Thermotolerant and Thermophilic Mycobiota in Different Steps of Compost Maturation
Abstract
1. Introduction
2. Materials and Methods
2.1. Composting Procedure and Sampling
2.2. Fungal Isolation
2.3. Molecular Identification
2.4. Data Analysis
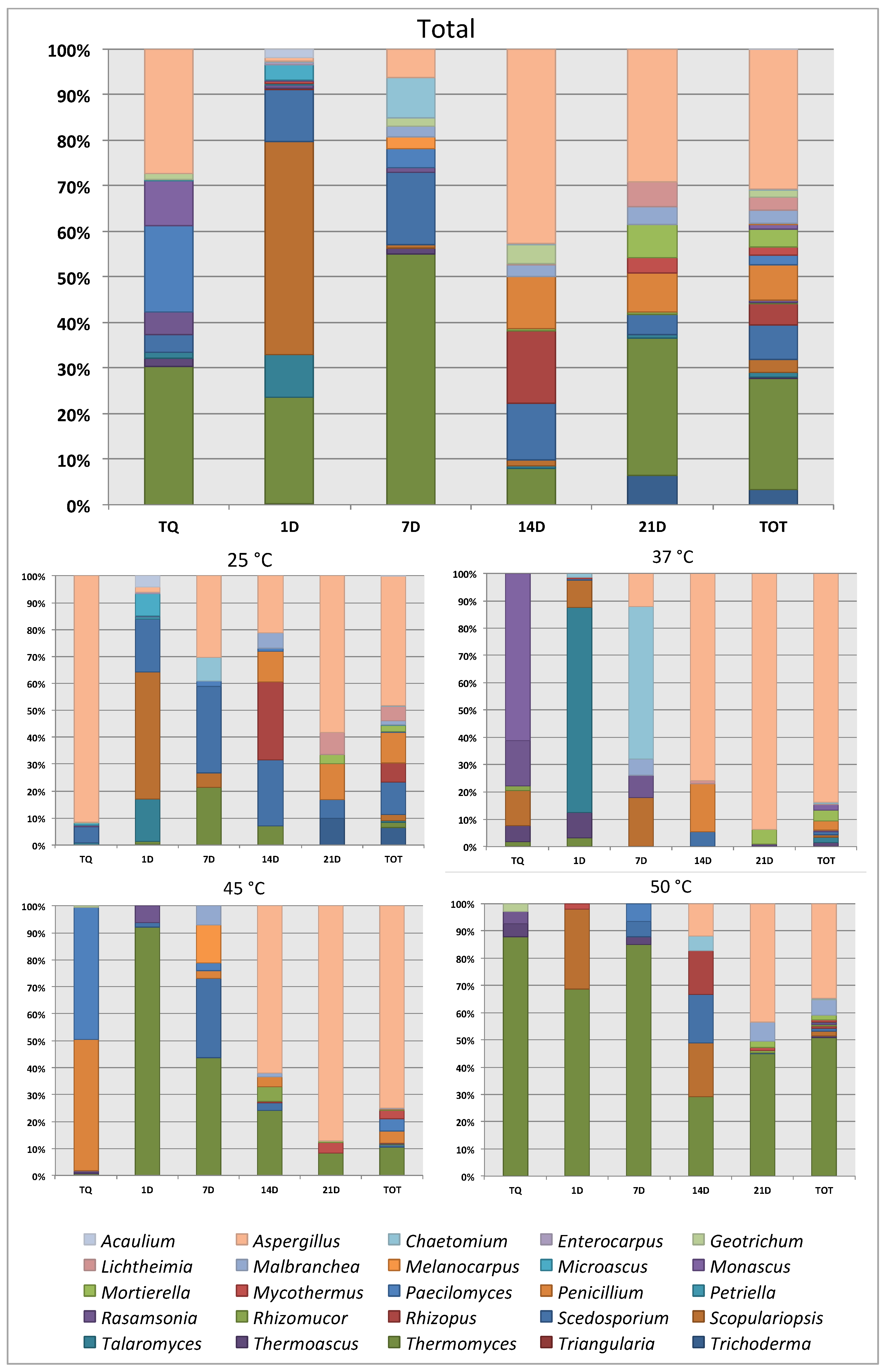
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cerda, A.; Artola, A.; Font, X.; Barrena, R.; Gea, T.; Sánchez, A. Composting of food wastes: Status and challenges. Bioresour. Technol. 2018, 248, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Torres, M.; Oviedo-Ocaña, E.R.; Dominguez, I.; Komilis, D.; Sánchez, A. A systematic review on the composting of green waste: Feedstock quality and optimization strategies. Waste Manag. 2018, 77, 486–499. [Google Scholar] [CrossRef] [PubMed]
- Partanen, P.; Hultman, J.; Paulin, L.; Auvinen, P.; Romantschuk, M. Bacterial diversity at different stages of the composting process. BMC Microbiol. 2010, 10, 94. [Google Scholar] [CrossRef]
- Wei, H.; Tucker, M.P.; Baker, J.O.; Harris, M.; Luo, Y.; Xu, Q.; Himmel, M.E.; Ding, S.Y. Tracking dynamics of plant biomass composting by changes in substrate structure, microbial community, and enzyme activity. Biotechnol. Biofuels 2012, 5, 20. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Li, M.; Jia, X.; Wei, Z.; Zhao, Y.; Xi, B.; Zhu, C.; Liu, D. Comparison of bacterial community structure and dynamics during the thermophilic composting of different types of solid wastes: Anaerobic digestion residue, pig manure and chicken manure. Microb. Biotechnol. 2014, 7, 424–433. [Google Scholar] [CrossRef]
- Onwosi, C.O.; Igbokwe, V.C.; Odimba, J.N.; Eke, I.E.; Nwankwoala, M.O.; Iroh, I.N.; Ezeogu, L.I. Composting technology in waste stabilization: On the methods, challenges and future prospects. J. Environ. Manag. 2017, 190, 140–157. [Google Scholar] [CrossRef]
- Liu, L.; Wang, S.; Guo, X.; Zhao, T.; Zhang, B. Succession and diversity of microorganisms and their association with physicochemical properties during green waste thermophilic composting. Waste Manag. 2018, 73, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Etesami, H.; Hemati, A.; Alikhani, H.A. Microbial Bioconversion of Agricultural Wastes for Rural Sanitation and Soil Carbon Enrichment. In Microbial Interventions in Agriculture and Environment; Singh, D., Gupta, V., Prabha, R., Eds.; Springer: Singapore, 2019; Volume 2, pp. 179–204. [Google Scholar]
