Hesperidin Helps Improve the Intestinal Structure, Maintain Barrier Function, and Reduce Inflammation in Yellow-Feathered Broilers Exposed to High Temperatures
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Animal Rearing and Experiments
2.3. Sample Collection
2.4. Intestinal Permeability Test
2.5. Serum Cytokine Levels
2.6. Intestinal Morphology Analysis
2.7. Real-Time PCR
2.8. Statistical Analysis
3. Results
3.1. Impact of Hesperidin on the Growth Performance of Yellow-Feathered Broilers in High Temperatures
3.2. Effect of Hesperidin on Serum Hormone Concentration of Yellow-Feathered Broilers under High Temperature
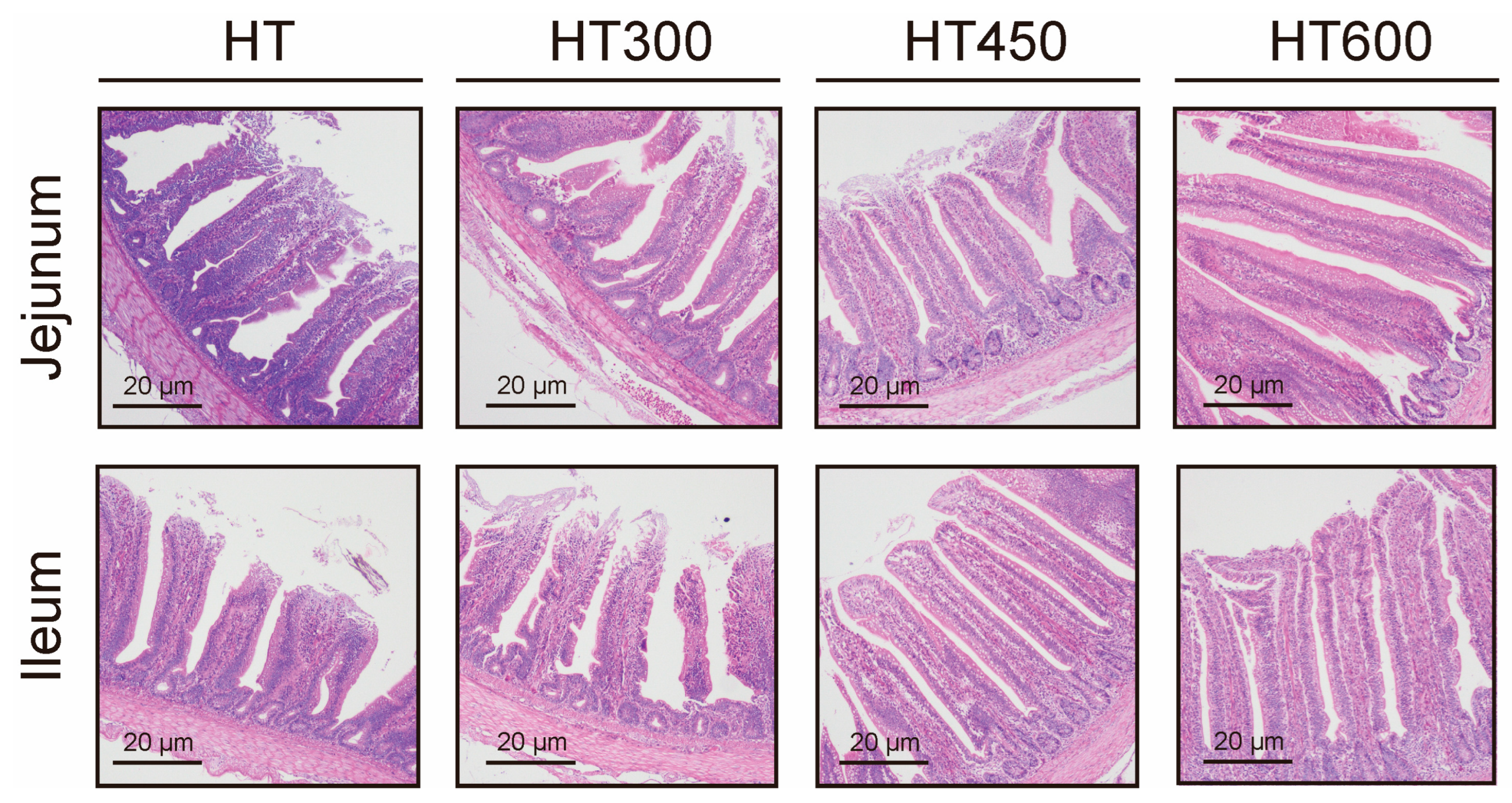
3.3. Impact of Hesperidin on the Structure of the Jejunum and Ileum in Yellow-Feathered Broilers Exposed to High Temperatures
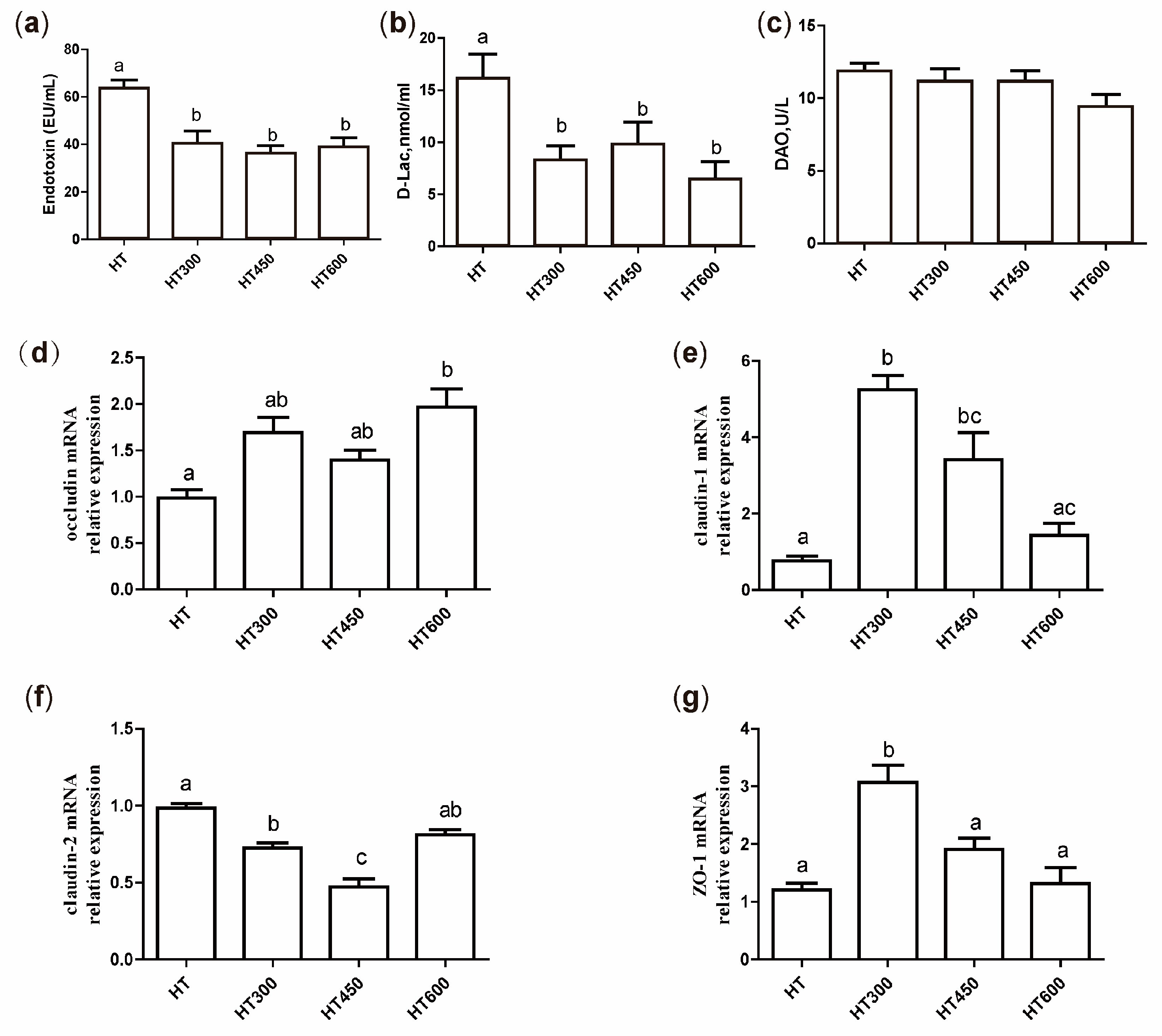
3.4. Impact of Hesperidin on Gut Permeability in Yellow-Feathered Broiler Chickens Exposed to High Temperatures
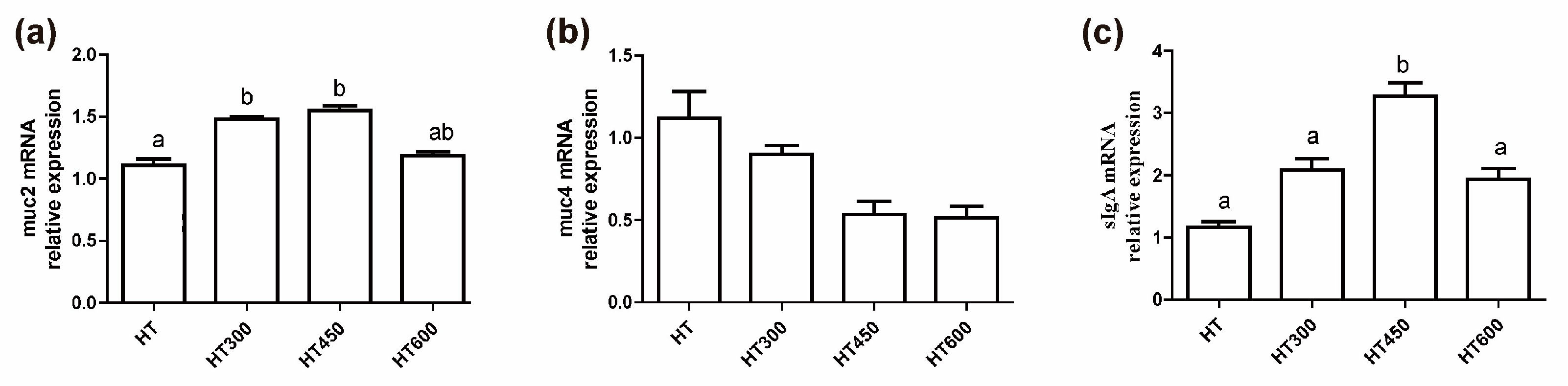
3.5. Impact of Hesperidin on the Intestinal Mucosa of Yellow-Feathered Broiler Chickens during Periods of Heat Stress
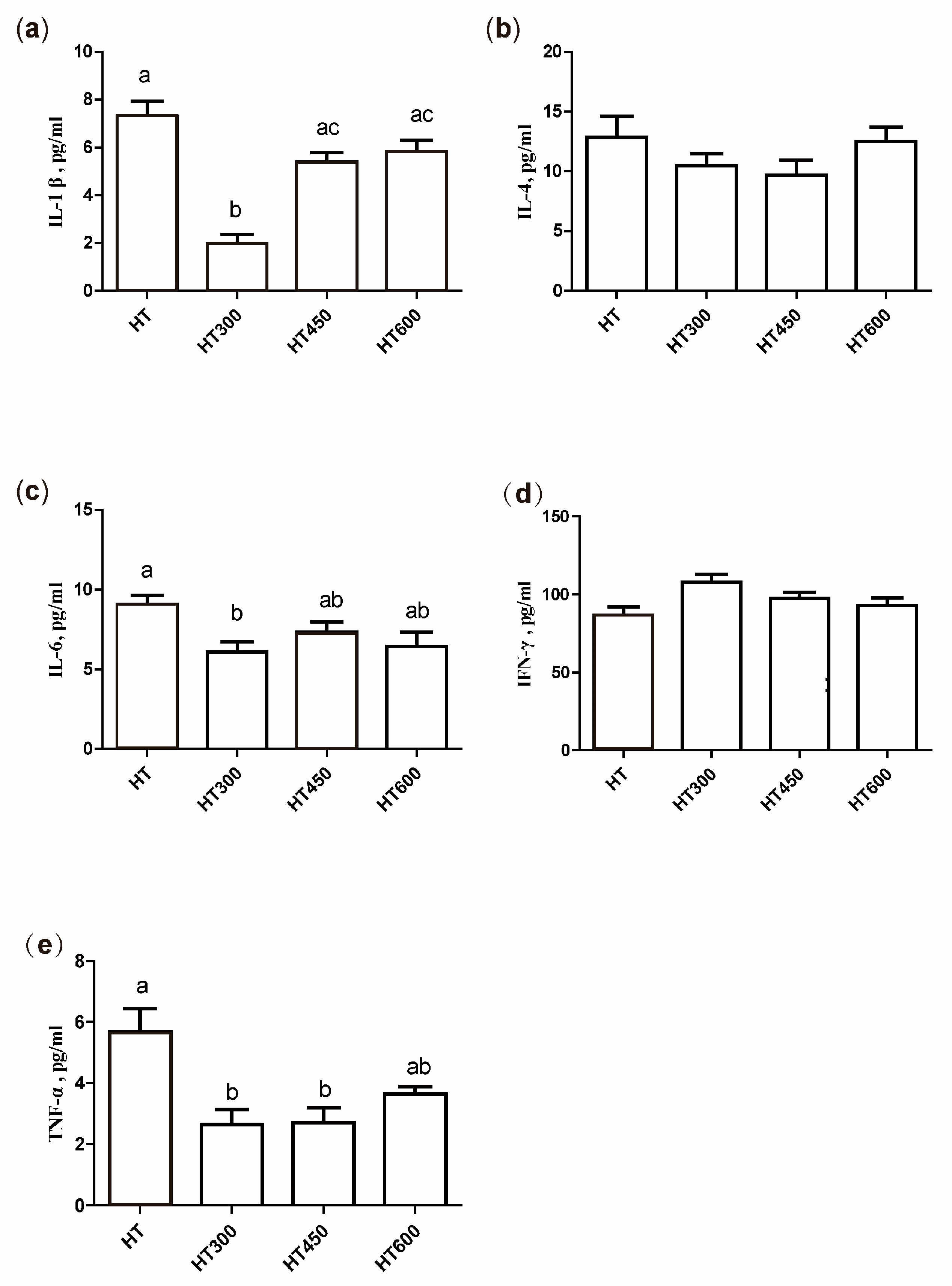
3.6. Impact of Hesperidin on Cytokine Levels in the Serum of Yellow-Feathered Broilers Exposed to High Temperatures
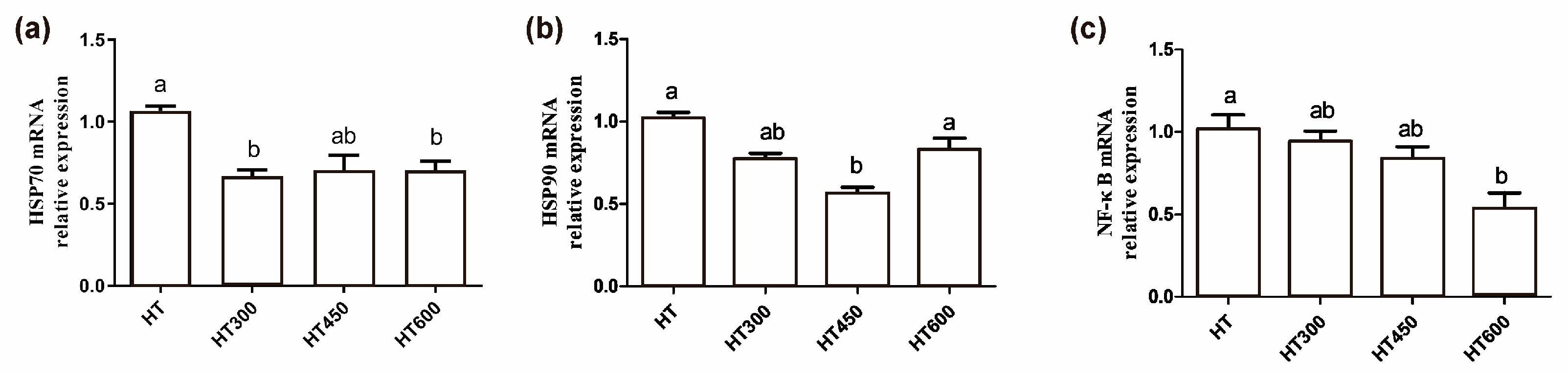
3.7. Impact of Hesperidin on the HSP70, HSP90, and NF-κB mRNA Levels in the Jejunum of Yellow-Feathered Broilers Exposed to High Temperatures
4. Discussion
4.1. The Influence of Hesperidin on the Growth Performance of Yellow-Feathered Broilers in High Temperatures
