Effects of Sanitizers on Microbiological Control of Hatching Eggshells and Poultry Health during Embryogenesis and Early Stages after Hatching in the Last Decade
Abstract
:Simple Summary
Abstract
1. Introduction
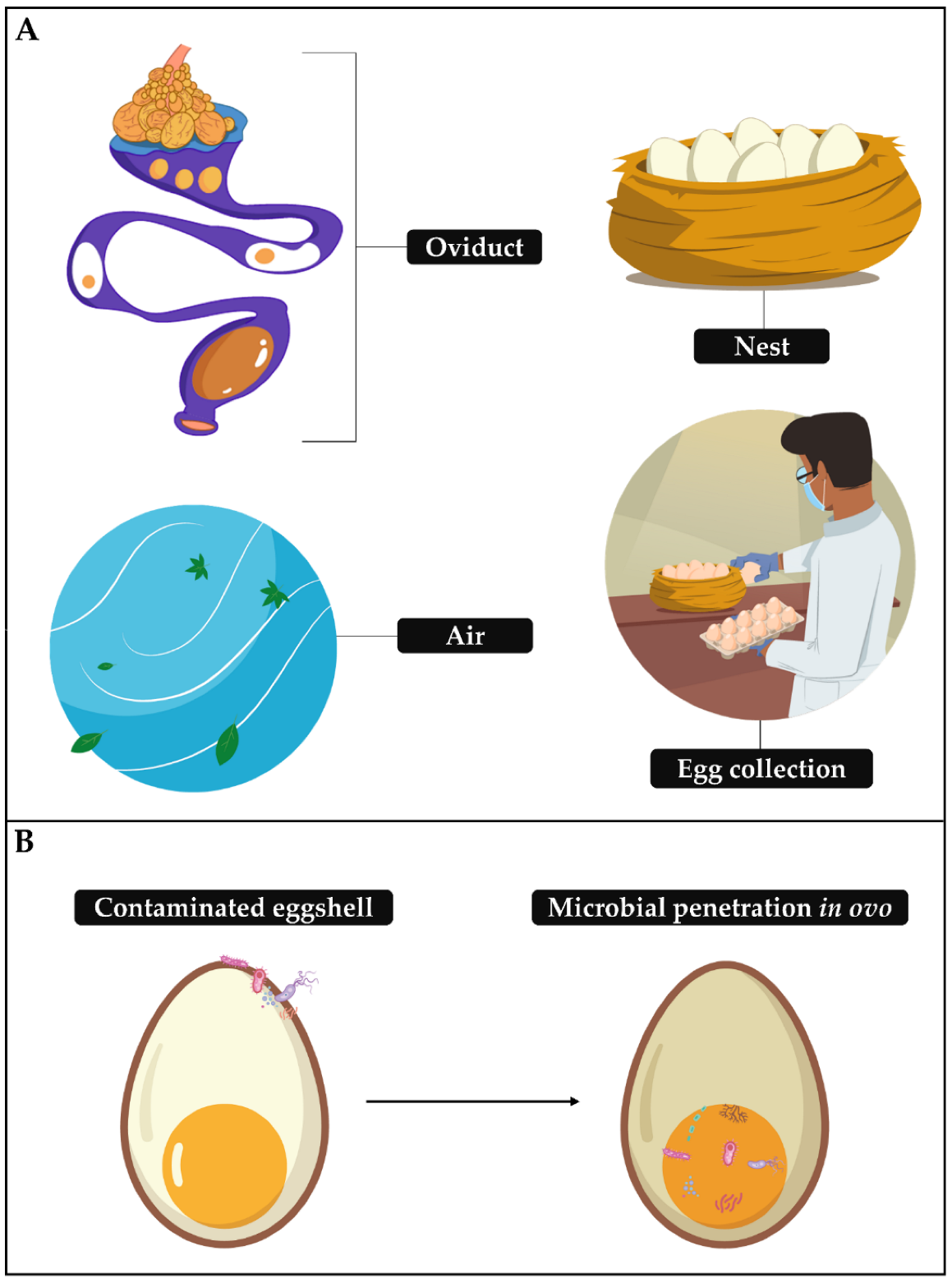
2. Eggshell and Its Contamination
3. Sanitizers and the Sanitization of Hatching Eggs
3.1. Articles and Search Criteria
3.2. Objective, Optimal Timing and Methods for Sanitizing Hatching Eggs
- Fumigation: the release of sanitizing vapors on the surface of hatching eggshells in an enclosed space.
- Spraying: the dispersion of a sanitizing mist on the surface of hatching eggs.
- Immersion: the act of immersing hatching eggs in sanitizer until there is an interaction between them.

