1. Introduction
The intestinal mucosa consists of a simple columnar layer of epithelial cells and the lamina propria and muscular mucosa beneath it, with epithelial cells on the mucosal surface creating a barrier between the sometimes hostile external and internal environments [
1,
2]. The largest exchange surface between the body and the external environment is the intestinal epithelial barrier (IEB), whose permeability plays a central role in nutrient intake and body fluid regulation and controlling the infection of pathogens [
3]. However, various factors, such as weaning stress, oxidative stress, and bacterial infection, can impair the IEB by inducing overproduction of pro-inflammatory cytokines and reactive oxygen species (ROS), which subsequently leads to decreases in nutrient digestion and absorption, systemic inflammatory response, and even the death of piglets [
4,
5,
6,
7].
Lipids can serve as an ideal energy source for piglets due to its high energy concentration (2.25 times more than carbohydrates and proteins), and have been usually added to the diet to solve the problem of insufficient energy intake during the weaning stage of piglets [
8]. A number of studies indicated that dietary oil supplementation can improve the growth performance of weaned piglets. For instance, dietary lipid supplementation significantly increased ADG and G:F of piglets from the second week to the fifth week post-weaning, and significantly increased ADG and G:F of the piglets during the first five weeks post-weaning [
9]. Moreover, dietary lard supplementation significantly increased the G:F of piglets from the second week to the third week and the third week post-weaning [
10].
In addition to acting as an energy source, the lipids can also provide a series of essential fatty acids (EFAs), such as linoleic acid and linolenic acid. Previous studies indicated that the EFAs can inhibit the overproduction of intestinal inflammatory mediators, especially various pro-inflammatory cytokines [
11,
12]. For instance, in vitro studies indicated that Eicosapentaenoic acid (EPA) and Docosahexaenoic acid (DHA) can inhibit the production of IL-1β and tumor necrosis factor (TNF)-α by monocytes [
13], and inhibit the production of IL-6 and IL-8 by venous endothelial cells [
14,
15]. Moreover, dietary supplementation of 2% Conjugated linoleic acid (CLA) in second-trimester sows significantly reduced intestinal inflammation, and increased the serum concentrations of immunoglobulin in neonatal pigs upon enterotoxigenic
Escherichia coli challenge [
16].
A previous study indicated that the effect of dietary lipids supplementation on animals may vary widely depending on their sources or chemical structures [
17]. For instance, dietary supplementation of fish oil (one of the major sources of EPA) significantly reduced the production of inflammatory cytokines such as the TNF-α, IL-1β, and IL-6 by macrophages in rodents [
18,
19]. Rice oil contains a large number of compounds with antioxidant properties, such as glutamine, tocopherol, and tocotrienol [
20]. Previous studies have shown that dietary supplementation of rice oil can improve the growth performance of broilers [
21,
22] and reduce the serum cholesterol concentration [
23]. Compared to long-chain fatty acids (LCFAs), medium-chain fatty acids (MCFAs) are more efficiently digested and absorbed in the small intestine of piglets [
24]. Coconut oil is a critical source of the MCFAs, and a previous study indicated that the ADG of the weaned pigs during the first four weeks post-weaning was higher in the coconut oil group than in the butter and corn oil group (the weaner diet contains 8% oil) [
25]. Moreover, both the ADG and ADFI of the weaned pigs were higher in the coconut oil group than in the soybean oil group during the three to four weeks after weaning [
9].
Although, these studies indicated a beneficial effect of different lipid sources on the weaned pigs, the combined effects of different lipid sources are just beginning to be explored. In the present study, we investigated the effects of different lipid sources on growth performance and intestinal health in weaned piglets. Pigs were fed with the corn–soybean meal diet containing 2% SO or oil mixtures (FPRO and CPRO). We observed improved digestibility and intestinal epithelium functions, as well as a reduced production of inflammatory cytokines in the FPRO and CPRO group, which may offer a theoretical basis for rational selection of lipid sources for the weaned pigs as well as other mammalian animals.