4.2. The Influence of Hesperidin on the Serum Hormone Concentration of Yellow-Feathered Broilers under High-Temperature
4.3. Impact of Hesperidin on Intestinal Injury in Yellow-Feathered Broiler Chickens Exposed to High Temperatures
4.4. The Mechanism by Which Hesperidin Facilitates the Repair of Intestinal Injury Induced by Heat Stress
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lara, L.J.; Rostagno, M.H. Impact of Heat Stress on Poultry Production. Animals 2013, 3, 356–369. [Google Scholar] [CrossRef]
- Vandana, G.D.; Sejian, V.; Lees, A.M.; Pragna, P.; Silpa, M.V.; Maloney, S.K. Heat stress and poultry production: Impact and amelioration. Int. J. Biometeorol. 2021, 65, 163–179. [Google Scholar] [CrossRef] [PubMed]
- Alhotan, R.A.; Al Sulaiman, A.R.; Alharthi, A.S.; Abudabos, A.M. Protective influence of betaine on intestinal health by regulating inflammation and improving barrier function in broilers under heat stress. Poult. Sci. 2021, 100, 101337. [Google Scholar] [CrossRef]
- Hu, R.Z.; He, Y.J.; Arowolo, M.A.; Wu, S.S.; He, J. Polyphenols as Potential Attenuators of Heat Stress in Poultry Production. Antioxidants 2019, 8, 67. [Google Scholar] [CrossRef] [PubMed]
- Rostagno, M.H. Effects of heat stress on the gut health of poultry. J. Anim. Sci. 2020, 98, skaa090P. [Google Scholar] [CrossRef] [PubMed]
- Lan, R.; Zhao, Z.; Li, S.; An, L. Sodium butyrate as an effective feed additive to improve performance, liver function, and meat quality in broilers under hot climatic conditions. Poult. Sci. 2020, 99, 5491–5500. [Google Scholar] [CrossRef]
