Outbreak of Tularemia in a Group of Hunters in Germany in 2018—Kinetics of Antibody and Cytokine Responses
Abstract
:1. Introduction
2. Methods and Materials
2.1. Hare Material
2.2. Samples from Hunting Dogs
2.3. Clinical Material from Humans: Blood Cultures, Throat Swabs, Serum
2.4. Cultivation of Sample Material from Humans and Dogs
2.5. Genomic DNA for Molecular Analysis
2.6. PCR Detection fopA, tul4, DD brucellosis mazG, IS711, Singleplex and Multiplex Real-Time PCRs, RD1-PCR
2.7. Whole Genome Sequencing
2.8. Enzyme Linked Immunosorbent Assay (ELISA) and Western blot (WB)
2.9. Cytokine and Chemokine Measurements
3. Results
3.1. Pathogen Detection in Hares, Humans, and Hunting Dogs
3.2. Antibody Detection in Hunting Dogs
3.3. Serology and Antibody Kinetics in Humans
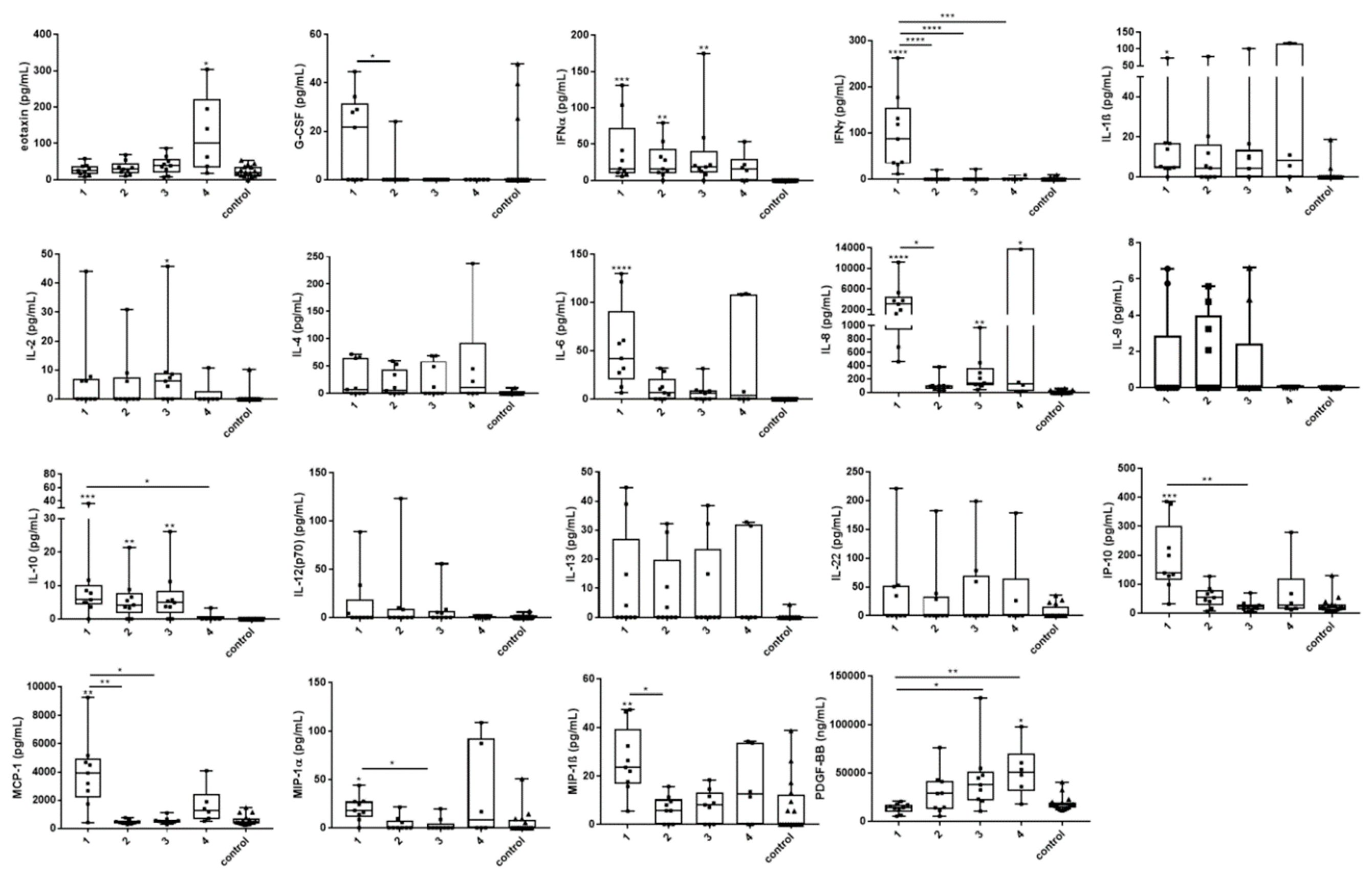
3.4. Cytokine and Chemokine Changes During Infection
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ellis, J.; Oyston, P.C.; Green, M.; Titball, R.W. Tularemia. Clin. Microbiol. Rev. 2002, 15, 631–646. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sjostedt, A. Special topic on francisella tularensis and tularemia. Front. Microbiol. 2011, 2, 86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Faber, M.; Heuner, K.; Jacob, D.; Grunow, R. Tularemia in Germany-A Re-emerging Zoonosis. Front. Cell. Infect. Microbiol. 2018, 8, 40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burckhardt, F.; Hoffmann, D.; Jahn, K.; Heuner, K.; Jacob, D.; Vogt, M.; Bent, S.; Grunow, R.; Zanger, P. Oropharyngeal tularemia from freshly pressed grape must. N. Engl. J. Med. 2018, 379, 197–199. [Google Scholar] [CrossRef] [PubMed]
- Jacob, D.; Koppen, K.; Radonic, A.; Haldemann, B.; Zanger, P.; Heuner, K.; Grunow, R. Molecular identification of the source of an uncommon tularaemia outbreak, Germany, autumn 2016. Eurosurveillance 2019, 24. [Google Scholar] [CrossRef]
- Hauri, A.M.; Hofstetter, I.; Seibold, E.; Kaysser, P.; Eckert, J.; Neubauer, H.; Splettstoesser, W.D. Investigating an airborne tularemia outbreak, Germany. Emerg. Infect. Dis. 2010, 16, 238–243. [Google Scholar] [CrossRef]
- Böhm, S.; Grunow, R.; vom Berge, K.; Böhmer, M. Tularaemia Outbreak After a Hare Hunt in Bavaria, Germany, 2018–Lacking awareness about the disease and its potential transmission routes? 2019. Available online: https://www.escaide.eu/sites/default/files/documents/ESCAIDE-abstract-book-2019.pdf (accessed on 22 October 2020).
- Broekhuijsen, M.; Larsson, P.; Johansson, A.; Bystrom, M.; Eriksson, U.; Larsson, E.; Prior, R.G.; Sjostedt, A.; Titball, R.W.; Forsman, M. Genome-wide DNA microarray analysis of Francisella tularensis strains demonstrates extensive genetic conservation within the species but identifies regions that are unique to the highly virulent F. tularensis subsp. tularensis. J. Clin. Microbiol. 2003, 41, 2924–2931. [Google Scholar] [CrossRef] [Green Version]
