Alpha-Difluoromethylornithine, an Irreversible Inhibitor of Polyamine Biosynthesis, as a Therapeutic Strategy against Hyperproliferative and Infectious Diseases
Abstract
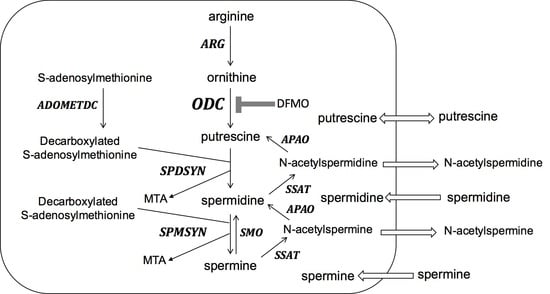
:1. Introduction
2. Pharmacodynamic and Pharmacokinetic Properties of DFMO
3. African Sleeping Sickness
4. Vaniqa
5. Colon Cancer
6. Neuroblastoma
7. Conclusions
Author Contributions
Conflicts of Interest
References
- Gerner, E.W.; Meyskens, F.L., Jr. Polyamines and cancer: Old molecules, new understanding. Nat. Rev. Cancer 2004, 4, 781–792. [Google Scholar] [CrossRef] [PubMed]
- Heby, O.; Persson, L.; Rentala, M. Targeting the polyamine biosynthetic enzymes: A promising approach to therapy of African sleeping sickness, Chagas’ disease, and leishmaniasis. Amino Acids 2007, 33, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Heby, O.; Roberts, S.C.; Ullman, B. Polyamine biosynthetic enzymes as drug targets in parasitic protozoa. Biochem. Soc. Trans. 2003, 31, 415–419. [Google Scholar] [CrossRef] [PubMed]
- Roberts, S.; Ullman, B. Parasite Polyamines as Pharmaceutical Targets. Curr. Pharm. Des. 2017, 23, 3325–3341. [Google Scholar] [CrossRef] [PubMed]
- Miller-Fleming, L.; Olin-Sandoval, V.; Campbell, K.; Ralser, M. Remaining Mysteries of Molecular Biology: The Role of Polyamines in the Cell. J. Mol. Biol. 2015, 427, 3389–3406. [Google Scholar] [CrossRef] [PubMed]
- Igarashi, K.; Kashiwagi, K. Modulation of cellular function by polyamines. Int. J. Biochem. Cell Biol. 2010, 42, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Bachrach, U.; Wang, Y.C.; Tabib, A. Polyamines: New cues in cellular signal transduction. News Physiol. Sci. 2001, 16, 106–109. [Google Scholar] [CrossRef] [PubMed]
- Rai, P.R.; Somani, R.R.; Kandpile, P.S. Ornithine Decarboxylase Inhibition: A strategy to combat various diseases. Mini Rev. Med. Chem. 2017. [Google Scholar] [CrossRef] [PubMed]
- Ramot, Y.; Pietila, M.; Giuliani, G.; Rinaldi, F.; Alhonen, L.; Paus, R. Polyamines and hair: A couple in search of perfection. Exp. Dermatol. 2010, 19, 784–790. [Google Scholar] [CrossRef] [PubMed]
- Smith, K.J.; Skelton, H. α-Difluoromethylornithine, a polyamine inhibitor: Its potential role in controlling hair growth and in cancer treatment and chemo-prevention. Int. J. Dermatol. 2006, 45, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Wallace, H.M.; Fraser, A.V. Inhibitors of polyamine metabolism: Review article. Amino Acids 2004, 26, 353–365. [Google Scholar] [CrossRef] [PubMed]
- Park, M.H.; Nishimura, K.; Zanelli, C.F.; Valentini, S.R. Functional significance of eIF5A and its hypusine modification in eukaryotes. Amino Acids 2010, 38, 491–500. [Google Scholar] [CrossRef] [PubMed]
- Meyskens, F.L., Jr.; Gerner, E.W. Development of difluoromethylornithine (DFMO) as a chemoprevention agent. Clin. Cancer Res. 1999, 5, 945–951. [Google Scholar] [PubMed]
- Pegg, A.E. Regulation of ornithine decarboxylase. J. Biol. Chem. 2006, 281, 14529–14532. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, B.W.; Bey, P.; Danzin, C.; Jung, M.J.; Casara, P.; Vevert, J.P. Catalytic Irreversible Inhibition of Mammalian Ornithine Decarboxylase (E.C.4.1.1.17) by Substrate and Product Analogs. J. Am. Chem. Soc. 1978, 100, 2551–2553. [Google Scholar] [CrossRef]
- Pegg, A.E.; McGovern, K.A.; Wiest, L. Decarboxylation of α-difluoromethylornithine by ornithine decarboxylase. Biochem. J. 1987, 241, 305–307. [Google Scholar] [CrossRef] [PubMed]
