L-phenylalanine Increased Gut Hormone Secretion through Calcium-Sensing Receptor in the Porcine Duodenum
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
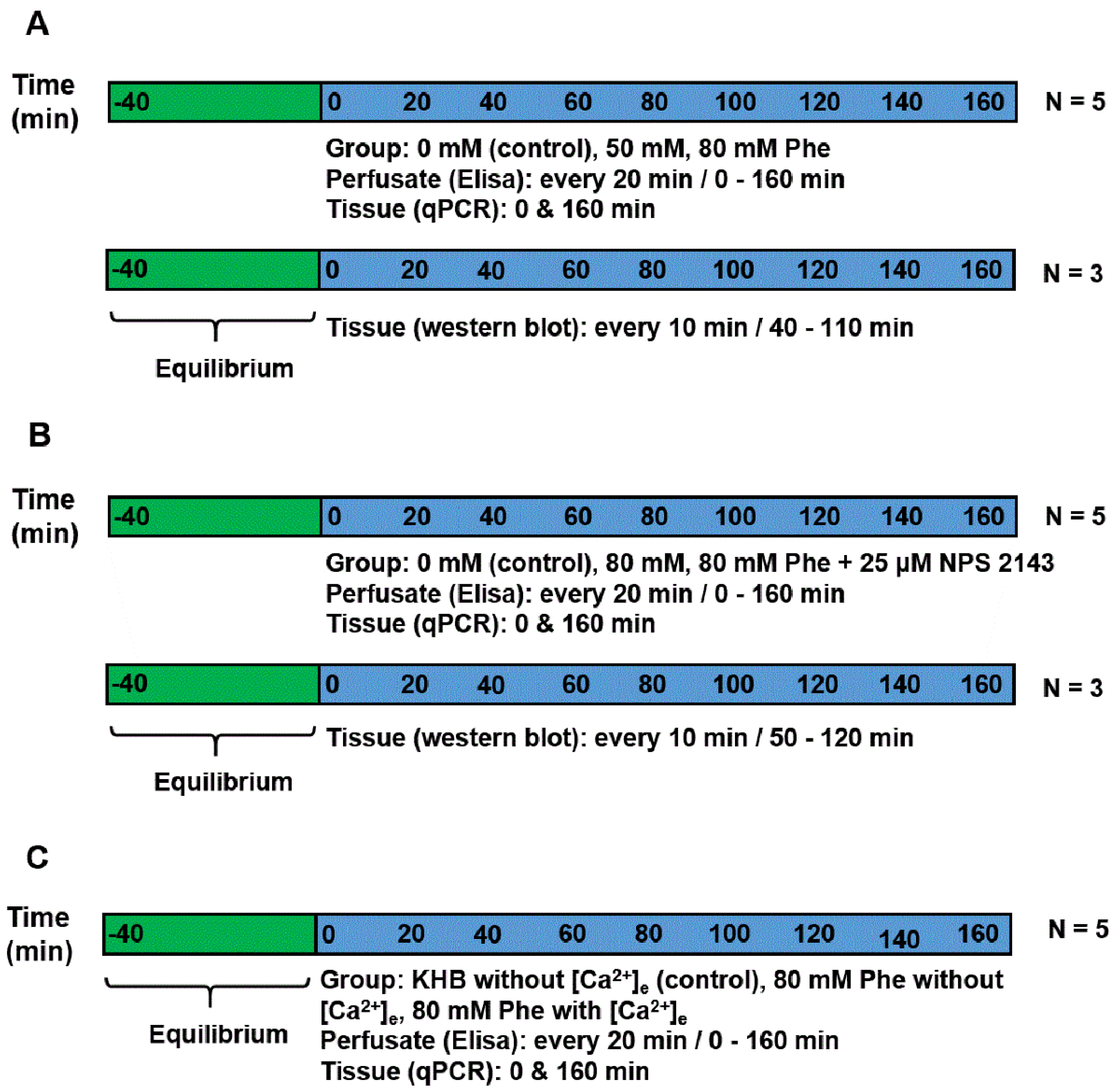
2.1. Sampling and Perfusion of the Pig Duodenum
2.2. Total RNA Extraction, Protocol of cDNA Formation, and RT-qPCR
2.3. Determination of Gut Hormone Concentration
2.4. Western Blotting Analysis
2.5. Statistical Analysis
3. Results
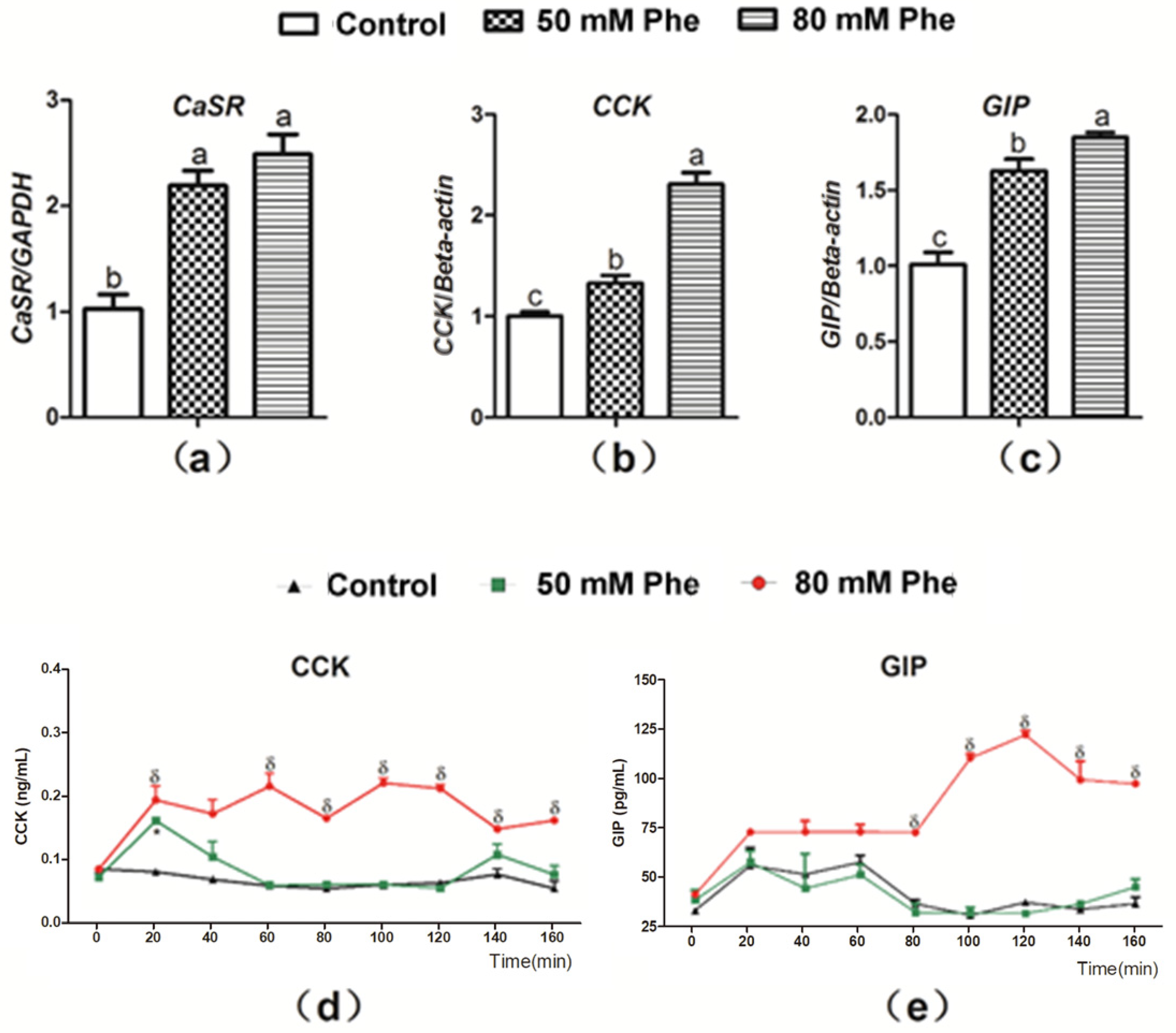
3.1. CaSR mRNA Expression and Gut Hormone Secretion after Treatment with Different Phe Concentrations
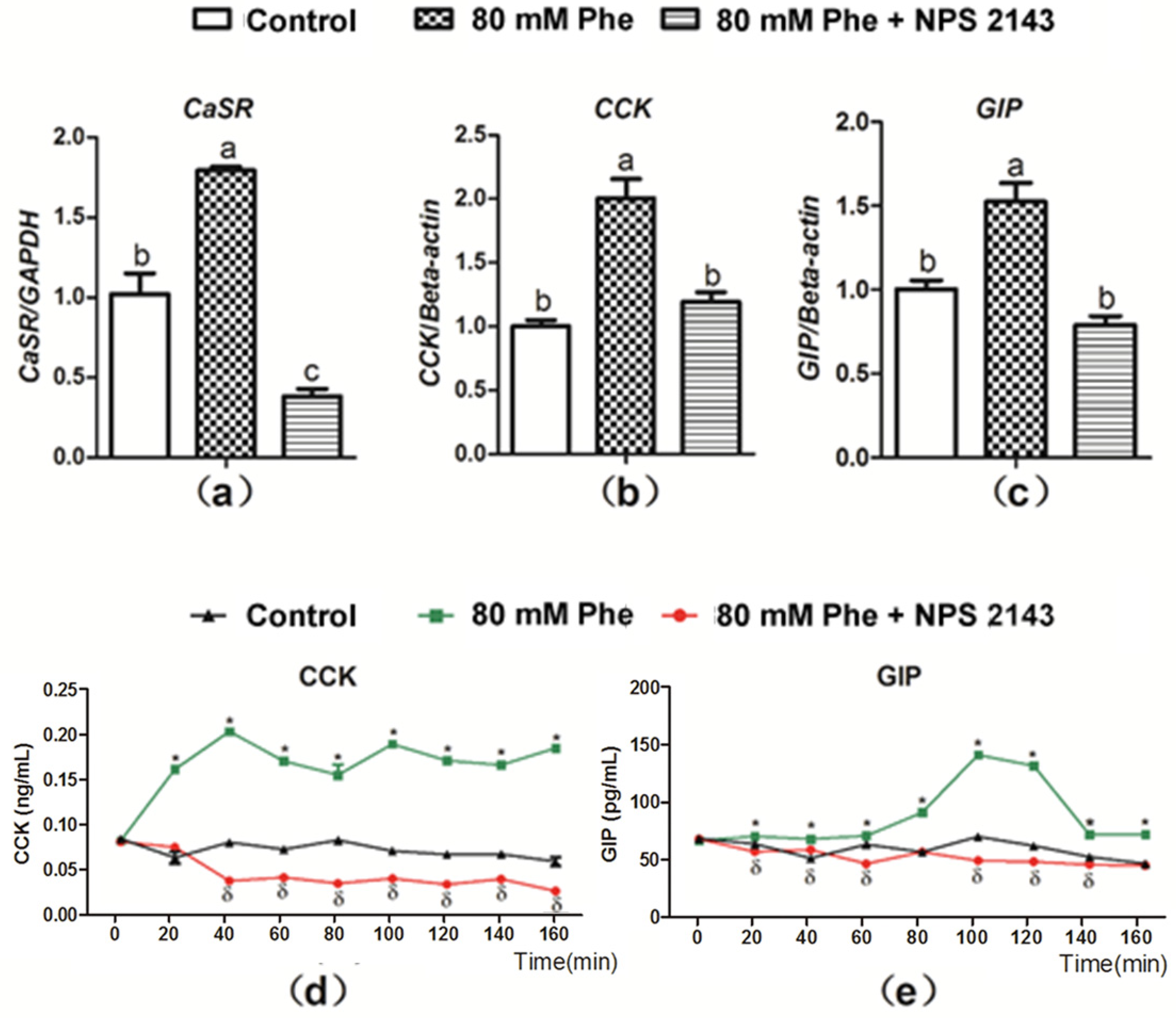
3.2. Phe-Regulated Expression and Secretion of Gut Hormones Was Mediated Via a CaSR-Dependent Mechanism
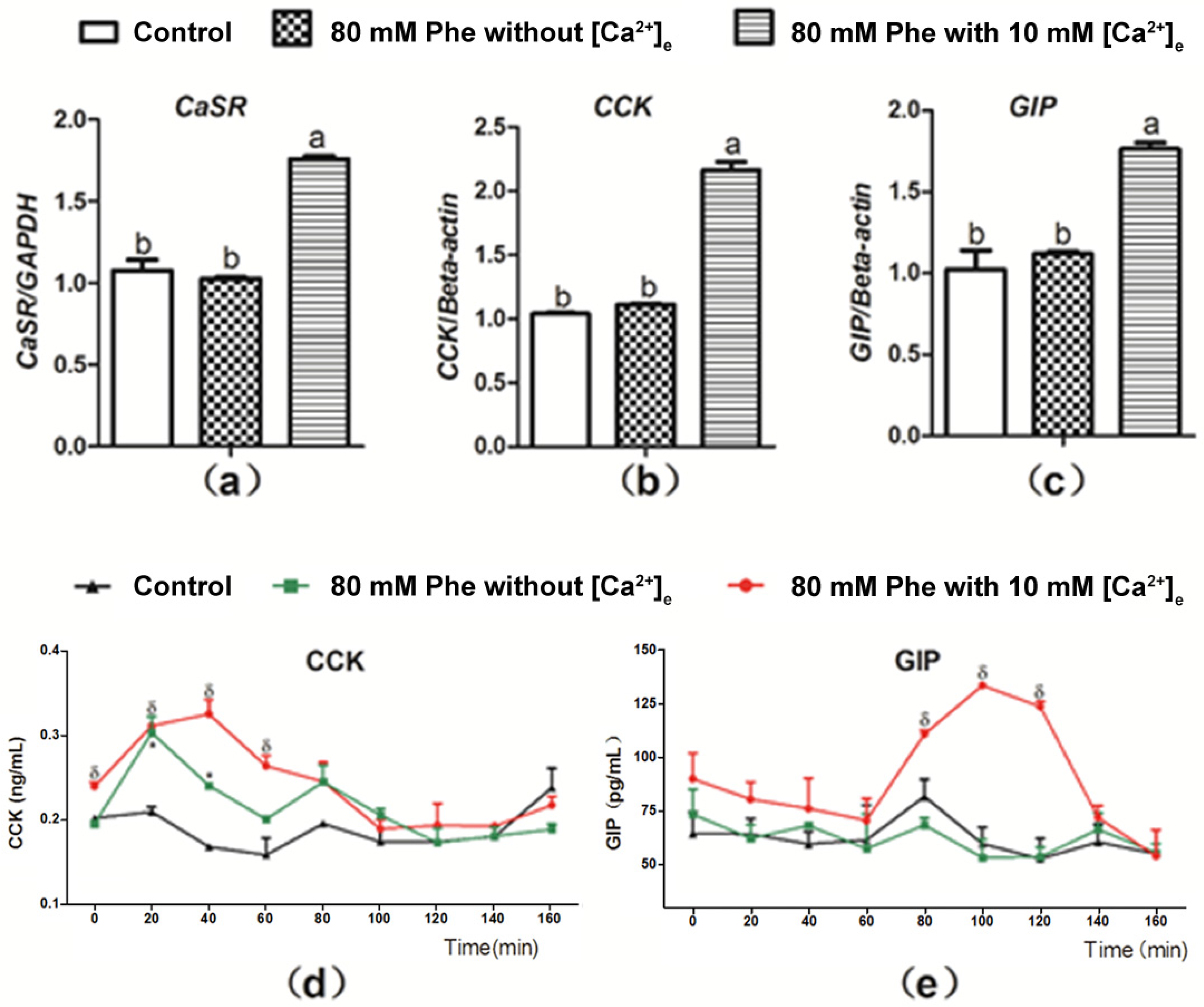
3.3. [Ca2+]e Modulated Phe-Induced CaSR and Gut Hormone Gene Expression and Gut Hormone Secretion
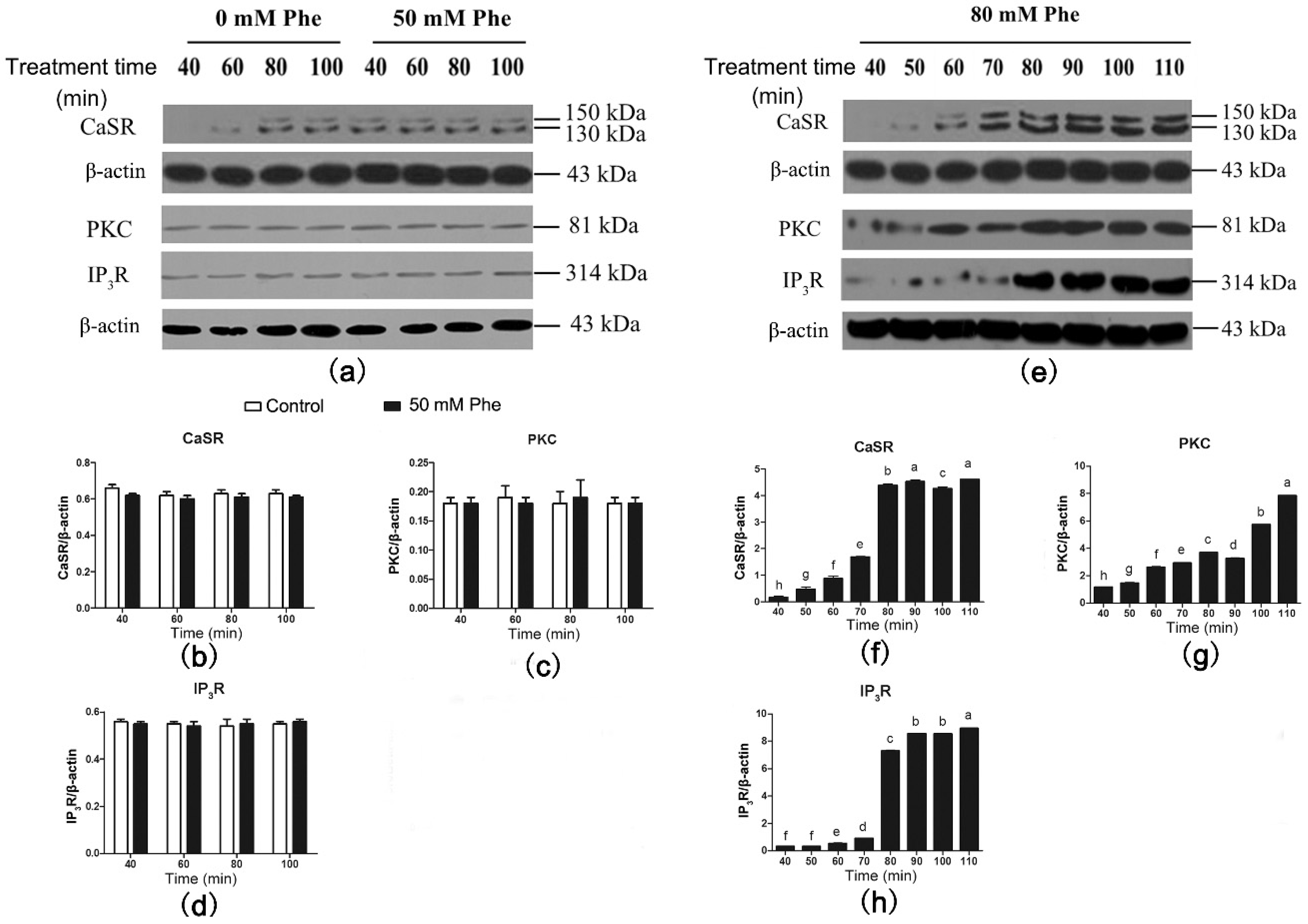
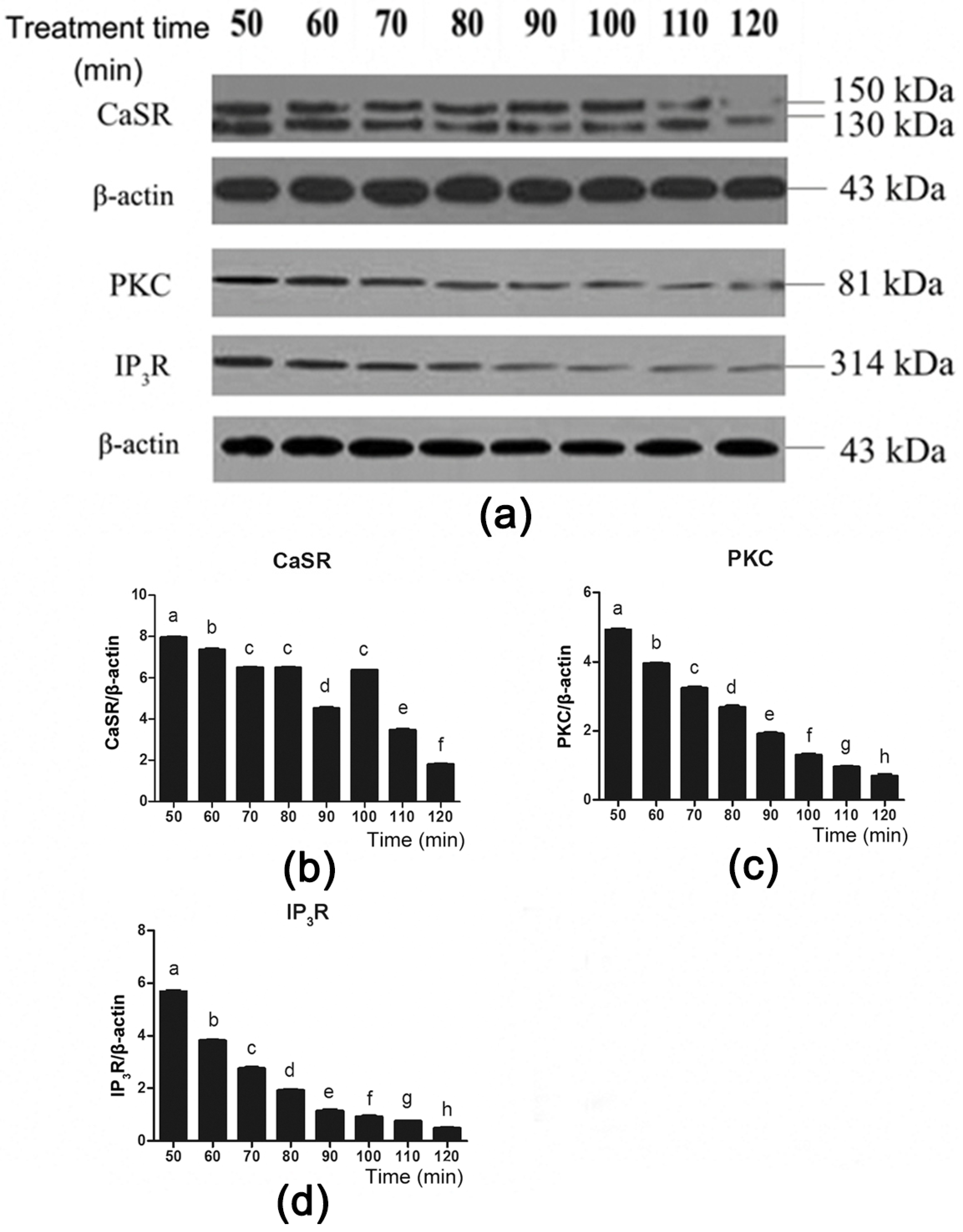
3.4. CaSR and Its Downstream Signaling Proteins Are Involved in Gut Hormone Secretion
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Li, Q.; Patience, J.F. Factors involved in the regulation of feed and energy intake of pigs. Anim. Feed Sci. Technol. 2017, 233, 22–33. [Google Scholar] [CrossRef]
