1. Introduction
Infertility and its associated issues is a major problem faced by couples [
1]. The most common reason for infertility in men is the inability to produce a sufficient number of active and healthy sperms [
2]. Numerous aspects can affect sperm development, including the use of antibiotics, toxins, chemotherapy drugs for cancer, pesticides, air pollution, radiation, stress, and inadequate vitamins. Agarwal et al. [
3] found that the causes noted decrease sperm levels by creating free radicals and oxidizing germ cells in the testicular tissue. Cisplatin (Cis) is a chemotherapeutic drug, a DNA-alkylating agent that exercises its anti-tumor function by prompting DNA crosslinks and DNA double-strand breaks; both destroy DNA transcription and replication, causing programmed cell death/apoptosis [
4]. Moreover, Fallahzadeh et al. [
5] stated that Cis triggers oxidative stress by generating reactive oxygen species (ROS) that stimulates cell destruction and necrosis through lipid peroxidation of tissues, protein denaturation, and DNA lesions.
Cisplatin (Cis) is one of the most widely used antitumor medications for the treatment of solid tumors, such as head and neck, testis, lung, ovarian, bladder cancer, and hematological malignancies [
6], However, Cis’ use in clinical commitments is limited because of its side-effects, such as neurotoxicity, hepatotoxicity, and even testicular toxicity [
5].
Cherry et al. [
7] found that there was impaired fertility and decay in reproductive organ weights along with modifications in the growth and advancement of following generations in male rats treated with Cisp [
7]. Both endocrine and exocrine compartments have an imbalance in spermatogenesis, gonadal dysfunction, androgenesis [
8] and also prevent testosterone secretion by damage of Leydig cells. Cis decreases sperm motility and normal morphology. Chromosomal abnormalities in spermatozoa and azoospermia are related to the side-effects of Cis treatment. These side-effects are consigned to nitrosative and oxidative damage produced by Cis [
9]. The physiological disturbance is due to increased lipid peroxidation and redox imbalance contributes to the apoptosis of the germ cells [
10].
Recent studies have shown the antioxidant function of stem cells and their application to restore the damaging effects of ROS tissues [
11]. Mesenchymal stem cells (MSCs) are multipotent stem cells originating from other tissues, such as the cord blood, adipose tissue, bone marrow, and amniotic membrane. MSCs are common in mature organisms and might be involved in tissue maintenance and repair, as well as in the regulation of immunological responses, function restoration, and homeostasis [
12]. Squillaro et al. [
13] stated that, in addition to chondrogenic, osteogenic, and adipogenic differentiation, MSCs are characterized by unique surface antigen expression; they are also able to self-renew themselves. Beside, stem cells will proliferate and create the daughter cell lines for the development of tissues [
14]. The bone marrow is the source of MSCs from which to receive other tissues. Adult MSCs’ ability to ‘transdifferentiate’ could reform regenerative medicine [
14]. Current studies have revealed the capacity of both embryonic and adult stem cells to distinguish into primordial germ cells and mature gametes. After the sixth day of birth, primordial germ cells (PGCs), the origin of male germ cells derived from the proximal epiblast, migrate to the seminiferous tubules’ basement membrane and become spermatogonial stem cells (SSCs) [
15]. SSCs are responsible for sustaining the mechanism of spermatogenesis during its lifespan; the correct diagnosis of impaired spermatogenesis is a major stage in investigating male infertility. Nayernia et al. [
15] proved that bone marrow stem cells are capable of distinguishing in vitro and in vivo between primordial germ cells and spermatogonia. Also, another study has confirmed that adult stem cells derived from the stroma bone marrow can differentiate into Leydig cells in rat testes [
16].
Red beetroot (
Beta vulgaris), a naturally occurring root vegetable, has numerous attentions as a health promoting functional food [
17]. Beetroot regularly used up as part of the normal diet as a salad or fresh juice, and is used in the food industry as a food coloring agent recognized as E162 [
18]. Recent studies have given convincing evidence that the ingestion of beetroot has beneficial physiological effects that could lead to enhanced clinical outcomes for many pathologies, such as hypertension, dementia, type II diabetes, and atherosclerosis. It is a rich source of phytochemical compounds that involves carotenoids, ascorbic acid, flavonoids, phenolic acids, and betalains [
19]. Clifford et al. [
20] well established the high antioxidant, anti-inflammatory, and anti-carcinogenic properties of betalains. Besides, beetroot has been reported to be mainly associated with human sex hormone enhancement as an aphrodisiac and also improves sexual weakness [
21].
The goal of this work was to study the influence of mesenchymal stem cells and beetroot on improving testicular damage that cisplatin induced in rats.
4. Discussion
Dysfunction or testis damage are the serious side-effects of chemotherapy drugs such as cisplatin. In the present study, we estimate the sperm count, several biochemical, hormonal, histological, and gene expression parameters related to testicular toxicity and oxidative stress in the testis tissue to evaluate the protective effect of BM-MSCs and beetroot against Cis-induced reproductive toxicity in rats. Cis treatments cause cytotoxicity as ROS formation, DNA damage leading to germ epithelial damage, sperm dysfunction, and germ cells apoptosis. It also leads to inflammation [
7].
