Diet-Induced Metabolic Syndrome Reduced Heart Rate Variability and Increased Irregularity and Complexity of Short-Term RR Time Series in Rabbits
Simple Summary
Abstract
1. Introduction
2. Material and Methods
2.1. Animals and Diets
2.2. Electrocardiographic Study
2.3. Measurements and Calculations
2.4. Statistical Analysis
3. Results
3.1. Time-Domain Analysis of Short-Term HRV
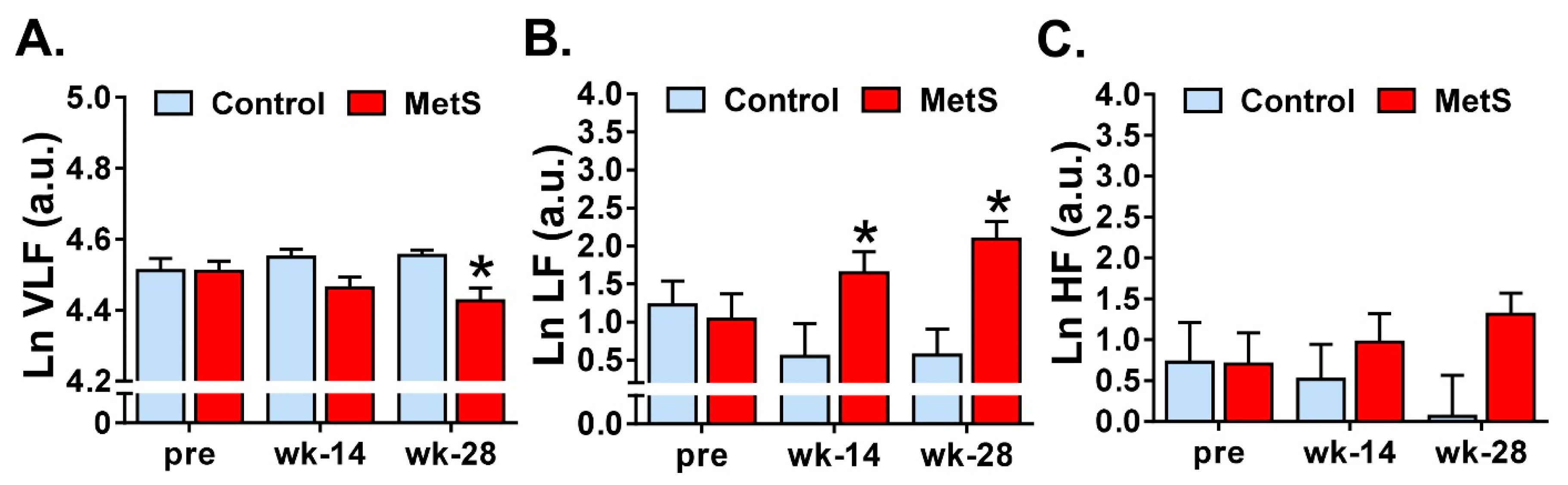
3.2. Modifications of Short-Term HRV in the Frequency Domain
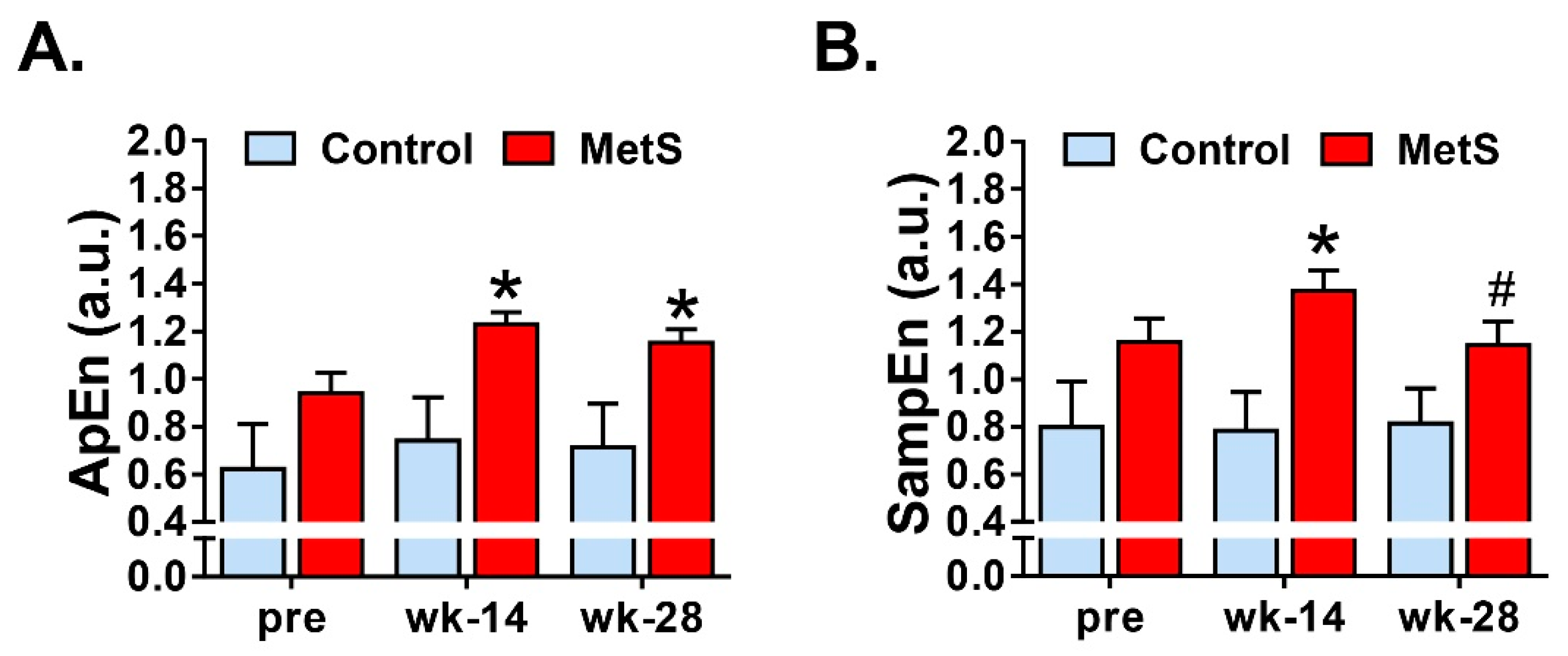
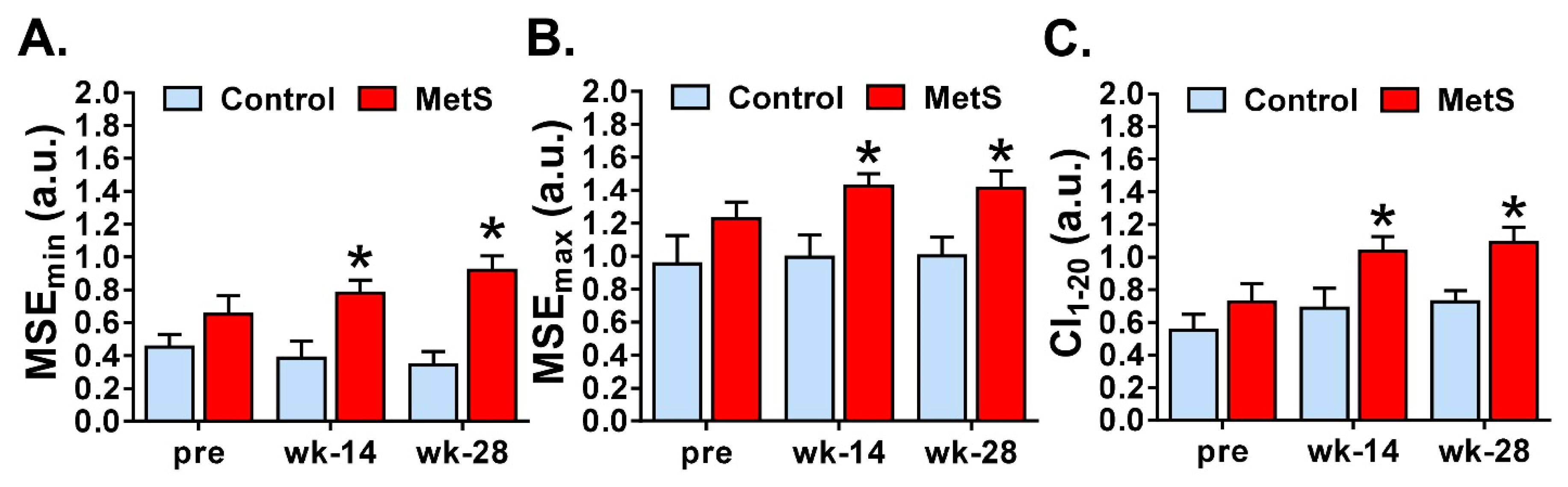
3.3. Nonlinear Analysis of Short-Term HRV
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Parameter | Description |
|---|---|
| NN (ms) | Mean of the selected R-R interval series. |
| SDNN (ms) | Standard deviation of the R-R interval series (overall variability). |
| HR (beats·min−1) | Mean heart rate. |
| SDHR (beats·min−1) | Standard deviation of spontaneous heart rate values. |
| RMSSD (ms) | The root mean square of differences of successive R-R intervals. |
| NN50 (count) | Number successive R-R interval pairs that differences between successive R-R intervals. |
| Triangular index (ms) | Integral of the sample density distribution of R-R intervals divided by the maximum of the density distribution. |
| TINN (ms) | Triangular interpolation of the R-R interval histogram. Baseline width of the minimum square difference triangular interpolation of the maximum of the sample density distribution of R-R intervals. |
| Parameter | Description |
|---|---|
| VLF, LF, HF power (%) | Relative powers in VLF, LF and HF ranges.
|
| Total power | Total spectral power |
| LF/HF | Ratio between powers in LF and HF bands. |
| Parameter | Description |
|---|---|
| SD1 (ms) | Standard deviation of the perpendicular point along the line of identity of the Poincaré plot. It represents the instantaneous beat-to-beat short-term variability. |
| SD2 (ms) | Standard deviation of the perpendicular point along the line of identity of the Poincaré plot. It represents the instantaneous beat-to-beat long-term variability. |