3.3. Formaldehyde
4. Sanitizers and Their Effects on the Microbiological Quality of Hatching Eggshells and the Health and Survival of Poultry in the Developmental Phase and Early Period after Hatching
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Al-Shemery, N.J.; Kamaluddin, Z.N. Effect of Using Different Concentrations of Hydrogen Peroxide and Formalin Compared to Formaldehyde Evaporation in Sterilization of Hatching Eggs of Broiler. Euphrates J. Agric. Sci. 2018, 10, 36–41. [Google Scholar]
- Oliveira, G.S.; Nascimento, S.T.; dos Santos, V.M.; Silva, M.G. Clove Essential Oil in the Sanitation of Fertile Eggs. Poult. Sci. 2020, 99, 5509–5516. [Google Scholar] [CrossRef] [PubMed]
- Gholami-Ahangaran, M.; Shahzamani, S.; Yazdkhasti, M. Comparison of Virkon S and Formaldehyde on Hatchabil-ity and Survival Rate of Chicks in Disinfection of Fertile Eggs. Rev. Med. Vet. 2016, 167, 45–49. [Google Scholar]
- Soares, C.E.S.; Cartabiano-Leite, C.E.; Ferreira, W.X.; Maiorka, A.; Dahlke, F.; Scussel, V.M.; de Dea Lindner, J. Peracetic Acid: Effect on the Chicken Eggshell Cuticle and Decontaminating Action on Filamentous Fungi. Jokull J. 2021, 71, 82–96. [Google Scholar]
- Oliveira, G.S.; dos Santos, V.M.; Nascimento, S.T.; Rodrigues, J.C. Alternative Sanitizers to Paraformaldehyde for Incubation of Fertile Eggs. Poult. Sci. 2020, 99, 2001–2006. [Google Scholar] [CrossRef]
- Nogueira, W.C.L.; Pena, A.C.S.; de Souza, C.N.; Azevedo, I.L.; Fariafilho, D.E.; Almeida, A.C. Disinfection of Fertile Eggs of Free-Range Poultry with Essential Oils. Rev. Bras. Saude Prod. Anim. 2019, 20, e0822019. [Google Scholar] [CrossRef] [Green Version]
- Al-Shammari, K.I.; Batkowska, J.; Gryzi, M.M. Assessment of Ultraviolet Light Effect in Hatching Eggs Disinfection on Hatchability Traits of Two Breeds of Quails and Chickens. Acta Sci. Pol. Zootec. 2015, 14, 33–44. [Google Scholar]
- Badran, A.M.M.; Osman, A.M.R.; Yassein, D.M.M. Comparative Study of the Effect of Some Disinfectants on Em-bryonic Mortality, Hatchability, and Some Blood Components. Egypt. Poult. Sci. J. 2018, 38, 1069–1081. [Google Scholar] [CrossRef]
- Tebrün, W.; Motola, G.; Hafez, M.H.; Bachmeier, J.; Schmidt, V.; Renfert, K.; Reichelt, C.; Brüggemann-Schwarze, S.; Pees, M. Preliminary Study: Health and Performance Assessment in Broiler Chicks Following Application of Six Different Hatching Egg Disinfection Protocols. PLoS ONE 2020, 15, e0232825. [Google Scholar] [CrossRef]
- Oliveira, G.S.; Nascimento, S.T.; dos Santos, V.M.; Dallago, B.S.L. Spraying Hatching Eggs with Clove Essential Oil Does Not Compromise the Quality of Embryos and One-Day-Old Chicks or Broiler Performance. Animals 2021, 11, 2045. [Google Scholar] [CrossRef]
- Zhang, L.; Steinmaus, C.; Eastmond, D.A.; Xin, X.K.; Smith, M.T. Formaldehyde Exposure and Leukemia: A New Meta-Analysis and Potential Mechanisms. Mut. Res./Rev. Mut. Res. 2009, 681, 150–168. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Guidelines for Indoor Air Quality: Selected Pollutants; World Health Organization: Geneva, Switzerland, 2010; pp. 103–156. [Google Scholar]
- Cadirci, S. Disinfection of Hatching Eggs by Formaldehyde Fumigation-a Review. Eur. Poult. Sci. 2009, 73, 116–123. [Google Scholar]
- Hincke, M.T.; Da Silva, M.; Guyot, N.; Gautron, J.; McKee, M.D.; Guabiraba-Brito, R.; Réhault-Godbert, S. Dynamics of Structural Barriers and Innate Immune Components during Incubation of the Avian Egg: Critical Interplay be-tween Autonomous Embryonic Development and Maternal Anticipation. J. Innate Immun. 2019, 11, 111–124. [Google Scholar] [CrossRef] [PubMed]
- Kulshreshtha, G.; D’Alba, L.; Dunn, I.C.; Rehault-Godbert, S.; Rodriguez-Navarro, A.B.; Hincke, M.T. Properties, Genetics and Innate Immune Function of the Cuticle in Egg-Laying Species. Front. Immunol. 2022, 13, 838525. [Google Scholar] [CrossRef]
- de Reu, K.; Grijspeerdt, K.; Messens, W.; Heyndrickx, M.; Uyttendaele, M.; Debevere, J.; Herman, L. Eggshell Factors Influencing Eggshell Penetration and Whole Egg Contamination by Different Bacteria, Including Salmonella Enteritidis. Int. J. Food Microbiol. 2006, 112, 253–260. [Google Scholar] [CrossRef]
- Gantois, I.; Ducatelle, R.; Pasmans, F.; Haesebrouck, F.; Gast, R.; Humphrey, T.J.; van Immerseel, F. Mechanisms of Egg Contamination by Salmonella Enteritidis. FEMS Microbiol. Rev. 2009, 33, 718–738. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shafey, T.M.; Hussein, E.O.S.; Al-Batshan, H.A. Effects of Ultrasonic Waves on Eggshell Strength and Hatchability of Layer-Type Breeder Eggs. S. Afr. J. Anim. Sci. 2013, 43, 56–63. [Google Scholar] [CrossRef] [Green Version]