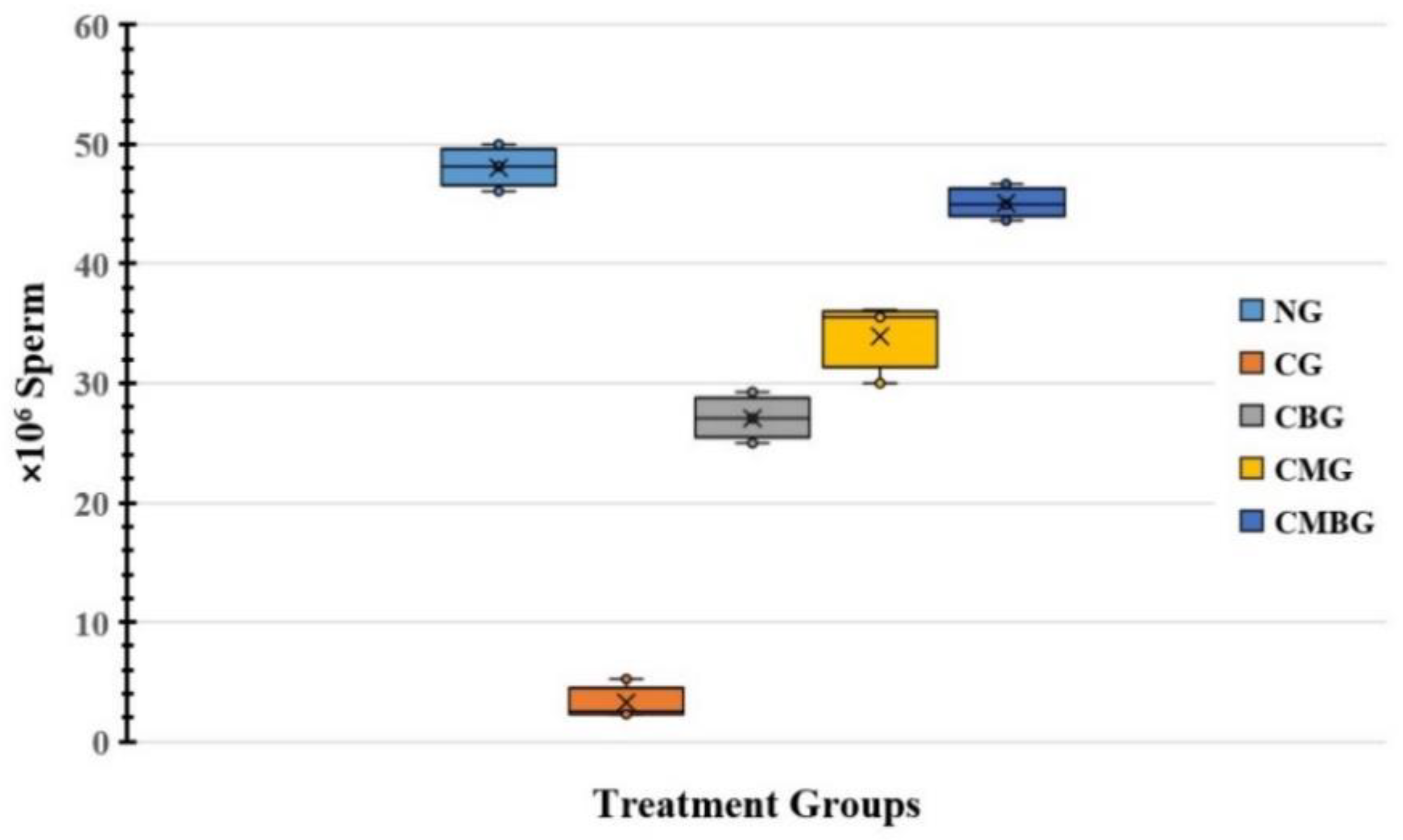
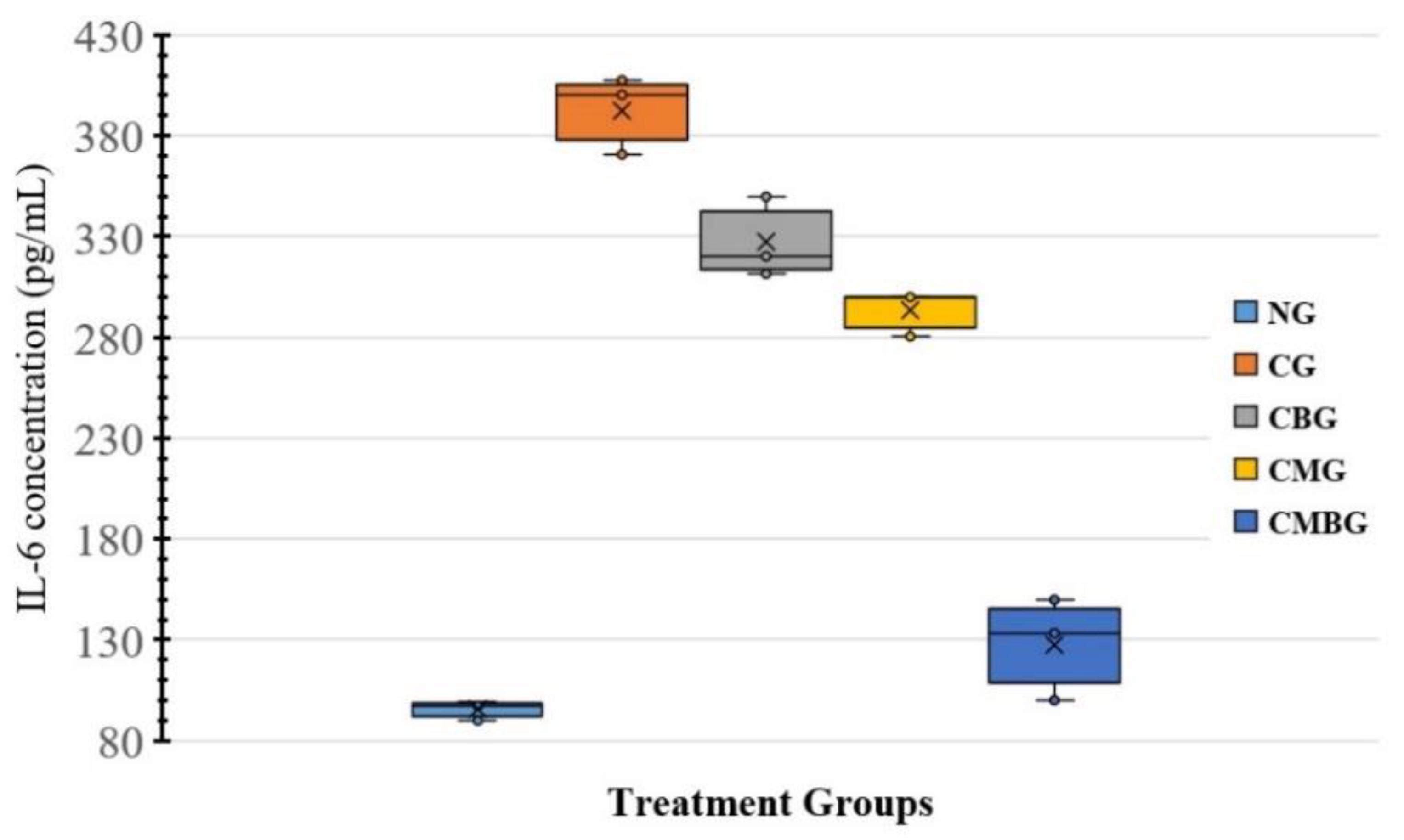
In the present investigation, Cis administration contributed to a drop in sperm count as a result of cisplatin toxic side-effects, resulting in a reduced testicular activity. The free radical development and decreasing cisplatin-induced antioxidant enzymes cause rapid loss of intracellular adenosine triphosphate (ATP), leading to loss of sperm motility and reduced sperm viability [
30]. In addition, reducing sperm motility and increasing abnormal sperm rate in rats treated with cisplatin could be caused by lipid peroxidation of unsaturated fatty acids in the sperm plasma membrane, resulting in loss of fluidity and function [
31].
Following the treatment of the Cis group with BM-MSCs and beetroot extract, there was a statistically significant improvement in sperm count. These findings were in accordance with Dillasamola et al. [
32], who found that beetroot extract restores natural sperm count and motility after xenobiotic toxicity. Beetroot’s mechanism of action can serve as a useful approach to reinforce endogenous antioxidant defenses and protect cellular constituents from xenobiotics [
33].
BM-MSCs are confirmed to have been used in experimental rodent infertility models treated with busulfan [
16]. In the current work, the MSC group was correlated with significantly higher sperm compared to the rat group treated with Cis. Hence, it could be concluded that MSCs might have a potential role in treating male infertility and testosterone deficiency. It could also be suggested that the beneficial effects of MSCs may be due to differentiation in male germ cells, as stated by Nayernia et al. [
15]. Yazawa et al. [
16] established that MSCs are able to differentiate into steroidogenic cells, such as Leydig cells, in vivo as well as in vitro.
