Fractionation of Cellulose-Rich Products from an Empty Fruit Bunch (EFB) by Means of Steam Explosion Followed by Organosolv Treatment
Abstract
:1. Introduction
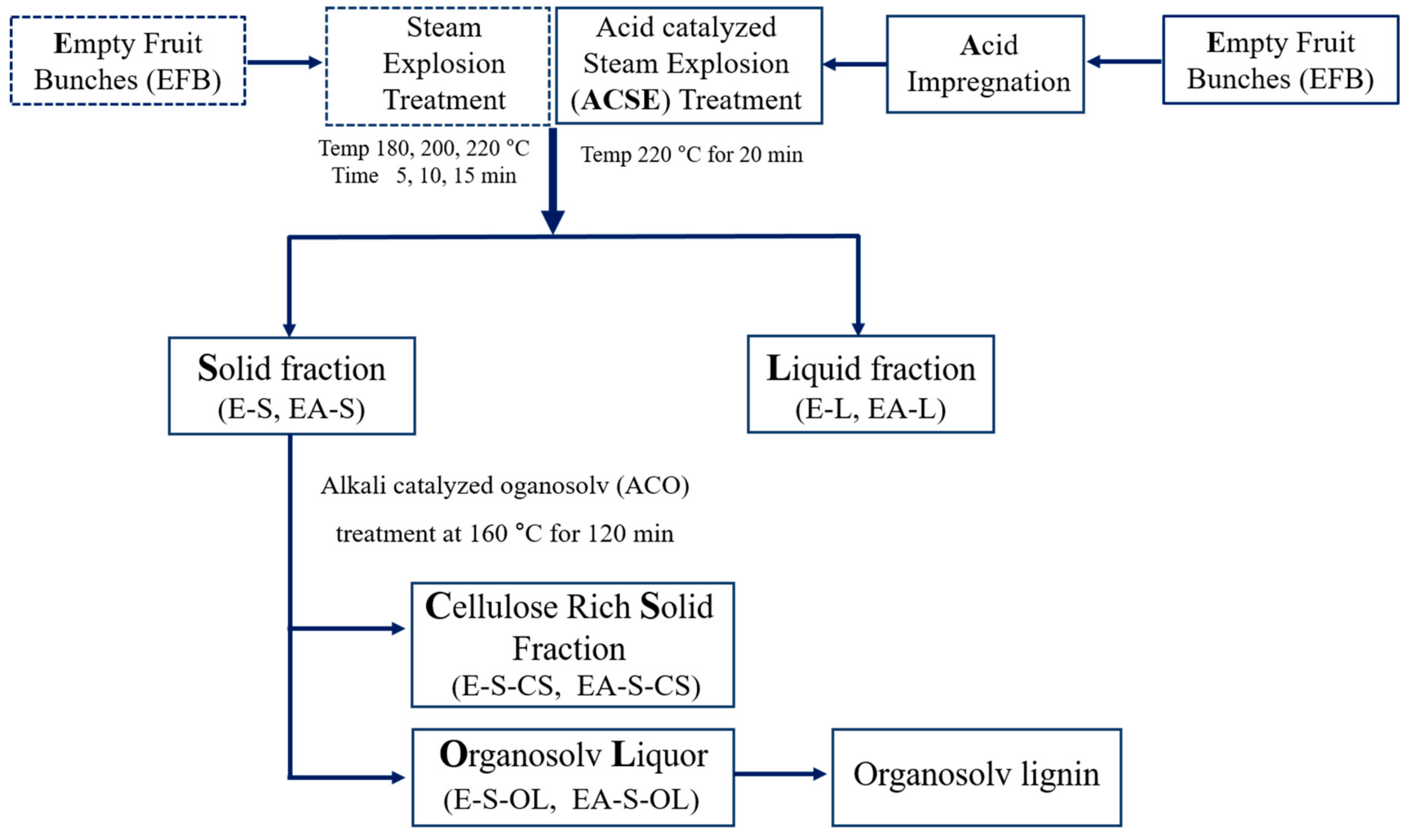
2. Materials and Methods
2.1. Feedstock Analysis
2.2. ACSE-First Pretreatment
2.3. ACO Second Pretreatment
2.4. Analysis
3. Results and Discussion
3.1. Mass Balance
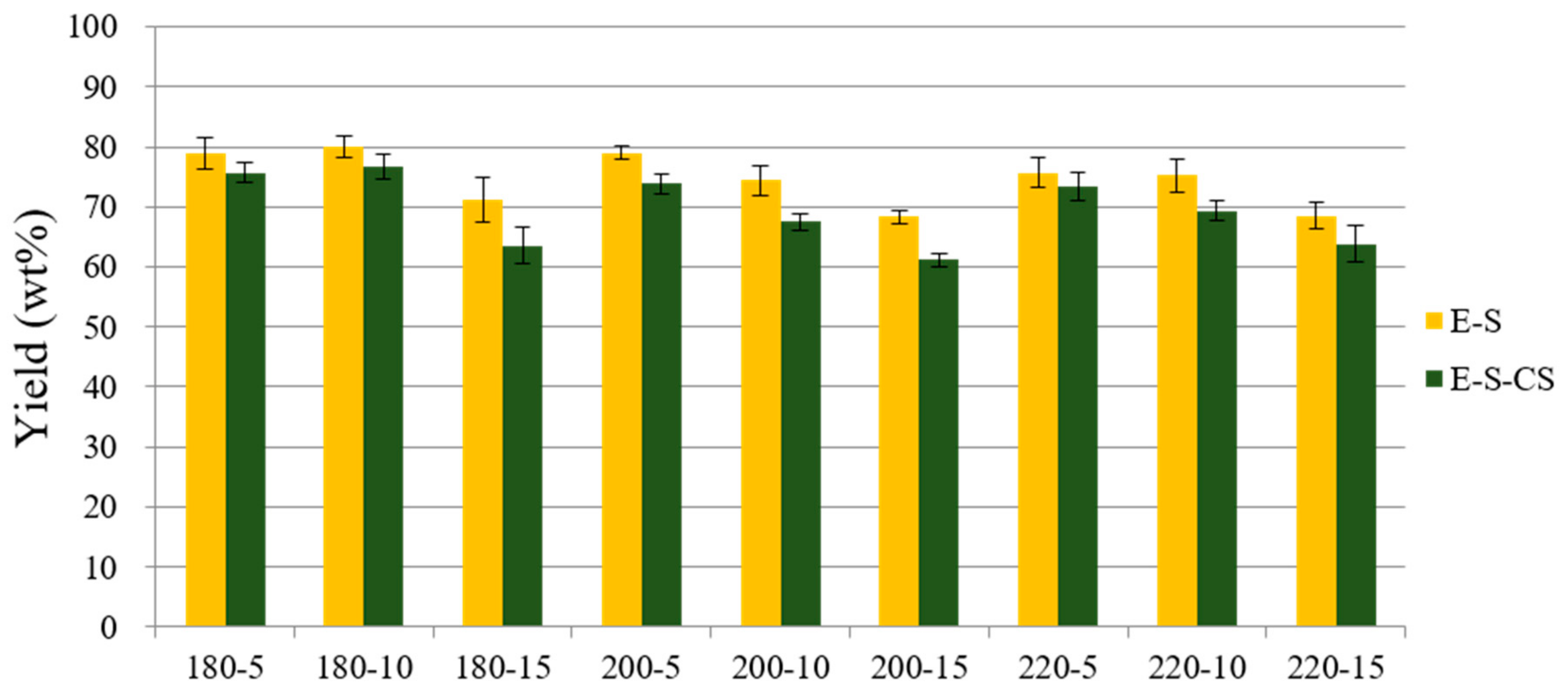
3.1.1. Effect of Residence Time and Temperature on Fractionation
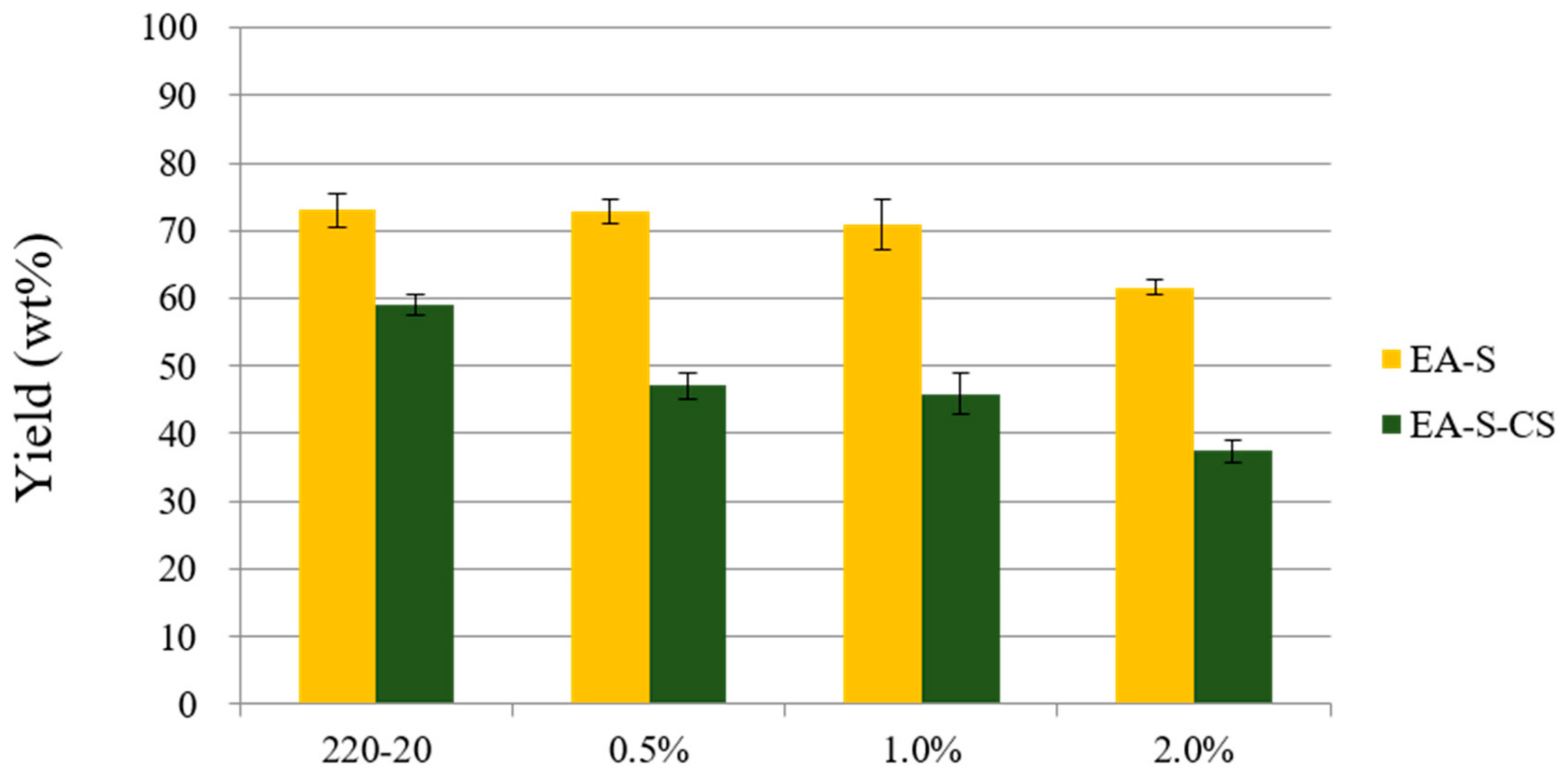
3.1.2. Effect of Dilute Acid Impregnation on Fractionation
3.1.3. Sugars Quantification
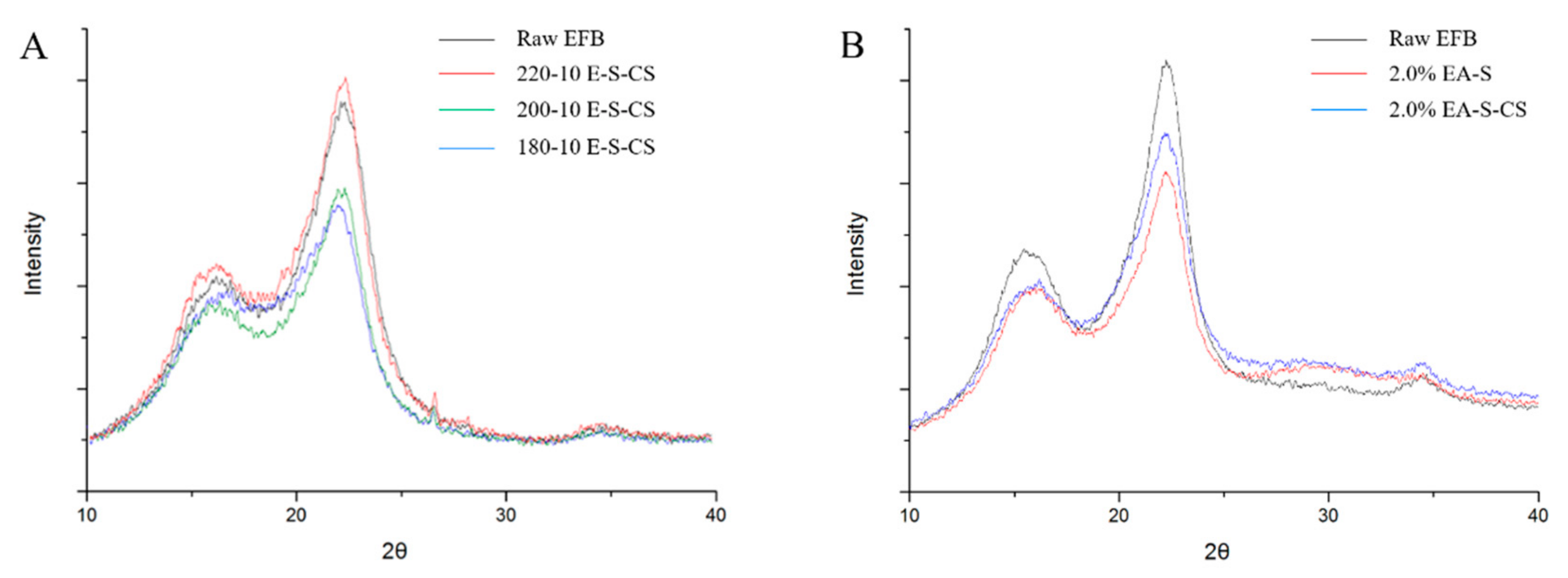
3.2. Cellulose Crystallinity
3.3. Chemical Properties of Organosolv Lignin
