Microbial Resource Management for Ex Situ Biomethanation of Hydrogen at Alkaline pH
Abstract
:1. Introduction
2. Materials and Methods
2.1. Inoculum
2.2. Growth Medium
2.3. Enrichment Setup
2.4. Cultivation Experiments
2.5. Microbial Community Analysis
2.6. Analytical Methods
2.7. Statistical Analysis
3. Results and Discussion
3.1. Enrichment of the Hydrogenotrophic Community and Biomethanation Performance
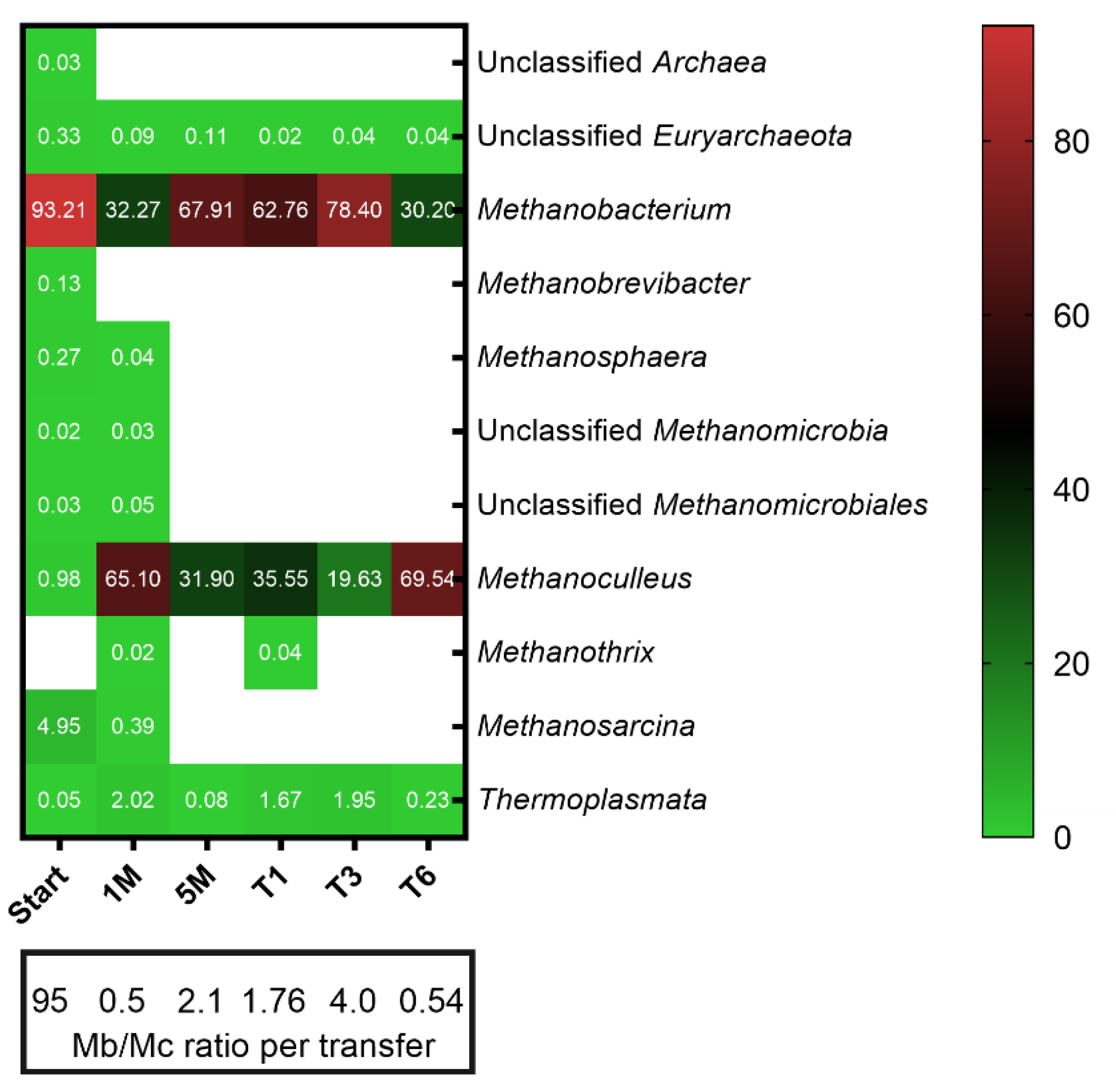
3.2. Microbial Community Structure and Dynamics
3.3. Microbial Resource Management for Selective Production of Methane
3.3.1. Effect of Medium Composition
3.3.2. Effect of Stirring Intensity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lecker, B.; Illi, L.; Lemmer, A.; Oechsner, H. Biological hydrogen methanation—A review. Bioresour. Technol. 2017, 245, 1220–1228. [Google Scholar] [CrossRef]
- Schiebahn, S.; Grube, T.; Robinius, M.; Tietze, V.; Kumar, B.; Stolten, D. Power to gas: Technological overview, systems analysis and economic assessment for a case study in Germany. Int. J. Hydrogen Energy 2015, 40, 4285–4294. [Google Scholar] [CrossRef]
- Luo, G.; Johansson, S.; Boe, K.; Xie, L.; Zhou, Q.; Angelidaki, I. Simultaneous hydrogen utilization and in situ biogas upgrading in an anaerobic reactor. Biotechnol. Bioeng. 2012, 109, 1088–1094. [Google Scholar] [CrossRef] [PubMed]
- Verbeeck, K.; Buelens, L.C.; Galvita, V.V.; Marin, G.B.; Van Geem, K.M.; Rabaey, K. Upgrading the value of anaerobic digestion via chemical production from grid injected biomethane. Energy Environ. Sci. 2018, 11, 1788–1802. [Google Scholar] [CrossRef]
- Muñoz, R.; Meier, L.; Diaz, I.; Jeison, D. A review on the state-of-the-art of physical/chemical and biological technologies for biogas upgrading. Rev. Environ. Sci. Biotechnol. 2015, 14, 727–759. [Google Scholar]
- Angelidaki, I.; Treu, L.; Tsapekos, P.; Luo, G.; Campanaro, S.; Wenzel, H.; Kougias, P.G. Biogas upgrading and utilization: Current status and perspectives. Biotechnol. Adv. 2018, 36, 452–466. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Villadsen, S.N.B.; Fosbøl, P.L.; Angelidaki, I.; Woodley, J.M.; Nielsen, L.P.; Møller, P. The Potential of Biogas; The Solution to Energy Storage. ChemSusChem 2019, 12(10), 2147–2153. [Google Scholar] [CrossRef]
- Rittmann, S.; Seifert, A.; Herwig, C. Essential prerequisites for successful bioprocess development of biological CH4 production from CO 2 and H2. Crit. Rev. Biotechnol. 2015, 35, 141–151. [Google Scholar] [CrossRef]
- Hoelzle, R.D.; Virdis, B.; Batstone, D.J. Regulation mechanisms in mixed and pure culture microbial fermentation. Biotechnol. Bioeng. 2014, 111, 2139–2154. [Google Scholar] [CrossRef]