- Veldhorst, M.; Postma-Smeets, A.; Soenen, S.; Hochstenbach-Waelen, A.; Hursel, R.; Diepvens, K.; Lejeune, M.; Luscombe, N.D.; Westerterp-Plantenga, M. Protein-induced satiety: Effects and mechanisms of different proteins. Physiol. Behav. 2008, 94, 300–307. [Google Scholar] [CrossRef] [PubMed]
- Qiu, W.J.; Pan, J.; Gu, X.F. Morphologic study of the effect of phenylalanine and its metabolites on the neuronal differentiation of P19 cell. Chin. J. Inorg. Chem. 2002, 18, 117–121. [Google Scholar]
- Santos-Hernández, M.; Miralles, B.; Amigo, L.; Recio, I. Intestinal Signaling of Proteins and Digestion-Derived Products Relevant to Satiety. J. Agric. Food Chem. 2018, 66, 10123–10131. [Google Scholar] [CrossRef] [PubMed]
- Colombel, J.F.; Sutton, A.; Chayvialle, J.A.; Modigliani, R. Cholecystokinin release and biliopancreatic secretion in response to selective perfusion of the duodenal loop with amino acids in man. Gut 1988, 29, 1158–1166. [Google Scholar] [CrossRef] [PubMed]
- Mace, O.J.; Schindler, M.; Patel, S. The regulation of K- and L- cell activity by GLUT2 and the calcium-sensing receptor CasR in rat small intestine. J. Physiol. 2012, 590, 2917–2936. [Google Scholar] [CrossRef]