- Sun, H.; Jiang, R.; Xu, S.; Zhang, Z.; Xu, G.; Zheng, J.; Qu, L. Transcriptome responses to heat stress in hypothalamus of a meat-type chicken. J. Anim. Sci. Biotechnol. 2015, 6, 6. [Google Scholar] [CrossRef]
- Saleh, K.M.M.; Al-Zghoul, M.B. Effect of Acute Heat Stress on the mRNA Levels of Cytokines in Broiler Chickens Subjected to Embryonic Thermal Manipulation. Animals 2019, 9, 499. [Google Scholar] [CrossRef] [PubMed]
- Seifi, K.; Rezaei, M.; Yansari, A.T.; Riazi, G.H.; Zamiri, M.J.; Heidari, R. Saturated fatty acids may ameliorate environmental heat stress in broiler birds by affecting mitochondrial energetics and related genes. J. Therm. Biol. 2018, 78, 1–9. [Google Scholar] [CrossRef]
- Grotz, M.R.; Deitch, E.A.; Ding, J.; Xu, D.; Huang, Q.; Regel, G. Intestinal cytokine response after gut ischemia: Role of gut barrier failure. Ann. Surg. 1999, 229, 478–486. [Google Scholar] [CrossRef]
- Pyrzynska, K. Hesperidin: A Review on Extraction Methods, Stability and Biological Activities. Nutrients 2022, 14, 2387. [Google Scholar] [CrossRef]
- Buzdağlı, Y.; Eyipınar, C.D.; Kacı, F.N.; Tekin, A. Effects of hesperidin on anti-inflammatory and antioxidant response in healthy people: A meta-analysis and meta-regression. Int. J. Environ. Health Res. 2023, 33, 1390–1405. [Google Scholar] [CrossRef] [PubMed]
- Hager-Theodorides, A.L.; Massouras, T.; Simitzis, P.E.; Moschou, K.; Zoidis, E.; Sfakianaki, E.; Politi, K.; Charismiadou, M.; Goliomytis, M.; Deligeorgis, S. Hesperidin and Naringin Improve Broiler Meat Fatty Acid Profile and Modulate the Expression of Genes Involved in Fatty Acid β-oxidation and Antioxidant Defense in a Dose Dependent Manner. Foods 2021, 10, 739. [Google Scholar] [CrossRef] [PubMed]