- Kougias, P.G.; Treu, L.; Benavente, D.P.; Boe, K.; Campanaro, S.; Angelidaki, I. Ex-situ biogas upgrading and enhancement in different reactor systems. Bioresour. Technol. 2017, 225, 429–437. [Google Scholar] [CrossRef]
- Rittmann, S.K.-M.R. A Critical Assessment of Microbiological Biogas to Biomethane Upgrading Systems. In Biogas Science and Technology; Guebitz, G.M., Bauer, A., Bochmann, G., Gronauer, A., Weiss, S., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 117–135. ISBN 978-3-319-21993-6. [Google Scholar]
- Verstraete, W.; Wittebolle, L.; Heylen, K.; Vanparys, B.; de Vos, P.; van de Wiele, T.; Boon, N. Microbial Resource Management: The road to go for environmental biotechnology. Eng. Life Sci. 2007, 7, 117–126. [Google Scholar] [CrossRef]
- Mu, D.S.; Liang, Q.Y.; Wang, X.M.; Lu, D.C.; Shi, M.J.; Chen, G.J.; Du, Z.J. Metatranscriptomic and comparative genomic insights into resuscitation mechanisms during enrichment culturing. Microbiome 2018, 6, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Garcia, S.L. Mixed cultures as model communities: Hunting for ubiquitous microorganisms, their partners, and interactions. Aquat. Microb. Ecol. 2016, 77, 79–85. [Google Scholar] [CrossRef]
- Stewart, E.J. Growing unculturable bacteria. J. Bacteriol. 2012, 194, 4151–4160. [Google Scholar] [CrossRef] [Green Version]
- Kaeberlein, T.; Lewis, K.; Epstein, S.S. Quot; Microorganisms in Pure Culture in a Simulated Natural Environment. Science 2002, 296, 1127–1129. [Google Scholar] [CrossRef] [Green Version]
- Luo, G.; Angelidaki, I. Integrated biogas upgrading and hydrogen utilization in an anaerobic reactor containing enriched hydrogenotrophic methanogenic culture. Biotechnol. Bioeng. 2012, 109, 2729–2736. [Google Scholar] [CrossRef]
- Savvas, S.; Donnelly, J.; Patterson, T.P.; Dinsdale, R.; Esteves, S.R. Closed nutrient recycling via microbial catabolism in an eco-engineered self regenerating mixed anaerobic microbiome for hydrogenotrophic methanogenesis. Bioresour. Technol. 2017, 227, 93–101. [Google Scholar] [CrossRef]
- Savvas, S.; Donnelly, J.; Patterson, T.; Chong, Z.S.; Esteves, S.R. Biological methanation of CO2 in a novel biofilm plug-flow reactor: A high rate and low parasitic energy process. Appl. Energy 2017, 202, 238–247. [Google Scholar] [CrossRef]
- Guneratnam, A.J.; Ahern, E.; FitzGerald, J.A.; Jackson, S.A.; Xia, A.; Dobson, A.D.W.; Murphy, J.D. Study of the performance of a thermophilic biological methanation system. Bioresour. Technol. 2017, 225, 308–315. [Google Scholar] [CrossRef]
