
13]. Control of
Salmonella in both food categories is thus crucial.
Bacteriophages (phages) are viruses of bacteria which are adapted to be very specific for infecting and killing bacterial hosts [
14]. The habitat of phages is normally related to their hosts as predator and prey [
15]. Phage application as a biocontrol agent against pathogens has been reported the outstanding properties over antibiotics (e.g., specificity to target hosts) [
14] or chemical agents which are commonly used in meat products and may have some negative effects on human health [
16]. Certain phages have been shown to have minimal effects on the change of quality and sensory properties of specific foods [
17]. To improve the antibacterial efficiency and avoid phage-resistant bacteria from using phage-based biocontrol, phage cocktail is an alternative approach. Several studies have reported the success in using effective phage cocktails to control
Salmonella serovars in various food products, including raw meat, fresh produce and ready-to-eat foods [
18,
19,
20].
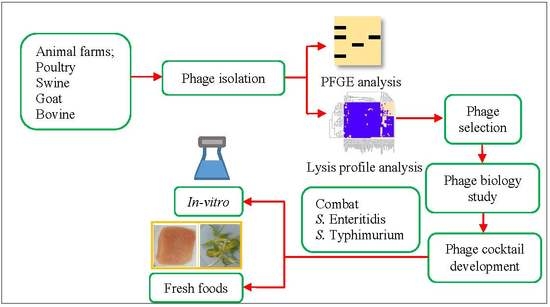
Phage abundance and diversity recovered from particular sources can provide a better understanding on the phage-host relationship. Phage lysis profiles obtained from testing phages against Salmonella strains from various sources are useful information for further development of phage-based control for targeting Salmonella serovars predominant in different sources. This study aimed to isolate Salmonella phages from animal farms (poultry, swine, goat and bovine) in Thailand and investigate phage lysis profiles on Salmonella strains from at least two geographical regions (Thailand and USA) isolated from different sources, including animal farms, food processing plants and humans that have a history of Salmonella infection. Phages presenting the highest lysis ability against the two most common Salmonella serovars (Enteritidis and Typhimurium) were further developed as a phage cocktail. One-step growth curves of all phages included in the cocktail were studied to determine phage biology including latent period and burst size. Our developed phage cocktail was evaluated the effectiveness in reducing S. Enteritidis and S. Typhimurium in vitro and in fresh foods (raw chicken meat and sunflower sprouts) during storage at 4 °C.
2. Materials and Methods
2.1. Sample Collection
A total of 12 samples were collected from six animal farms, including poultry, swine, goat, and bovine farms in Songkhla province, Thailand between January 2014 and October 2016. Of six farms, three of which were commercial farms (poultry, swine and goat) and three others were free range farms (poultry and bovine). Approximately 500 g of each randomly selected sample (feces, feed, soil and drinking water) was collected using a sterile spatula and transferred to a sterile bag. Feces samples were collected from the floor inside the pens of the commercial farms where animals resided. For free range farms, feces samples were collected from random open areas where animals resided. Feed samples were taken from the storage containers freshly prepared for feeding animals. Soil samples were taken from the free range farms only and from random open areas where animals resided. Drinking water in farms was also collected. Each sample was kept in a cooler box containing ice packs and transferred to a laboratory. Collected samples were stored at 4 °C until analysis in the laboratory.
2.2. Bacterial Strains Used in the Study
Salmonella strains used in this study consisted of 47
Salmonella strains presenting 28 common serovars (
Table 1). All
Salmonella strains tested were the representative of predominant serovars isolated from various sources, including animal farms, food processing plants and humans in Thailand and USA. A total of 23 strains of
Salmonella isolated from animal farms and human in the USA were obtained from Food Safety Laboratory, Cornell University (indicated with a prefix of “FSL”). Other 24 strains of
Salmonella were previously isolated from animal farms, food processing plants and humans in Thailand. These were obtained from the Faculty of Microbiology, Mahidol University and the Faculty of Agro-Industry, Prince of Songkla University, Thailand (indicated with a prefix of “PSU-BS-”).