- Wang, C.; Dong, D.; Wang, H.; Müller, K.; Qin, Y.; Wang, H.; Wu, W. Metagenomic analysis of microbial consortia enriched from compost: New insights into the role of Actinobacteria in lignocellulose decomposition. Biotechnol. Biofuels 2016, 9, 22. [Google Scholar] [CrossRef]
- Wang, X.; Pan, S.; Zhang, Z.; Lin, X.; Zhang, Y.; Chen, S. Effects of the feeding ratio of food waste on fed-batch aerobic composting and its microbial community. Bioresour. Technol. 2017, 224, 397–404. [Google Scholar] [CrossRef]
- Franke-Whittle, I.H.; Klammer, S.H.; Insam, H. Design and application of an oligonucleotide microarray for the investigation of compost microbial communities. J. Microbiol. Methods 2005, 62, 37–56. [Google Scholar] [CrossRef]
- Franceschini, S.; Chitarra, W.; Pugliese, M.; Gisi, U.; Garibaldi, A.; Gullino, M.L. Quantification of Aspergillus fumigatus and enteric bacteria in European compost and biochar. Compost Sci. Util. 2016, 24, 20–29. [Google Scholar] [CrossRef]
- Fuchs, J.G. Interactions between Beneficial and Harmful Microorganisms: From the Composting Process to Compost Application. In Microbes at Work from Wastes to Resources; Insam, H., Franke-Whittle, I., Goberna, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 213–229. [Google Scholar]
- Bonito, G.; Isikhuemhen, O.S.; Vilgalys, R. Identification of fungi associated with municipal compost using DNA-based techniques. Bioresour. Technol. 2010, 101, 1021–1027. [Google Scholar] [CrossRef] [PubMed]
- Antunes, L.P.; Martins, L.F.; Pereira, R.V.; Thomas, A.M.; Barbosa, D.; Lemos, L.N.; Silva, G.M.M.; Moura, L.M.S.; Epamino, G.W.C.; Digiampietri, L.A.; et al. Microbial community structure and dynamics in thermophilic composting viewed through metagenomics and metatranscriptomics. Sci. Rep. 2016, 6, 1–13. [Google Scholar] [CrossRef]
- Tian, X.; Yang, T.; He, J.; Chu, Q.; Jia, X.; Huang, J. Fungal community and cellulose-degrading genes in the composting process of Chinese medicinal herbal residues. Bioresour. Technol. 2017, 241, 374–383. [Google Scholar] [CrossRef]
- Anastasi, A.; Varese, G.C.; Marchisio, V.F. Isolation and identification of fungal communities in compost and vermicompost. Mycologia 2005, 97, 33–44. [Google Scholar] [CrossRef]
- Langarica-Fuentes, A.; Handley, P.S.; Houlden, A.; Fox, G.; Robson, G.D. An investigation of the biodiversity of thermophilic and thermotolerant fungal species in composts using culture-based and molecular techniques. Fungal Ecol. 2014, 11, 132–144. [Google Scholar] [CrossRef]