- Bassani, I.; Kougias, P.G.; Treu, L.; Porté, H.; Campanaro, S.; Angelidaki, I. Optimization of hydrogen dispersion in thermophilic up-flow reactors for ex situ biogas upgrading. Bioresour. Technol. 2017, 234, 310–319. [Google Scholar] [CrossRef]
- Alfaro, N.; Fdz-Polanco, M.; Fdz-Polanco, F.; Díaz, I. Evaluation of process performance, energy consumption and microbiota characterization in a ceramic membrane bioreactor for ex-situ biomethanation of H2 and CO2. Bioresour. Technol. 2018, 258, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Corbellini, V.; Kougias, P.G.; Treu, L.; Bassani, I.; Malpei, F.; Angelidaki, I. Hybrid biogas upgrading in a two-stage thermophilic reactor. Energy Convers. Manag. 2018, 168, 1–10. [Google Scholar] [CrossRef]
- Yun, Y.M.; Sung, S.; Kang, S.; Kim, M.S.; Kim, D.H. Enrichment of hydrogenotrophic methanogens by means of gas recycle and its application in biogas upgrading. Energy 2017, 135, 294–302. [Google Scholar] [CrossRef]
- Bassani, I.; Kougias, P.G.; Treu, L.; Angelidaki, I. Biogas Upgrading via Hydrogenotrophic Methanogenesis in Two-Stage Continuous Stirred Tank Reactors at Mesophilic and Thermophilic Conditions. Environ. Sci. Technol. 2015, 49, 12585–12593. [Google Scholar] [CrossRef] [PubMed]
- Rachbauer, L.; Beyer, R.; Bochmann, G.; Fuchs, W. Characteristics of adapted hydrogenotrophic community during biomethanation. Sci. Total Environ. 2017, 595, 912–919. [Google Scholar] [CrossRef]
- Luo, G.; Angelidaki, I. Co-digestion of manure and whey for in situ biogas upgrading by the addition of H2: Process performance and microbial insights. Appl. Microbiol. Biotechnol. 2013, 97, 1373–1381. [Google Scholar] [CrossRef]
- Szuhaj, M.; Ács, N.; Tengölics, R.; Bodor, A.; Rákhely, G.; Kovács, K.L.; Bagi, Z. Conversion of H2 and CO2 to CH4 and acetate in fed-batch biogas reactors by mixed biogas community: A novel route for the power-to-gas concept. Biotechnol. Biofuels 2016, 9, 102. [Google Scholar] [CrossRef] [Green Version]
- Mohd Yasin, N.H.; Maeda, T.; Hu, A.; Yu, C.P.; Wood, T.K. CO2 sequestration by methanogens in activated sludge for methane production. Appl. Energy 2015, 142, 426–434. [Google Scholar] [CrossRef]
- Rachbauer, L.; Voitl, G.; Bochmann, G.; Fuchs, W. Biological biogas upgrading capacity of a hydrogenotrophic community in a trickle-bed reactor. Appl. Energy 2016, 180, 483–490. [Google Scholar] [CrossRef]
- Díaz, I.; Pérez, C.; Alfaro, N.; Fdz-Polanco, F. A feasibility study on the bioconversion of CO2 and H2 to biomethane by gas sparging through polymeric membranes. Bioresour. Technol. 2015, 185, 246–253. [Google Scholar] [CrossRef]