- Appelt, S.; Koppen, K.; Radonic, A.; Drechsel, O.; Jacob, D.; Grunow, R.; Heuner, K. Genetic diversity and spatial segregation of francisella tularensis subspecies holarctica in Germany. Front. Cell. Infect. Microbiol. 2019, 9, 376. [Google Scholar] [CrossRef] [Green Version]
- Becker, S.; Lochau, P.; Jacob, D.; Heuner, K.; Grunow, R. Successful re-evaluation of broth medium T for the growth of Francisella tularensis ssp. and other higly pathogenic bacteria. J. Microbiolol. Methods 2016, 121, 5–7. [Google Scholar] [CrossRef]
- Jenzora, A.; Jansen, A.; Ranisch, H.; Lierz, M.; Wichmann, O.; Grunow, R. Seroprevalence study of Francisella tularensis among hunters in Germany. FEMS Immunol. Med. Microbiol. 2008, 53, 183–189. [Google Scholar] [CrossRef]
- Gehringer, H.; Schacht, E.; Maylaender, N.; Zeman, E.; Kaysser, P.; Oehme, R.; Pluta, S.; Splettstoesser, W.D. Presence of an emerging subclone of Francisella tularensis holarctica in Ixodes ricinus ticks from south-western Germany. Ticks Tick Borne Dis. 2013, 4, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Müller, W.; Hotzel, H.; Otto, P.; Karger, A.; Bettin, B.; Bocklisch, H.; Braune, S.; Eskens, U.; Hormansdorfer, S.; Konrad, R.; et al. German Francisella tularensis isolates from European brown hares (Lepus europaeus) reveal genetic and phenotypic diversity. BMC Microbiol. 2013, 13, 61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaysser, P.; Seibold, E.; Matz-Rensing, K.; Pfeffer, M.; Essbauer, S.; Splettstoesser, W.D. Re-emergence of tularemia in Germany: presence of Francisella tularensis in different rodent species in endemic areas. BMC Infect. Dis. 2008, 8, 157. [Google Scholar] [CrossRef] [Green Version]
- Schulze, C.; Heuner, K.; Myrtennas, K.; Karlsson, E.; Jacob, D.; Kutzer, P.; K, G.R.; Forsman, M.; Grunow, R. High and novel genetic diversity of Francisella tularensis in Germany and indication of environmental persistence. Epidemiol. Infect. 2016, 144, 3025–3036. [Google Scholar] [CrossRef] [Green Version]
- Karlsson, E.; Golovliov, I.; Larkeryd, A.; Granberg, M.; Larsson, E.; Ohrman, C.; Niemcewicz, M.; Birdsell, D.; Wagner, D.M.; Forsman, M.; et al. Clonality of erythromycin resistance in Francisella tularensis. J. Antimicrob. Chemother. 2016, 71, 2815–2823. [Google Scholar] [CrossRef] [Green Version]
- Svensson, K.; Back, E.; Eliasson, H.; Berglund, L.; Granberg, M.; Karlsson, L.; Larsson, P.; Forsman, M.; Johansson, A. Landscape epidemiology of tularemia outbreaks in Sweden. Emerg. Infect. Dis. 2009, 15, 1937–1947. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vogler, A.J.; Birdsell, D.; Price, L.B.; Bowers, J.R.; Beckstrom-Sternberg, S.M.; Auerbach, R.K.; Beckstrom-Sternberg, J.S.; Johansson, A.; Clare, A.; Buchhagen, J.L.; et al. Phylogeography of Francisella tularensis: global expansion of a highly fit clone. J. Bacteriol. 2009, 191, 2474–2484. [Google Scholar] [CrossRef] [Green Version]