- Kamboh, A.A.; Zhu, W.Y. Individual and combined effects of genistein and hesperidin supplementation on meat quality in meat-type broiler chickens. J. Sci. Food Agric. 2013, 93, 3362–3367. [Google Scholar] [CrossRef] [PubMed]
- Kamboh, A.A.; Hang, S.Q.; Bakhetgul, M.; Zhu, W.Y. Effects of genistein and hesperidin on biomarkers of heat stress in broilers under persistent summer stress. Poult. Sci. 2013, 92, 2411–2418. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Li, S.; Arowolo, M.A.; Yu, Q.; Chen, F.; Hu, R.; He, J. Effect of resveratrol on growth performance, rectal temperature and serum parameters of yellow-feather broilers under heat stress. Anim. Sci. J. Nihon Chikusan Gakkaiho 2019, 90, 401–411. [Google Scholar] [CrossRef]
- He, S.; Yu, Q.; He, Y.; Hu, R.; Xia, S.; He, J. Dietary resveratrol supplementation inhibits heat stress-induced high-activated innate immunity and inflammatory response in spleen of yellow-feather broilers. Poult. Sci. 2019, 98, 6378–6387. [Google Scholar] [CrossRef]
- Hu, R.; Yang, X.; Gong, J.; Lv, J.; Yuan, X.; Shi, M.; Fu, C.; Tan, B.; Fan, Z.; Chen, L.; et al. Patterns of alteration in boar semen quality from 9 to 37 months old and improvement by protocatechuic acid. J. Anim. Sci. Biotechnol. 2024, 15, 78. [Google Scholar] [CrossRef]