| DFA α1 (a.u.) | Short terms fluctuations (4–12 beats) of detrended fluctuation analysis. The slopes of a log-log plot (correlation measure as a function of segment length). |
| DFA α2 (a.u.) | Long-terms fluctuations (13–64 beats) of detrended fluctuation analysis. The slopes of a log-log plot (correlation measure as a function of segment length). |
| ApEn (a.u.) | Approximate entropy, measures the complexity of RR time series (m = 2, r = 0.2) [29]. |
| SampEn (a.u.) | Sample entropy, measures the irregularity RR time series (m = 2, r = 0.2) [30]. |
| MSEmin (a.u.) | Minimum value of multiscale entropy [31]. |
| MSEmax (a.u.) | Maximum value of multiscale entropy [31]. |
| CI1–20 (a.u.) | Mean of entropies on all 20 scales of MSE [25]. |
| DET (%) | Determinism (percentage of recurrence points which form diagonal lines in recurrence plot) |
| REC (%) | Recurrence rate (percentage of recurrence points in recurrence plot) |
References
- Zimmet, P.; Alberti, K.G.M.M.; Stern, N.; Bilu, C.; El-Osta, A.; Einat, H.; Kronfeld-Schor, N. The Circadian Syndrome: Is the Metabolic Syndrome and much more! J. Int. Med. 2019, 286, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Alberti, K.G.; Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z.; Cleeman, J.I.; Donato, K.A.; Fruchart, J.C.; James, W.P.; Loria, C.M.; Smith, S.C., Jr. Harmonizing the metabolic syndrome: A joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 2009, 120, 1640–1645. [Google Scholar] [PubMed]
- Nalliah, C.J.; Sanders, P.; Kottkamp, H.; Kalman, J.M. The role of obesity in atrial fibrillation. Eur. Heart J. 2016, 37, 1565–1572. [Google Scholar] [CrossRef] [PubMed]
- Calvo, C.J.; Lozano, W.M.; Arias-Mutis, Ó.J.; Such-Miquel, L.; Such, L.; Genovés, P.; Guill, A.; Millet, J.; Chorro, F.J.; Alberola, A.; et al. Modifications of short-term intrinsic pacemaker variability in diet-induced metabolic syndrome: A study on isolated rabbit heart. J. Physiol. Biochem. 