- Daly, K.; Al-Rammahi, M.; Moran, A.W.; Marcello, M.; Ninomiya, Y.; Shirazi-Beechey, S.P. Sensing of amino acids by the gut-expressed taste receptor T1R1-T1R3 stimulates CCK secretion. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 304, G271–G282. [Google Scholar] [CrossRef]
- Acar, I.; Cetinkaya, A.; Lay, I.; Ileri-Gurel, E. The role of calcium sensing receptors in GLP-1 and PYY secretion after acute intraduodenal administration of L-Tryptophan in rats. Nutr. Neurosci. 2018, 17, 1–9. [Google Scholar] [CrossRef]
- Janssen, S.; Depoortere, I. Nutrient sensing in the gut: New roads to therapeutics? Trends Endocrin. Met. 2013, 24, 92–100. [Google Scholar] [CrossRef]
- Brown, E.M.; Gamba, G.; Riccardi, D.; Lombardi, M.; Butters, R.; Kifor, O.; Sun, A.; Hediger, M.A.; Lytton, J.; Hebert, S.C. Cloning and characterization of an extracellular Ca2+-sensing receptor from bovine parathyroid. Nature 1993, 366, 575–580. [Google Scholar] [CrossRef]
- Alamshah, A.; Spreckley, E.; Norton, M.; Kinsey-Jones, J.S.; Amin, A.; Ramgulam, A.; Cao, Y.; Johnson, R.; Saleh, K.; Akalestou, E.; et al. L-phenylalanine modulates gut hormone release and glucose tolerance and suppresses food intake through the calcium-sensing receptor in rodents. Int. J. Obes. 2017, 41, 1693–1701. [Google Scholar] [CrossRef] [PubMed]