- Williams, J.E.; Dillard, L.H.; Hall, G.O. The Penetration Patterns of Salmonella Typhimurium through the Outer Structures of Chicken Eggs. Avian Dis. 1968, 12, 445–466. [Google Scholar] [CrossRef] [PubMed]
- Berrang, M.E.; Frank, J.F.; Buhr, R.J.; Bailey, J.S.; Cox, N.A.; Mauldin, J. Eggshell Characteristics and Penetration by Salmonella Through the Productive Life of a Broiler Breeder Flock. Poult. Sci. 1998, 77, 1446–1450. [Google Scholar] [CrossRef]
- Rezaee, M.S.; Liebhart, D.; Hess, C.; Hess, M.; Paudel, S. Bacterial Infection in Chicken Embryos and Consequences of Yolk Sac Constitution for Embryo Survival. Vet. Pathol. 2021, 58, 71–79. [Google Scholar] [CrossRef]
- Orajaka, L.J.E.; Mohan, K. Aerobic Bacterial Flora from Dead-in-Shell Chicken Embryos from Nigeria. Avian Dis. 1985, 29, 583–589. [Google Scholar] [CrossRef] [PubMed]
- Higenyi, J. Microbial Contamination Load of Hatching Eggs in Butaleja, Eastern Uganda. Anim. Vet. Sci. 2014, 2, 22. [Google Scholar] [CrossRef] [Green Version]
- Zhelev, G.; Lyutzkanov, M.; Urumova, V.; Mihaylov, G.; Petrov, V.; Marutsov, P. Microbial Contamination in a Duck Hatchery. Rev. Med. Vet. 2012, 163, 319–322. [Google Scholar]
- Oliveira, G.D.S.; McManus, C.; dos Santos, V.M. Garlic as Active Principle of Sanitiser for Hatching Eggs. Worlds Poult. Sci. J. 2022, 78, 1–16. [Google Scholar] [CrossRef]
- Araújo, W.A.G.; Albino, L.F.T. Incubação Comercial [Commercial Incubation]; Transworld Research Network.: Trivandrum, India, 2011. [Google Scholar]
- Oliveira, G.D.S.; dos Santos, V.M.; Nascimento, S.T. Essential Oils as Sanitisers for Hatching Eggs. Worlds Poult. Sci. J. 2021, 77, 605–617. [Google Scholar] [CrossRef]
- Oliveira, G.D.S.; dos Santos, V.M.; McManus, C. Propolis: Effects on the Sanitisation of Hatching Eggs. Worlds Poult. Sci. J. 2022, 78, 261–272. [Google Scholar] [CrossRef]
- Mousa-Balabel, T.M.; Mohamed, R.A.; Al-Midani, S.A.; El-Samad, M.S.A. Impact of Boiler Breeders Hatching Eggs Disinfection Time on Some Hatchability Parameters. Int. J. Sci. Basic Appl. Res. 2016, 30, 230–240. [Google Scholar]
- Ge, J.; Yang, H.; Lu, X.; Wang, S.; Zhao, Y.; Huang, J.; Xi, Z.; Zhang, L.; Li, R. Combined Exposure to Formaldehyde and PM2.5: Hematopoietic Toxicity and Molecular Mechanism in Mice. Environ. Int. 2020, 144, 106050. [Google Scholar] [CrossRef]
- Hrnčár, C.; Prachárová, S.; Bujko, J. The Effect of Disinfection of Hatching Eggs on Hatchability of Oravka Chickens. Sci. Pap. Anim. Sci. Biotechnol. 2012, 45, 411–414. [Google Scholar]
- Durmus, I. Determining Effects of Use of Various Disinfecting Materials on Hatching Results and Total Bacterial Count. Asian J. Anim. Vet. Adv. 2012, 7, 739–744. [Google Scholar] [CrossRef] [Green Version]
- Fidan, E.D.; Turkyilmaz, M.K.; Ahmet Nazligul, A. The Effects of Different Storage and Fumigation Lengths on Hatchability and Hatching Weight in Japanese Quails (Coturnix coturnix Japonica). J. Anim. Vet. Adv. 2012, 11, 1400–1404. [Google Scholar] [CrossRef] [Green Version]
- Shahein, E.H.A.; Sedeek, E.K. Role of Spraying Hatching Eggs with Natural Disinfectants on Hatching Characteris-tics and Eggshell Bacterial Counts. Egypt. Poult. Sci. J. 2014, 34, 213–230. [Google Scholar] [CrossRef] [Green Version]
- Keïta, A.; Huneau-Salaün, A.; Guillot, A.; Galliot, P.; Tavares, M.; Puterflam, J. A Multi-Pronged Approach to the Search for an Alternative to Formaldehyde as an Egg Disinfectant without Affecting Worker Health, Hatching, or Broiler Production Parameters. Poult. Sci. 2016, 95, 1609–1616. [Google Scholar] [CrossRef] [PubMed]
- Bekele, M.W.; Leta, M.U. Effect of Egg Storage Temperature and Fumigation on Hatchability of Cobb 500 and Hub-bard Broiler Strains. Afr. J. Agric. Res. 2016, 11, 3418–3424. [Google Scholar] [CrossRef] [Green Version]
- Clímaco, W.L.D.S.; Melo, É.D.F.; Vaz, D.P.; Saldanha, M.M.; Pinto, M.F.V.D.S.; Fernandes, L.C.C.; Baião, N.C.; Oliveira, L.G.D.; Sant’Anna, F.M.D.; Souza, M.R.D.; et al. Eggshell Microbiology and Quality of Hatching Eggs Subjected to Different Sanitizing Procedures. Pesq. Agrop. Bras. 2018, 53, 1177–1183. [Google Scholar] [CrossRef]
- Pees, M.; Motola, G.; Hafez, M.H.; Bachmeier, J.; Brüggemann-Schwarze, S.; Tebrün, W. Use of Electron Irradiation versus Formaldehyde Fumigation as Hatching Egg Disinfectants-Efficacy and Impact on Hatchability and Broiler Performance. Tierärztliche Prax. Ausg. G Großtiere/Nutztiere 2020, 48, 406–413. [Google Scholar] [CrossRef]