2019, 75, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Camm, A.J.; Malik, M.; Bigger, J.T.; Breithardt, G.; Cerutti, S.; Cohen, R.J.; Coumel, P.; Fallen, E.L.; Kennedy, H.L.; Kleiger, R.E.; et al. Heart rate variability. Standards of measurement, physiological interpretation, and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Eur. Heart J. 1996, 17, 354–381. [Google Scholar]
- Franciosi, S.; Perry, F.K.G.; Roston, T.M.; Armstrong, K.R.; Claydon, V.E.; Sanatani, S. The role of the autonomic nervous system in arrhythmias and sudden cardiac death. Auton. Neurosci. 2017, 205, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Zarzoso, M.; Rysevaite, K.; Milstein, M.L.; Calvo, C.J.; Kean, A.C.; Atienza, F.; Pauza, D.H.; Jalife, J.; Noujaim, S.F. Nerves projecting from the intrinsic cardiac ganglia of the pulmonary veins modulate sinoatrial node pacemaker function. Cardiovasc. Res. 2013, 99, 566–575. [Google Scholar] [CrossRef]
- Arias-Mutis, O.J.; Marrachelli, V.G.; Ruiz-Saurí, A.; Alberola, A.; Morales, J.M.; Such-Miquel, L.; Monleon, D.; Chorro, F.J.; Such, L.; Zarzoso, M. Development and characterization of an experimental model of diet-induced metabolic syndrome in rabbit. PLoS ONE 2017, 12, e0178315. [Google Scholar] [CrossRef] [PubMed]
- Lozano, W.M.; Arias-Mutis, O.J.; Calvo, C.J.; Chorro, F.J.; Zarzoso, M. Diet-Induced Rabbit Models for the Study of Metabolic Syndrome. Animals 2019, 9, E463. [Google Scholar] [CrossRef]
- Carroll, J.F.; Dwyer, T.M.; Grady, A.W.; Reinhart, G.A.; Montani, J.P.; Cockrell, K.; Meydrech, E.F.; Mizelle, H.L. Hypertension; cardiac hypertrophy; and neurohumoral activity in a new animal model of obesity. Am. J. Physiol 1996, 271, H373–H378. [Google Scholar] [CrossRef]
- Tarvainen, M.P.; Niskanen, J.P.; Lipponen, J.A.; Ranta-Aho, P.O.; Karjalainen, P.A. Kubios HRV—Heart rate variability analysis software. Comput. Methods Programs Biomed. 2014, 113, 210–220. [Google Scholar] [CrossRef]
- Varga, B.; Gergely, A.; Galambos, Á.; Kis, A. Heart Rate and Heart Rate Variability during Sleep in Family Dogs (Canis familiaris). Moderate Effect of Pre-Sleep Emotions. Animals 2018, 8, E107. [Google Scholar] [CrossRef]