- Voelklein, M.A.; Rusmanis, D.; Murphy, J.D. Biological methanation: Strategies for in-situ and ex-situ upgrading in anaerobic digestion. Appl. Energy 2019, 235, 1061–1071. [Google Scholar] [CrossRef]
- Tian, H.; Fotidis, I.A.; Mancini, E.; Treu, L.; Mahdy, A.; Ballesteros, M.; González-Fernández, C.; Angelidaki, I. Acclimation to extremely high ammonia levels in continuous biomethanation process and the associated microbial community dynamics. Bioresour. Technol. 2018, 247, 616–623. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nikolausz, M.; Walter, R.F.H.; Sträuber, H.; Liebetrau, J.; Schmidt, T.; Kleinsteuber, S.; Bratfisch, F.; Günther, U.; Richnow, H.H. Evaluation of stable isotope fingerprinting techniques for the assessment of the predominant methanogenic pathways in anaerobic digesters. Appl. Microbiol. Biotechnol. 2013, 97, 2251–2262. [Google Scholar] [CrossRef] [PubMed]
- Lv, Z.; Leite, A.F.; Harms, H.; Glaser, K.; Liebetrau, J.; Kleinsteuber, S.; Nikolausz, M. Microbial community shifts in biogas reactors upon complete or partial ammonia inhibition. Appl. Microbiol. Biotechnol. 2019, 103, 519–533. [Google Scholar] [CrossRef]
- Porsch, K.; Wirth, B.; Tóth, E.M.; Schattenberg, F.; Nikolausz, M. Characterization of wheat straw-degrading anaerobic alkali-tolerant mixed cultures from soda lake sediments by molecular and cultivation techniques. Microb. Biotechnol. 2015, 8, 801–814. [Google Scholar] [CrossRef]
- Bonk, F.; Popp, D.; Weinrich, S.; Sträuber, H.; Becker, D.; Kleinsteuber, S.; Harms, H.; Centler, F. Determination of Microbial Maintenance in Acetogenesis and Methanogenesis by Experimental and Modeling Techniques. Front. Microbiol. 2019, 10, 1–13. [Google Scholar] [CrossRef]
- Klindworth, A.; Pruesse, E.; Schweer, T.; Peplies, J.; Quast, C.; Horn, M.; Glöckner, F.O. Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res. 2013, 41, e1. [Google Scholar] [CrossRef]
- Steinberg, L.M.; Regan, J.M. Phylogenetic comparison of the methanogenic communities from an acidic, oligotrophic fen and an anaerobic digester treating municipal wastewater sludge. Appl. Environ. Microbiol. 2008, 74, 6663–6671. [Google Scholar] [CrossRef] [Green Version]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads kenkyuhi hojokin gan rinsho kenkyu jigyo. EMBnet.journal 2013, 17, 10–12. [Google Scholar] [CrossRef]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McIlroy, S.J.; Kirkegaard, R.H.; McIlroy, B.; Nierychlo, M.; Kristensen, J.M.; Karst, S.M.; Albertsen, M.; Nielsen, P.H. MiDAS 2.0: An ecosystem-specific taxonomy and online database for the organisms of wastewater treatment systems expanded for anaerobic digester groups. Database 2017, 2017, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Fish, J.A.; Chai, B.; Wang, Q.; Sun, Y.; Brown, C.T.; Tiedje, J.M.; Cole, J.R. FunGene: The functional gene pipeline and repository. Front. Microbiol. 