- Hrnčár, C.; Hanusová, E.; Hanus, A.; Arpášová, H.; Kokoszyński, D.; Bujko, J. The Effect of Various Disinfectants on Hatching Results in Chickens. Sci. Pap. Anim. Sci. Biotechnol. 2021, 54, 193–196. [Google Scholar]
- Zeweil, H.S.; Rizk, R.E.; Bekhet, G.M.; Ahmed, M.R. Comparing the Effectiveness of Egg Disinfectants against Bacte-ria and Mitotic Indices of Developing Chick Embryos. J. Basic Appl. Zool. 2015, 70, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Baylan, M.; Akpınar, G.C.; Canogullari, S.D.; Ayasan, T. The Effects of Using Garlic Extract for Quail Hatching Egg Disinfection on Hatching Results and Performance. Rev. Bras. Cienc. Avic. 2018, 20, 343–350. [Google Scholar] [CrossRef]
- Batkowska, J.; Al-Shammari, K.I.A.; Gryzinska, M.M.; Brodacki, A.; Wlazlo, L.; Nowakowicz-Debek, B. Effect of Us-ing Colloidal Silver in the Disinfection of Hatching Eggs on Some Microbial, Hatchability and Performance Traits in Japanese Quail (Coturnix Cot. Japonica). Eur. Poult. Sci. 2017, 81, 211. [Google Scholar] [CrossRef]
- Batkowska, J.; Al-Shammari, K.I.A.; Lukasz, W.; Nowakowicz-Debek, B.; Gryzinska, M. Evaluation of Propolis Ex-tract as a Disinfectant of Japanese Quail (Coturnix coturnix Japonica) Hatching Eggs. Poult. Sci. 2018, 97, 2372–2377. [Google Scholar] [CrossRef] [PubMed]
- Wlazlo, L.; Drabik, K.; Al-Shammari, K.I.A.; Batkowska, J.; Nowakowicz-Debek, B.; Gryzińska, M. Use of Reactive Oxygen Species (Ozone, Hydrogen Peroxide) for Disinfection of Hatching Eggs. Poult. Sci. 2020, 99, 2478–2484. [Google Scholar] [CrossRef] [PubMed]
- Bekhet, G.M. Impact of Hatching Egg Disinfection on Hatching Characteristics and Chick Embryos. Indian J. Anim. Res. 2021, 55, 353–358. [Google Scholar] [CrossRef]
- Taşdemir, A.N.; Onbaşılar, E.E.; Yalçın, S.; Boyalı, B.; Aygören, H.; Tülek, E.; Sarıçam, S.; Akan, M. Effects of Oregano Juice on Eggshell Microbial Load, Layer Embryo Development, Hatching Results, and Growth at the First 2 Weeks after Hatch. Trop. Anim. Health Prod. 2021, 53, 404. [Google Scholar] [CrossRef]
- Harikrishnan, S.; Narayanankutty, K.; Chacko, B.; Anitha, P. The Effect of Various Sanitizing Agents on the Economics of Hatching Kuttanad Duck Eggs. Indian J. Vet. Sci. Biotechnol. 2014, 9, 67–68. [Google Scholar]
- Vilela, C.O.; Vargas, G.D.; Fischer, G.; Ladeira, S.; de Faria, R.O.; Nunes, C.F.; de Lima, M.; Hübner, S.O.; Luz, P.; Osório, L.G.; et al. Propolis: A Natural Product as an Alternative for Disinfection of Embryonated Eggs for Incubation. Arq. Inst. Biol. 2012, 79, 161–167. [Google Scholar] [CrossRef] [Green Version]
- Aygun, A.; Sert, D.; Copur, G. Effects of Propolis on Eggshell Microbial Activity, Hatchability, and Chick Performance in Japanese Quail (Coturnix coturnix Japonica) Eggs. Poult. Sci. 2012, 91, 1018–1025. [Google Scholar] [CrossRef]
- Uçan, U.S.; Gök, A. Efficacy of a Water-Based Disinfectant on Reduction of Eggshell Bacterial Contamination. Eur. J. Vet. Sci. 2012, 28, 57–59. [Google Scholar]
- Nowaczewski, S.; Szablewski, T.; Cegielska-Radziejewska, R.; Kontecka, H. Microbiological Response of Japanese Quail Eggs to Disinfection and Location in the Setter during Incubation. Folia Biol. 2013, 61, 119–124. [Google Scholar] [CrossRef] [Green Version]
- Aygun, A.; Sert, D. Effects of Prestorage Application of Propolis and Storage Time on Eggshell Microbial Activity, Hatchability, and Chick Performance in Japanese Quail (Coturnix coturnix Japonica) Eggs. Poult. Sci. 2013, 92, 3330–3337. [Google Scholar] [CrossRef]
- Lowman, Z.S.; Parkhurst, C.R. Effects of Bac-D on Total Aerobic Bacteria Naturally Found on Broiler Breeder Eggs. Int. J. Poult. Sci. 2013, 12, 505–508. [Google Scholar] [CrossRef]
- Buhr, R.J.; Spickler, J.L.; Ritter, A.R.; Bourassa, D.V.; Cox, N.A.; Richardson, L.J.; Wilson, J.L. Efficacy of Combination Chemicals as Sanitizers of Salmonella-Inoculated Broiler Hatching Eggshells. J. Appl. Poul. Res. 2013, 22, 27–35. [Google Scholar] [CrossRef]
- Musgrove, M.T.; Stephens, C.B.; Bourassa, D.V.; Cox, N.A.; Mauldin, J.M.; Berrang, M.E.; Buhr, R.J. Enterobacteri-aceae and Salmonella Recovered from Nonsanitized and Sanitized Broiler Hatching Eggs. J. Appl. Poult. Res. 2014, 23, 516–522. [Google Scholar] [CrossRef]
- Hanafy, A.M.; Ahmed, E.G.; Abdel-Ghany, A.M.; Nashaat, M.H. Effect of Disinfectants, Flock Physiological Status, Season and Storage Period of Broiler Breeder Eggs on Field Hatchability and Hatchery Operation Quality under New-Salheya City Situation. Egypt J. Anim. Prod. 2015, 52, 115–125. [Google Scholar]