- Quinteiro-Filho, W.M.; Rodrigues, M.V.; Ribeiro, A.; Ferraz-de-Paula, V.; Pinheiro, M.L.; Sá, L.R.; Ferreira, A.J.; Palermo-Neto, J. Acute heat stress impairs performance parameters and induces mild intestinal enteritis in broiler chickens: Role of acute hypothalamic-pituitary-adrenal axis activation. J. Anim. Sci. 2012, 90, 1986–1994. [Google Scholar] [CrossRef]
- Akbarian, A.; Michiels, J.; Degroote, J.; Majdeddin, M.; Golian, A.; De Smet, S. Association between heat stress and oxidative stress in poultry; mitochondrial dysfunction and dietary interventions with phytochemicals. J. Anim. Sci. Biotechnol. 2016, 7, 37. [Google Scholar] [CrossRef]
- Fang, X.; Nong, K.; Qin, X.; Liu, Z.; Gao, F.; Jing, Y.; Fan, H.; Wang, Z.; Wang, X.; Zhang, H. Effect of purple sweet potato-derived anthocyanins on heat stress response in Wenchang chickens and preliminary mechanism study. Poult. Sci. 2023, 102, 102861. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Gao, X.; Wu, J.; Chen, M. The Correlation between Endotoxin, D-Lactate, and Diamine Oxidase with Endoscopic Activity in Inflammatory Bowel Disease. Dis. Markers 2022, 2022, 9171436. [Google Scholar] [CrossRef]
- Mani, V.; Harris, A.J.; Keating, A.F.; Weber, T.E.; Dekkers, J.C.; Gabler, N.K. Intestinal integrity, endotoxin transport and detoxification in pigs divergently selected for residual feed intake. J. Anim. Sci. 2013, 91, 2141–2150. [Google Scholar] [CrossRef]
- Sun, X.Q.; Fu, X.B.; Zhang, R.; Lu, Y.; Deng, Q.; Jiang, X.G.; Sheng, Z.Y. Relationship between plasma D(-)-lactate and intestinal damage after severe injuries in rats. World J. Gastroenterol. 2001, 7, 555–558. [Google Scholar] [CrossRef]