Salmonella strains were kept in 15% glycerol at −80 °C as working stocks. For overnight culture, an isolated colony of
Salmonella from Tryptone Soya Agar (TSA; Oxoid, Hampshire, UK) was transferred in 5 mL of Tryptone Soya Broth (TSB; Oxoid, Hampshire, UK) and incubated at 37 °C for 16–18 h.
2.3. Isolation and Purification of Salmonella Phages
Salmonella phages were isolated using enrichment isolation with a multi-strain
Salmonella cocktail whose serovars are shown to be predominant in Thai animal farms (Agona H2-016, Anatum A4-525, Give H2-018, Kentucky W1-010, Stanley H2-002, Typhimurium H2-001, and Virchow H2-117) [
11,
21]. Briefly, 25 g of each sample was enriched with 225 mL of TSB and 2.5 mL of host mixture. After filtration of the mixture through 0.45 µm and 0.22 µm syringe filters, filtrate (100 µL) was used to prepare the double layer by mixing with each host strain previously grown in TSB for 16–18 h at 37 °C. For each overlay, 300 µL of the 1:10 dilution of the overnight host strain was mixed with 4 mL of 0.7% TSA, followed by incubation at 37 °C for 18–24 h. Plaques were observed on each host lawn. A distinct isolated plaque was selected and suspended in 300 µL of Phosphate Buffered Saline (PBS, (pH 7.4), 137 mM NaCl, 2.7 mM KCl, 4.3 mM Na
2HPO
4, 1.4 mM KH
2PO
4) for purification. Serial dilutions were performed and appropriate dilution was subjected to three passages with a specific host that showed a positive result, using a double layer agar technique [
4].
2.4. Lysate Preparation and Titer Determination of Salmonella Phages
An isolated plaque from the third purification passage was used to prepare 10-fold serial dilutions in PBS. Appropriate dilutions were used to prepare the overlay with a given host to yield semi-confluent lysis. Overlay was harvested with 10 mL of Salt-Magnesium buffer (SM buffer), followed by centrifugation at 3213×
g for 15 min at 4 °C. Supernatant was filtered through a 0.22 µm syringe filter and phage lysates were kept at 4 °C. Each phage lysate was serially diluted in PBS and 100 µL of each dilution was mixed with 300 µL of host, then the mixture was poured on bottom agar [
22]. Phage titers were determined after incubation at 25 °C for 16–18 h by counting plaques present on each plate of a given dilution [
22].
2.5. Determination of Lysis Profiles of Salmonella Phages
Lysis profile for each phage was determined by a spot test on bacterial lawn of a given
Salmonella strain in the collection included in this study. Briefly, 5 µL of each phage lysate representing 10
8 PFU/mL were spotted on the bacterial host lawn prepared as mentioned above but without filtrate. Phage lysis patterns were determined after 18–24 h of incubation at 25 °C. The experiment was performed in independent triplicates. Phage lysis patterns were analyzed by converting a positive lysis (zone of lysis on a spot) to a score of 1 and negative results were converted to a score of 0. A heatmap representing lysis groups was generated by cluster analysis, following Vongkamjan et al. [
22] with Ward’s method of binary distance, using the R software program (R development Core Team 2012 [
23].
2.6. Genome Size Determination of Salmonella Phages
Representative
Salmonella phages from each sample source were selected for genome size determination. A total of 17 phage isolates were included for Pulsed-Field Gel Electrophoresis (PFGE) analysis. Agarose plugs were prepared by mixing equal volume (55 µL) of a given phage lysate with high titer of approximately 10
6–10
8 PFU/mL and 1.3% low melting point agarose. Plugs were kept at low temperature (4 °C) in order to solidify for 1 h. Plugs were loaded into 1% agarose gel and electrophoresis was performed in 0.5X TBE buffer using CHEF-DR III system (Bio-Rad, Hercules, CA, USA). PFGE was performed for 20 h with 0.5–5 s of switch time. Two size markers were included; CHEF DNA size standard of 8–48 kb ladder and CHEF DNA size standard lambda λ ladder 0.05–1 Mb (both from Bio-Rad, Hercules, CA, USA) [
4,
22].