- Shaffer, F.; McCraty, R.; Zerr, C.L. A healthy heart is not a metronome: An integrative review of the heart’s anatomy and heart rate variability. Front. Psychol. 2014, 5, 1040. [Google Scholar] [CrossRef]
- Koskinen, T.; Kähönen, M.; Jula, A.; Mattsson, N.; Laitinen, T.; Keltikangas-Järvinen, L.; Viikari, J.; Välimäki, I.; Rönnemaa, T.; Raitakari, O.T. Metabolic syndrome and short-term heart rate variability in young adults. The cardiovascular risk in young Finns study. Diabet. Med. 2009, 26, 354–361. [Google Scholar] [CrossRef]
- Brunner, E.J.; Hemingway, H.; Walker, B.R.; Page, M.; Clarke, P.; Juneja, M.; Shipley, M.J.; Kumari, M.; Andrew, R.; Seckl, J.R.; et al. Adrenocortical; autonomic; and inflammatory causes of the metabolic syndrome: Nested case-control study. Circulation 2002, 106, 2659–2665. [Google Scholar] [CrossRef]
- Min, K.B.; Min, J.Y.; Paek, D.; Cho, S.I. The impact of the components of metabolic syndrome on heart rate variability: Using the NCEP-ATP III and IDF definitions. Pacing Clin. Electrophysiol. 2008, 31, 584–591. [Google Scholar] [CrossRef]
- Chang, C.J.; Yang, Y.C.; Lu, F.H.; Lin, T.S.; Chen, J.J.; Yeh, T.L.; Wu, C.H.; Wu, J.S. Altered cardiac autonomic function may precede insulin resistance in metabolic syndrome. Am. J. Med. 2010, 123, 432–438. [Google Scholar] [CrossRef]
- Ma, Y.; Tseng, P.H.; Ahn, A.; Wu, M.S.; Ho, Y.L.; Chen, M.F.; Peng, C.K. Cardiac Autonomic Alteration and Metabolic Syndrome: An Ambulatory ECG-based Study in A General Population. Sci. Rep. 2017, 7, 44363. [Google Scholar] [CrossRef]
- Stuckey, M.I.; Tulppo, M.P.; Kiviniemi, A.M.; Petrella, R.J. Heart rate variability and the metabolic syndrome: A systematic review of the literature. Diabetes Metab. Res. Rev. 2014, 30, 784–793. [Google Scholar] [CrossRef]
- Assoumou, H.G.; Pichot, V.; Barthelemy, J.C.; Dauphinot, V.; Celle, S.; Gosse, P.; Kossovsky, M.; Gaspoz, J.M.; Roche, F. Metabolic syndrome and short-term and long-term heart rate variability in elderly free of clinical cardiovascular disease: The PROOF study. Rejuvenation Res. 2010, 13, 653–663. [Google Scholar] [CrossRef]