- López-González, J.A.; del Carmen Vargas-García, M.; López, M.J.; Suárez-Estrella, F.; del Mar Jurado, M.; Moreno, J. Biodiversity and succession of mycobiota associated to agricultural lignocellulosic waste-based composting. Bioresour. Technol. 2015, 187, 305–313. [Google Scholar] [CrossRef]
- Haas, D.; Lesch, S.; Buzina, W.; Galler, H.; Gutschi, A.M.; Habib, J.; Pfeifer, B.; Luxner, J.; Reinthaler, F.F. Culturable fungi in potting soils and compost. Med. Mycol. 2016, 54, 825–834. [Google Scholar] [CrossRef]
- Kane, E.; Mullins, J.T. Thermophilic Fungi in a Municipal Waste Compost System. Mycologia 1973, 65, 1087–1100. [Google Scholar] [CrossRef]
- Laich, F.; Andrade, J. Penicillium pedernalense sp. nov., isolated from whiteleg shrimp heads waste compost. Int. J. Syst. Evol. Microb. 2016, 66, 4382–4388. [Google Scholar] [CrossRef]
- Thanh, V.N.; Thuy, N.T.; Huong, H.T.T.; Hien, D.D.; Hang, D.T.M.; Anh, D.T.K.; Hüttner, S.; Larsbrink, J.; Olsson, L. Surveying of acid-tolerant thermophilic lignocellulolytic fungi in Vietnam reveals surprisingly high genetic diversity. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef]
- Zhang, L.; Sun, X. Addition of fish pond sediment and rock phosphate enhances the composting of green waste. Bioresour. Technol. 2017, 233, 116–126. [Google Scholar] [CrossRef]
- Atif, K.; Haouas, A.; Aziz, F.; Jamali, M.Y.; Tallou, A.; Amir, S. Pathogens Evolution During the Composting of the Household Waste Mixture Enriched with Phosphate Residues and Olive Oil Mill Wastewater. Waste Biomass Valori. 2020, 11, 1789–1797. [Google Scholar] [CrossRef]
- Gams, W.; Aa, H.A.; van der Plaats-Niterink, A.J.; Samson, R.A.; Stalpers, J.A. CBS Course of Mycology, 3rd ed.; Centraalbureau voor Schimmelcultures: Baarn, The Netherlands, 1987; p. 136. [Google Scholar]
- Samson, R.A.; Houbraken, J.; Thrane, U.; Frisvad, J.C.; Andersen, B. Food and Indoor Fungi. CBS Laboratory Manual Series; Westerdijk Fungal Biodiversity Institute: Utrecht, The Netherlands, 2010; p. 390. [Google Scholar]
- Shannon, C.E. A mathematical theory of communication. Bell Syst. Tech. J. 1948, 27, 623–656. [Google Scholar] [CrossRef]
- Magurran, A.E. Measuring Biological Diversity; Blackwell: Oxford, UK, 2004; p. 230. [Google Scholar]
- Mueller, G.M.; Foster, M.; Bills, G.F. Biodiversity of Fungi Inventory and Monitoring Methods; Academic Press: Burlington, MA, USA, 2004; p. 777. [Google Scholar]