2013, 4, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- RStudio Team. RStudio: Integrated Development for R; RStudio, Inc.: Boston, MA, USA, 2016. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016; ISBN 978-3-319-24277-4. [Google Scholar]
- Kern, T.; Theiss, J.; Röske, K.; Rother, M. Assessment of hydrogen metabolism in commercial anaerobic digesters. Appl. Microbiol. Biotechnol. 2016, 100, 4699–4710. [Google Scholar] [CrossRef]
- Shieh, J.S.; Whitman, W.B. Pathway of acetate assimilation in autotrophic and heterotrophic methanococci. J. Bacteriol. 1987, 169, 5327–5329. [Google Scholar] [CrossRef] [Green Version]
- Goyal, N.; Padhiary, M.; Karimi, I.A.; Zhou, Z. Flux measurements and maintenance energy for carbon dioxide utilization by Methanococcus maripaludis. Microb. Cell Fact. 2015, 14, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Feist, A.M.; Scholten, J.C.M.; Palsson, B.; Brockman, F.J.; Ideker, T. Modeling methanogenesis with a genome-scale metabolic reconstruction of Methanosarcina barkeri. Mol. Syst. Biol. 2006, 2, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Treu, L.; Campanaro, S.; Kougias, P.G.; Sartori, C.; Bassani, I.; Angelidaki, I. Hydrogen-fueled microbial pathways in biogas upgrading systems revealed by genome-centric metagenomics. Front. Microbiol. 2018, 9, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Jensen, M.B.; Strübing, D.; de Jonge, N.; Nielsen, J.L.; Ottosen, L.D.M.; Koch, K.; Kofoed, M.V.W. Stick or leave—Pushing methanogens to biofilm formation for ex situ biomethanation. Bioresour. Technol. 2019, 291, 121784. [Google Scholar] [CrossRef]
- Dupnock, T.L.; Deshusses, M.A. High-Performance Biogas Upgrading Using a Biotrickling Filter and Hydrogenotrophic Methanogens. Appl. Biochem. Biotechnol. 2017, 183, 488–502. [Google Scholar] [CrossRef]
- Schuchmann, K.; Müller, V. Autotrophy at the thermodynamic limit of life: A model for energy conservation in acetogenic bacteria. Nat. Rev. Microbiol. 2014, 12, 809–821. [Google Scholar] [CrossRef] [PubMed]
- Westerholm, M.; Moestedt, J.; Schnürer, A. Biogas production through syntrophic acetate oxidation and deliberate operating strategies for improved digester performance. Appl. Energy 2016, 179, 124–135. [Google Scholar] [CrossRef] [Green Version]