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- United States Department of Agriculture Economics, Statistics and Market Information System. Oilseeds: World Markets and Trade. Available online: https://downloads.usda.library.cornell.edu/usda-esmis/files/tx31qh68h/sq87bv06k/9z903025j/oilseed-trade-09-12-2018.pdf (accessed on 21 December 2018).
- Pleanjai, S.; Gheewala, S.H.; Garivait, S. Environmental evaluation of biodiesel production from palm oil in a life cycle perspective. Asian J. Energy Environ. 2007, 8, 15–32. [Google Scholar]
- Shrotri, A.; Kobayashi, H.; Fukuoka, A. Catalytic conversion of structural carbohydrates and lignin to chemicals. Adv. Catal. 2017, 60, 59–123. [Google Scholar]
- Brownell, H.; Yu, E.; Saddler, J. Steam-explosion pretreatment of wood: Effect of chip size, acid, moisture content and pressure drop. Biotechnol. Bioeng. 1986, 28, 792–801. [Google Scholar] [CrossRef] [PubMed]
- Choi, W.-I.; Park, J.-Y.; Lee, J.-P.; Oh, Y.-K.; Park, Y.C.; Kim, J.S.; Park, J.M.; Kim, C.H.; Lee, J.-S. Optimization of NaOH-catalyzed steam pretreatment of empty fruit bunch. Biotechnol. Biofuels 2013, 6, 170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Emmel, A.; Mathias, A.L.; Wypych, F.; Ramos, L.P. Fractionation of eucalyptus grandis chips by dilute acid-catalysed steam explosion. Bioresour. Technol. 2003, 86, 105–115. [Google Scholar] [CrossRef]
- Alvira, P.; Tomas-Pejo, E.; Ballesteros, M.; Negro, M.J. Pretreatment technologies for an efficient bioethanol production process based on enzymatic hydrolysis: A review. Bioresour. Technol. 2010, 101, 4851–4861. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Cheng, K.; Liu, D. Organosolv pretreatment of lignocellulosic biomass for enzymatic hydrolysis. Appl. Microbiol. Biotechnol. 2009, 82, 815. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, M.; Canton, L.; Rodriguez, A.; Labidi, J. Effect of organosolv and soda pulping processes on the metals content of non-woody pulps. Bioresour. Technol. 2008, 99, 6621–6625. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Moon, J.G.; Choi, I.-G.; Choi, J.W. Study on the thermochemical degradation features of empty fruit bunch on the function of pyrolysis temperature. J. Korean Wood Sci. Technol. 2016, 44, 350–359. [Google Scholar] [CrossRef] [Green Version]
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D. Determination of Ash in Biomass; Laboratory Analytical Procedure (LAP) Technical Report NREL/TP-510-42622; National Renewable Energy Laboratory: Golden, CO, USA, 2005.