2. Materials and Methods
2.1. Experimental Design and Diets
A total of 72 healthy weaned pigs (average 7.80 ± 0.11 kg) with similar body conditions and breeds were randomly divided into three treatment groups with eight replicates per treatment, and three piglets housed in a pen. Pigs were fed corn- and soybean-meal-based diets containing 2% soybean oil (SO), fish–palm–rice oil mixture (FPRO, consisting of 10% fish oil, 50% palm oil, and 40% rice oil), or coconut–palm–rice oil mixture (CPRO, consisting of 5% coconut oil, 80% palm oil, and 15% rice oil), respectively. The composition and nutrition level of the basic diet was prepared in reference to the NRC (2012) pig feeding standard (
Table 1) [
26]. The basal diet was supplemented with 2% soybean oil and the FPRO and CPRO groups were supplemented with 2% FPRO and CPRO, respectively. The feeding management and immunization of the experimental group were carried out according to the field regulations. The pre-feeding period lasted for 3 days. The experiment lasted 28 days. Pigs were supplied with feed and fresh water freely and room temperature was controlled between 25~28 °C, relative humidity 65% ± 5% [
27]. On days 0 and 29 of the experimental period, the feed volume and weight of piglets were recorded to calculate the average daily feed volume (ADFI), average daily gain (ADG), and feed conversion rate (FCR) for the entire experimental period.
2.2. Sample Collection
Feed samples were collected at the beginning of the experiment and stored at −20 °C for nutritional analysis. Fresh fecal samples were collected immediately after excretion on days 25–28 of the experiment, weighed, and 10 mL 10% H
2SO
4 solution was added to every 100 g of fresh fecal matter [
28]. Feed and fecal samples were dried for 2 days at 65 °C, ground through a one-mesh sieve, and then stored at −20 °C until nutrient digestibility analysis. On the morning of the 29th day, we selected one piglet from each cage that is closest to the average weight for blood collection and slaughter, and collected 15 mL of venous blood. After 30 min, the blood was centrifuged at 3500 rpm and 4 °C for 15 min to prepare the serum. After centrifugation, the serum was packed separately (1.5 mL EP tube, 300 μL/tube) and put it on ice. All blood samples were repackaged and then transferred to −20 °C refrigerator for storage. After slaughter, about 2 cm of the middle segment of the duodenum, jejunum, and ileum were taken and rinsed gently with precooled normal saline to remove the contents, and were then immersed in 4% paraformaldehyde for fixation and preservation, which was used to determine the histomorphology [
29]. Next, another segment of the duodenum, jejunum, and ileum was cut open with a scissor, then the contents of the intestine were washed with normal saline and the water was absorbed with filter paper. The intestinal mucosa samples were gently scraped and immediately frozen with liquid nitrogen, and then stored at −80 °C. The cecum and colon chyme were squeezed into a paper cup and quickly collected with the corresponding sampling spoon. The chyme was put into the corresponding cryopreservation tube (sterilized) [
30]. Each kind of intestine was put into four tubes, wrapped with tin foil, and quickly put into liquid nitrogen for preservation.
2.3. Apparent Total Tract Nutrient Digestibility Analysis
Using Acid insoluble ash (AIA) as an endogenous indicator, the nutrient digestibility levels of dried, ground, and fecal samples were analyzed. The contents of dry matter (DM), crude protein (CP), crude fat (EE), and ash were determined according to AOAC [
31]. The energy (GE) was measured directly by a bomb calorimeter.
2.4. Serum Biochemical Indicators
Immunoglobulin (IgA, IgG, IgM), glycolipid substitute (triglyceride, cholesterol, low-density lipoprotein, blood glucose, etc.), inflammatory factor (IL-6, TNF-α, IL-1β), antioxidant indexes (T-AOC, CAT, SOD, GSH-PX, MDA), hormone and growth factors (insulin, IGF-1) were tested in special kits built in Nanjing. The specific operation was carried out according to the kit instructions.