- Babokhov, P.; Sanyaolu, A.O.; Oyibo, W.A.; Fagbenro-Beyioku, A.F.; Iriemenam, N.C. A current analysis of chemotherapy strategies for the treatment of human African trypanosomiasis. Pathog. Glob. Health 2013, 107, 242–252. [Google Scholar] [CrossRef] [PubMed]
- Coyne, P.E., Jr. The eflornithine story. J. Am. Acad. Dermatol. 2001, 45, 784–786. [Google Scholar] [CrossRef] [PubMed]
- Ebikeme, C. The death and life of the resurrection drug. PLoS Negl. Trop. Dis. 2014, 8, e2910. [Google Scholar] [CrossRef] [PubMed]
- Jackson, J.; Caro, J.J.; Caro, G.; Garfield, F.; Huber, F.; Zhou, W.; Lin, C.S.; Shander, D.; Schrode, K.; Eflornithine, H.S.G. The effect of eflornithine 13.9% cream on the bother and discomfort due to hirsutism. Int. J. Dermatol. 2007, 46, 976–981. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, R.T.; Nare, B.; Phillips, M.A. State of the art in African trypanosome drug discovery. Curr. Top. Med. Chem. 2011, 11, 1255–1274. [Google Scholar] [CrossRef] [PubMed]
- Steverding, D. The development of drugs for treatment of sleeping sickness: A historical review. Parasit Vectors 2010, 3, 15. [Google Scholar] [CrossRef] [PubMed]
- Carbone, P.P.; Douglas, J.A.; Thomas, J.; Tutsch, K.; Pomplun, M.; Hamielec, M.; Pauk, D. Bioavailability study of oral liquid and tablet forms of α-difluoromethylornithine. Clin. Cancer Res. 2000, 6, 3850–3854. [Google Scholar] [PubMed]
- Legros, D.; Ollivier, G.; Gastellu-Etchegorry, M.; Paquet, C.; Burri, C.; Jannin, J.; Buscher, P. Treatment of human African trypanosomiasis—Present situation and needs for research and development. Lancet Infect. Dis. 2002, 2, 437–440. [Google Scholar] [CrossRef]
- Milord, F.; Pepin, J.; Loko, L.; Ethier, L.; Mpia, B. Efficacy and toxicity of eflornithine for treatment of Trypanosoma brucei gambiense sleeping sickness. Lancet 1992, 340, 652–655. [Google Scholar] [CrossRef]
- Jobanputra, K.S.; Rajpal, A.V.; Nagpur, N.G. Eflornithine. Indian J. Dermatol. Venereol. Leprol. 2007, 73, 365–366. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, B.; Noveck, R.; Behr, D.; Palmisano, M. Percutaneous absorption and pharmacokinetics of eflornithine HCl 13.9% cream in women with unwanted facial hair. J. Clin. Pharmacol. 2001, 41, 972–978. [Google Scholar] [CrossRef] [PubMed]
- Burke, C.A.; Dekker, E.; Samadder, N.J.; Stoffel, E.; Cohen, A. Efficacy and safety of eflornithine (CPP-1X)/sulindac combination therapy versus each as monotherapy in patients with familial adenomatous polyposis (FAP): Design and rationale of a randomized, double-blind, Phase III trial. BMC Gastroenterol. 2016, 16, 87. [Google Scholar] [CrossRef] [PubMed]
- Lynch, P.M.; Burke, C.A.; Phillips, R.; Morris, J.S.; Slack, R.; Wang, X.; Liu, J.; Patterson, S.; Sinicrope, F.A.; Rodriguez-Bigas, M.A.; et al. An international randomised trial of celecoxib versus celecoxib plus difluoromethylornithine in patients with familial adenomatous polyposis. Gut 2016, 65, 286–295. [Google Scholar] [CrossRef] [PubMed]
- Meyskens, F.L., Jr.; McLaren, C.E.; Pelot, D.; Fujikawa-Brooks, S.; Carpenter, P.M.; Hawk, E.; Kelloff, G.; Lawson, M.J.; Kidao, J.; McCracken, J.; et al. Difluoromethylornithine plus sulindac for the prevention of sporadic colorectal adenomas: A randomized placebo-controlled, double-blind trial. Cancer Prev. Res. 2008, 1, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Zell, J.A.; Pelot, D.; Chen, W.P.; McLaren, C.E.; Gerner, E.W.; Meyskens, F.L. Risk of cardiovascular events in a randomized placebo-controlled, double-blind trial of difluoromethylornithine plus sulindac for the prevention of sporadic colorectal adenomas. Cancer Prev. Res. 2009, 2, 209–212. [Google Scholar] [CrossRef] [PubMed]