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D.; Crocker, D. Determination of Structural Carbohydrates and Lignin in Biomass; Laboratory Analytical Procedure (LAP) Technical Report NREL/TP-510-42618; National Renewable Energy Laboratory: Golden, CO, USA, 2008.
- Sluiter, A.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D. Determination of Extractives in Biomass; Laboratory Analytical Procedure (LAP) Technical Report NREL/TP-510-42619; National Renewable Energy Laboratory: Golden, CO, USA, 2005.
- Lee, J.H.; Hwang, H.; Moon, J.; Choi, J.W. Characterization of hydrothermal liquefaction products from coconut shell in the presence of selected transition metal chlorides. J. Anal. Appl. Pyrol. 2016, 122, 415–421. [Google Scholar] [CrossRef]
- Moon, J.; Lee, J.H.; Hwang, H.; Choi, I.-G.; Choi, J.W. Effect of inorganic constituents existing in empty fruit bunch (EFB) on features of pyrolysis products. J. Korean Wood Sci. Technol. 2016, 44, 629–638. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.-Y.; Lee, J.H.; Park, J.; Kim, J.K.; An, D.; Song, I.K.; Choi, J.W. Catalytic pyrolysis of lignin over HZSM-5 catalysts: Effect of various parameters on the production of aromatic hydrocarbon. J. Anal. Appl. Pyrolysis 2015, 114, 273–280. [Google Scholar] [CrossRef]
- Overend, R.P.; Chornet, E. Fractionation of lignocellulosics by steam-aqueous pretreatment. Philos Trans R Soc Lond A 1987, 321, 523–536. [Google Scholar] [CrossRef]
- Haghighi Mood, S.; Hossein Golfeshan, A.; Tabatabaei, M.; Salehi Jouzani, G.; Najafi, G.H.; Gholami, M.; Ardjmand, M. Lignocellulosic biomass to bioethanol, a comprehensive review with a focus on pretreatment. Renew. Sust. Energ. Rev. 2013, 27, 77–93. [Google Scholar] [CrossRef]
- Kapoor, M.; Raj, T.; Vijayaraj, M.; Chopra, A.; Gupta, R.P.; Tuli, D.K.; Kumar, R. Structural features of dilute acid, steam exploded, and alkali pretreated mustard stalk and their impact on enzymatic hydrolysis. Carbohydr. Polym. 2015, 124, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.H.; Pen, B.L.; Yu, C.T.; Hwang, W.S. Pretreatment efficiency and structural characterization of rice straw by an integrated process of dilute-acid and steam explosion for bioethanol production. Bioresour. Technol. 2011, 102, 2916–2924. [Google Scholar] [CrossRef] [PubMed]
- Zimbardi, F.; Viola, E.; Nanna, F.; Larocca, E.; Cardinale, M.; Barisano, D. Acid impregnation and steam explosion of corn stover in batch processes. Ind. Crops Prod. 2007, 26, 195–206. [Google Scholar] [CrossRef]