- Upadhyaya, I.; Yin, H.B.; Nair, M.S.; Chen, C.H.; Upadhyay, A.; Darre, M.J.; Venkitanarayanan, K. Efficacy of Fumi-gation with Trans-Cinnamaldehyde and Eugenol in Reducing Salmonella enterica serovar Enteritidis on Embryonated Egg Shells. Poult. Sci. 2015, 94, 1685–1690. [Google Scholar] [CrossRef]
- Yildirim, I.; Aygun, A.; Sert, D. Effects of Preincubation Application of Low and High Frequency Ultrasound on Eggshell Microbial Activity, Hatchability, Supply Organ Weights at Hatch, and Chick Performance in Japanese Quail (Coturnix coturnix Japonica) Hatching Eggs. Poult. Sci. 2015, 94, 1678–1684. [Google Scholar] [CrossRef]
- Gottselig, S.M.; Dunn-Horrocks, S.L.; Woodring, K.S.; Coufal, C.D.; Duong, T. Advanced Oxidation Process Sanitiza-tion of Eggshell Surfaces. Poult. Sci. 2016, 95, 1356–1362. [Google Scholar] [CrossRef]
- Rehkopf, A.C.; Byrd, J.A.; Coufal, C.D.; Duong, T. Advanced Oxidation Process Sanitization of Hatching Eggs Re-duces Salmonella in Broiler Chicks. Poult. Sci. 2017, 96, 3709–3716. [Google Scholar] [CrossRef]
- Mousa-Balabel, T.M.; Al-Midani, S.A.; Al-Refaay, M.A.; Kerady, S.M. Coliform Bacteria and Hatching Egg Disinfect-ants. Int. J. Sci. Basic Appl. Res. 2017, 33, 151–163. [Google Scholar]
- Fouad, W.; Abdel-Hafez, M.S. Effect of Spraying Hatching Eggs of Japanese Quails by Live Yeast on Physiological Changes in the Embryonic Development, Hatchability and Total Bacterial Count. Egypt. Poult. Sci. J. 2017, 37, 1303–1321. [Google Scholar] [CrossRef]
- Olsen, R.; Kudirkiene, E.; Thøfner, I.; Pors, S.; Karlskov-Mortensen, P.; Li, L.; Papasolomontos, S.; Angastiniotou, C.; Christensen, J. Impact of Egg Disinfection of Hatching Eggs on the Eggshell Microbiome and Bacterial Load. Poult. Sci. 2017, 96, 3901–3911. [Google Scholar] [CrossRef] [PubMed]
- Korowiecka, K.; Trela, M.; Tombarkiewicz, B.; Pawlak, K.; Niedziółka, J.W.; Swadźba, M.; Lis, M.W. Assessment of the Effect of Selected Substances Used for Disinfection of Hatching Eggs on Hatching Results in Chickens. Sci. Ann. Pol. Soc. Anim. Prod. 2017, 13, 25–35. [Google Scholar] [CrossRef]
- Kusstatscher, P.; Cernava, T.; Liebminger, S.; Berg, G. Replacing Conventional Decontamination of Hatching Eggs with a Natural Defense Strategy Based on Antimicrobial, Volatile Pyrazines. Sci. Rep. 2017, 7, 13253. [Google Scholar] [CrossRef] [PubMed]
- Sokovnin, S.Y.; Donnik, I.M.; Shkuratova, I.A.; Krivonogova, A.S.; Isaeva, A.G.; Balezin, M.E.; Vazirov, R.A. The Use of Nanosecond Electron Beam for the Eggs Surface Disinfection in Industrial Poultry. J. Phys. Conf. Ser. 2018, 1115, 022034. [Google Scholar] [CrossRef]
- Fouad, W.; Abdel-Hafez, M.S.; El-Halim, H.A.H.A. Influence of Spraying Garlic Oil on Embryonic Development, Hatchability, Physiological Parameters, Post-Hatch Chick Growth and Bacterial Contamination of Fertile Quail Eggs. Egypt. Poult. Sci. J. 2018, 38, 877–893. [Google Scholar] [CrossRef]
- Li, X.; Anderson, D.; Rathgeber, B.; McLean, N.; MacIsaac, J. Fumigating Broiler Hatching Eggs with Lysozyme Product (Inovapure) to Reduce Eggshell Microbial Load. Poult. Sci. 2018, 97, 4252–4261. [Google Scholar] [CrossRef]
- Chung, H.; Kim, H.; Myeong, D.; Kim, S.; Choe, N.H. Effect of Chlorine Dioxide Gas Application to Egg Surface: Microbial Reduction Effect, Quality of Eggs, and Hatchability. Korean J. Food Sci. Anim. Resour. 2018, 38, 487–497. [Google Scholar] [CrossRef]
- Batkowska, J.; Wlazlo, L.; Drabik, K.; Nowakowicz-Debek, B.; Al-Shammari, K.I.A.; Gryzinska, M. Evaluation of Grapefruit Juice (Citrus paradisi) as an Alternative Disinfectant for Hatching Eggs. Pak. J. Zool. 2018, 50, 647–653. [Google Scholar] [CrossRef]
- Gatea, S.M.; Altaie, S.M.S.; Khafaji, S.S.; Aljanabi, T.K.; Shatti, D.H.; Hussain, M.A. Influence of Spraying Different Solutions at Different Incubation Periods on Hatchability Parameters of Local Iraqi’s Eggs. IOP Conf. Ser. Earth Environ. Sci. 2019, 388, 012034. [Google Scholar] [CrossRef]
- Cantu, K.; Archer, G.S.; Tucker, Z.S.; Coufal, C.D. Effectiveness of Duck Hatching Egg Sanitization with the Combi-nation of Hydrogen Peroxide and Ultraviolet Light. J. Appl. Poult. Res. 2019, 28, 301–306. [Google Scholar] [CrossRef]
- Hidayat, M.N.; Thaha, A.H.; Mayanti, R. Effect of the Use of Noni Leaf Extract as a Natural Disinfectant on the Per-centage of Hatchability and Day Old Quail (DOQ) Hatching. Chal. J. Anim. Husb. 2020, 4, 48–53. [Google Scholar] [CrossRef]