2.5. Intestinal Morphology
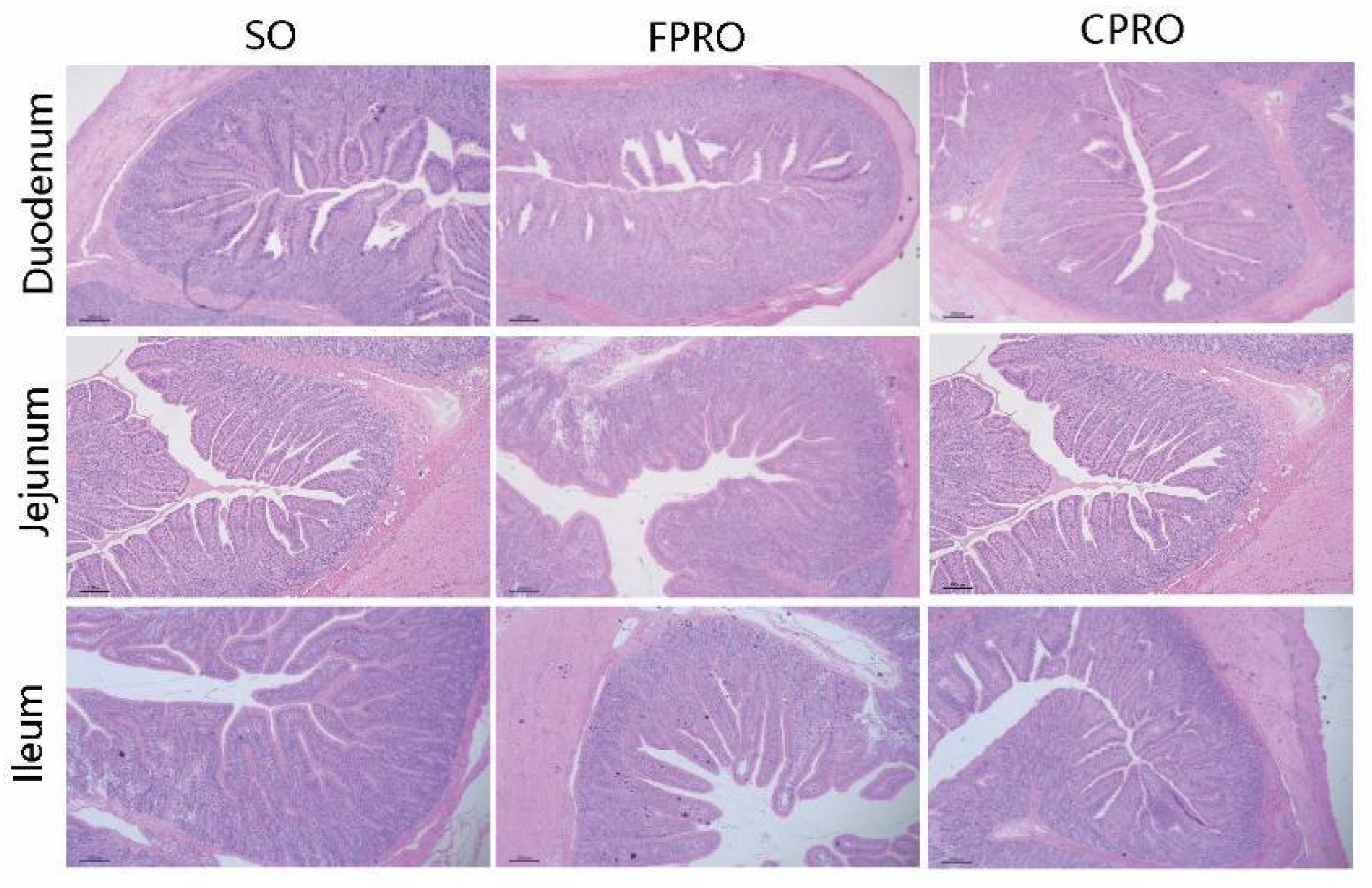
The intestinal segments fixed with 4% paraformaldehyde were dehydrated through a graded series of ethanol and then embedded in paraffin [
32]. The slices (2 µm) were stained with hematoxylin and eosin, observed with Leica DM1000 biological microscope under 100 times of field of vision, selected 10 appropriate fields of vision (complete villi) and photographed, and then measured the villus height and corresponding crypt depth of a single villus [
33].
2.6. Intestinal Disaccharidase Content
The frozen duodenum, jejunum, and ileal mucosa were homogenized with a ratio of 1:9 (w/v) frozen normal saline for 15 min. Homogenates were centrifuged at 3500× g at 4 °C for 15 min, and enzyme activities were measured in the supernatant. The content of disaccharidase (sucrase, lactase, and maltase) in the supernatant was determined with the kit of Nanjing Jiancheng Bioengineering Research Institute.
2.7. Intestinal Flora
We weighed 0.2 g caecum and colon thawed chyme, and used EZNA® Stool DNA kit (Omega Bio Tek, Norcross, GA, USA) to extract total DNA in accordance with the specific operation per the instructions of the kit. The Taqman probe was used for real-time fluorescence quantitative PCR reaction, and RealMasterMix (Probe) was used for detection. The number of copies contained in each sample was calculated by the Ct value and the standard curve with each gram of content as the detection unit. The result was expressed by the common logarithm of the number of bacteria in each gram of content (lg (copies/g)). Construction of the 20 μL reaction system: 8 μL 2 × RealMasterMix, 1 μL of forward and 1 μL of reverse primers (100 nM), 1 μL 20 × Probe Enhancer Solution, 0.3 μL probe, 1 μL DNA, and 7.7 μL of RNase-Free ddH2O. Reaction procedure: pre denaturation at 95 °C for 10 s, denaturation at 95 °C for 15 s, annealing at 60 °C for 25 s, extension at 72 °C for 60 s, 40 cycles.