2.7. Development of a Phage Cocktail Targeting Two Major Salmonella Serovars
To develop a
Salmonella phage cocktail against
S. Enteritidis and
S. Typhimurium, nine phages which showed the strong lysis ability on
S. Enteritidis and
S. Typhimurium (KP1, KP2, KP4, KP5, KP9, KP34, KP36, KP49, and KP50) were ordered their lysis ability by a spotting test and efficiency of plating (EOP) using method modified from previous study [
24]. In this study,
S. Enteritidis FSL S5-371 and
S. Typhimurium H2-001 were used as the target hosts.
S. Anatum FSL A4-525 was used as the reference host of phages KP1, KP2, KP5 and KP9.
S. Virchow H2-117 was used as the reference host of phages KP34 and KP36.
S. Agona H2-016 was used as the reference host of phages KP49 and KP50. Three
Salmonella phages which showed the highest lysis ability were selected to prepare a cocktail using a ratio of 1:1:1 for each phage.
2.7.1. Spotting Assay
Each Salmonella phage dilutions ranging in concentration from 103–107 PFU/mL were spotted on bacterial lawn of S. Enteritidis or S. Typhimurium prepared as mentioned above. Immediately after spotting, the plates were incubated at 25 °C for 16–18 h. The clear zone or visible plaques formed by serial dilutions on the plate were determined as +++, confluent lysis (clear spot); ++, semi-confluent lysis (semi-clear); +, turbidity without plaque formation. The experiment was repeated three times for each phage.
2.7.2. Efficiency of Plating (EOP) Assay
Similar to a spotting assay, each Salmonella phage dilution ranging in concentrations from 103–107 PFU/mL were spotted on bacterial lawn of S. Enteritidis or S. Typhimurium. EOP assay replicates for a particular phage were done in parallel on both reference and target hosts. The EOP was calculated by the ratio of the average PFU on a target host to the average PFU on a corresponding reference host. EOP values were presented in 3 levels; high production (EOP ≥ 0.5), medium production (0.01 ≤ EOP < 0.5) and low production (0.0001 < EOP < 0.01).
2.8. One-Step Growth Curve
A one-step growth curve for three phages included in the phage cocktail were investigated following a protocol of Bao et al. [
25] with modifications. The bacterial hosts
S. Enteritidis and
S. Typhimurium grown overnight (10
7 CFU/mL) in TSB as mentioned above were mixed with 10
8 PFU/mL and 10
9 PFU/mL of phage to a final volume of 30 mL to represent the multiplicity of infection (MOI) of 10 and 100, respectively. The co-culture was incubated at 37 °C (220 rpm) for an initial attachment for 20 min, followed by centrifugation of the sample at 6000×
g for 10 min at 4 °C to remove the excess phage as the supernatant. Cell pellets were re-suspended with the same volume (30 mL) as pre-centrifugation with TSB and resumed to incubation for additional 60 min. Lysate (1 mL) was taken every 5 min for the standard plaque count assay (in triplicates) to determine the number of phages obtained from each period. Latent period was defined as the time interval between the adsorption (not including 20 min of pre-incubation) and the beginning of the first burst, as indicated by the initial rise in the phage titer. Burst size was calculated as the ratio of the final count of liberated phage particles to the initial count of infected bacterial cells during the latent period.
2.9. Efficiency of Phage Cocktail to Reduce S. Enteritidis and S. Typhimurium In Vitro and Evaluation of Phage-Resistance in Salmonella after Treated with Phage Cocktail
An overnight culture of S. Enteritidis and S. Typhimurium prepared as mentioned above (10 mL) was separately centrifuged at 6000× g for 10 min at 4 °C. To wash cell pellets, 5 mL of PBS were added and centrifuged at the same conditions three times. Washed Salmonella pellets were suspended in TSB and diluted to approximately 105 CFU/mL. Phage cocktail stock was diluted with TSB to achieve phage concentration at 107 PFU/mL. Each Salmonella strain suspension and phage cocktail preparation were mixed at a ratio of 1:1 by volume and incubated at 37 °C in a shaking incubator (ThermoStableTM IS-30 model, DAIHAN Scientific, Gangwon-do, Korea) at 220 rpm for 12 h. Controls included in the study were only S. Enteritidis and S. Typhimurium cultured in TSB. The cell numbers of S. Enteritidis and S. Typhimurium from the treatments and controls at each temperature tested were enumerated every 4 h interval by a spread plate on TSA. After 12 h of experiment, samples were collected for the analyses of phage resistance.