- Fockink, D.H.; Sánchez, J.H.; Ramos, L.P. Comprehensive analysis of sugarcane bagasse steam explosion using autocatalysis and dilute acid hydrolysis (H3PO4 and H2SO4) at equivalent combined severity factors. Ind. Crops Prod. 2018, 123, 563–572. [Google Scholar] [CrossRef]
- Park, S.; Baker, J.O.; Himmel, M.E.; Parilla, P.A.; Johnson, D.K. Cellulose crystallinity index: measurement techniques and their impact on interpreting cellulase performance. Biotechnol. biofuels 2010, 3, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Properties | Value |
|---|---|
| Elemental analysis (wt%) | |
| Carbon | 45.4 ± 0.4 |
| Hydrogen | 5.0 ± 0.2 |
| Nitrogen | 0.6 ± 0.0 |
| Oxygen 1 | 49.0 ± 0.2 |
| Component analysis (wt%) | |
| Holocellulose | 70.6 ± 0.2 |
| Arabinose | 6.1 |
| Galactose | 3.1 |
| Glucose | 39.0 |
| Xylose | 18.8 |
| Lignin | 18.0 ± 0.2 |
| Extractives | 4.6 ± 0.1 |
| Ash | 6.2 ± 0.3 |
| Inorganic element (ppm) | |
| Aluminum | 1760 |
| Calcium | 2170 |
| Iron | 2680 |
| Magnesium | 790 |
| Potassium | 11,930 |
| Phosphorus | 750 |
| Silicon | 760 |
| Thermogravimetric analysis | |
| Volatiles (wt%) | 80.5 |
| Char (wt%) | 19.5 |
| Temperature at max. degradation rate (°C) | 318.8 |
| Holocellulose (wt%) | Lignin (wt%) | ||
|---|---|---|---|
| Raw EFB | 70.6 ± 0.2 | 18.0 ± 0.2 | |
| E-S | 180-5 | 70.2 ± 0.1 | 23.3 ± 0.0 |
| 180-10 | 69.3 ± 0.2 | 22.9 ± 0.1 | |
| 180-15 | 70.8 ± 0.2 | 25.1 ± 0.2 | |
| 200-5 | 71.2 ± 0.1 | 24.2 ± 1.0 | |
| 200-10 | 69.9 ± 0.1 | 27.9 ± 0.8 | |
| 200-15 | 71.6 ± 0.4 | 29.3 ± 0.8 | |
| 220-5 | 68.3 ± 0.1 | 29.4 ± 0.8 | |
| 220-10 | 67.5 ± 0.0 | 33.7 ± 0.1 | |
| 220-15 | 67.9 ± 0.3 | 34.6 ± 0.2 | |
| E-S-CS | 180-5 | 80.8 ± 0.3 | 20.1 ± 0.1 |
| 180-10 | 80.3 ± 0.2 | 20.2 ± 0.1 | |
| 180-15 | 81.1 ± 0.2 | 21.6 ± 0.3 | |
| 200-5 | 83.6 ± 0.1 | 20.6 ± 0.1 | |
| 200-10 | 80.2 ± 0.2 | 22.8 ± 0.5 | |
| 200-15 | 81.5 ± 0.1 | 22.9 ± 0.9 | |
| 220-5 | 83.8 ± 0.2 | 21.5 ± 0.6 | |
| 220-10 | 74.9 ± 0.8 | 24.6 ± 0.5 | |
| 220-15 | 76.2 ± 0.1 | 22.8 ± 0.2 | |
| Holocellulose (wt%) | Lignin (wt%) | ||
|---|---|---|---|
| Raw EFB | 70.6 ± 0.2 | 18.0 ± 0.2 | |
| EA-S | 220-20 | 69.0 ± 0.2 | 24.4 ± 0.8 |
| 0.5% | 56.7 ± 0.2 | 33.7 ± 0.2 | |
| 1.0% | 52.0 ± 0.1 | 38.0 ± 0.1 | |
| 2.0% | 45.6 ± 0.1 | 45.9 ± 0.8 | |
| EA-S-CS | 220-20 | 64.1 ± 0.1 | 23.2 ± 0.8 |
| 0.5% | 56.5 ± 0.4 | 25.3 ± 0.4 | |
| 1.0% | 49.3 ± 0.3 | 27.9 ± 0.4 | |
| 2.0% | 42.8 ± 0.1 | 30.1 ± 0.3 | |
| Total Sugar (mg/g Fraction) | Lignin (mg/g Fraction) | ||||||
|---|---|---|---|---|---|---|---|
| Glucose | Xylose | Arabinose | Galactose | Mannose | |||
| EA-S | 220-20 | 483 | 170 | 23 | 10 | 4 | 244 |
| 0.5% | 433 | 104 | - | - | - | 337 | |
| 1.0% | 421 | 76 | - | - | - | 380 | |
| 2.0% | 373 | 33 | - | - | - | 459 | |