2.8. Gene Expression
About 0.1 g of the intestinal sample was put into a pre-cooled 1.5 mL centrifuge tube, and 1 mL RNAiso Plus reagent (Takara, Kyoto, Japan) was added. The samples were homogenized on ice until dissolved. After standing for 5 min, the samples were centrifuged for 15 min at 12,000×
g/4 °C. An amount of 400 μL supernatant was absorbed into a new aseptic centrifuge tube, and isopropyl alcohol was added in the same volume, inverted, and mixed gently. After standing on ice for 10 min, the samples were centrifuged at 12,000×
g/4 °C for 10 min to precipitate RNA. The supernatant was discarded, and 1 mL of pre-cooled 75% ethanol (freshly mixed with DEPC water) was added, then the suspended RNA was precipitated and centrifuged for 5 min at 7500×
g/4 °C. Discard the supernatant and leave it at room temperature for 1–2 min to dry the precipitate. When the RNA was slightly dry, 40 μL DEPC water was added, and the pipette gun was used to gently blow and suck 50 times to fully dissolve the RNA. By using a spectrophotometer (NanoDrop 2000, Thermo Fisher Scientific, Inc., Waltham, MA, USA) to detect the purity and concentration of total RNA, samples with an OD260/OD280 ratio of 1.8–2.0 were considered suitable. Total RNA was used as template to synthesize cDNA by reverse transcription kit, referring to the manual of PrimeScript™ RT Reagent (Takara, Japan) for specific operation. Reverse transcription consists of two steps: 42 °C, 2 min (removal of genomic DNA); 37 °C for 15 min, 85 °C for 5 s. Based on the procedure and reaction system of the TB Green™ Premix Ex Taq™ II (TaKaRa, Japan) kit, QuanStudio™ 6 Flex real-time quantitative PCR was used to detect the expression of target genes in the small intestine. The reaction system of qPCRas follows: TB Green Premix Ex Taq II (Tli RNaseH Plus, 2×) 5 μL, ROX ReferenceDye II (50×) 0.2 μL, forward primer 0.4 μL, reverse primer 0.4 μL, cDNA 1 μL, Double Steaming Water 3 μL. The reaction procedure as follows: 95 °C predenaturation for 30 s, 95 °C denaturation for 5 s, 60 °C annealing for 34 s, 40 cycles. Quantitative results used the GAPDH gene as the internal reference gene. The relative expression levels of each target gene in tissues were calculated by2
−ΔΔCt [
34].
2.9. Data Statistics and Analysis
After all experimental data were analyzed by Microsoft Excel 2019, SPSS 24.0 statistical software was used to perform one-way ANOVA on the experimental data of the three treatment groups. When the differences were significant, Duncan’s multiple comparisons were performed. All experimental data were expressed as “mean ± standard error”, p < 0.05 was considered significant, and 0.05 ≤ p ≤ 0.10 was considered a trend.
4. Discussion
Fat and oil are commonly considered as effective energy sources for animals, including pigs [
35]. The digestion and metabolism of lipids are dependent on their sources, dietary proportions, and intermolecular distribution of saturated and unsaturated fatty acids [
36,
37]. Previous studies on the weaned pigs (5 to 20 kg) indicated that dietary fat supplementation reduced feed intake, but significantly improved the feed conversion efficiency [
8]. Compared to other vegetable oils, palm oil and coconut oil are mainly composed of MCFA (the fatty acids below 16 carbon atoms in coconut oil account for 66.2% of the total fatty acids) [
38], and their digestibility levels range from 80 to 95% [
39]. Long-chain fatty acids can delay stomach emptying and increase feelings of fullness, which may decrease the feed intake for pigs [
40]. In the present study, we discussed the effects of different lipid mixtures on the growth performance and intestinal health of weaned piglets. As expected, the digestibility levels of DM, GE, and ash were higher in the FPRO and CPRO group than in the SO. Moreover, the FPRO group had a higher digestibility of EE than other groups, which may be associated with the presence of a considerable amount of unsaturated fatty acids in the fish oil [
41].