- Bassiri, H.; Benavides, A.; Haber, M.; Gilmour, S.K.; Norris, M.D.; Hogarty, M.D. Translational development of difluoromethylornithine (DFMO) for the treatment of neuroblastoma. Transl. Pediatr. 2015, 4, 226–238. [Google Scholar] [PubMed]
- Priotto, G.; Kasparian, S.; Mutombo, W.; Ngouama, D.; Ghorashian, S.; Arnold, U.; Ghabri, S.; Baudin, E.; Buard, V.; Kazadi-Kyanza, S.; et al. Nifurtimox-eflornithine combination therapy for second-stage African Trypanosoma brucei gambiense trypanosomiasis: A multicentre, randomised, phase III, non-inferiority trial. Lancet 2009, 374, 56–64. [Google Scholar] [CrossRef] [Green Version]
- Priotto, G.; Pinoges, L.; Fursa, I.B.; Burke, B.; Nicolay, N.; Grillet, G.; Hewison, C.; Balasegaram, M. Safety and effectiveness of first line eflornithine for Trypanosoma brucei gambiense sleeping sickness in Sudan: Cohort study. BMJ 2008, 336, 705–708. [Google Scholar] [CrossRef] [PubMed]
- Eperon, G.; Balasegaram, M.; Potet, J.; Mowbray, C.; Valverde, O.; Chappuis, F. Treatment options for second-stage gambiense human African trypanosomiasis. Expert Rev. Antiinfect. Ther. 2014, 12, 1407–1417. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.C. A novel suicide inhibitor strategy for antiparasitic drug development. J. Cell. Biochem. 1991, 45, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Hua, S.B.; Wang, C.C.; Gottesdiener, K.M. Trypanosoma brucei brucei: Characterization of an ODC null bloodstream form mutant and the action of alpha-difluoromethylornithine. Exp. Parasitol. 1998, 88, 255–257. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; McCloskey, D.E.; Phillips, M.A. RNA interference-mediated silencing of ornithine decarboxylase and spermidine synthase genes in Trypanosoma brucei provides insight into regulation of polyamine biosynthesis. Eukaryot. Cell. 2009, 8, 747–755. [Google Scholar] [CrossRef] [PubMed]
- Iten, M.; Mett, H.; Evans, A.; Enyaru, J.C.; Brun, R.; Kaminsky, R. Alterations in ornithine decarboxylase characteristics account for tolerance of Trypanosoma brucei rhodesiense to d,l-α-difluoromethylornithine. Antimicrob. Agents Chemother. 1997, 41, 1922–1925. [Google Scholar] [PubMed]
- Bacchi, C.J.; Nathan, H.C.; Hutner, S.H.; McCann, P.P.; Sjoerdsma, A. Polyamine metabolism: A potential therapeutic target in trypanosomes. Science 1980, 210, 332–334. [Google Scholar] [CrossRef] [PubMed]
- Van Nieuwenhove, S.; Schechter, P.J.; Declercq, J.; Bone, G.; Burke, J.; Sjoerdsma, A. Treatment of gambiense sleeping sickness in the Sudan with oral DFMO (DL-α-difluoromethylornithine), an inhibitor of ornithine decarboxylase; first field trial. Trans. R. Soc. Trop. Med. Hyg. 1985, 79, 692–698. [Google Scholar] [CrossRef]
- Doua, F.; Boa, F.Y.; Schechter, P.J.; Miezan, T.W.; Diai, D.; Sanon, S.R.; De Raadt, P.; Haegele, K.D.; Sjoerdsma, A.; Konian, K. Treatment of human late stage gambiense trypanosomiasis with α-difluoromethylornithine (eflornithine): Efficacy and tolerance in 14 cases in Cote d’Ivoire. Am. J. Trop. Med. Hyg. 1987, 37, 525–533. [Google Scholar] [CrossRef] [PubMed]
- Kuzoe, F.A. Perspectives in research on and control of African trypanosomiasis. Ann. Trop. Med. Parasitol. 1991, 85, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Bacchi, C.J. Progress in Anti-Polyamine Drug Development/Chemotherapy vs. Protozoan-Caused Diseases: The DFMO Story. 2006. Available online: http://wizard.musc.edu/dfmostory.pdf (accessed on 27 December 2017).