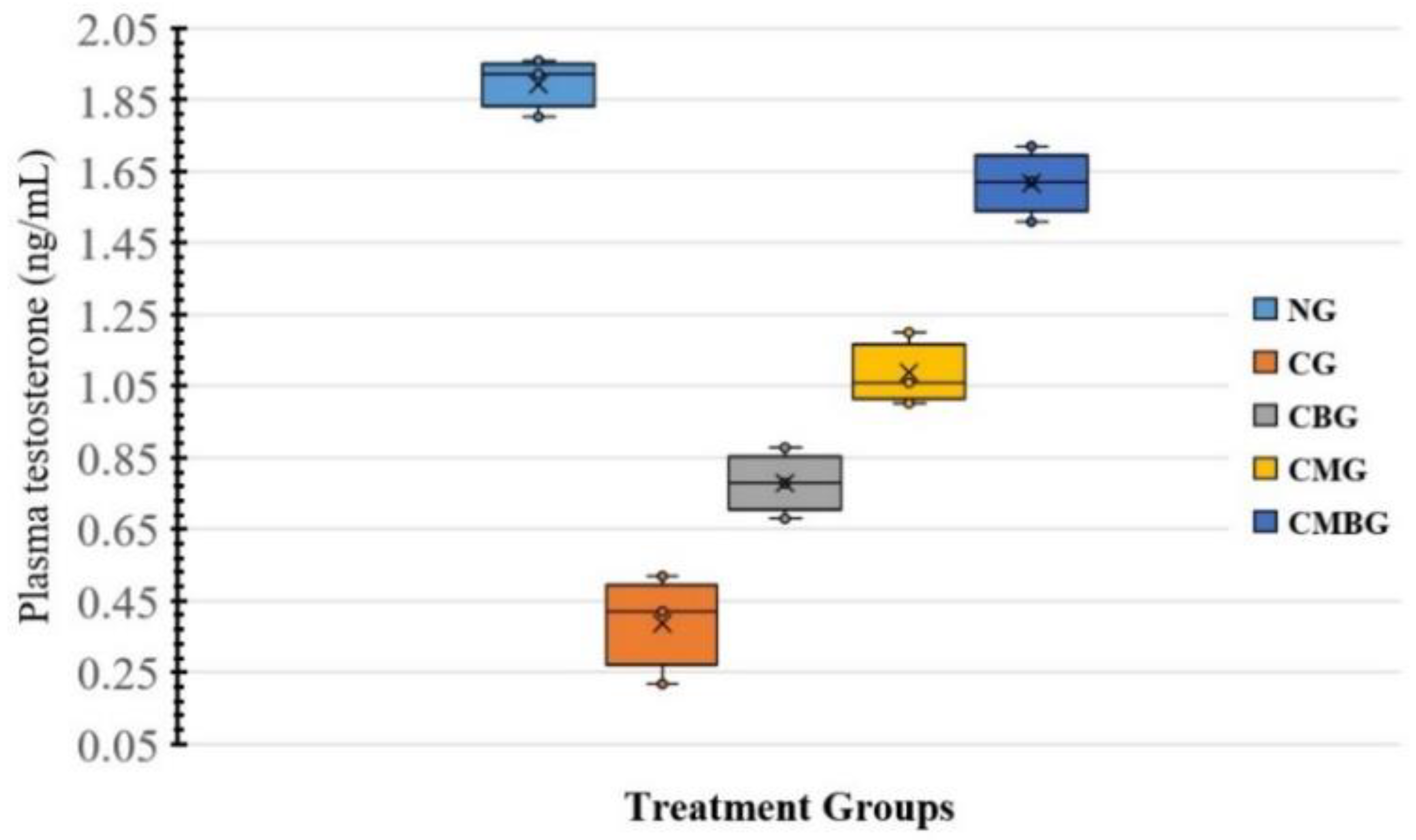
Testosterone plays an important role in controlling spermatogenesis. Concurrent tests after cisplatin administration indicated a substantial decrease in serum testosterone levels. Recent research showed that cisplatin harms the testis with decreased testosterone levels [
5]. This remarkable reduction in hormonal level may be explained by the serious damage caused by cisplatin on Leydig and Sertoli cells as one of the possible mechanisms resulting from the increased generation of free radicals [
34]. Moreover, several researchers have reported that interference with the expression of the Luteinizing Hormone (LH) receptor and cholesterol mobilization inhibition to mitochondrial cytochrome P450 interferes with the initial steps in testosterone production. Such results are consistent with those suggested by [
34].
The administration of BM-MSCs and beetroot extract to Cis-treated animals led to significant increase in serum testosterone concentration, which corresponded to Mohammadi et al. [
35], who informed that improvement in testosterone level achieved by beetroot extract was due to its antioxidant activity. Regarding BM-MSCs, this was used as a therapeutic tool and elevated levels of testosterone, leading to the transformation of MSCs into steroid cells as Leydig cells containing testosterone [
14].