The changes in resistance phenotype of S. Enteritidis and S. Typhimurium after treated with a phage cocktail were evaluated by a spotting test. Five colonies of S. Enteritidis and S. Typhimurium from controls and treatments of phage cocktail recovered from TSA after 12 h of experiment above were re-cultured in TSB, followed by incubation at 37 °C for 16–18 h. Each culture was used to prepare an overlay for a spotting test. Serial dilutions (103–107 PFU/mL) of phage cocktail and each individual phage mixed in a cocktail were spotted on each bacterial lawn. Phage lysis patterns were determined after 18–24 h of incubation at 25 °C.
2.10. Efficiency of Phage Cocktail to Reduce S. Enteritidis and S. Typhimurium in Foods
Chicken meat and sunflower sprouts were selected as representative foods which commonly have
Salmonella contamination. Samples were purchased from the supermarkets in Hat Yai, Thailand. To eliminate
Salmonella that may be present, the samples were soaked in 50 ppm free chlorine concentration solution for 5 min [
26]. Subsequently, the samples were soaked and washed in sterile distilled water for 5 min for 3 times. Chicken breast was aseptically cut into pieces of approximately 5 × 5 cm
2 and sunflower sprout was aseptically weight approximately 5 g. Overnight culture of
S. Enteritidis and
S. Typhimurium was prepared. Cell pellets were suspended with PBS and diluted to obtain the final concentration of 10
5 CFU/mL. One milliliter of each
Salmonella strain was evenly spiked to the surface of each piece of chicken breast and sunflower sprout in a sterile Whirl-Pak bag to achieve the inoculation level of approximately 10
5 CFU/piece or 10
5 CFU/5 g. Samples were left on clean bench for 10 min to allow the cells to adapt to the conditions on food samples tested. A phage cocktail preparation (1 mL containing 10
7 PFU/mL) was evenly spiked to each piece of chicken and sunflower sprout in a sterile Whirl-Pak bag. PBS buffer (1 mL) was added to sample inoculated with
Salmonella as control. Treatments and control were stored at 4 °C. Number of
Salmonella cells was enumerated on day 0, 1, 2, 3, and 4 on Xylose-Lysine-Desoxycholate agar (XLD; Oxoid, Hampshire, UK).
2.11. Statistical Analysis
Differences of the bacterial counts between control and treatment by a phage cocktail in this study were analyzed by Student’s t-test for both in vitro and in food models. The Analysis of variance (ANOVA) was used to compare differences between the storage period for a given strain. Comparison of means was carried out by Duncan’s multiple range tests. Significance was declared at p < 0.05 using the statistical Package for Social Science (SPSS 10.0 for windows, SPSS Inc., Chicago, IL, USA).
4. Discussion
The recovery of high number of
Salmonella phages in this study suggests that animal farm environments in Songkhla province, Thailand represent an important source of abundant
Salmonella phages. Typically,
Salmonella phages have been isolated from diverse animal farms such as poultry farms [
25,
27], swine farms [
28,
29], goat farms [
30] and bovine farms [
4,
31]. Our study also suggests that free range farms could yield many phages, especially from soil and feed samples. Similar to previously reported sources of phages, Wongsuntornpoj et al. [
31] isolated a number of
Salmonella phages from small-scale free range cattle farms in Thailand [
31]. As the presence of bacteriophage is normally related to bacterial host population [
32], samples from free range farms including soil in an open land may have high opportunity to be exposed to various important sources of
Salmonella such as wild birds, insects, rodent droppings and other carriers [
33].