- Yun, O.; Priotto, G.; Tong, J.; Flevaud, L.; Chappuis, F. NECT is next: Implementing the new drug combination therapy for Trypanosoma brucei gambiense sleeping sickness. PLoS Negl. Trop. Dis. 2010, 4, e720. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gradoni, L.; Iorio, M.A.; Gramiccia, M.; Orsini, S. In Vivo effect of eflornithine (DFMO) and some related compounds on Leishmania infantum preliminary communication. Farmaco 1989, 44, 1157–1166. [Google Scholar] [PubMed]
- Mukhopadhyay, R.; Madhubala, R. Effect of a bis(benzyl)polyamine analogue, and DL-α-difluoromethylornithine on parasite suppression and cellular polyamine levels in golden hamster during Leishmania donovani infection. Pharmacol. Res. 1993, 28, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Olenyik, T.; Gilroy, C.; Ullman, B. Oral putrescine restores virulence of ornithine decarboxylase-deficient Leishmania donovani in mice. Mol. Biochem. Parasitol. 2011, 176, 109–111. [Google Scholar] [CrossRef] [PubMed]
- Boitz, J.M.; Yates, P.A.; Kline, C.; Gaur, U.; Wilson, M.E.; Ullman, B.; Roberts, S.C. Leishmania donovani ornithine decarboxylase is indispensable for parasite survival in the mammalian host. Infect. Immun. 2009, 77, 756–763. [Google Scholar] [CrossRef] [PubMed]
- Wolf, J.E., Jr.; Shander, D.; Huber, F.; Jackson, J.; Lin, C.S.; Mathes, B.M.; Schrode, K.; Eflornithine, H.S.G. Randomized, double-blind clinical evaluation of the efficacy and safety of topical eflornithine HCl 13.9% cream in the treatment of women with facial hair. Int. J. Dermatol. 2007, 46, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Pepin, J.; Milord, F.; Guern, C.; Schechter, P.J. Difluoromethylornithine for arseno-resistant Trypanosoma brucei gambiense sleeping sickness. Lancet 1987, 2, 1431–1433. [Google Scholar] [CrossRef]
- Jansen, C.; Mattox, D.E.; Miller, K.D.; Brownell, W.E. An animal model of hearing loss from α-difluoromethylornithine. Arch. Otolaryngol. Head Neck Surg. 1989, 115, 1234–1237. [Google Scholar] [CrossRef] [PubMed]
- Salzer, S.J.; Mattox, D.E.; Brownell, W.E. Cochlear damage and increased threshold in α-difluoromethylornithine (DFMO) treated guinea pigs. Hear. Res. 1990, 46, 101–112. [Google Scholar] [CrossRef]
- Wheeler, D.L.; Ness, K.J.; Oberley, T.D.; Verma, A.K. Inhibition of the development of metastatic squamous cell carcinoma in protein kinase C ε transgenic mice by α-difluoromethylornithine accompanied by marked hair follicle degeneration and hair loss. Cancer Res. 2003, 63, 3037–3042. [Google Scholar] [PubMed]
- Janne, J.; Alhonen, L.; Pietila, M.; Keinanen, T.A. Genetic approaches to the cellular functions of polyamines in mammals. Eur. J. Biochem. 2004, 271, 877–894. [Google Scholar] [CrossRef] [PubMed]
- Pietila, M.; Parkkinen, J.J.; Alhonen, L.; Janne, J. Relation of skin polyamines to the hairless phenotype in transgenic mice overexpressing spermidine/spermine N1-acetyltransferase. J. Investig. Dermatol. 2001, 116, 801–805. [Google Scholar] [CrossRef] [PubMed]
- Soler, A.P.; Gilliard, G.; Megosh, L.C.; O’Brien, T.G. Modulation of murine hair follicle function by alterations in ornithine decarboxylase activity. J. Investig. Dermatol. 1996, 106, 1108–1113. [Google Scholar] [CrossRef] [PubMed]
- Hamzavi, I.; Tan, E.; Shapiro, J.; Lui, H. A randomized bilateral vehicle-controlled study of eflornithine cream combined with laser treatment versus laser treatment alone for facial hirsutism in women. J. Am. Acad. Dermatol. 2007, 57, 54–59. [Google Scholar] [CrossRef] [PubMed]
- Alberts, D.S.; Dorr, R.T.; Einspahr, J.G.; Aickin, M.; Saboda, K.; Xu, M.J.; Peng, Y.M.; Goldman, R.; Foote, J.A.; Warneke, J.A.; et al. Chemoprevention of human actinic keratoses by topical 2-(difluoromethyl)-DL-ornithine. Cancer Epidemiol. Biomark. Prev. 2000, 9, 1281–1286. [Google Scholar]
- Bartels, P.; Yozwiak, M.; Einspahr, J.; Saboda, K.; Liu, Y.; Brooks, C.; Bartels, H.; Alberts, D.S. Chemopreventive efficacy of topical difluoromethylornithine and/or triamcinolone in the treatment of actinic keratoses analyzed by karyometry. Anal. Quant. Cytol. Histol. 2009, 31, 355–366. [Google Scholar] [PubMed]