- Wolvekamp, M.C.; de Bruin, R.W. Diamine oxidase: An overview of historical, biochemical and functional aspects. Dig. Dis. 1994, 12, 2–14. [Google Scholar] [CrossRef]
- Shan, Q.; Ma, F.; Wei, J.; Li, H.; Ma, H.; Sun, P. Physiological Functions of Heat Shock Proteins. Curr. Protein Pept. Sci. 2020, 21, 751–760. [Google Scholar] [CrossRef] [PubMed]
- Camilleri, M.; Madsen, K.; Spiller, R.; Greenwood-Van Meerveld, B.; Verne, G.N. Intestinal barrier function in health and gastrointestinal disease. Neurogastroenterol. Motil. 2012, 24, 503–512. [Google Scholar] [CrossRef]
- Ma, N.; Guo, P.; Zhang, J.; He, T.; Kim, S.W.; Zhang, G.; Ma, X. Nutrients Mediate Intestinal Bacteria-Mucosal Immune Crosstalk. Front. Immunol. 2018, 9, 5. [Google Scholar] [CrossRef]
- Zhang, C.; Zhao, X.H.; Yang, L.; Chen, X.Y.; Jiang, R.S.; Jin, S.H.; Geng, Z.Y. Resveratrol alleviates heat stress-induced impairment of intestinal morphology, microflora, and barrier integrity in broilers. Poult. Sci. 2017, 96, 4325–4332. [Google Scholar] [CrossRef] [PubMed]
- Hu, R.; Wu, S.; Li, B.; Tan, J.; Yan, J.; Wang, Y.; Tang, Z.; Liu, M.; Fu, C.; Zhang, H.; et al. Dietary ferulic acid and vanillic acid on inflammation, gut barrier function and growth performance in lipopolysaccharide-challenged piglets. Anim. Nutr. 2022, 8, 144–152. [Google Scholar] [CrossRef]
- Suzuki, T. Regulation of the intestinal barrier by nutrients: The role of tight junctions. Anim. Sci. J. Nihon Chikusan Gakkaiho 2020, 91, e13357. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Liu, B.; Wang, Y.; Huang, X.; Yan, Z.; Jiang, Q.; Chen, Q. Ellagic Acid Improves Antioxidant Capacity and Intestinal Barrier Function of Heat-Stressed Broilers via Regulating Gut Microbiota. Animals 2022, 12, 1180. [Google Scholar] [CrossRef]