Variations in the genome size of
Salmonella phages obtained in this study indicate the potential diverse phage groups that may be distributed in farm environments in Thailand. Some identical genome sizes were observed among phages from various farms in different countries (
Table 8), suggesting presence of common phage types from these animal farm-related sources. Among
Salmonella phages isolated from dairy farms in USA, the genome sizes ranging from 22 kb to 156 kb [
4], while some phages from dairy farms in Thailand exhibited the genome sizes of 40 kb to 200 kb [
31]. Lysis profiles indicated that our isolated phages showed better ability to lyse
Salmonella strains from animal farms in Thailand as compared to strains from USA. Our findings indicate the relationship between phages and
Salmonella hosts is due to geological isolation. This phenomenon can be explained by the mechanisms of the phage-host receptors which are related to the evolution as pray-predator [
34]. However, the spectrum of phage lysis can be extended across different sources or regions of host habitats. In this study, our isolated phages showed lysis on
Salmonella strains from diverse sources including environments related to animal farms, animal slaughterhouses, food processing plants and from humans. Phages could also lyse bacterial hosts from different continents. Overall, phages isolated from animal farms in this study could lyse most serovars of
Salmonella that have been reported as the predominant and prevalent serovars in humans, foods and animal farms in Thailand including
Salmonella serovars Agona, Anatun, Give, Enteritidis, Kedougou, Kentucky, Typhimurium and Weltevreden [
11,
21,
35,
36]. Findings here suggests a potential for phage applications against bacterial hosts from various environments in different food chains or countries.
Phage latent period and burst size are major parameters which play an important role in the host lysis system [
42]. Phages which present a short latent period and large burst size will be replicated more quickly and the new phage particles (progeny) could be released more efficiently [
25]. Our phages combined in a phage cocktail showed a short latent period and large burst size of up to 97.7 and 173.7 PFU/cell on both
S. Enteritidis and
S. Typhimurium, respectively. The range of
Salmonella phage latent periods has been reported within the range of 15 to 45 min [
43]. Abedon et al. [
44] reported that shorter phage latent periods could be obtained by higher bacterial densities. It is also suggested that phage exhibiting very short latent periods maybe viewed as specialists for propagation when bacteria within cultures are highly prevalent [
44]. Phage burst size has been reported with large variations (5 to 250 PFU/cell) depending on the bacterial strain infected [
45]. Overall, the short latent period and large burst size of our phages suggests a rapid replication and effective release of new phage particles from both
S. Enteritidis and
S. Typhimurium hosts, thus appropriate for using to control bacterial hosts as reported by previous other studies [
46,
47].
Our developed phage cocktail showed high efficiency to control
S. Enteritidis and
S. Typhimurium in both in vitro study and different food categories that are typically linked to
Salmonella and foodborne outbreaks. Our phage cocktail could decrease
S. Enteritidis on chicken meat and sunflower sprouts by 0.66 log CFU/cm
2 and 1.27 log CFU/g, respectively.
S. Typhimurium on chicken meat and sunflower sprouts were decreased by 1.73 log CFU/cm
2 and 1.17 log CFU/g, respectively. In a previous study, Grant et al. [
48] demonstrated a < 1 log reduction of
Salmonella on ground chicken after treatment with the commercial
Salmonella phage cocktail (Salmonelex
TM, Micreos Food Safety, Wageningen, The Netherlands) [
48]. Another phage cocktail showed a reduction of
S. Enteritidis and
S. Typhimurium on chicken breast at 0.9 and 2.2 log CFU/g, respectively within 7 days at 4 °C [
20]. Our finding showed no evidence of phage resistance in both
Salmonella serovars upon phage cocktail treatment. This phenomenon might be related to the biology (latent period and burst size) of the phages included in our phage cocktail. Short periods of time to kill bacterial hosts for phages is an important factor that prevents the occurrence of bacteria resistance to phage [
43].

 and MOI 10 presented by
and MOI 10 presented by  . Bars represent the mean standard deviation (n = 3).