- Babbar, N.; Gerner, E.W. Targeting polyamines and inflammation for cancer prevention. Recent Results Cancer Res. 2011, 188, 49–64. [Google Scholar] [PubMed]
- Gerner, E.W.; Meyskens, F.L., Jr. Combination chemoprevention for colon cancer targeting polyamine synthesis and inflammation. Clin. Cancer Res. 2009, 15, 758–761. [Google Scholar] [CrossRef] [PubMed]
- Love, R.R.; Jacoby, R.; Newton, M.A.; Tutsch, K.D.; Simon, K.; Pomplun, M.; Verma, A.K. A randomized, placebo-controlled trial of low-dose α-difluoromethylornithine in individuals at risk for colorectal cancer. Cancer Epidemiol. Biomark. Prev. 1998, 7, 989–992. [Google Scholar]
- Meyskens, F.L., Jr.; Gerner, E.W.; Emerson, S.; Pelot, D.; Durbin, T.; Doyle, K.; Lagerberg, W. Effect of α-difluoromethylornithine on rectal mucosal levels of polyamines in a randomized, double-blinded trial for colon cancer prevention. J. Natl. Cancer Inst. 1998, 90, 1212–1218. [Google Scholar] [CrossRef] [PubMed]
- Rial, N.S.; Meyskens, F.L.; Gerner, E.W. Polyamines as mediators of APC-dependent intestinal carcinogenesis and cancer chemoprevention. Essays Biochem. 2009, 46, 111–124. [Google Scholar] [CrossRef] [PubMed]
- Thompson, P.A.; Wertheim, B.C.; Zell, J.A.; Chen, W.P.; McLaren, C.E.; LaFleur, B.J.; Meyskens, F.L.; Gerner, E.W. Levels of rectal mucosal polyamines and prostaglandin E2 predict ability of DFMO and sulindac to prevent colorectal adenoma. Gastroenterology 2010, 139, 797–805. [Google Scholar] [CrossRef] [PubMed]
- Paz, E.A.; LaFleur, B.; Gerner, E.W. Polyamines are oncometabolites that regulate the LIN28/let-7 pathway in colorectal cancer cells. Mol. Carcinog. 2014, 53, E96–E106. [Google Scholar] [CrossRef] [PubMed]
- Gerner, E.W.; Meyskens, F.L., Jr.; Goldschmid, S.; Lance, P.; Pelot, D. Rationale for, and design of, a clinical trial targeting polyamine metabolism for colon cancer chemoprevention. Amino Acids 2007, 33, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Ignatenko, N.A.; Zhang, H.; Watts, G.S.; Skovan, B.A.; Stringer, D.E.; Gerner, E.W. The chemopreventive agent α-difluoromethylornithine blocks Ki-ras-dependent tumor formation and specific gene expression in Caco-2 cells. Mol. Carcinog. 2004, 39, 221–233. [Google Scholar] [CrossRef] [PubMed]
- LeGendre-McGhee, S.; Rice, P.S.; Wall, R.A.; Sprute, K.J.; Bommireddy, R.; Luttman, A.M.; Nagle, R.B.; Abril, E.R.; Farrell, K.; Hsu, C.H.; et al. Time-serial Assessment of Drug Combination Interventions in a Mouse Model of Colorectal Carcinogenesis Using Optical Coherence Tomography. Cancer Growth Metastasis 2015, 8, 63–80. [Google Scholar] [CrossRef] [PubMed]
- Ibanez, C.; Simo, C.; Valdes, A.; Campone, L.; Piccinelli, A.L.; Garcia-Canas, V.; Cifuentes, A. Metabolomics of adherent mammalian cells by capillary electrophoresis-mass spectrometry: HT-29 cells as case study. J. Pharm. Biomed. Anal. 2015, 110, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Witherspoon, M.; Chen, Q.; Kopelovich, L.; Gross, S.S.; Lipkin, S.M. Unbiased metabolite profiling indicates that a diminished thymidine pool is the underlying mechanism of colon cancer chemoprevention by α-difluoromethylornithine. Cancer Discov. 2013, 3, 1072–1081. [Google Scholar] [CrossRef] [PubMed]
- Meyskens, F.L., Jr.; Emerson, S.S.; Pelot, D.; Meshkinpour, H.; Shassetz, L.R.; Einspahr, J.; Alberts, D.S.; Gerner, E.W. Dose de-escalation chemoprevention trial of α-difluoromethylornithine in patients with colon polyps. J. Natl. Cancer Inst. 1994, 86, 1122–1130. [Google Scholar] [CrossRef] [PubMed]
- Jacoby, R.F.; Cole, C.E.; Tutsch, K.; Newton, M.A.; Kelloff, G.; Hawk, E.T.; Lubet, R.A. Chemopreventive efficacy of combined piroxicam and difluoromethylornithine treatment of APC mutant Min mouse adenomas, and selective toxicity against APC mutant embryos. Cancer Res. 2000, 60, 1864–1870. [Google Scholar] [PubMed]