- Wittwer, M.; Altpeter, E.; Pilo, P.; Gygli, S.M.; Beuret, C.; Foucault, F.; Ackermann-Gaumann, R.; Karrer, U.; Jacob, D.; Grunow, R.; et al. Population genomics of francisella tularensis subsp. holarctica and its implication on the eco-epidemiology of tularemia in Switzerland. Front. Cell. Infect. Microbiol. 2018, 8, 89. [Google Scholar] [CrossRef]
- Kudelina, R.I. [Change in the properties of the causative agent of tularemia due to erythromycin]. Zh. Mikrobiol. Epidemiol. Immunobiol. 1973, 50, 98–101. [Google Scholar]
- Kwit, N.A.; Schwartz, A.; Kugeler, K.J.; Mead, P.S.; Nelson, C.A. Human tularaemia associated with exposure to domestic dogs-United States, 2006–2016. Zoonoses Public Health 2019, 66, 417–421. [Google Scholar] [CrossRef]
- Schmitt, P.; Splettstosser, W.; Porsch-Ozcurumez, M.; Finke, E.J.; Grunow, R. A novel screening ELISA and a confirmatory Western blot useful for diagnosis and epidemiological studies of tularemia. Epidemiol. Infect. 2005, 133, 759–766. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.rki.de/DE/Content/Kommissionen/Stakob/Stellungnahmen/Stellungnahme_Tularaemie.pdf? (accessed on 22 October 2020).
- Koskela, P.; Salminen, A. Humoral immunity against Francisella tularensis after natural infection. J. Clin. Microbiol. 1985, 22, 973–979. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wetzstein, N.; Karcher, I.; Kupper-Tetzel, C.P.; Kann, G.; Hogardt, M.; Jozsa, K.; Jacob, D.; Grunow, R.; Just-Nubling, G.; Wolf, T. Clinical characteristics in a sentinel case as well as in a cluster of tularemia patients associated with grape harvest. Int. J. Infect. Dis. 2019, 84, 116–120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lübbert, C.; Taege, C.; Seufferlein, T.; Grunow, R. [Prolonged course of tick-borne ulceroglandular tularemia in a 20-year-old patient in Germany--case report and review of the literature]. Dtsch. Med. Wochenschr. 2009, 134, 1405–1410. [Google Scholar] [CrossRef]
- Cowley, S.C.; Elkins, K.L. Immunity to Francisella. Front. Microbiol. 2011, 2, 26. [Google Scholar] [CrossRef] [Green Version]
- Telepnev, M.; Golovliov, I.; Sjostedt, A. Francisella tularensis LVS initially activates but subsequently down-regulates intracellular signaling and cytokine secretion in mouse monocytic and human peripheral blood mononuclear cells. Microb. Pathog. 2005, 38, 239–247. [Google Scholar] [CrossRef]
- Paranavitana, C.; Pittman, P.; Velauthapillai, M.; DaSilva, L. Temporal cytokine profiling of Francisella tularensis-infected human peripheral blood mononuclear cells. J. Microbiol. Immunol. Infect. 2008, 41, 192–199. [Google Scholar]
- Bolger, C.E.; Forestal, C.A.; Italo, J.K.; Benach, J.L.; Furie, M.B. The live vaccine strain of Francisella tularensis replicates in human and murine macrophages but induces only the human cells to secrete proinflammatory cytokines. J. Leukoc. Biol. 2005, 77, 893–897. [Google Scholar] [CrossRef]