- Hira, T.; Nakajima, S.; Eto, Y.; Hara, H. Calcium-sensing receptor mediates phenylalanine-induced cholecystokinin secretion in enteroendocrine STC-1 cells. FEBS J. 2008, 275, 4620–4626. [Google Scholar] [CrossRef] [PubMed]
- Conigrave, A.D.; Franks, A.H.; Brown, E.M.; Quinn, S.J. L-Amino acid sensing by the calcium-sensing receptor: A general mechanism for coupling protein and calcium metabolism? Eur. J. Clin. Nutr. 2002, 56, 1072–1080. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, Y.; Imai, S. Structure and function of inositol 1,4,5-trisphosphate receptor. Ann. N.Y. Acad. Sci. 2010, 707, 178–197. [Google Scholar] [CrossRef]
- Zhang, C.; Miller, C.L.; Brown, E.M.; Yang, J.J. The calcium sensing receptor: From calcium sensing to signaling. Sci. China Life Sci. 2015, 58, 14–27. [Google Scholar] [CrossRef]
- Li, J.; Yang, S.; Yu, F.; Ji, E.; Woodrow-Weiss, J. Endothelin-1 enhanced carotid body chemosensory activity in chronic intermittent hypoxia through PLC, PKC and p38MAPK signaling pathways. Neuropeptides 2019, 74, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Shigeto, M.; Ramracheya, R.; Tarasov, A.I.; Cha, C.Y.; Chibalina, M.V.; Hastoy, B.; Philippaert, K.; Reinbothe, T.; Rorsman, N.; Salehi, A.; et al. GLP-1 stimulates insulin secretion by PKC-dependent TRPM4 and TRPM5 activation. J. Clin. Investig. 2015, 125, 4714–4728. [Google Scholar] [CrossRef]
- Zhao, X.; Xian, Y.; Wang, C.; Ding, L.; Meng, X.; Zhu, W.; Hang, S. Calcium-sensing receptor-mediated L-tryptophan-induced secretion of cholecystokinin and glucose-dependent insuliotropic peptide in swine duodenum. J. Vet. Sci. 2018, 19, 179–187. [Google Scholar] [CrossRef]
- Tan, Y.F.; Chen, W.H.; Zou, S.X. The effect of β-casomorphin on piglets’ stomach antrum under superfusion. J. Nanjing Agric. Univ. 2000, 23, 72–75. [Google Scholar]
- Xian, Y.; Zhao, X.; Wang, C.; Kang, C.; Ding, L.; Zhu, W.; Hang, S. Phenylalanine and tryptophan stimulate gastrin and somatostatin secretion and H+-K+-ATPase activity in pigs through calcium-sensing receptor. Gen. Comp. Endcor. 2018, 267, 1–8. [Google Scholar] [CrossRef]