- Jaccard, P. Nouvelles recherches sur la distribution florale. Bull. Soc. Vaud. Sci. Nat. 1908, 44, 223–270. [Google Scholar]
- Houbraken, J.; de Vries, R.P.; Samson, R.A. Modern taxonomy of biotechnologically important Aspergillus and Penicillium species. Adv. Appl. Microbiol. 2014, 86, 199–249. [Google Scholar]
| Samples | ||||||
|---|---|---|---|---|---|---|
| TQ | 1 | 7 | 14 | 21 | ||
| Incubation Temperature | 25 °C | |||||
| 37 °C | ||||||
| 45 °C | ||||||
| 50 °C | ||||||
| Scale bar ×104 CFU g−1 | |||||
| 1 | 30 | ||||
| Samples | GenBank Accession Numbers | ||||||
|---|---|---|---|---|---|---|---|
| Species | TQ | 1D | 7D | 14D | 21D | ITS Barcode | Secondary Barcode |
| Acaulium acremonium | MT316336 | MT312849, MT433447 | |||||
| Aspergillus chevalieri | MT316337 | MT420413 | |||||
| Aspergillus fumigatus | MT316338 | MT420414 | |||||
| Aspergillus nidulans | MT316339 | MT420415 | |||||
| Aspergillus niger | MT316340 | MT420412 | |||||
| Aspergillus pseudoglaucus | MT316341 | MT420416 | |||||
| Aspergillus quadrilineatus | MT316342 | MT420417 | |||||
| Aspergillus terreus | MT316343 | MT420418 | |||||
| Aspergillus sp. | MT316344 | MT420419 | |||||
| Chaetomium thermophilum | MT316345 | MT433448 | |||||
| Chaetomium uniapiculatum | MT316346 | MT433449 | |||||
| Enterocarpus grenotii | MT316347 | MT312850 | |||||
| Geotrichum sp. | MT316348 | MT312851 | |||||
| Lichtheimia corymbifera | MT316349 | MT312852 | |||||
| Malbranchea cinnamomea | MT316350 | MT312853 | |||||
| Melanocarpus albomyces | MT316351 | MT433450 | |||||
| Microascus paisii | MT316352 | MT433451 | |||||
| Microascus restrictus | MT316353 | MT433452 | |||||
| Monascus ruber | MT316354 | MT433453 | |||||
| Mortierella wolfii | MT316355 | MT312854 | |||||
| Mycothermus thermophilus | MT316356 | MT433454 | |||||
| Paecilomyces variotii | MT316357 | MT433455, MT312848 | |||||
| Penicillium crustosum | MT316358 | MT433456 | |||||
| Penicillium sp. 1 | MT316359 | - | |||||
| Penicillium sp. 2 | MT316360 | - | |||||
| Petriella sp. | MT316361 | MT433457 | |||||
| Rasamsonia emersonii | MT316362 | MT433458 | |||||
| Rasamsonia sp. | MT316363 | MT433459 | |||||
| Rhizomucor miehei | MT316364 | MT312855 | |||||
| Rhizopus microsporus | MT316365 | MT312856 | |||||
| Rhizopus oryzae | MT316366 | MT312857 | |||||
| Scedosporium apiospermum | MT316367 | MT433460 | |||||
| Scedosporium aurantiacum | MT316368 | MT433461 | |||||
| Scedosporium dehoogii | MT316369 | MT433462 | |||||
