1. Introduction
In recent years, the demands of consumers have shifted from quantity to quality and, as a result, the intramuscular fat (IMF) content and fatty acid profile of poultry meat have become important to both producers and consumers [
1]. Due to its relatively higher proportion of polyunsaturated fatty acids, poultry meat is usually better than other meats [
2]. However, unsaturated fatty acids are susceptible to oxidation, and malonaldehyde (MDA) is well known as one of the end products of this process [
3]. Thus, lipid oxidation is one of the main factors leading to the deterioration of meat quality, which can occur both before and after death [
4]. Furthermore, lipid oxidation causes adverse changes in glycolysis that increase the drip loss and thereby lead to meat spoilage and a shorter shelf life [
5,
6]. Thus, an effective approach for improving the IMF and protecting intramuscular lipids against oxidative damage is needed.
Thiazolidinediones (TZDs) are a class of medicines currently been used to treat type 2 diabetes [
7]. Although TZDs have beneficial effects on glycemic control, insulin sensitivity, inflammation and oxidative stress, and reduction of triglycerides [
8,
9,
10], TZDs are clinically at risk for hepatic insufficiency, thus the use of TZDs in the animal diet is prohibited in some countries [
11]. However, regarding the effects of TZDs on skeletal muscle lipid metabolism, many studies have found that intramuscular lipid content and the proportion of unsaturated fatty acids are increased after TZD treatment [
12,
13,
14]. As adipogenic compounds, TZDs play a crucial role in adipocyte differentiation and have the potential to alter the formation of intramuscular or marbled fat [
15,
16]. As one of the TZDs, pioglitazone hydrochloride (PGZ) is a high-affinity ligand for peroxisome proliferator-activated receptor gamma (
PPARγ) [
17], and previous study has found that PGZ can promote adipocyte differentiation to modify intramuscular adipogenesis [
15]. More adipocytes and larger cell sizes have been observed in mice supplemented with PGZ [
13,
14,
18]. According to previous reports, PGZ has the potential to promote fat deposition in animal muscle [
19,
20]. Consistent with these findings, our previous studies confirmed that dietary supplementation with 15 mg/kg PGZ can increase the IMF in the longissimus thoracis muscle of pigs [
21,
22]. Therefore, PGZ has potential as a new feed additive for improving meat quality of animals. However, few studies have explored the effects of PGZ on poultry, such as chickens.
The inclusion of an antioxidant additive in broiler chicken diets is necessary to protect the PGZ supplementation-induced increase in the IMF of the thigh muscle from oxidation. Resveratrol (RES) is a botanical polyphenolic compound, and previous studies have indicated that dietary supplemental with RES could promote antioxidant activity in quail eggs and broiler chickens [
23,
24]. Furthermore, the impairment in the antioxidant status caused by heat stress is attenuated in chickens fed a diet supplemented with RES [
25]. As an important plant-derived bioactive polyphenol, RES can eliminate reactive oxygen species (ROS) and thereby improve antioxidant activity [
26]. Consequently, RES has the potential to work as an antioxidant and preservative to extend the shelf life of meat and thereby avoid huge economic losses.
Because RES can improve the antioxidant capacity and PGZ can enhance the IMF and proportions of unsaturated fatty acids, the aim of this study was to explore whether the combination of PGZ and RES exerts effects on the muscle antioxidant status, IMF content, and fatty acid profile of broiler chickens.
2. Materials and Methods
The protocols used for the feeding and management of broiler chickens in this study were approved by the Animal Ethics Committee of South China Agricultural University and were in accordance to the Guidelines for the Care and Use of Laboratory Animals of South China Agricultural University (Guangzhou, China).
2.1. Materials
PGZ (purity ≥ 99%) and RES (purity ≥ 98%) was obtained from Sichuan Jisheng Biopharmaceutical Co., Ltd. (Leshan, China).
2.2. Experimental Design, Diets and Growth Performance
A total of 500 female yellow-feathered broiler chickens, approximately 72 days of age (28 days before slaughter), were randomly assigned to four groups: a control group (fed with basic diet,
Table S1), and the PGZ, RES, or PGZ+RES groups, which were fed the basic diet and supplemented with 15 mg/kg PGZ, 400 mg/kg RES, or 15 mg/kg PGZ + 400 mg/kg RES, respectively, for 28 days. The basic diet was formulated according to the Nutrient Requirements of Poultry (Ministry of Agriculture of the People’s Republic of China 2004).