- Liddle, R.A.; Goldfine, I.D.; Rosen, M.S.; Taplitz, R.A.; Williams, J.A. Cholecystokinin bioactivity in human plasma. Molecular forms, responses to feeding, and relationship to gallbladder contraction. J. Clin. Investig 1985, 75, 1144–1152. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, C.; Göke, R.; Richter, G.; Fehmann, H.C.; Arnold, R.; Göke, B. Glucagon-like peptide-1 and glucose-dependent insulin-releasing polypeptide plasma levels in response to nutrients. Digestion 1995, 56, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, N.; Cheng, I.; Rogers, K.; Riccardi, D.; Hall, A.; Diaz, R.; Hebert, S.C.; Soybel, D.; Brown, E.M. Identification and localization of extracellular Ca2+-sensing receptor in rat intestine. Am. J. Physiol. 1998, 274, G122–G130. [Google Scholar] [CrossRef] [PubMed]
- Sheinin, Y.; Kállay, E.; Wrba, F.; Kriwanek, S.; Peterlik, M.; Cross, H.S. Immunocytochemical localization of the extracellular calcium-sensing receptor in normal and malignant human large intestinal mucosa. J. Histochem. Cytochem. 2000, 48, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Kang, C.; Xian, Y.; Zhang, M.; Chen, X.; Pei, M.; Zhu, W.; Hang, S. Sensing of L-Arginine by gut-expressed calcium sensing receptor stimulates cholecystokinin and glucose-dependent insulinotropic peptide secretion in pig model. J. Food Sci. 2018, 83, 2394–2401. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, S.; Hira, T.; Hara, H. Calcium-sensing receptor mediates dietary peptide-induced CCK secretion in enteroendocrine STC-1 cells. Mol. Nutr. Food Res. 2012, 56, 753–760. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Zhou, D.Y.; Yang, J.F.; Song, S.; Zhang, T.; Zhu, C.; Song, Y.Q.; Yu, C.; Zhu, B.W. Effects of abalone (Haliotis discus hannai Ino) gonad polysaccharides on cholecystokinin release in STC-1 cells and its signaling mechanism. Carbohydr. Polym. 2016, 151, 268–273. [Google Scholar] [CrossRef]
- Zhou, H.R.; Pestka, J.J. Deoxynivalenol (vomitoxin)-induced cholecystokinin and glucagon-like peptide-1 release in the STC-1 enteroendocrine cell model is mediated by calcium-sensing receptor and transient receptor potential ankyrin-1 channel. Toxicol. Sci. 2015, 145, 407–417. [Google Scholar] [CrossRef]
- Nakajima, S.; Hira, T.; Eto, Y.; Asano, K.; Hara, H. Soybean β51–63 peptide stimulates cholecystokinin secretion via a calcium-sensing receptor in enteroendocrine STC-1 cells. Regul. Pept. 2010, 159, 148–155. [Google Scholar] [CrossRef]