| EA-S-CS | 220-20 | 447 | 161 | 20 | 8 | 5 | 232 |
| 0.5% | 426 | 89 | - | - | - | 253 | |
| 1.0% | 405 | 50 | - | - | - | 279 | |
| 2.0% | 380 | 48 | - | - | - | 301 | |
| Total Sugar (mg/mL Liquid) | Lignin (mg/mL Liquid) | ||||||
|---|---|---|---|---|---|---|---|
| Glucose | Xylose | Arabinose | Galactose | Mannose | |||
| EA-L | 220-20 | 12.2 | 10.1 | 3.6 | 0.3 | - | n.d. |
| 0.5% | 0.3 | 13.4 | 0.7 | 0.3 | - | n.d. | |
| 1.0% | 1.4 | 20.6 | 0.8 | 0.4 | 0.2 | n.d. | |
| 2.0% | 7.9 | 21.9 | 1.1 | 0.7 | 1.0 | n.d. | |
| EA-S-OL | 220-20 | - | 2.2 | 3.0 | - | 0.4 | 38.2 |
| 0.5% | 0.8 | 3.2 | - | - | - | 19.0 | |
| 1.0% | 0.1 | 1.4 | - | - | - | 20.5 | |
| 2.0% | - | 2.4 | - | - | - | 12.2 | |
| Crystallinity Index (%) | ||
|---|---|---|
| Raw | 58.3 | |
| E-S-CS | 180-10 | 35.9 |
| 200-10 | 47.8 | |
| 220-10 | 60.0 | |
| EA-S 2.0% | 47.8 | |
| EA-S-CS 2.0% | 57.0 | |
| No. | Compound | Concentration (Area/IS Area) | |
|---|---|---|---|
| 220-5 | 220-10 | ||
| 1 | Toluene | 0.45 | 0.15 |
| 2 | Phenol | 4.66 | 1.67 |
| 3 | o-Cresol | 0.44 | 0.16 |
| 4 | p-Cresol | 0.54 | 0.19 |
| 5 | Guaiacol | 0.51 | 0.33 |
| 6 | Creosol | 0.53 | 0.25 |
| 7 | 2-Methoxy-4-vinylphenol | 0.40 | 0.12 |
| 8 | Syringol- | 1.01 | 0.61 |
| 9 | 3,5-Dimethoxy-4-hydroxytoluene | 1.05 | 0.33 |
| 10 | Isoeugenol | 0.30 | 0.10 |
| 11 | 2,6-Dimethyl-3-(methoxymethyl)-benzoquinone | 0.55 | 0.12 |
| 12 | (E)-4-Propenylsyringol | 0.68 | 0.19 |
| 13 | n-Hexadecanoic acid | 1.07 | 0.36 |
| 14 | Octadecanoic acid | 0.33 | 0.15 |
| 12.50 | 4.72 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, J.H.; Ahmed, M.A.; Choi, I.-G.; Choi, J.W. Fractionation of Cellulose-Rich Products from an Empty Fruit Bunch (EFB) by Means of Steam Explosion Followed by Organosolv Treatment. Appl. Sci. 2020, 10, 835. https://doi.org/10.3390/app10030835
Lee JH, Ahmed MA, Choi I-G, Choi JW. Fractionation of Cellulose-Rich Products from an Empty Fruit Bunch (EFB) by Means of Steam Explosion Followed by Organosolv Treatment. Applied Sciences. 2020; 10(3):835. https://doi.org/10.3390/app10030835
Chicago/Turabian StyleLee, Jae Hoon, Muhammad Ajaz Ahmed, In-Gyu Choi, and Joon Weon Choi. 2020. "Fractionation of Cellulose-Rich Products from an Empty Fruit Bunch (EFB) by Means of Steam Explosion Followed by Organosolv Treatment" Applied Sciences 10, no. 3: 835. https://doi.org/10.3390/app10030835
APA StyleLee, J. H., Ahmed, M. A., Choi, I.-G., & Choi, J. W. (2020). Fractionation of Cellulose-Rich Products from an Empty Fruit Bunch (EFB) by Means of Steam Explosion Followed by Organosolv Treatment. Applied Sciences, 10(3), 835. https://doi.org/10.3390/app10030835