- Rasic-Milutinovic, Z.R.; Milicevic, D.R.; Milovanovic, B.D.; Perunicic-Pekovic, G.B.; Pencic, B.D. Do components of metabolic syndrome contribute to cardiac autonomic neuropathy in non-diabetic patients? Saudi Med. J. 2010, 31, 650–657. [Google Scholar]
- Usui, H.; Nishida, Y. The very low-frequency band of heart rate variability represents the slow recovery component after a mental stress task. PLoS ONE 2017, 12, e0182611. [Google Scholar] [CrossRef]
- Guzzetti, S.; La Rovere, M.T.; Pinna, G.D.; Maestri, R.; Borroni, E.; Porta, A.; Mortara, A.; Malliani, A. Different spectral components of 24 h heart rate variability are related to different modes of death in chronic heart failure. Eur. Heart J. 2005, 26, 357–362. [Google Scholar] [CrossRef]
- Bigger, J.T., Jr.; Fleiss, J.L.; Steinman, R.C.; Rolnitzky, L.M.; Kleiger, R.E.; Rottman, J.N. Frequency domain measures of heart period variability and mortality after myocardial infarction. Circulation 1992, 85, 164–171. [Google Scholar] [CrossRef]
- Stuckey, M.I.; Kiviniemi, A.; Gill, D.P.; Shoemaker, J.K.; Petrella, R.J. Associations between heart rate variability; metabolic syndrome risk factors; and insulin resistance. Appl. Physiol. Nutr. Metab. 2015, 40, 734–740. [Google Scholar] [CrossRef]
- DiRienzo, M.; Parati, G.; Castiglioni, P.; Omboni, S.; Ferrari, A.U.; Ramirez, A.J.; Pedotti, A.; Mancia, G. Role of sino-aortic afferents in modulating BP and pulse–interval characteristics in unanesthetized cats. Am. J. Physiol. 1991, 261, H1811–H1818. [Google Scholar]
- Mäkikallio, T.H.; Seppänen, T.; Niemelä, M.; Airaksinen, K.E.; Tulppo, M.; Huikuri, H.V. Abnormalities in beat to beat complexity of heart rate dynamics in patients with a previous myocardial infarction. J. Am. Coll. Cardiol. 1996, 28, 1005–1011. [Google Scholar] [CrossRef]
- Yaniv, Y.; Lyashkov, A.E.; Lakatta, E.G. Impaired signaling intrinsic to sinoatrial node pacemaker cells affects heart rate variability during cardiac disease. J. Clin. Trials 2014, 4, 152. [Google Scholar]