- Diekert, G.; Wohlfarth, G. Metabolism of homoacetogens. Antonie Van Leeuwenhoek 1994, 66, 209–221. [Google Scholar] [CrossRef] [PubMed]
- Ljungdahl, L. The Autotrophic Pathway of Acetate Synthesis in Acetogenic Bacteria. Annu. Rev. Microbiol. 1986, 40, 415–450. [Google Scholar] [CrossRef]
- Bengelsdorf, F.R.; Beck, M.H.; Erz, C.; Hoffmeister, S.; Karl, M.M.; Riegler, P.; Wirth, S.; Poehlein, A.; Weuster-Botz, D.; Dürre, P. Bacterial Anaerobic Synthesis Gas (Syngas) and CO2 + H2 Fermentation. Adv. Appl. Microbiol. 2018, 103, 143–221. [Google Scholar]
- Peng, J.; Wegner, C.E.; Bei, Q.; Liu, P.; Liesack, W. Metatranscriptomics reveals a differential temperature effect on the structural and functional organization of the anaerobic food web in rice field soil. Microbiome 2018, 6, 1–16. [Google Scholar] [CrossRef]
- Mosbæk, F.; Kjeldal, H.; Mulat, D.G.; Albertsen, M.; Ward, A.J.; Feilberg, A.; Nielsen, J.L. Identification of syntrophic acetate-oxidizing bacteria in anaerobic digesters by combined protein-based stable isotope probing and metagenomics. ISME J. 2016, 10, 2405–2418. [Google Scholar] [CrossRef] [Green Version]
- De Vrieze, J.; Boon, N.; Verstraete, W. Taking the technical microbiome into the next decade. Environ. Microbiol. 2018, 20, 1991–2000. [Google Scholar] [CrossRef]
- Stams, A.J.M.; Van Dijk, J.B.; Dijkema, C.; Plugge, C.M. Growth of syntrophic propionate-oxidizing bacteria with fumarate in the absence of methanogenic bacteria. Appl. Environ. Microbiol. 1993, 59, 1114–1119. [Google Scholar] [CrossRef] [Green Version]
- Schink, B.; Montag, D.; Keller, A.; Müller, N. Hydrogen or formate: Alternative key players in methanogenic degradation. Environ. Microbiol. Rep. 2017, 9, 189–202. [Google Scholar] [CrossRef]
- Woods, D.D. Hydrogenlyases: The synthesis of formic acid by bacteria. Biochem. J. 1936, 30, 515–51527. [Google Scholar] [PubMed]
- Peters, V.; Janssen, P.H.; Conrad, R. Transient production of formate during chemolithotrophic growth of anaerobic microorganisms on hydrogen. Curr. Microbiol. 1999, 38, 285–289. [Google Scholar] [CrossRef]
- Wu, W.M.; Hickey, R.F.; Jain, M.K.; Zeikus, J.G. Energetics and regulations of formate and hydrogen metabolism by Methanobacterium formicicum. Arch. Microbiol. 1993, 159, 57–65. [Google Scholar] [CrossRef]
- Wintsche, B.; Glaser, K.; Sträuber, H.; Centler, F.; Liebetrau, J.; Harms, H.; Kleinsteuber, S. Trace elements induce predominance among methanogenic activity in anaerobic digestion. Front. Microbiol. 2016, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Agneessens, L.M.; Ottosen, L.D.M.; Voigt, N.V.; Nielsen, J.L.; de Jonge, N.; Fischer, C.H.; Kofoed, M.V.W. In-situ biogas upgrading with pulse H2 additions: The relevance of methanogen adaption and inorganic carbon level. Bioresour. Technol. 2017, 233, 256–263. [Google Scholar] [CrossRef]