- Fouad, W.; Abdelfattah, M.G.; Abdelnabi, M.A. Effect of Spraying Hatching Eggs by Different Levels of Vinegar on Embryological Development, Hatchability and Physiological Performance of Dandarwi Chicks. Egypt. Poult. Sci. J. 2019, 39, 291–309. [Google Scholar] [CrossRef]
- Melo, E.F.; Clímaco, W.L.S.; Triginelli, M.V.; Vaz, D.P.; de Souza, M.R.; Baião, N.C.; Pompeu, M.A.; Lara, L.J.C. An Evaluation of Alternative Methods for Sanitizing Hatching Eggs. Poult. Sci. 2019, 98, 2466–2473. [Google Scholar] [CrossRef]
- Al-Asadi, K.J.T.; Ibrahim, B.M. Effect of the Use of Immersion and Injection Methods for Eggs Hatching of Broiler Breeders in the Aquatic Extracts of Some Plant Seeds as Early Feeding: 2-Subsequent Production Performance. Plant Arch. 2020, 20, 2123–2130. [Google Scholar]
- Toghyani, P.; Shahzamani, S.; Gholami Ahangaran, M.; Ali Mousavi Firouzabadi, S. Comparison of Eucalyptus Ex-tract and Formaldehyde on Hatchability and Survival Rate of Chicks in Disinfection of Fertile Eggs. Int. J. Pharm. Res. All. Sci. 2020, 9, 105–109. [Google Scholar]
- Stegniy, B.T.; Paliy, A.P.; Pavlichenko, O.V.; Stegniy, O.O.; Palii, A.P. Comparative Assessment of the Effect of Dis-infectants on the Level of Biotic Contamination and Hatchability of Chicken Eggs. J. Vet. Med. Biotechnol. Biosaf. 2020, 6, 17–22. [Google Scholar] [CrossRef]
- Patrzałek, M.; Kosecka-Strojek, M.; Lisowska-Łysiak, K.; Trela, M.; Kot, M.; Gawlak, M.; Liszka, D.; Sajewicz, M.; Tombarkiewicz, B.; Pawlak, K.; et al. Preliminary Evaluation of Application of a 3-Dimensional Network Structure of Siloxanes Dergall Preparation on Chick Embryo Development and Microbiological Status of Eggshells. Poult. Sci. 2020, 99, 1581–1590. [Google Scholar] [CrossRef] [PubMed]
- Melo, E.F.; McElreath, J.S.; Wilson, J.L.; Lara, L.J.C.; Cox, N.A.; Jordan, B.J. Effects of a Dry Hydrogen Peroxide Dis-infection System Used in an Egg Cooler on Hatchability and Chick Quality. Poult. Sci. 2020, 99, 5487–5490. [Google Scholar] [CrossRef]
- Cassar, J.R.; Bright, L.M.; Patterson, P.H.; Mills, E.W.; Demirci, A. The Efficacy of Pulsed Ultraviolet Light Processing for Table and Hatching Eggs. Poult. Sci. 2021, 100, 100923. [Google Scholar] [CrossRef]
- Bekhet, G.M.; Sayed, A.A. Oregano-Oil Antagonist Lipopolysaccharide (LPS) Induced Toxicity in Pre- and Post-Hatch Chick Embryo. J. Appl. Anim. Res. 2021, 49, 211–220. [Google Scholar] [CrossRef]
- Branco, J.R.O.; Dallago, B.S.L.; Bernal, F.E.M. Efficiency of Ultraviolet Light for Disinfection of Fertile Broiler Eggs. Arq. Bras. Med. Vet. Zootec. 2021, 73, 1137–1146. [Google Scholar] [CrossRef]
- Koc, S.; Aygun, A. Effects of Ozone on Eggshell Microbial Load, Hatching Traits and Chick Performance in Quail Eggs. Innoriginal Int. J. Sci. 2013, 8, 47–52. [Google Scholar]
- Abo-Samaha, M.I.; Basha, H.A. Effect of Spraying Japanese Quail Eggs with Garlic Oil on Hatching Performance and Hatch Weight. Adv. Anim. Vet. Sci. 2021, 9, 156–161. [Google Scholar] [CrossRef]
- Liu, C.; Zheng, W.; Li, Z.; Zhou, L.; Sun, Y.; Han, S. Slightly Acidic Electrolyzed Water as an Alternative Disinfection Technique for Hatching Eggs. Poult. Sci. 2021, 101, 101643. [Google Scholar] [CrossRef]
- Asad, N.R.; Buarque, L.M.; Asad, O.; Bonacossa De Almeida, C.E.; Felzenszwalb, I.; Bispo Cabral-Neto, J.; Leitão, A.C. Several Pathways of Hydrogen Peroxide Action That Damage the E. Coli Genome General Aspects. Genet. Mol. Biol. 2004, 27, 291–303. [Google Scholar] [CrossRef] [Green Version]
- Patil, S.; Valdramidis, V.P.; Karatzas, K.A.G.; Cullen, P.J.; Bourke, P. Assessing the Microbial Oxidative Stress Mechanism of Ozone Treatment through the Responses of Escherichia Coli Mutants. J. Appl. Microbiol. 2011, 111, 136–144. [Google Scholar] [CrossRef]
- Dunowska, M.; Morley, P.S.; Hyatt, D.R. The Effect of Virkon®S Fogging on Survival of Salmonella enterica and Staphylococcus Aureus on Surfaces in a Veterinary Teaching Hospital. Vet. Microbiol. 2005, 105, 281–289. [Google Scholar] [CrossRef]
- Ventura, M.; Wemimont, E. Review of Hydrogen Peroxide Material Safety Data Sheets. In Proceedings of the 38th AIAA/ASME/SAE/ASEE Joint Propulsion Conference & Exhibit, Indianapolis, Indiana, 7–10 July 2002; pp. 1–13. [Google Scholar]
- Baysan, A.; Lynch, E. The Use of Ozone in Dentistry and Medicine. Part 2. Ozone and Root Caries. Prim. Dent. Care 2006, 13, 37–41. [Google Scholar] [CrossRef]