- Li, H.; Schut, H.A.; Conran, P.; Kramer, P.M.; Lubet, R.A.; Steele, V.E.; Hawk, E.E.; Kelloff, G.J.; Pereira, M.A. Prevention by aspirin and its combination with α-difluoromethylornithine of azoxymethane-induced tumors, aberrant crypt foci and prostaglandin E2 levels in rat colon. Carcinogenesis 1999, 20, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Laukaitis, C.M.; Erdman, S.H.; Gerner, E.W. Chemoprevention in patients with genetic risk of colorectal cancers. Colorectal Cancer 2012, 1, 225–240. [Google Scholar] [CrossRef] [PubMed]
- Raj, K.P.; Zell, J.A.; Rock, C.L.; McLaren, C.E.; Zoumas-Morse, C.; Gerner, E.W.; Meyskens, F.L. Role of dietary polyamines in a phase III clinical trial of difluoromethylornithine (DFMO) and sulindac for prevention of sporadic colorectal adenomas. Br. J. Cancer 2013, 108, 512–518. [Google Scholar] [CrossRef] [PubMed]
- Sporn, M.B.; Hong, W.K. Concomitant DFMO and sulindac chemoprevention of colorectal adenomas: A major clinical advance. Nat. Clin. Pract. Oncol. 2008, 5, 628–629. [Google Scholar] [CrossRef] [PubMed]
- Martinez, M.E.; O’Brien, T.G.; Fultz, K.E.; Babbar, N.; Yerushalmi, H.; Qu, N.; Guo, Y.; Boorman, D.; Einspahr, J.; Alberts, D.S.; et al. Pronounced reduction in adenoma recurrence associated with aspirin use and a polymorphism in the ornithine decarboxylase gene. Proc. Natl. Acad. Sci. USA 2003, 100, 7859–7864. [Google Scholar] [CrossRef] [PubMed]
- Hubner, R.A.; Muir, K.R.; Liu, J.F.; Logan, R.F.; Grainge, M.J.; Houlston, R.S. Members of the, U.C. Ornithine decarboxylase G316A genotype is prognostic for colorectal adenoma recurrence and predicts efficacy of aspirin chemoprevention. Clin. Cancer Res. 2008, 14, 2303–2309. [Google Scholar] [CrossRef] [PubMed]
- Janakiram, N.B.; Mohammed, A.; Bryant, T.; Zhang, Y.; Brewer, M.; Duff, A.; Biddick, L.; Singh, A.; Lightfoot, S.; Steele, V.E.; et al. Potentiating NK cell activity by combination of Rosuvastatin and Difluoromethylornithine for effective chemopreventive efficacy against Colon Cancer. Sci. Rep. 2016, 6, 37046. [Google Scholar] [CrossRef] [PubMed]
- Lao, C.D.; Backoff, P.; Shotland, L.I.; McCarty, D.; Eaton, T.; Ondrey, F.G.; Viner, J.L.; Spechler, S.J.; Hawk, E.T.; Brenner, D.E. Irreversible ototoxicity associated with difluoromethylornithine. Cancer Epidemiol. Biomark. Prev. 2004, 13, 1250–1252. [Google Scholar]
- Pasic, T.R.; Heisey, D.; Love, R.R. α-difluoromethylornithine ototoxicity. Chemoprevention clinical trial results. Arch. Otolaryngol. Head Neck Surg. 1997, 123, 1281–1286. [Google Scholar] [CrossRef] [PubMed]
- Gamble, L.D.; Hogarty, M.D.; Liu, X.; Ziegler, D.S.; Marshall, G.; Norris, M.D.; Haber, M. Polyamine pathway inhibition as a novel therapeutic approach to treating neuroblastoma. Front. Oncol. 2012, 2, 162. [Google Scholar] [CrossRef] [PubMed]
- Saulnier Sholler, G.L.; Gerner, E.W.; Bergendahl, G.; MacArthur, R.B.; VanderWerff, A.; Ashikaga, T.; Bond, J.P.; Ferguson, W.; Roberts, W.; Wada, R.K.; et al. A Phase I Trial of DFMO Targeting Polyamine Addiction in Patients with Relapsed/Refractory Neuroblastoma. PLoS ONE 2015, 10, e0127246. [Google Scholar]
- Louis, C.U.; Shohet, J.M. Neuroblastoma: Molecular pathogenesis and therapy. Annu. Rev. Med. 2015, 66, 49–63. [Google Scholar] [CrossRef] [PubMed]
- Evageliou, N.F.; Hogarty, M.D. Disrupting polyamine homeostasis as a therapeutic strategy for neuroblastoma. Clin. Cancer Res. 2009, 15, 5956–5961. [Google Scholar] [CrossRef] [PubMed]
- Rasmuson, A.; Segerstrom, L.; Nethander, M.; Finnman, J.; Elfman, L.H.; Javanmardi, N.; Nilsson, S.; Johnsen, J.I.; Martinsson, T.; Kogner, P. Tumor development, growth characteristics and spectrum of genetic aberrations in the TH-MYCN mouse model of neuroblastoma. PLoS ONE 2012, 7, e51297. [Google Scholar] [CrossRef] [PubMed]
- Rounbehler, R.J.; Li, W.; Hall, M.A.; Yang, C.; Fallahi, M.; Cleveland, J.L. Targeting ornithine decarboxylase impairs development of MYCN-amplified neuroblastoma. Cancer Res. 2009, 69, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Hogarty, M.D.; Norris, M.D.; Davis, K.; Liu, X.; Evageliou, N.F.; Hayes, C.S.; Pawel, B.; Guo, R.; Zhao, H.; Sekyere, E.; et al. ODC1 is a critical determinant of MYCN oncogenesis and a therapeutic target in neuroblastoma. Cancer Res. 2008, 68, 9735–9745. [Google Scholar] [CrossRef] [PubMed]