- Savvas, S.; Donnelly, J.; Patterson, T.; Chong, Z.S.; Esteves, S.R. Methanogenic capacity and robustness of hydrogenotrophic cultures based on closed nutrient recycling via microbial catabolism: Impact of temperature and microbial attachment. Bioresour. Technol. 2018, 257, 164–171. [Google Scholar] [CrossRef] [PubMed]





| Sample | Sludge b | T1 c | T2 c | T3 c | T4 c | T5 c | T6 c |
|---|---|---|---|---|---|---|---|
| Days of incubation | 167 | 56 | 27 | 22 | 24 | 28 | 40 |
| CH4 (%) a | 85.17 ± 4.5 | 86.99 ± 5.4 | 88.04 ± 3.2 | 87.48 ± 4.7 | 88.00 ± 2.3 | 87.21 ± 4.1 | 87.47 ± 2.5 |
| Biomass concentration | |||||||
| start (mg L−1) d | - | - | - | 92.2 ± 11.73 | 91.4 ± 2.56 | 90.3 ± 17.79 | 88.6 ± 10.48 |
| end (mg L−1) d | - | - | - | 609.1 ± 33.57 | 548.2 ± 30.22 | 570.7 ± 31.46 | 588.2 ± 32.42 |
| pH (start) | 8.0 ± 0.1 | 9.0 ± 0.1 | 9.0 ± 0.1 | 9.0 ± 0.1 | 9.0 ± 0.1 | 9.0 ± 0.1 | 9.0 ± 0.1 |
| pH (end) | 8.5 ± 0.2 | 8.4 ± 0.1 | 8.3 ± 0.2 | 7.9 ± 0.1 | 8.1 ± 0.1 | 8.0 ± 0.1 | 8.1 ± 0.1 |
| Acetate (mg L−1) | 123.5 ± 7.8 | 2089 ± 194 | 970.8 ± 54.6 | 1922 ± 398 | 2053 ± 156 | 886.9 ± 170.9 | 122.7 ± 20.1 |
| Propionate (mg L−1) | 0.0 ± 0.0 | 26.03 ± 2.3 | 492.1 ± 16.4 | 185.9 ± 18.3 | 188.3 ± 37.9 | 135.9 ± 3.9 | 92.15 ± 3.7 |
| Butyrate (mg L−1) | 0.0 ± 0.0 | 56.33 ± 19.2 | 0.0 ± 0.0 | 147.2 ± 13.2 | 84.04 ± 10.26 | 28.1 ± 0.84 | 25.89 ± 1.4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Logroño, W.; Popp, D.; Kleinsteuber, S.; Sträuber, H.; Harms, H.; Nikolausz, M. Microbial Resource Management for Ex Situ Biomethanation of Hydrogen at Alkaline pH. Microorganisms 2020, 8, 614. https://doi.org/10.3390/microorganisms8040614
Logroño W, Popp D, Kleinsteuber S, Sträuber H, Harms H, Nikolausz M. Microbial Resource Management for Ex Situ Biomethanation of Hydrogen at Alkaline pH. Microorganisms. 2020; 8(4):614. https://doi.org/10.3390/microorganisms8040614
Chicago/Turabian StyleLogroño, Washington, Denny Popp, Sabine Kleinsteuber, Heike Sträuber, Hauke Harms, and Marcell Nikolausz. 2020. "Microbial Resource Management for Ex Situ Biomethanation of Hydrogen at Alkaline pH" Microorganisms 8, no. 4: 614. https://doi.org/10.3390/microorganisms8040614
APA StyleLogroño, W., Popp, D., Kleinsteuber, S., Sträuber, H., Harms, H., & Nikolausz, M. (2020). Microbial Resource Management for Ex Situ Biomethanation of Hydrogen at Alkaline pH. Microorganisms, 8(4), 614. https://doi.org/10.3390/microorganisms8040614