Prior to the experiment, the experimental room was cleaned and disinfected. Immunization was performed using a routine procedure. The broiler chickens were housed in a room with a concrete floor covered with wheat shavings and allowed access to feed and water ad libitum. The room was illuminated for 23 h, and the temperature was maintained at approximately 25 ± 2 °C. Before the experiment, the animals were pre-fed with the assigned diet for 5 days, and all weak broiler chickens were eliminated. The weight and feed intake of weak broiler chickens were recorded, and these data were excluded in the final statistics. The experiment spanned 28 days, and the average daily feed intake (ADFI) was recorded. All broiler chickens were weighed at 1, 14, and 28 days to calculate the average daily gain (ADG) and feed/gain (F/G) ratio.
2.3. Serum Biochemical Indices
After feeding for 28 days, 3 broiler chickens were selected from each replicate (totaling 15 broiler chickens per diet group) and fasted overnight for 12 h. The broiler chickens were bled from the jugular vein, and the blood samples from the broiler chickens of the formal experiment were collected, then maintained at an oblique position for 30 min. The serum samples were then collected after centrifugation of blood samples at 3000 rpm for 10 min and stored at −20 °C. Various serum biochemical indices, including total protein (TP), triglyceride (TG), cholesterol (CHO), low-density lipoprotein cholesterol (LDL-C), serum urea nitrogen (SUN), and high-density lipoprotein cholesterol (HDL-C), were then detected using kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China). The kits and their sensitivity and detection limits used in this study were as follows: TP assay kit A045-2-2, 5.0-122.1 U/mL; TG assay kit A110-1-1; Total CHO assay kit A111-1-1; LDL-C assay kit A113-1-1; SUN assay kit C013-2-1; and HDL-C assay kit A112-1-1.
2.4. Carcass Performance and Meat Quality
After blood was harvested and feathers were removed, carcass weights were calculated. The carcass traits and meat quality were measured as previously described [
27]. Dressing percentage was calculated by dividing carcass weight by live weight. The semi-eviscerated yield (feet, head, and all the organs except the lungs and kidneys were removed), eviscerated yield (lung and kidney were removed), breast muscle yield (2 × right left muscle), thigh muscle yield (2 × left thigh muscle), and abdominal fat were expressed relative to carcass weight.
The entire left breast muscle and thigh muscle (the skin was removed) were collected immediately after slaughter and used for detection of the meat quality. The pH was detected using a meter (HI99161, HANNA, Rome, Italy) equipped with an insertion glass electrode, and the color was detected at the same part of the front of all muscle using a colorimeter (CR410, MINOLTA, Tokyo, Japan) after 45 min and 24 h slaughter. The drip loss was determined with the muscle samples were suspended in a zip-lock bag and stored at 4 °C for 24 h. The cooking loss was determined using muscle samples sealed in a zip-lock bag; the muscle samples were cooked in a 75 °C water bath and maintained at 72 °C for 20 min. The shear force was detected using the muscle cooked in a water bath at 70 °C for 30 min with a digital muscle tenderness tester (C-LM3B, TENOVO, Beijing, China) at 24 h after slaughter. A piece of muscle from the same location on the right side of the body was obtained and stored in liquid nitrogen for histological analysis, determination of the fatty acid composition, and RT-PCR assay. The remaining of the thigh muscle samples of each chicken were divided into 5 sections and stored at 4 °C. They were then collected for the detection of the total volatile basic nitrogen (TVB-N) at 0, 3, 5, 7, and 9 days. As a crucial indicator used for evaluating the shelf life of meat, the TVB-N values were detected using the procedure described by Goulas [
28] and expressed as mg of TVB-N/100 g.
Six sections of thigh muscle were cut at 8 μm and embedded in 4% paraformaldehyde to protect against dehydration. The sections were then stained with Oil Red O. The whole staining procedure lasted approximately 10 min. Specifically, the slices were immersed in isopropanol for 2 min, washed under running tap water for 1 min, covered with working solution for 5 min, and then washed in clean isopropanol for 2 min to adjust the slice color. A working solution composed of 0.5 g of Oil Red O diluted with 100 mL of isopropanol was prepared and stored at 4 °C [
29].