- Heled, Y.; Fleischmann, C.; Epstein, Y. Cytokines and their role in hyperthermia and heat stroke. J. Basic. Clin. Physiol. Pharmacol. 2013, 24, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, T. The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harb. Perspect. Biol. 2009, 1, a001651. [Google Scholar] [CrossRef]
- Poma, P. NF-κB and Disease. Int. J. Mol. Sci. 2020, 21, 9181. [Google Scholar] [CrossRef] [PubMed]

| Item | Amount |
|---|---|
| Ingredient (%) | |
| Corn | 59.50 |
| Soybean meal | 24.70 |
| Sunflower meal | 3.00 |
| Soybean oil | 6.20 |
| Corn gluten meal | 2.00 |
| Dicalcium phosphate | 1.10 |
| Limestone | 1.40 |
| Methionine | 0.2 |
| Salt | 0.30 |
| Vitamin-mineral-premix a | 1.6 |
| Total | 100.00 |
| Nutrient level b | |
| ME/(MJ/kg) | 13.00 |
| CP (%) | 19.00 |
| Ca (%) | 0.85 |
| Available P (%) | 0.40 |
| Lysine (%) | 1.1 |
| Methionine (%) | 0.50 |
| Item | HT | HT300 | HT450 | HT600 | SEM | p-Value |
|---|---|---|---|---|---|---|
| Initial weight | 390.33 | 391.83 | 393.00 | 390.66 | 12.81 | 0.879 |
| ADG, g | 41.93 | 42.18 | 41.91 | 41.22 | 2.48 | 0.73 |
| ADFI, g | 98.73 | 96.3 | 96.73 | 96.25 | 2.48 | 0.37 |
| F/G | 2.33 | 2.28 | 2.32 | 2.33 | 0.11 | 0.70 |
| Mortality, % | 98.75 | 99.58 | 99.16 | 99.16 | 0.30 | 0.14 |
| Items | HT | HT300 | HT450 | HT600 | SEM | p-Value |
|---|---|---|---|---|---|---|
| Jejunum | ||||||