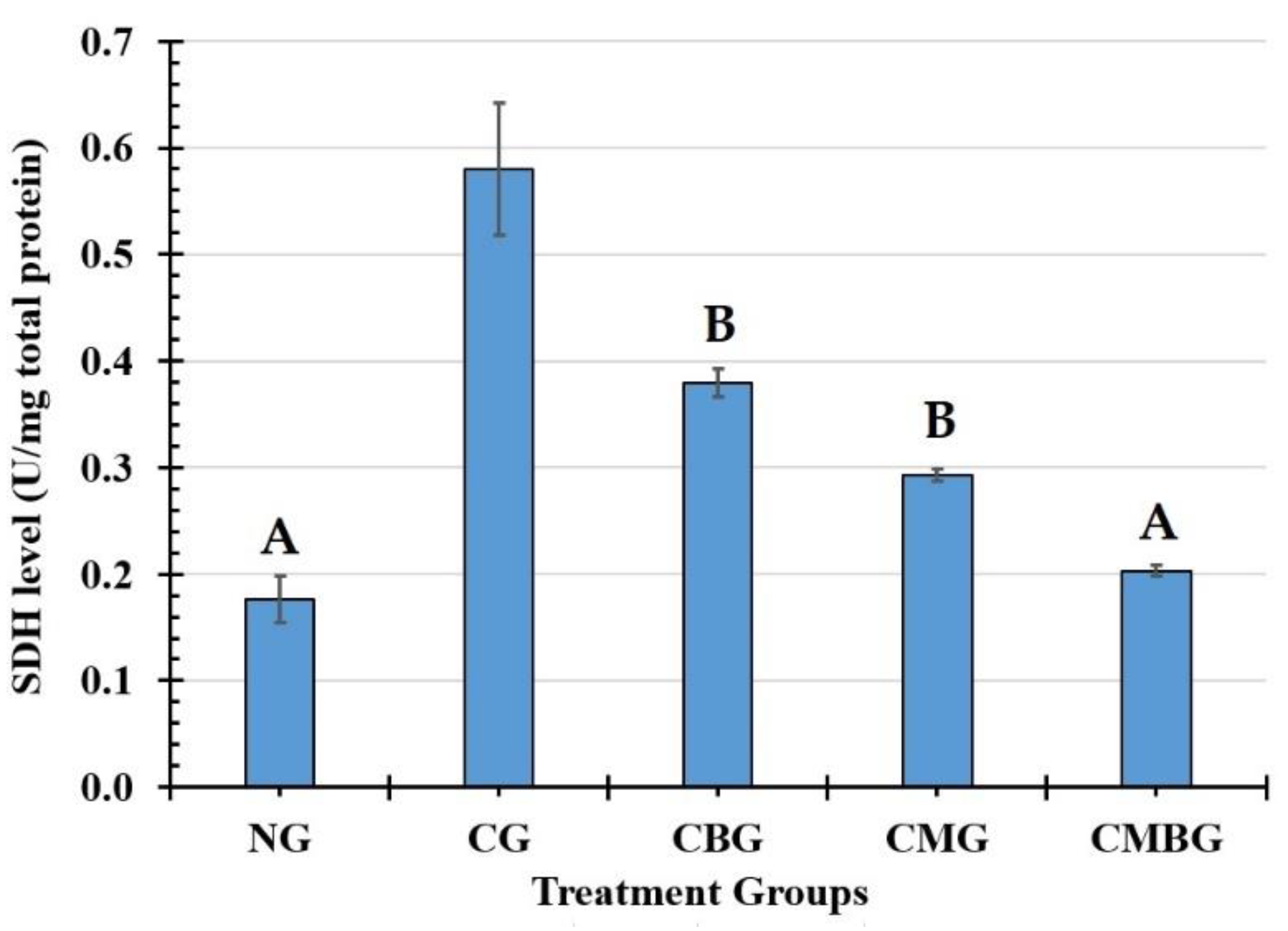
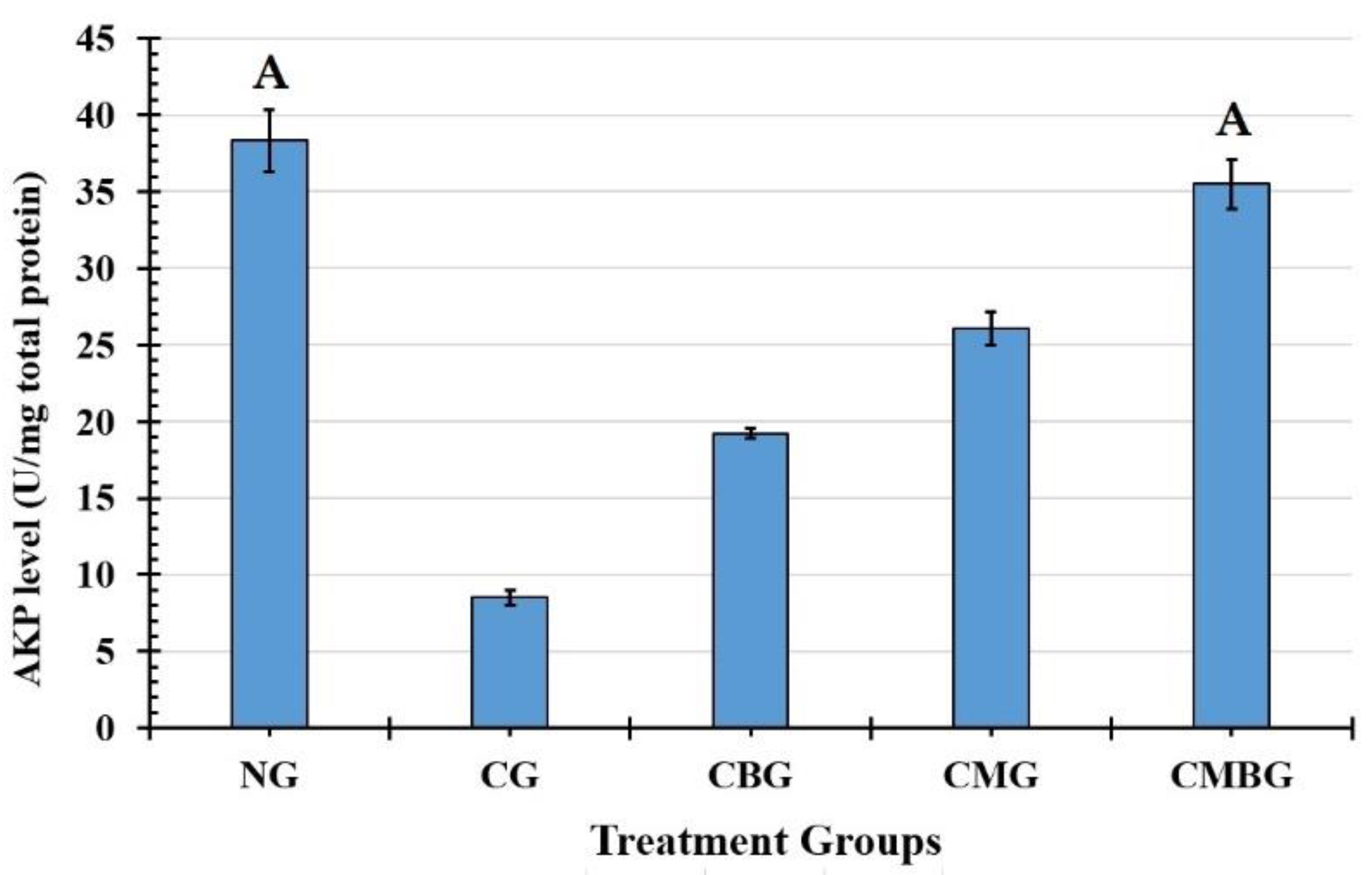
In the testis, AKP is associated with sperm cell division and glucose transfer to spermatogenic cells and SDH, biomarkers of the testicular enzyme that relate to sperm metabolism. SDH is widely distributed and found in germ cells and seminiferous tubules, which relate to sperm cell maturation, testis, and spermatozoa, and the spermatozoa energy metabolism [
36]. The present data shows the levels of testicular AKP and SDH significantly decreased or increased in the Cis groups, which indicates that Cis may influence sperm cell energy metabolism. In the current research, co-administration with BM-MSCs and beetroot extract to the Cis group induced improvement in the function of those enzymes.
Cytokines perform important roles in normal cell physiology. These are linked to or healed by inflammation, immune response, and tissue damage. Cisplatin stimulated inflammatory cells and consequently magnified the inflammatory response by releasing various cytokines such as TNF and IL-6, resulting in damage to the testes. In this study, the treatment of rats with BM-MSCs combined with beetroot significantly reduced serum levels of IL-6. Therefore, it may be recommended that BM-MSCs with beetroot may relieve cisplatin-caused testis injury by suppressing the inflammatory response. In addition, these results were in agreement with Winkler et al. [
37] who reported beetroot extract to be capable of counteracting proinflammatory cascades in the periphery mononuclear blood cells [
37]. Due to the strong role of inflammation in the development and progression of many clinical conditions, including coronary heart disease and cancer, the beneficial effect of beetroot extract may be associated with this anti-inflammatory capability [
38]. Therefore, BM-MSCs with beetroot may be suggested to mitigate cisplatin-caused injury to the testis by suppressing the inflammatory response. Albasher et al. [
39] mentioned that beetroot treatment reduced TNF-α and IL-6 in renal tissue exposed to gentamicin by the inactivation of NF-κB.