- Irshad, M.; Ali Subhani, M.; Ali, S.; Hussain, A. Biological Importance of Essential Oils. In Essential Oils-Oils of Nature; El-Shemy, H.A., Ed.; IntechOpen: London, UK, 2019. [Google Scholar]
- Stojanović, S.; Najman, S.J.; Bogdanova-Popov, B.; Najman, S.S. Propolis: Chemical Composition, Biological and Pharmacological Activity–a Review. Acta Med. Median. 2020, 59, 108–113. [Google Scholar] [CrossRef]
- Scripnic, E.; Eremia, N. Propolis Extract Use in Incubation Technology for Hens’ Eggs Treatment. Sci. Pap. Ser. D. Anim. Sci. 2015, 53, 330–333. [Google Scholar]
- Sasi, M.; Kumar, S.; Kumar, M.; Thapa, S.; Prajapati, U.; Tak, Y.; Changan, S.; Saurabh, V.; Kumari, S.; Kumar, A.; et al. Garlic (Allium sativum L.) Bioactives and Its Role in Alleviating Oral Pathologies. Antioxidants 2021, 10, 1847. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Liu, C.H.; Cai, J.; Zhang, W.; Qi, W.L.; Wang, Z.; Liu, Z.B.; Yang, Y. Broad-Spectrum Antimicrobial Ac-tivity, Chemical Composition and Mechanism of Action of Garlic (Allium sativum) Extracts. Food Control 2018, 86, 117–125. [Google Scholar] [CrossRef]
- Tariq, S.; Wani, S.; Rasool, W.; Shafi, K.; Bhat, M.A.; Prabhakar, A.; Shalla, A.H.; Rather, M.A. A Comprehensive Re-view of the Antibacterial, Antifungal and Antiviral Potential of Essential Oils and Their Chemical Constituents against Drug-Resistant Microbial Pathogens. Microb. Pathog. 2019, 134, 103580. [Google Scholar] [CrossRef] [PubMed]
- Vadillo-Rodríguez, V.; Cavagnola, M.A.; Pérez-Giraldo, C.; Fernández-Calderón, M.C. A Physico-Chemical Study of the Interaction of Ethanolic Extracts of Propolis with Bacterial Cells. Coll. Surf. B Bioint. 2021, 200, 111571. [Google Scholar] [CrossRef]
- Stähli, A.; Schröter, H.; Bullitta, S.; Serralutzu, F.; Dore, A.; Nietzsche, S.; Milia, E.; Sculean, A.; Eick, S. In Vitro Activity of Propolis on Oral Microorganisms and Biofilms. Antibiotics 2021, 10, 1045. [Google Scholar] [CrossRef]
- Nolan, L.K.; John Barnes, H.; Vaillancourt, J.-P.; Abdul-Aziz, T.; Logue, C.M. Colibacillosis. In Diseases of Poultry; Swayne, D.E., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2013. [Google Scholar]
- Hasyim, A.R.; Lase, J.A.; Alwiyah, A.; Suroto, S.; Khairiyah, K.; Hutagalung, M.; Harahap, S.M.; el Ramija, K.; Lestari, D.; Ardiarini, N.; et al. The Effectiveness of Cherry Leaf Extract (Muntingia calabura L.) as an Anti-Bacterial Against Hatchability of Kub Chicken Eggs in Artificial Hatchery. Bul. Peternak. 2021, 45, 214. [Google Scholar] [CrossRef]
- Patterson, P.H.; Ricke, S.C.; Sunde, M.L.; Schaefer, D.M. Hatching Eggs Sanitized with Chlorine Dioxide Foam: Egg Hatchability and Bactericidal. Avian Dis. 1990, 34, 1–6. [Google Scholar] [CrossRef]

| Study (Reference) | Effect on Eggshell Microbial Count * | Effect on Hatchability * |
| [31] | Non-evaluated | No effects |
| [32] | No effects | No effects |
| [33] | - | No effects |
| [34] | Reduced | Increased |
| [35] | Reduced | Non-evaluated |
| [36] | - | Increased |
| [37] | Reduced | Non-evaluated |
| [38] | Reduced | No effects |
| [2] | Reduced | Increased |
| [39] | Non-evaluated | Increased |
| Study (Reference) | Some Reports of Adverse Effects on Embryos and Chicks | |
| [40] | Underweight, underdeveloped and malformed embryos. | |
| [41] | Increased embryonic mortality in the early stage. | |
| [42,43,44] | Reduced chick survival rate in the first post-hatch week. | |
| [45] | Increased embryonic mortality in early, mid and late stages. | |
| [46] | Reduced chick quality score as a result of slow activities and high number of unclosed navels. | |
| Study (Reference) | Sanitizer | Effect on Eggshell Microbial Count * | Effect on Hatchability * |
|---|---|---|---|
| [31] | Ozone | - | No effects |
| [48,49] | Propolis | Reduced | No effects |
| [50] | PotoClean | Reduced | - |
| [32] | Orthophenylphenol | Reduced | No effects |
| Stabilized hydrogen peroxide + peracetic acid + acetic acid | |||
| Sodium hypochlorite + chlorine dioxide + sodium chlorite + ozone + water | |||
| [51] | Ethanol | Reduced | No effects |
| [52] | Propolis | Reduced | No effects |
| [18] | Ultrasonic waves | - | Increased |
| [53] | Bac-D | Reduced | No effects |
| [54] | Quaternary ammoniums + bronopol + biguanide | Reduced | - |
| Quaternary ammoniums + polyhexamethylenebiguanide hydrochloride moiety | |||
| Hydrogen peroxide | |||
| Ammonium chlorides + hydrogen peroxide | |||
| Quaternary ammoniums | |||
| [34] | Propolis | Reduced | Increased |
| Thyme essential oil | |||