| villus height (um) | 75.31 | 92.94 | 104.84 | 88.95 | 56.73 | 0.34 |
| Crypt depth (um) | 119.30 a | 102.10 ab | 88.60 b | 108.60 ab | 3.48 | 0.008 |
| V/C | 6.42 b | 9.51 ab | 12.0 a | 8.17 ab | 0.73 | 0.04 |
| Ileum | ||||||
| villus height (um) | 98.75 | 123.65 | 108.81 | 122.64 | 45.68 | 0.16 |
| Crypt depth (um) | 16.24 a | 10.46 b | 10.27 b | 13.07 ab | 8.19 | 0.02 |
| V/C | 6.63 b | 12.16 a | 10.69 a | 9.62 ab | 0.63 | 0.006 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, S.; Bian, G.; Guo, Y.; Guo, J. Hesperidin Helps Improve the Intestinal Structure, Maintain Barrier Function, and Reduce Inflammation in Yellow-Feathered Broilers Exposed to High Temperatures. Animals 2024, 14, 2585. https://doi.org/10.3390/ani14172585
He S, Bian G, Guo Y, Guo J. Hesperidin Helps Improve the Intestinal Structure, Maintain Barrier Function, and Reduce Inflammation in Yellow-Feathered Broilers Exposed to High Temperatures. Animals. 2024; 14(17):2585. https://doi.org/10.3390/ani14172585
Chicago/Turabian StyleHe, Shaoping, Guozhi Bian, Yuming Guo, and Jiyu Guo. 2024. "Hesperidin Helps Improve the Intestinal Structure, Maintain Barrier Function, and Reduce Inflammation in Yellow-Feathered Broilers Exposed to High Temperatures" Animals 14, no. 17: 2585. https://doi.org/10.3390/ani14172585
APA StyleHe, S., Bian, G., Guo, Y., & Guo, J. (2024). Hesperidin Helps Improve the Intestinal Structure, Maintain Barrier Function, and Reduce Inflammation in Yellow-Feathered Broilers Exposed to High Temperatures. Animals, 14(17), 2585. https://doi.org/10.3390/ani14172585