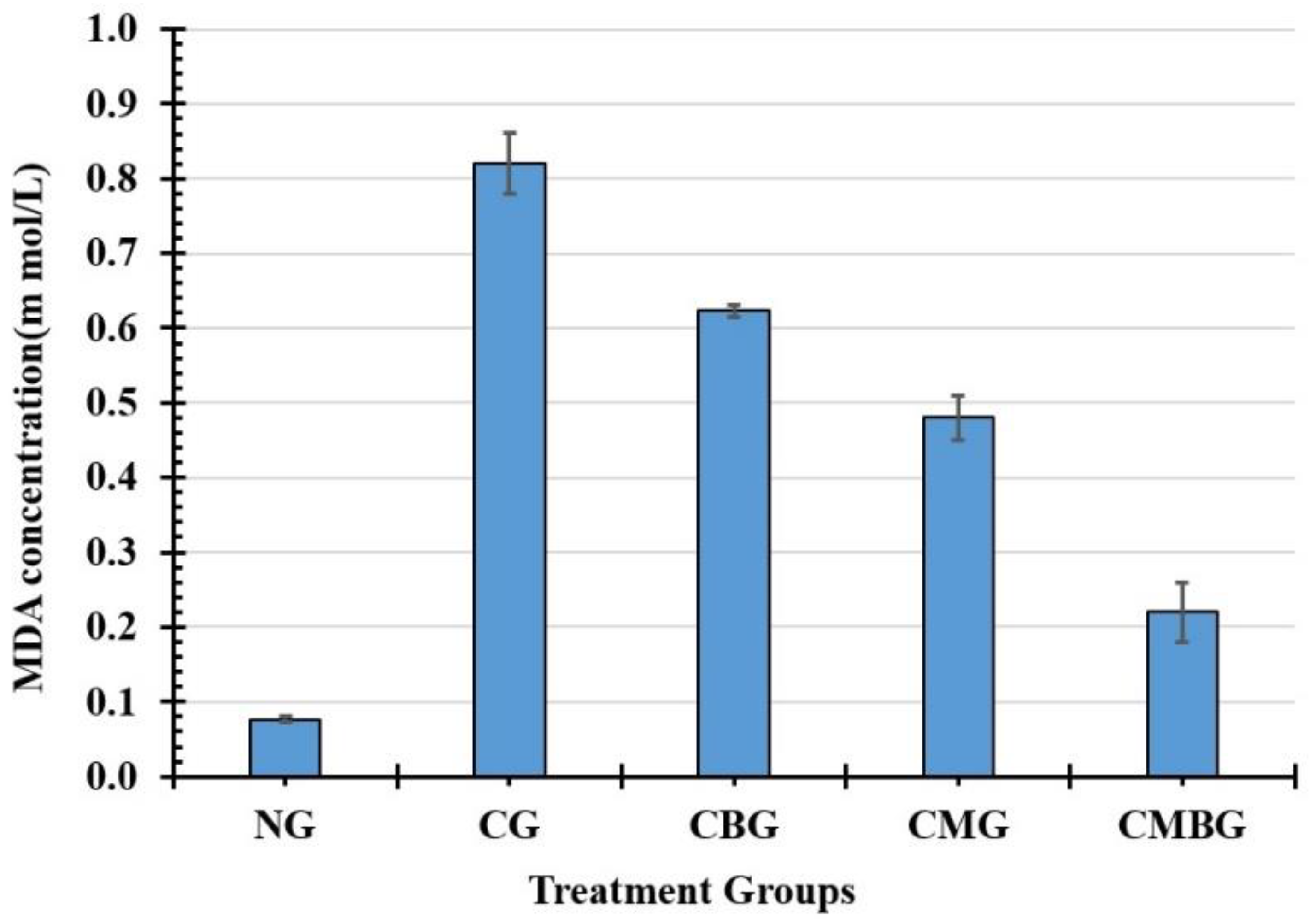
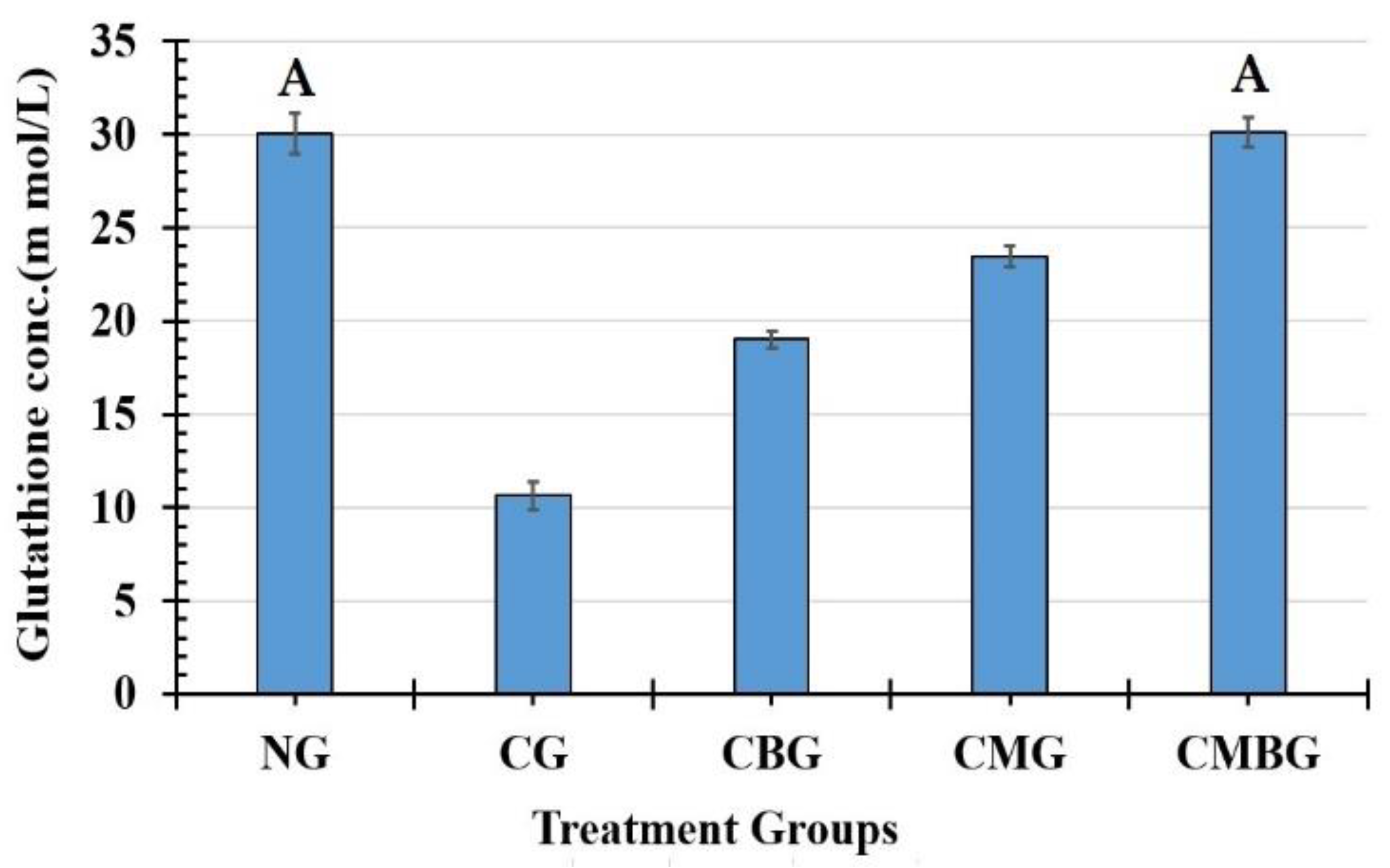
Previous studies have shown that Cis toxicity induces oxidative stress, leading to the generation of free radicals with an increase in ROS and reduced antioxidant molecules of the enzymatic and non-enzymatic testicular protection. In the current study, Cis showed that rat’s antioxidant profile changed as testicular MDA increased and TAC decreased, with GSH demonstrating that enzymatic and non-enzymatic antioxidant molecules were insufficient to scavenge free radicals generated from cisplatin [
40]. The increase in the amount of MDA may be attributed to the excess output of ROS attacking the cell membrane due to a deficiency of antioxidant enzymes [
41]. The decrease in these enzymes’ activity could be explained either by their consumption during the transformation of free radicals into less hazardous or harmless metabolites, or by the direct inhibitory effect of cisplatin on the enzyme function [
42]. The testis is one of the main target organs for oxidative stress and peroxidative destruction due to the high polyunsaturated fatty acids and low antioxidant concentration efficiency, which results in reduced sperm vitality, motility, and consequently infertility [
43].
Elshiekh et al. [
44] reported results on beetroot improvements to cisplatin damage in rats; these were in line with the current work. They recorded significant improvements in sperm count, sperm morphology, histopathology, serum testosterone, MDA level, glutathione (GSH), and catalase activities [
44].