2.5. Determination of the Fatty Acid Composition
The fatty acid contents and compositions were measured as previously described [
30,
31]. Samples of 30 g thigh muscle were baked at constant temperature (at 70 °C). 2 g portion of each sample was weighed and then placed in a degreasing filter paper bag and dried to constant weight for extraction. After extraction, 2 mL methylation reagent (DongYing HeYi Chemical Co., LTD., Shandong, China) was taken in a 10 mL screw cap centrifuge tube, and accurately added to 200 mL of fat and gently mixed by pipetting. After sealing, the screw cap centrifuge tube was placed in a boiling water bath at 100 °C for 30 min. The upper organic phase was transferred to a 2 mL centrifuge tube for centrifuge. The upper organic phase was transferred to a 1.5 mL gas chromatography sample vial (Jinpu Analytical Instrument Co., Ltd., Shandong, China) for later use. The fatty acid profile was detected by GC 7890A gas chromatograph (Agilent, Santa Clara, CA, USA). Nitrogen was used as carrier gas (2 mL/min). Injector and detector temperature were 270 °C. The temperature of the column was operated as follows: 75 °C for 0.5 min, from 75 °C to 190 °C at 20 °C/min, and from 190 °C to 250 °C at 5 °C/min. The types of fatty acids were identified by comparing the retention times with FAME Mix 37 (18919–1 AMP, Supelco, Bellefonte, PA, USA). Contents of various fatty acids were calculated by calculating retention times and peak area percentages.
2.6. Determination of Muscle Antioxidant Abilities and ROS
The MDA, catalase (CAT), total superoxide dismutase (T-SOD) activity, glutathione peroxidase (GSH-Px), and total antioxidant capacity (T-AOC) of thigh muscle were determined using commercial kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China). The thigh muscles slaughtered within two hours were cut into frozen sections of 5 μm, washed twice with phosphate buffer saline, and returned to room temperature for incubation with 10 μM dihydroethidium (DHE, 50102ES02, YEASEN, Shanghai, China) at 37 °C for 60 min. Fluorescence intensity was measured using Varioskan LUX (Thermo Fisher Scientific, Waltham, MA, USA).
2.7. Determination of mRNA Abundances
Total RNA from the thigh muscle was extracted using TRIzol (Ambion, Austin, TX, USA) [
32]. A quantity of 50–100 mg of frozen thigh muscle tissue was cut and placed in a mortar, poured into liquid nitrogen, and crushed. A quantity of 1 mL TRIZOL was added to separate the nucleoprotein complex. A quantity of 0.2 mL of chloroform was then added and the upper aqueous phase was transferred into 1.5 mL centrifuge tubes, to which were added 0.5 mL of isopropanol. A quantity of 98 μL of Rnase-free water was then added after washing the RNA pellet (GeneStar, Beijing, China). The concentration of RNA was determined by NanoDrop 2000 (Waltham, MA, USA), and first-strand cDNA was synthesized with Primer Script™ (GeneStar, Beijing, China) and used for PCR amplification with T100™ Thermal Cycler (Bio-Rad, Hercules, CA, USA). The IQ5 Real-Time PCR Detection System (Bio-Rad, CA, USA) was used to determine the mRNA abundances, and the sequences of the primers used in this study are listed in
Table S2.
2.8. Statistical Analysis
All the data are shown as means. SPSS software (version 20.0) was used for the statistical analyses, and the statistical significance levels (p < 0.05) were determined by one-way analysis of variance (ANOVA) followed by Duncan’s multiple range test.
4. Discussion
The yellow-feathered broiler chicken is highly accepted in the local market of Asian countries due to its delicious meat [
33]. However, the yellow-feathered broiler chicken is a slow-growing breed. The ADG and feed/gain of yellow feather broilers are approximately 18.0 g/d and 4.8 (feed/gain, g/g), respectively [
27,
34]. Therefore, optimization of their feed nutrients is important for improving the growth performance of these slow-growing broiler chickens.
Our previous studies in pigs showed that dietary PGZ and PGZ + RES supplementation improved the ADG and reduced the F/G ratio, the results being consistent with this study [
21,
22]. The increased ADG of pig may be due to the ability of PGZ to promote adipocyte differentiation and store more fat in the body [
35]. In the present study, we used low dose of PGZ as an adipogenic additive rather than as a drug, and in order to minimize the impact of PGZ on the liver and prevent drug residue in chicken products, we started to add a low dose (15 mg/kg) of PGZ to the diets one month before the chicken was released. Furthermore, broiler chickens supplemented with PGZ + RES exhibited an increased dressing percentage and a decreased abdominal fat percentage. Previous studies showed RES reduced the subcutaneous fat mass of pigs and obese rats through the inhibition of fat formation and lipolysis, which was due to the enhanced expression of silent information regulator 1 in adipocytes [
36,
37]. Our results show that the PGZ and PGZ + RES groups present decreased CHO and LDL-C levels but increased HDL-C concentrations, indicating that these broiler chickens exhibit an increased lipid utilization and an improved fat metabolism in vivo, which is more beneficial for IMF deposition rather than simply shifting toward abdominal fat [
22,
38,
39].