. Bars represent the mean standard deviation (n = 3).
 and MOI 10 presented by
and MOI 10 presented by  . Bars represent the mean standard deviation (n = 3).
. Bars represent the mean standard deviation (n = 3).
 and treatment of a phage cocktail presented by
and treatment of a phage cocktail presented by  . Bars represent the mean standard deviation (n = 3). The sign (*) on the lines indicates significant differences (p < 0.05) of bacterial count between control and treatment during storage time for a given Salmonella strain. Different uppercase letters on the lines indicate significant differences (p < 0.05) of bacterial count among days of storage for a given Salmonella strain.
. Bars represent the mean standard deviation (n = 3). The sign (*) on the lines indicates significant differences (p < 0.05) of bacterial count between control and treatment during storage time for a given Salmonella strain. Different uppercase letters on the lines indicate significant differences (p < 0.05) of bacterial count among days of storage for a given Salmonella strain.
 and treatment of a phage cocktail presented by
and treatment of a phage cocktail presented by  . Bars represent the mean standard deviation (n = 3). The sign (*) on the lines indicates significant differences (p < 0.05) of bacterial count between control and treatment during storage time for a given Salmonella strain. Different uppercase letters on the lines indicate significant differences (p < 0.05) of bacterial count among days of storage for a given Salmonella strain.
. Bars represent the mean standard deviation (n = 3). The sign (*) on the lines indicates significant differences (p < 0.05) of bacterial count between control and treatment during storage time for a given Salmonella strain. Different uppercase letters on the lines indicate significant differences (p < 0.05) of bacterial count among days of storage for a given Salmonella strain.
 ) and S. Typhimurium (
) and S. Typhimurium (  ) artificially contaminated in (a) chicken meat and (b) sunflower sprouts treated with a phage cocktail and stored at 4 °C for 4 days. Control (non-phage cocktail treated) of S. Enteritidis and S. Typhimurium inoculated on both samples presented by solid line and dashed line, respectively. Bars represent the mean standard deviation (n = 3). The sign (*) on the bars indicates significant differences (p < 0.05) of bacterial count between control and treatment during storage time for a given Salmonella strain. Different uppercase letters on the bars indicate significant differences (p < 0.05) of bacterial count among days of storage for a given Salmonella strain.
) artificially contaminated in (a) chicken meat and (b) sunflower sprouts treated with a phage cocktail and stored at 4 °C for 4 days. Control (non-phage cocktail treated) of S. Enteritidis and S. Typhimurium inoculated on both samples presented by solid line and dashed line, respectively. Bars represent the mean standard deviation (n = 3). The sign (*) on the bars indicates significant differences (p < 0.05) of bacterial count between control and treatment during storage time for a given Salmonella strain. Different uppercase letters on the bars indicate significant differences (p < 0.05) of bacterial count among days of storage for a given Salmonella strain.
 ) and S. Typhimurium (
) and S. Typhimurium (  ) artificially contaminated in (a) chicken meat and (b) sunflower sprouts treated with a phage cocktail and stored at 4 °C for 4 days. Control (non-phage cocktail treated) of S. Enteritidis and S. Typhimurium inoculated on both samples presented by solid line and dashed line, respectively. Bars represent the mean standard deviation (n = 3). The sign (*) on the bars indicates significant differences (p < 0.05) of bacterial count between control and treatment during storage time for a given Salmonella strain. Different uppercase letters on the bars indicate significant differences (p < 0.05) of bacterial count among days of storage for a given Salmonella strain.
) artificially contaminated in (a) chicken meat and (b) sunflower sprouts treated with a phage cocktail and stored at 4 °C for 4 days. Control (non-phage cocktail treated) of S. Enteritidis and S. Typhimurium inoculated on both samples presented by solid line and dashed line, respectively. Bars represent the mean standard deviation (n = 3). The sign (*) on the bars indicates significant differences (p < 0.05) of bacterial count between control and treatment during storage time for a given Salmonella strain. Different uppercase letters on the bars indicate significant differences (p < 0.05) of bacterial count among days of storage for a given Salmonella strain.