The administration of BM-MSCs and beetroot extract to the Cis group in the current study induced significant improvements in the lipid peroxidation marker and antioxidant enzymes (TAC and GSH). These results agreed with Lorizola et al. [
45], who indicated that the administration of beetroot juice (BRJ) shows a substantial decline in the toxic carbon tetrachloride group for serum MDA levels. Interestingly, apigenin-derived flavonoid glycosides may have acted as natural exogenous antioxidants in beet stalks and leaves, reducing or decreasing oxidative stress in mice. Beetroot is moreover an extremely rich source of antioxidants. Several studies have shown that betalain pigments, in particular, protect cellular components against oxidative damages [
46]. The disturbance in prior oxidative stress levels was reversed after the injection of MSCs. Such results agree with other studies that demonstrated the antioxidant activities of MSCs [
47].
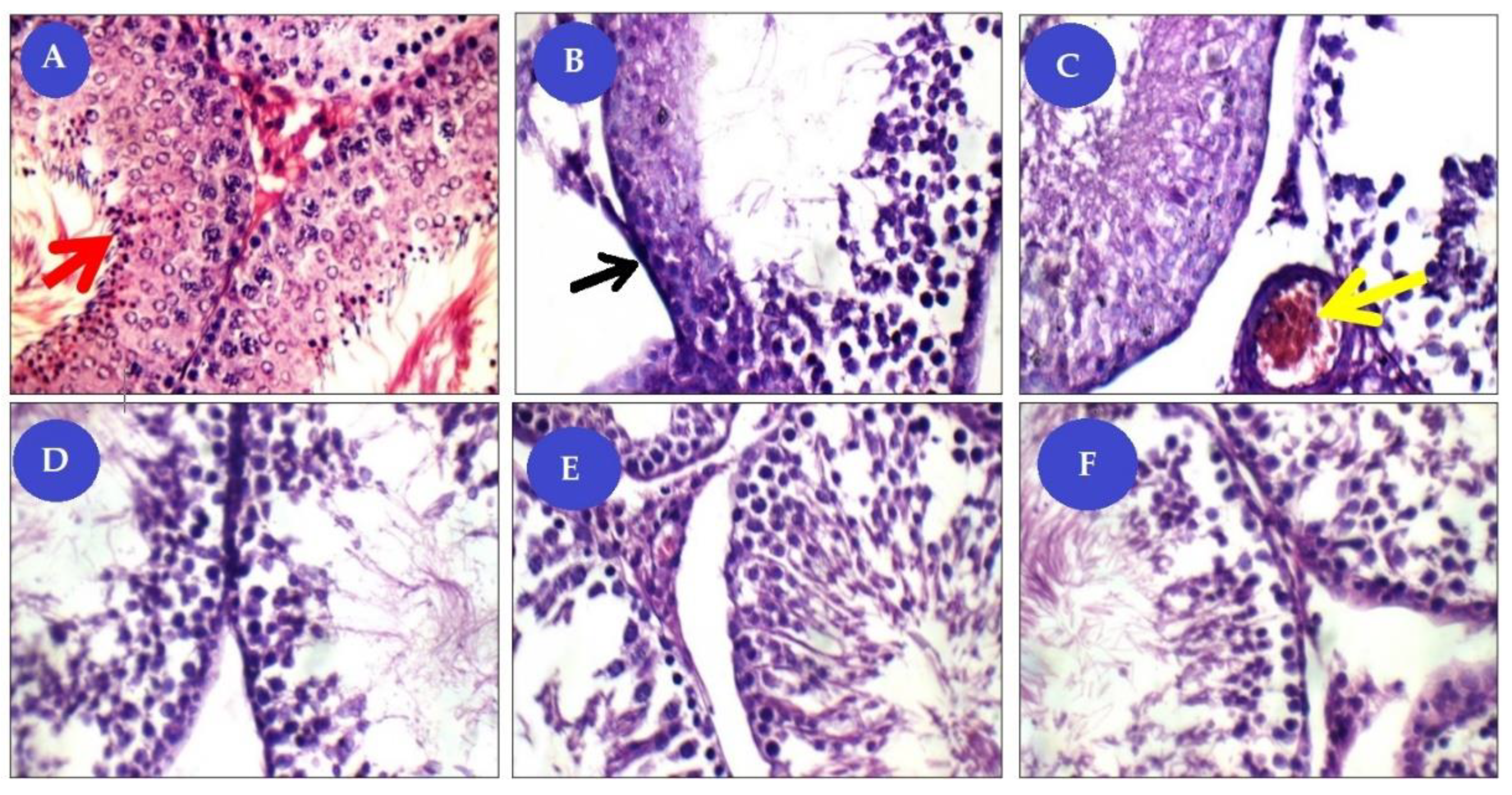
Great improvement was noticed in combined BM-MSCs and beetroot extract to Cis toxic group. The histopathological lesions observed in the present results are in corroboration with the observed biochemical and cytogenetic changes. The number of spermatogenic cells is decreased and the seminiferous tubule with vacuoles underwent degenerative changes in the Cis group. It has been documented that mesenchymal stem cells (MSCs) were able to identify and migrate to injury sites when transplanted systemically, indicating they had migratory ability. Homing to the damaged site and subsequently differentiating into tissue-specific cells, MSCs act as an essential tissue member and thus contribute to tissue repair [
48].