- Dang, C.V.; Reddy, E.P.; Shokat, K.M.; Soucek, L. Drugging the ‘undruggable’ cancer targets. Nat. Rev. Cancer 2017, 17, 502–508. [Google Scholar] [CrossRef] [PubMed]
- Wallick, C.J.; Gamper, I.; Thorne, M.; Feith, D.J.; Takasaki, K.Y.; Wilson, S.M.; Seki, J.A.; Pegg, A.E.; Byus, C.V.; Bachmann, A.S. Key role for p27Kip1, retinoblastoma protein Rb, and MYCN in polyamine inhibitor-induced G1 cell cycle arrest in MYCN-amplified human neuroblastoma cells. Oncogene 2005, 24, 5606–5618. [Google Scholar] [CrossRef] [PubMed]
- Herr, H.W.; Kleinert, E.L.; Relyea, N.M.; Whitmore, W.F., Jr. Potentiation of methylglyoxal-bis-guanylhydrazone by α-difluoromethylornithine in rat prostate cancer. Cancer 1984, 53, 1294–1298. [Google Scholar] [CrossRef]
- Evageliou, N.F.; Haber, M.; Vu, A.; Laetsch, T.W.; Murray, J.; Gamble, L.D.; Cheng, N.C.; Liu, K.; Reese, M.; Corrigan, K.A.; et al. Polyamine Antagonist Therapies Inhibit Neuroblastoma Initiation and Progression. Clin. Cancer Res. 2016, 22, 4391–4404. [Google Scholar] [CrossRef] [PubMed]
- Alhonen-Hongisto, L.; Seppanen, P.; Janne, J. Intracellular putrescine and spermidine deprivation induces increased uptake of the natural polyamines and methylglyoxal bis(guanylhydrazone). Biochem. J. 1980, 192, 941–945. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Weeks, R.S.; Burns, M.R.; Boorman, D.W.; Klein-Szanto, A.; O’Brien, T.G. Combination therapy with 2-difluoromethylornithine and a polyamine transport inhibitor against murine squamous cell carcinoma. Int. J. Cancer 2006, 118, 2344–2349. [Google Scholar] [CrossRef] [PubMed]
- Sunkara, P.S.; Prakash, N.J.; Rosenberger, A.L. An essential role for polyamines in tumor metastases. FEBS Lett. 1982, 150, 397–399. [Google Scholar] [CrossRef]
- Burns, M.R.; Graminski, G.F.; Weeks, R.S.; Chen, Y.; O’Brien, T.G. Lipophilic lysine-spermine conjugates are potent polyamine transport inhibitors for use in combination with a polyamine biosynthesis inhibitor. J. Med. Chem. 2009, 52, 1983–1993. [Google Scholar] [CrossRef] [PubMed]
- Hayes, C.S.; Burns, M.R.; Gilmour, S.K. Polyamine blockade promotes antitumor immunity. Oncoimmunology 2014, 3, e27360. [Google Scholar] [CrossRef] [PubMed]
- Hayes, C.S.; Shicora, A.C.; Keough, M.P.; Snook, A.E.; Burns, M.R.; Gilmour, S.K. Polyamine-blocking therapy reverses immunosuppression in the tumor microenvironment. Cancer Immunol. Res. 2014, 2, 274–285. [Google Scholar] [CrossRef] [PubMed]
- Nowotarski, S.L.; Woster, P.M.; Casero, R.A., Jr. Polyamines and cancer: Implications for chemotherapy and chemoprevention. Expert Rev. Mol. Med. 2013, 15, e3. [Google Scholar] [CrossRef] [PubMed]
- Samal, K.; Zhao, P.; Kendzicky, A.; Yco, L.P.; McClung, H.; Gerner, E.; Burns, M.; Bachmann, A.S.; Sholler, G. AMXT-1501, a novel polyamine transport inhibitor, synergizes with DFMO in inhibiting neuroblastoma cell proliferation by targeting both ornithine decarboxylase and polyamine transport. Int. J. Cancer 2013, 133, 1323–1333. [Google Scholar] [CrossRef] [PubMed]
- Casero, R.A., Jr.; Marton, L.J. Targeting polyamine metabolism and function in cancer and other hyperproliferative diseases. Nat. Rev. Drug Discov. 2007, 6, 373–390. [Google Scholar] [CrossRef] [PubMed]
- Alexander, E.T.; Minton, A.; Peters, M.C.; Phanstiel, O.T.; Gilmour, S.K. A novel polyamine blockade therapy activates an anti-tumor immune response. Oncotarget 2017, 8, 84140–84152. [Google Scholar] [CrossRef] [PubMed]
- Devens, B.H.; Weeks, R.S.; Burns, M.R.; Carlson, C.L.; Brawer, M.K. Polyamine depletion therapy in prostate cancer. Prostate Cancer Prostatic Dis. 2000, 3, 275–279. [Google Scholar] [CrossRef] [PubMed]