- Pincus, S.M. Assessing serial irregularity and its implications for health. Ann. N. Y. Acad. Sci. 2001, 954, 245–267. [Google Scholar] [CrossRef]
- Richman, J.S.; Moorman, J.R. Physiological time-series analysis using approximate entropy and sample entropy. Am. J. Physiol. Heart Circ. Physiol. 2000, 278, H2039–H2049. [Google Scholar] [CrossRef]
- Costa, M.; Goldberger, A.L.; Peng, C.K. Multiscale entropy analysis of complex physiologic time series. Phys. Rev. Lett. 2002, 89, 068102. [Google Scholar] [CrossRef]
| Parameter | Pre-Test | 14 Weeks | 28 Weeks | |||
|---|---|---|---|---|---|---|
| Control | MetS | Control | MetS | Control | MetS | |
| NN (ms) | 229 (26) | 237 (28) | 226 (35) | 242 (42) | 226 (17) | 254 (24) |
| SDNN (ms) | 10 (7.2) | 10 (5.9) | 9 (6.5) | 8 (4.8) | 12 (8.3) | 8 (3.7) |
| HR (beats/min−1) | 265 (25) | 257 (32) | 271 (37) | 254 (42) | 268 (19) | 238 (24) |
| SDHR (beats/min−1) | 10.8 (5.2) | 11.3 (6) | 9.8 (3) | 8.4 (4) | 13.2 (7) | 6.9 (2.6) *,# |
| RMSSD (ms) | 4.3 (3.3) | 7.4 (5.8) | 3.8 (3.2) | 4.1 (2.8) | 6.6 (7.9) | 4.1 (2.9) |
| NN50 (count) | 1.9 (1.6) | 18 (48.8) | 1 (1.5) | 0.5 (0.8) | 43.6 (125) | 2.8 (6.8) |
| Triangular index (ms) | 2.5 (0.5) | 2.9 (0.9) | 2.4 (0.8) | 2.5 (0.8) | 3.1 (1) | 2.4 (0.6) |
| TINN (ms) | 59 (17) | 73 (47) | 65 (40) | 45 (20) | 59 (37) | 55 (39) |
| Parameter | Pre-Diet | Week 14 | Week 28 | |||
|---|---|---|---|---|---|---|
| Control | MetS | Control | MetS | Control | MetS | |
| LF (n.u.) | 66.0 (26.4) | 56.3 (20.1) | 56.0 (18.8) | 64.8 (13.1) | 60.4 (20.1) | 68.3 (6.7) |
| HF (n.u.) | 30.8 (19.2) | 43.4 (20.4) | 43.4 (18.5) | 35.1 (13.1) | 38.9 (19.8) | 31.4 (6.6) |
| LF/HF (a.u.) | 2.4 (1.6) | 1.8 (1.3) | 1.1 (0.4) | 2.3 (1.4) | 2.4 (1.6) | 2.3 (0.6) |
| Ln total power (ms2) | 3.7 (1.3) | 3.2 (1.6) | 3.6 (1.3) | 3.2 (1.2) | 3.6 (1.8) | 3.3 (1.1) |
| Parameter | Pre-Diet | Week 14 | Week 28 | |||
|---|---|---|---|---|---|---|
| Control | MetS | Control | MetS | Control | MetS | |