Immunoglobulins, also known as antibodies, are glycoprotein molecules produced by plasma cells, which act as a critical part of the immune response by specifically recognizing and binding to particular antigens, such as bacteria or viruses, and aiding in their destruction [
42]. In this study, dietary supplementation of FPRO can significantly increase the serum IgA concentration, indicating an elevated immunity in the pigs. Pro-inflammatory cytokines, such as IL-1β and IL-6, can mediate host inflammatory processes and produce a rapid immune response after infection with pathogenic microorganisms [
43]. In this study, the serum IL-1β and IL-6 concentrations were decreased in the FPRO and CPRO group. This is probably due to the fish and coconut oil in the two lipid mixtures, as previous studies indicated that dietary supplementation of coconut oil and fish oil can reduce the occurrence of inflammatory responses in the weaned pigs [
44,
45]. Interestingly, the serum activity of CAT was also higher in the FPRO and CPRO group than in the SO group. CAT, as a key enzyme in the antioxidant defense system, plays an important role in clearing hydrogen peroxide in the body and avoiding oxidative stress [
46]. A previous study indicated that palm oil contains a large number of antioxidants, which can improve the activity of antioxidant enzymes [
47], whereas rice oil contains a large number of compounds with antioxidant properties, such as glutamine, tocopherol, and tocotrienol [
21].
The digestion and absorption of various nutrients are basically carried out in the small intestine. Villus height and crypt depth of intestinal mucosal epithelium are closely related to the absorption capacity of intestinal mucosa [
48]. In this study, the villus height of the jejunum and ileum in the FPRO and CPRO groups was higher than in the SO group, respectively, and the crypt depth in FPRO and CPRO groups was also lower than in the SO group. Coconut oil and palm oil are rich in medium-chain fatty acids [
49], MCFA can be directly utilized by intestinal epithelial cells for energy production, thereby helping to support the integrity of the piglets’ intestines [
50]. Previous studies have shown that MCFA supplementation for the weaned pigs can improve the intestinal morphology and integrity, as indicated by the significant increase in intestinal villus length, and decrease in crypt depth and the number of intraepithelial lymphocytes [
51]. Fish oil has also been reported to increase the villus height and villus height/crypt depth ratio in the weaned pigs [
6]. Changes of intestinal morphology are usually accompanied by brush margins of intestinal epithelium digestive enzyme activity [
52]. In this study, the maltase and lactase activities of duodenal and jejunal mucosa in FPRO and CPRO groups were higher than that in the SO group.
The gut microbiota is an important component of the gut ecosystem, and is closely related to the integrity of intestinal epithelial cells, the digestion and metabolism of nutrients, immune responses, and diseases [
17,
53]. Some difficult-to-digest fats can reach the hindgut and be a source of nutrients for bacteria, which may regulate the gut bacteria balance [
54]. Different sources of oils have different compositions, so their effects on gut microbes are also different [
55]. In this study, the abundance of lactobacillus in the colon and cecum in the FPRO group was higher than in the SO group. Moreover, the abundance of cecal lactobacillus i and colonic bifidobacterium in the CPRO group was also higher than in the SO group. This result is consistent with previous studies that found that dietary supplementation of rice and coconut oil significantly increased the abundances of beneficial bacteria such as
bifidobacterium and
Lactobacillus in the intestine [
56,
57].
Tight junctions play a crucial role in maintaining intestinal permeability, consisting of transmembrane barrier proteins (such as claudin and occludin), cytoplasmic scaffold proteins (such as the ZO family), and adhesion molecules [
58]. A previous study indicated that fish oil can enhance the expression levels of occludin and claudin-1 in the weaned pigs [
6]. In this study, the expression levels of ZO-1, Claudin-1, and Occludin in the duodenum and ileum were higher in the FPRO group than in the SO and CPRO group. Moreover, the expression levels of nutrient transporters such as the CAT-1, PepT1, FATP1, and SGLT1 were higher in the FPRO group than in the SO group. PepT1 is a small peptide transporter, while CAT1 is mainly responsible for the transport of basic amino acids [
59,
60]. GLUT-2 is one of the major transporters of glucose absorption, while FATP-1 is responsible for the transport of long-chain fatty acids into intestinal cells [
61]. These results indicated improved epithelium functions in the weaned pigs upon FPRO supplementation. IL-1β is considered to be an important pro-inflammatory cytokine involved in innate immunity; in general, down-regulating the expression of IL-1β protein in intestinal tissue can reduce inflammatory damage [
62]. NF-κB is a promoter of inflammation-associated tumors, and it is essential for promoting inflammation-associated cancers [
63]. Previous studies indicated that dietary supplementation of coconut oil and fish oil can reduce the occurrence of inflammatory responses in the weaned pigs [
44,
45]. In this study, the expression levels of IL-1β in the jejunum and ileum were down-regulated in the FPRO and CPRO group. Moreover, the expression levels of duodenal ERK1 and ileal NF-κB were decreased in the FPRO and CPRO groups, respectively. These results may suggest an anti-inflammatory effect of a lipid mixture containing the fish and coconut oil.