- Natrajan, M.S.; Rouphael, N.; Lai, L.; Kazmin, D.; Jensen, T.L.; Weiss, D.S.; Ibegbu, C.; Sztein, M.B.; Hooper, W.F.; Hill, H.; et al. Systems vaccinology for a live attenuated tularemia vaccine reveals unique transcriptional Signatures that predict humoral and cellular immune responses. Vaccines 2019, 8. [Google Scholar] [CrossRef] [Green Version]

| Dog No/Sample Identification | Serum 1 (Day 1) | Serum 2 (Day 8) | ||
|---|---|---|---|---|
| OD | Result | OD | Result | |
| 1/A-1278 | 0.021 | negative | 0.026 | negative |
| 2/A-1279 | 0.017 | negative | 0.018 | negative |
| 3/A-1280 | 1.581 | strongly positive | 1.409 | strongly positive |
| 4/A-1281 | 0.013 | negative | 0.035 | negative |
| 5/A-1282 | 0.156 | weakly positive | 0.121 | weakly positive |
| 6/A-1283 | 1.479 | strongly positive | 1.839 | strongly positive |
| 7/A-1284 | 0.448 | weakly positive | 0.382 | weakly positive |
| 8/A-1285 | 0.014 | negative | 0.018 | negative |
| 9/A-1286 | 0.022 | negative | 0.030 | negative |
| 10/A-1301 | 0.026 | negative | Not tested | Not tested |
| Index | Date | Action | Time After Exposure (Day/Weeks) | Time After Clinical Signs (Day/Weeks) |
|---|---|---|---|---|
| 0 | 27 October 2018 | Exposure | 0 | 0 |
| 1 | 01 November 2018 | Flu-like clinical signs, hospitalization | 5/1 | 0 |
| 2 | 04 November 2018 | First set of sera | 8/1 | 3/0 |
| 3 | 08 November 2018 | Second set of sera, discharge of patients from hospital in good health conditions | 12/2 | 7/1 |
| 4 | 14 November 2018 | Third set of sera | 18/3 | 13/2 |
| 5 | April/May 2019 | Fourth set of sera | Approx. 150/21 | Approx. 150/21 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jacob, D.; Barduhn, A.; Tappe, D.; Rauch, J.; Heuner, K.; Hierhammer, D.; vom Berge, K.; Riehm, J.M.; Hanczaruk, M.; Böhm, S.; et al. Outbreak of Tularemia in a Group of Hunters in Germany in 2018—Kinetics of Antibody and Cytokine Responses. Microorganisms 2020, 8, 1645. https://doi.org/10.3390/microorganisms8111645
Jacob D, Barduhn A, Tappe D, Rauch J, Heuner K, Hierhammer D, vom Berge K, Riehm JM, Hanczaruk M, Böhm S, et al. Outbreak of Tularemia in a Group of Hunters in Germany in 2018—Kinetics of Antibody and Cytokine Responses. Microorganisms. 2020; 8(11):1645. https://doi.org/10.3390/microorganisms8111645
Chicago/Turabian StyleJacob, Daniela, Anne Barduhn, Dennis Tappe, Jessica Rauch, Klaus Heuner, Daniela Hierhammer, Katharina vom Berge, Julia M. Riehm, Matthias Hanczaruk, Stefanie Böhm, and et al. 2020. "Outbreak of Tularemia in a Group of Hunters in Germany in 2018—Kinetics of Antibody and Cytokine Responses" Microorganisms 8, no. 11: 1645. https://doi.org/10.3390/microorganisms8111645
APA StyleJacob, D., Barduhn, A., Tappe, D., Rauch, J., Heuner, K., Hierhammer, D., vom Berge, K., Riehm, J. M., Hanczaruk, M., Böhm, S., Böhmer, M. M., Konrad, R., Bouschery, B., Dauer, M., Schichtl, E., Hossain, H., & Grunow, R. (2020). Outbreak of Tularemia in a Group of Hunters in Germany in 2018—Kinetics of Antibody and Cytokine Responses. Microorganisms, 8(11), 1645. https://doi.org/10.3390/microorganisms8111645