| [55] | Quaternary ammoniums + a polyhexamethylenebiguanide hydrochloride moiety | Reduced | Reduced (commercial facility testing) and no effects (lab testing) |
| [56] | Biosentry 904 | - | No effects |
| Egg-Washer-Pro | |||
| Virkon S | |||
| [57] | Trans-cinnamaldehyde | Reduced | - |
| Eugenol | |||
| [7] | Ultraviolet light | - | No effects |
| [58] | Ultrasonic waves | Reduced | No effects |
| [40] | Hydrogen peroxide | Reduced | - |
| Sodium chloride | |||
| Betadine | |||
| Virkon S | |||
| Cumin essential oil | |||
| Oregano essential oil | |||
| Cumin + oregano essential oils | |||
| [59] | Hydrogen peroxide + ultraviolet irradiation | Reduced | - |
| [35] | Sodium dichlorocyanurate | Reduced | - |
| Hydrogen peroxide | Reduced | ||
| Electrolyzed oxidizing water | No effects | ||
| [3] | Virkon S | - | No effects |
| [29] | Propolis | Reduced | - |
| TH4 | |||
| Virkon S | |||
| [60] | Hydrogen peroxide + ultraviolet irradiation | Reduced | - |
| [61] | Hydrogen peroxide | Reduced | Increased |
| TH4 | |||
| Virkon S | |||
| [42] | Colloidal silver | Reduced | No effects |
| [62] | Live yeast | Reduced | Increased |
| [63] | Virocid | Reduced | - |
| [64] | Amino-Steril | - | No effects |
| Oxydion | |||
| Viron FF | |||
| Hydro-Clean | |||
| [65] | Volatile pyrazines | Reduced | - |
| [1] | Hydrogen peroxide | Reduced | Increased |
| [66] | Nanosecond electron beam | Reduced | No effects |
| [67] | Garlic oil | Reduced | Increased |
| [8] | Hydrogen peroxide | Reduced | Reduced |
| TH4 | |||
| [68] | Lysozyme | Reduced | No effects |
| [69] | Chlorine dioxide gas | Reduced | No effects |
| [41] | Garlic extract | - | No effects |
| [70] | Grapefruit juice | Reduced | No effects |
| [37] | Ozone | No effects | - |
| Ultraviolet light | Reduced | ||
| Hydrogen peroxide | No effects | ||
| Peracetic acid | No effects | ||
| [43] | Propolis | No effects | No effects |
| [71] | Olive oil | - | Reduced |
| Albumin | |||
| [72] | Hydrogen peroxide + ultraviolet light | Reduced | Increased |
| [73] | Noni leaf extract | - | No effects |
| [6] | Lemongrass essential oil | Reduced | Reduced |
| Pedestrian tea essential oil | |||
| Lemongrass + pedestrian tea essential oils | |||
| [74] | Vinegar | Reduced | Increased |
| [75] | Ozone | No effects | No effects |
| Ultraviolet light | Reduced | ||
| Hydrogen peroxide | No effects | ||
| Peracetic acid | Reduced | ||
| [2] | Clove essential oil | Reduced | Increased |
| [5] | Propolis | - | Reduced |
| Clove essential oil | No effects | ||
| [76] | Fenugreek seed extract | - | No effects |
| Oat seed extract | |||
| Basil seed extract | |||
| [77] | Eucalyptus alcoholic extract | Reduced | Increased |
| [78] | Polydez | Reduced | - |
| Sterylii AB | No effects | ||
| Virosan | Reduced | ||
| [79] | Dergall | Reduced | No effects |
| [9] | Hydrogen peroxide | - | No effects |
| Low-energy electron irradiation | |||
| Peracetic acid | |||
| Essential oil (not specified) | |||
| [38] | Low-energy electron irradiation | Reduced | No effects |
| [44] | Hydrogen peroxide | Reduced | No effects |
| Ozone | Reduced | ||
| [80] | Hydrogen peroxide | - | No effects |
| [81] | Pulsed ultraviolet light | Reduced | No effects |
| [82] | Oregano essential oil | - | Increased |
| [83] | Ultraviolet light | Reduced | - |
| [39] | Ozone | - | Increased |
| Aldekol | |||
| Virkon S | |||
| [84] | Ozone | Reduced | No effects |
| [85] | Garlic oil | - | Increased |
| [46] | Oregano juice | Reduced | No effects |
| [86] | Slightly acidic electrolysis | Reduced | No effects |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oliveira, G.d.S.; McManus, C.; Salgado, C.B.; dos Santos, V.M. Effects of Sanitizers on Microbiological Control of Hatching Eggshells and Poultry Health during Embryogenesis and Early Stages after Hatching in the Last Decade. Animals 2022, 12, 2826. https://doi.org/10.3390/ani12202826
Oliveira GdS, McManus C, Salgado CB, dos Santos VM. Effects of Sanitizers on Microbiological Control of Hatching Eggshells and Poultry Health during Embryogenesis and Early Stages after Hatching in the Last Decade. Animals. 2022; 12(20):2826. https://doi.org/10.3390/ani12202826
Chicago/Turabian StyleOliveira, Gabriel da Silva, Concepta McManus, Cristiane Batista Salgado, and Vinícius Machado dos Santos. 2022. "Effects of Sanitizers on Microbiological Control of Hatching Eggshells and Poultry Health during Embryogenesis and Early Stages after Hatching in the Last Decade" Animals 12, no. 20: 2826. https://doi.org/10.3390/ani12202826