In addition, the ameliorative effect of MSCs may be by preventing testicular apoptosis, reducing intra-testicular oxidative stress, and promoting the development of testosterone which stimulates spermatogenesis. In previous research, this suggested the impact of MSC on ischemic injury in models of the kidney, heart, and lung [
49].
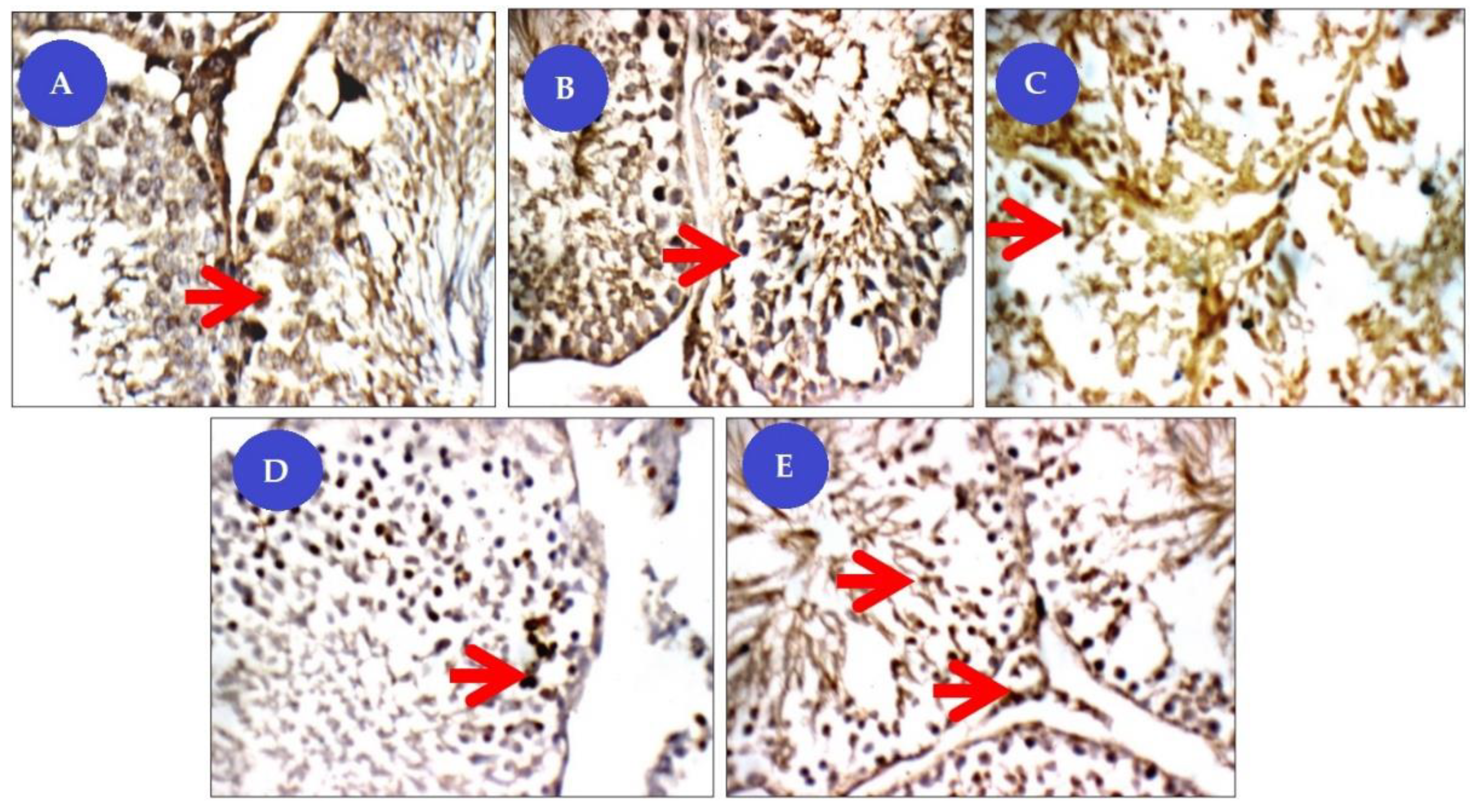
Our immunohistochemical studies revealed that the Cis group showed significantly increased anti-caspase expression−3 in the spermatogenic cell cytoplasm, which plays a vital role in seminiferous tubules in apoptosis regulation [
50]. Increased caspase activity in the testis was also associated with various pathologies such as impaired spermatogenesis, fragmentation of sperm DNA, reduced sperm motility varicocele, immune infertility, and testicular torsion. It serves as a junction point for different signaling pathways in the execution of cell apoptosis. Its activation leads to DNA degradation, chromatin condensation, and membrane protein destruction. Cis administration was shown to result in upward control of caspase-3 and germ cells and Leydig cell apoptosis by triggering extrinsic, intrinsic, and other metazoan apoptotic pathways [
51]. These results were due to the oxidative stress condition caused by the administration of cisplatin and its concomitant impact on the apoptosis of germ cells. Therefore, BM-MSCs with beetroot can be suggested to attenuate the level of caspase-3 immunoreactivity in the testes. The BM-MSC defense mechanism of action is believed to be mainly due to the ability of MSCs to defend testis against oxidative stress through their antioxidant ROS-scavenging properties [
52]. Besides, MSCs can modify the immune and inflammatory state induced by Cis administration [
53]. MSC ultimately exercised anti-apoptotic trophic, and tissue regeneration properties. In this sense, MSCs will stimulate the remaining sperm cells to proliferate and complete their division, releasing some growth factors and cytokines [
54]. This was also confirmed by our immunohistochemical tests, which demonstrated weak caspase-3 expression in the Cis treated group with a combination of BM-MSCs and beetroot extract.
PCNA is an intranuclear polypeptide and a cofactor of DNA polymerase delta, essential for repair, excision, and replication [
55]. As spermatogenesis is a complex cell cycle of fast proliferating cells ending with the formation of sperms, we used PCNA in this study as a tool to evaluate spermatogenesis. We observed that PCNA positive cells were strongly expressed in spermatogonia and early-stage spermatocytes of normal rat, whereas, in Cis-untreated rats, the number of PCNA positive testicular germ cells was substantially reduced, which is an indicator of disturbance in proliferation and spermatogenesis. Earlier studies have suggested that increased PCNA expression in testicular tissue indicates high proliferative activity and stimulation of the sperm [
56]. This study showed that the adverse effect of lowered PCNA expression in the Cis group is improved by treatment with a combination of BM-MSCs and beetroot extract. Thus, the upregulation of PCNA led to the promotion of cell cycle progress and the decline of apoptosis.