| SD1(ms) | 3.1 (2.3) | 5.2 (4.1) | 2.7 (2.3) | 2.9 (2) | 4.7 (5.6) | 2.9 (2) |
| SD2 (ms) | 13.8 (10.5) | 12.4 (8.5) | 12.5 (9.4) | 11.3 (6.9) | 15.8 (11.2) | 10.5 (5) |
| DFA-α1 (a.u.) | 0.8 (0.6) | 0.6 (0.3) | 0.9 (0.6) | 0.8 (0.2) | 0.8 (0.4) | 1 (0.2) |
| DFA-α2 (a.u.) | 0.8 (0.5) | 0.7 (0.4) | 0.8 (0.5) | 0.8 (0.3) | 0.7 (0.4) | 0.9 (0.2) |
| ShanEn (a.u.) | 3.3 (1) | 3.2 (0.6) | 3.7 (1) | 3.5 (0.4) | 3.7 (0.8) | 3.7 (0.5) |
| DET (%) | 98.2 (1.9) | 97.6 (1.2) | 98.1 (1.6) | 98.2 (1.3) | 98 (2.2) | 98.1 (3) |
| REC (%) | 39.3 (13.6) | 31.9 (10.8) | 38.7 (11.2) | 33.9 (11.9) | 36.2 (13.9) | 38.2 (12.8) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lozano, W.M.; Calvo, C.J.; Arias-Mutis, O.J.; Díaz, A.; Such-Miquel, L.; Zhao, J.; Alberola, A.; Chorro, F.J.; Zarzoso, M. Diet-Induced Metabolic Syndrome Reduced Heart Rate Variability and Increased Irregularity and Complexity of Short-Term RR Time Series in Rabbits. Animals 2019, 9, 572. https://doi.org/10.3390/ani9080572
Lozano WM, Calvo CJ, Arias-Mutis OJ, Díaz A, Such-Miquel L, Zhao J, Alberola A, Chorro FJ, Zarzoso M. Diet-Induced Metabolic Syndrome Reduced Heart Rate Variability and Increased Irregularity and Complexity of Short-Term RR Time Series in Rabbits. Animals. 2019; 9(8):572. https://doi.org/10.3390/ani9080572
Chicago/Turabian StyleLozano, Wilson M., Conrado J. Calvo, Oscar J. Arias-Mutis, Ana Díaz, Luis Such-Miquel, Jichao Zhao, Antonio Alberola, Francisco J. Chorro, and Manuel Zarzoso. 2019. "Diet-Induced Metabolic Syndrome Reduced Heart Rate Variability and Increased Irregularity and Complexity of Short-Term RR Time Series in Rabbits" Animals 9, no. 8: 572. https://doi.org/10.3390/ani9080572
APA StyleLozano, W. M., Calvo, C. J., Arias-Mutis, O. J., Díaz, A., Such-Miquel, L., Zhao, J., Alberola, A., Chorro, F. J., & Zarzoso, M. (2019). Diet-Induced Metabolic Syndrome Reduced Heart Rate Variability and Increased Irregularity and Complexity of Short-Term RR Time Series in Rabbits. Animals, 9(8), 572. https://doi.org/10.3390/ani9080572