- Gitto, S.B.; Pandey, V.; Oyer, J.L.; Copik, A.J.; Hogan, F.C.; Phanstiel, O., 4th; Altomare, D.A. Difluoromethylornithine Combined with a Polyamine Transport Inhibitor Is Effective against Gemcitabine Resistant Pancreatic Cancer. Mol. Pharm. 2018. [Google Scholar] [CrossRef] [PubMed]
- Muth, A.; Madan, M.; Archer, J.J.; Ocampo, N.; Rodriguez, L.; Phanstiel, O., 4th. Polyamine transport inhibitors: Design, synthesis, and combination therapies with difluoromethylornithine. J. Med. Chem. 2014, 57, 348–363. [Google Scholar] [CrossRef] [PubMed]
- Massaro, C.; Thomas, J.; Phanstiel Iv, O. Investigation of Polyamine Metabolism and Homeostasis in Pancreatic Cancers. Med. Sci. 2017, 5, 32. [Google Scholar] [CrossRef] [PubMed]
- Phanstiel, O., 4th. An overview of polyamine metabolism in pancreatic ductal adenocarcinoma. Int. J. Cancer 2017. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, A.; Janakiram, N.B.; Madka, V.; Ritchie, R.L.; Brewer, M.; Biddick, L.; Patlolla, J.M.; Sadeghi, M.; Lightfoot, S.; Steele, V.E.; et al. Eflornithine (DFMO) prevents progression of pancreatic cancer by modulating ornithine decarboxylase signaling. Cancer Prev. Res. 2014, 7, 1198–1209. [Google Scholar] [CrossRef] [PubMed]
- Bailey, H.H.; Kim, K.; Verma, A.K.; Sielaff, K.; Larson, P.O.; Snow, S.; Lenaghan, T.; Viner, J.L.; Douglas, J.; Dreckschmidt, N.E.; et al. A randomized, double-blind, placebo-controlled phase 3 skin cancer prevention study of α-difluoromethylornithine in subjects with previous history of skin cancer. Cancer Prev. Res. 2010, 3, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Jeter, J.M.; Alberts, D.S. Difluoromethylornithine: The proof is in the polyamines. Cancer Prev. Res. 2012, 5, 1341–1344. [Google Scholar] [CrossRef] [PubMed]
- Kreul, S.M.; Havighurst, T.; Kim, K.; Mendonca, E.A.; Wood, G.S.; Snow, S.; Borich, A.; Verma, A.; Bailey, H.H. A phase III skin cancer chemoprevention study of DFMO: Long-term follow-up of skin cancer events and toxicity. Cancer Prev. Res. 2012, 5, 1368–1374. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.I.; Schultz, C.R.; Buras, A.L.; Friedman, E.; Fedorko, A.; Seamon, L.; Chandramouli, G.V.R.; Maxwell, G.L.; Bachmann, A.S.; Risinger, J.I. Ornithine decarboxylase as a therapeutic target for endometrial cancer. PLoS ONE 2017, 12, e0189044. [Google Scholar] [CrossRef] [PubMed]
- Arisan, E.D.; Obakan, P.; Coker, A.; Palavan-Unsal, N. Inhibition of ornithine decarboxylase alters the roscovitine-induced mitochondrial-mediated apoptosis in MCF-7 breast cancer cells. Mol. Med. Rep. 2012, 5, 1323–1329. [Google Scholar] [PubMed]
- Zhu, Q.; Jin, L.; Casero, R.A.; Davidson, N.E.; Huang, Y. Role of ornithine decarboxylase in regulation of estrogen receptor alpha expression and growth in human breast cancer cells. Breast Cancer Res. Treat. 2012, 136, 57–66. [Google Scholar] [CrossRef] [PubMed]




© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
LoGiudice, N.; Le, L.; Abuan, I.; Leizorek, Y.; Roberts, S.C. Alpha-Difluoromethylornithine, an Irreversible Inhibitor of Polyamine Biosynthesis, as a Therapeutic Strategy against Hyperproliferative and Infectious Diseases. Med. Sci. 2018, 6, 12. https://doi.org/10.3390/medsci6010012
LoGiudice N, Le L, Abuan I, Leizorek Y, Roberts SC. Alpha-Difluoromethylornithine, an Irreversible Inhibitor of Polyamine Biosynthesis, as a Therapeutic Strategy against Hyperproliferative and Infectious Diseases. Medical Sciences. 2018; 6(1):12. https://doi.org/10.3390/medsci6010012
Chicago/Turabian StyleLoGiudice, Nicole, Linh Le, Irene Abuan, Yvette Leizorek, and Sigrid C. Roberts. 2018. "Alpha-Difluoromethylornithine, an Irreversible Inhibitor of Polyamine Biosynthesis, as a Therapeutic Strategy against Hyperproliferative and Infectious Diseases" Medical Sciences 6, no. 1: 12. https://doi.org/10.3390/medsci6010012
APA StyleLoGiudice, N., Le, L., Abuan, I., Leizorek, Y., & Roberts, S. C. (2018). Alpha-Difluoromethylornithine, an Irreversible Inhibitor of Polyamine Biosynthesis, as a Therapeutic Strategy against Hyperproliferative and Infectious Diseases. Medical Sciences, 6(1), 12. https://doi.org/10.3390/medsci6010012