Inducible Nitric Oxide Synthase (iNOS) is one of three known isoforms responsible for the synthesis of nitric oxide (NO) in mammalian nitric oxide synthase (NOS). The main iNOS control mechanism seems to be at the genetic induction level, hence its common name as an inducible enzyme [
57]. It is responsible for the production of high levels of nitric oxide (NO) under oxidative stress, which results in auto-cytotoxicity [
58]. Studies have demonstrated that NO’s oxidation products induce lipid peroxidation [
59]. Transcription factors such as NFkB stimulated under oxidative stress can also prompt the expression iNOS. Blocking NF-kB and therefore iNOS with antioxidants has already been shown to be effective in alleviating testicular injury caused by Cis [
60]. Numerous studies have shown that nitric oxide reacts with superoxide anion, forming radical peroxynitrite, which therefore oxidizes cellular structures and induces lipid peroxidation [
58]. It is extremely toxic, causing oxidizing damage to cell components, including lipids, nucleic +acids, and proteins.
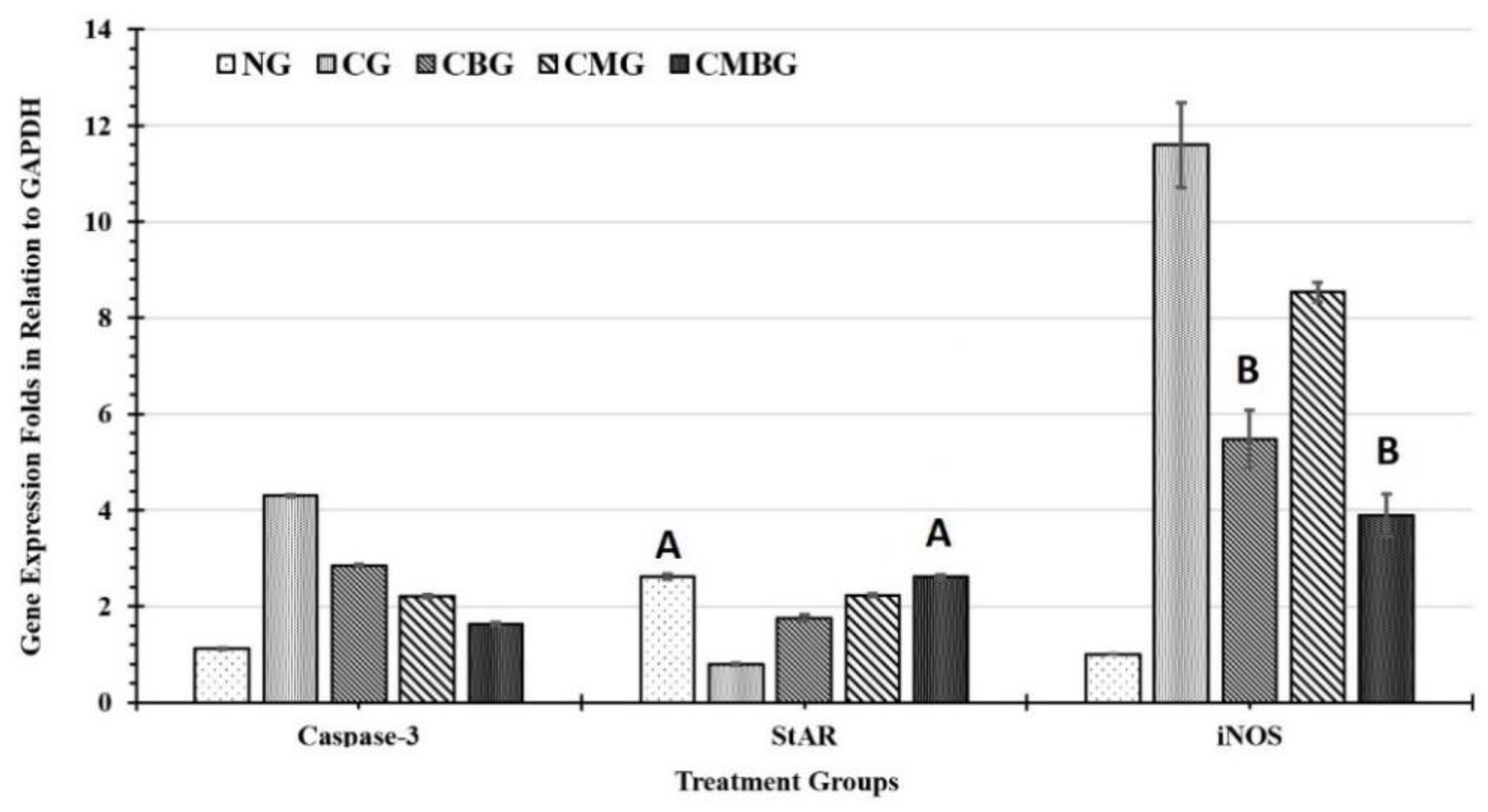
The research results suggest that cisplatin significantly increased the level of iNOS gene mRNA. Additionally, MSC and beetroot extract treatment of cisplatin-induced damaged testicular tissues causes substantial diminution of the iNOS gene regulation relative to cisplatin-treated rats. This result suggests that the protective effect of BM-MSCs and beetroot extract may be closely linked to its cisplatin-induced inhibition of nitric oxide production. Some earlier studies have stated the possible antioxidant properties of both beetroot and MSC against several forms of free radicals [
61].
One of the most important molecular biomarkers of fertility is the steroidogenic acute regulatory (StAR) protein. It is responsible for regulation of steroid hormone biosynthesis in steroidogenic cells of testis. The StAR protein is important to transport cholesterol from the outer membrane to the inner side of the mitochondria [
62]. StAR protein expression is regulated by LH-mediated activation of cAMP-dependent pathways, leading to transcriptional activation in Leydig cells [
63]. StAR expression is correlated to stress on testis, which affect its efficiency [
64,
65,
66]. The current results recorded an improvement in testis efficiency in the groups treated with stem cells and beetroot extract after Cis treatment considering the expression levels of the StAR gene.
Caspase-3 is a member of intrinsic protein factors for programmed cell death (apoptosis). Inactivation of caspase-3 dramatically reduces apoptosis in diverse settings, including activation-induced cell death (AICD). It is an essential component in apoptotic events that are remarkably system- and stimulus-dependent [
67]. The current results indicated overexpression in caspase-3 after treatment with Cis, which activates apoptosis. However, the current cell therapy combined with beetroot extract was effective in improvement of Cis side-effects. This can be explained by previous studies, which stated that dying cells provide instructive cues that can influence surrounding cells to proliferate and showed that even dying stem cells facilitate communication with adjacent stem cells by caspase-dependent production of Wnt8a-containing apoptotic bodies to drive cellular turnover [
68].