- Liu, G.; Cao, W.; Jia, G.; Zhao, H.; Chen, X.; Wang, J. Calcium-sensing receptor in nutrient sensing: An insight into the modulation of intestinal homoeostasis. Brit. J. Nutr. 2018, 120, 1–11. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feng, J.; Kang, C.; Wang, C.; Ding, L.; Zhu, W.; Hang, S. L-phenylalanine Increased Gut Hormone Secretion through Calcium-Sensing Receptor in the Porcine Duodenum. Animals 2019, 9, 476. https://doi.org/10.3390/ani9080476
Feng J, Kang C, Wang C, Ding L, Zhu W, Hang S. L-phenylalanine Increased Gut Hormone Secretion through Calcium-Sensing Receptor in the Porcine Duodenum. Animals. 2019; 9(8):476. https://doi.org/10.3390/ani9080476
Chicago/Turabian StyleFeng, Jiangyin, Cuicui Kang, Chao Wang, Liren Ding, Weiyun Zhu, and Suqin Hang. 2019. "L-phenylalanine Increased Gut Hormone Secretion through Calcium-Sensing Receptor in the Porcine Duodenum" Animals 9, no. 8: 476. https://doi.org/10.3390/ani9080476
APA StyleFeng, J., Kang, C., Wang, C., Ding, L., Zhu, W., & Hang, S. (2019). L-phenylalanine Increased Gut Hormone Secretion through Calcium-Sensing Receptor in the Porcine Duodenum. Animals, 9(8), 476. https://doi.org/10.3390/ani9080476