| Scedosporium minutisporum | MT316370 | MT433463 | |||||
| Scedosporium prolificans | MT316371 | MT433464 | |||||
| Scopulariopsis brevicaulis | MT316372 | MT433465, MT420423 | |||||
| Talaromyces trachyspermus | MT316373 | MT433466 | |||||
| Talaromyces tratensis | MT316374 | MT420420 | |||||
| Talaromyces wortmannii | MT316375 | MT420421 | |||||
| Thermoascus aurantiacus | MT316376 | MT433467, MT420422 | |||||
| Thermomyces dupontii | MT316377 | MT433468 | |||||
| Thermomyces lanuginosus | MT316378 | MT433469 | |||||
| Triangularia sp. | MT316379 | MT433470 | |||||
| Trichoderma longibrachiatum | MT316380 | MT420424 | |||||
| Scale bar ×104 CFU g−1 | ||||||||||
| 0 | 10 | |||||||||
| TQ | 1D | 7D | 14D | 21D | ||
|---|---|---|---|---|---|---|
| 25 °C | H’ | 0.69 | 2.56 | 2.83 | 2.91 | 2.70 |
| E | 0.22 | 0.67 | 0.89 | 0.84 | 0.90 | |
| 37 °C | H’ | 1.69 | 1.41 | 2.21 | 1.96 | 2.14 |
| E | 0.65 | 0.44 | 0.74 | 0.70 | 0.76 | |
| 45 °C | H’ | 0.33 | 1.15 | 2.12 | 1.61 | 1.96 |
| E | 0.14 | 0.58 | 0.82 | 0.57 | 0.70 | |
| 50 °C | H’ | 0.72 | 1.45 | 1.12 | 2.82 | 1.77 |
| E | 0.36 | 0.72 | 0.48 | 0.94 | 0.53 | |
| TOT | H’ | 2.60 | 2.67 | 2.65 | 3.39 | 3.44 |
| E | 0.67 | 0.62 | 0.65 | 0.77 | 0.84 |
| TQ | 1D | 7D | 14D | 21D | |
|---|---|---|---|---|---|
| TQ | 1 | 0.25 | 0.45 | 0.24 | 0.19 |
| 1D | 1 | 0.28 | 0.24 | 0.16 | |
| 7D | 1 | 0.36 | 0.21 | ||
| 14D | 1 | 0.46 | |||
| 21D | 1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Piazza, S.; Houbraken, J.; Meijer, M.; Cecchi, G.; Kraak, B.; Rosa, E.; Zotti, M. Thermotolerant and Thermophilic Mycobiota in Different Steps of Compost Maturation. Microorganisms 2020, 8, 880. https://doi.org/10.3390/microorganisms8060880
Di Piazza S, Houbraken J, Meijer M, Cecchi G, Kraak B, Rosa E, Zotti M. Thermotolerant and Thermophilic Mycobiota in Different Steps of Compost Maturation. Microorganisms. 2020; 8(6):880. https://doi.org/10.3390/microorganisms8060880
Chicago/Turabian StyleDi Piazza, Simone, Jos Houbraken, Martin Meijer, Grazia Cecchi, Bart Kraak, Ester Rosa, and Mirca Zotti. 2020. "Thermotolerant and Thermophilic Mycobiota in Different Steps of Compost Maturation" Microorganisms 8, no. 6: 880. https://doi.org/10.3390/microorganisms8060880
APA StyleDi Piazza, S., Houbraken, J., Meijer, M., Cecchi, G., Kraak, B., Rosa, E., & Zotti, M. (2020). Thermotolerant and Thermophilic Mycobiota in Different Steps of Compost Maturation. Microorganisms, 8(6), 880. https://doi.org/10.3390/microorganisms8060880