The cooking loss and drip loss are significantly decreased by PGZ + RES supplementation, which suggests an improved water holding capacity [
40]. The enhanced antioxidant capacity of the PGZ + RES group is beneficial for reducing glycolysis and increasing the water storage capacity. In addition, the glycogen level largely affects the pH value, and we thus find that PGZ + RES supplementation increases
A* and pH at 24 h after slaughter [
21,
30]. During storage, most nutrients, such as carbohydrates, proteins, and fats, decompose into various volatile organic compounds following microbial spoilage and biochemical reactions, and these basic nitrogenous compounds increase the TVB-N value, which is an important index for evaluating the shelf life of meat [
41,
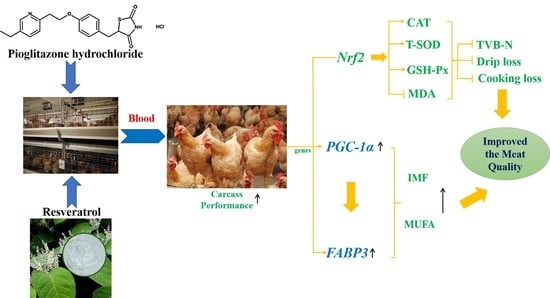
42]. We find that the TVB-N values at 5, 7, and 9 days are decreased in the RES and PGZ + RES groups, which indicates that PGZ+RES addition has positive effects on extending the meat shelf life and improving the meat quality. PGZ targets the adipose tissue through
PPARγ and
PGC-1α, and the latter is used to measure the effect of PGZ [
12,
43]. Moreover, PGZ has potential as a new feed additive for improving meat quality because it can increase the meat IMF [
30]. In this study, PGZ activates
PGC-1α and its downstream genes, such as
FABP3, and may thus induce a more active fat metabolism. In addition, the Oil Red O staining results shown in
Figure 1 also confirm that increased amounts of IMF are deposited in the muscles of the chickens belonging to the PGZ and PGZ + RES groups.
Higher unsaturated fatty acid proportions in meat are beneficial for human health [
1]. Early studies had demonstrated that PGZ could enhance the proportions of polyunsaturated fatty acids in serum phospholipids and longissimus thoracis muscle, and MUFAs in rodent muscle [
12,
21,
30,
44]. Similar to previous studies, our present study finds that both the PGZ and PGZ+RES supplementation can increase the C18:1ω-9, C18:3ω-6, and C20:3ω3 proportions in thigh muscle, and the latter supplementation results in higher proportions. The changes may be associated with the roles of PGZ as an affinity ligand of
PPARγ, and PGZ affects several specific genes, not only
PPARγ, but also
PGC-1α and
FABP3, to ensure its normal function [
45,
46]. Our results support the hypothesis that PGZ improves the IMF contents and MUFA proportions by activating critical genes involved in fat metabolism. In addition, free radicals in meat can attack unsaturated fatty acids, and MDA is well known as one of the end products [
3]. Our study demonstrates that dietary supplementation with RES and PGZ + RES decreases the MDA content and enhances the proportion of MUFAs in thigh muscle, which indicates that unsaturated fatty acids are protected from oxidation [
47].
Antioxidant enzyme defensive systems, composed of SOD, GSH-Px, and CAT, can directly eliminate ROS in meat [
36,
44]. The content of ROS determines the degree of oxidative stress response [
48]. Excessive ROS can cause cholesterol peroxidation, which in turn reduces membrane fluidity and receptor activity, thereby impairing membrane function [
49]. In general, decreased lipid peroxidation or improved antioxidant ability in muscle is beneficial to meat quality. Mujahid et al. showed that excessive MDA accumulation could inhibit the activities of antioxidant enzymes and accelerate oxidative damage to proteins and DNA [
50]. RES is a naturally occurring polyphenol compound, and its biological activities, including antioxidant and lipogenesis regulation, have been well researched in vitro and in vivo [
51,
52]. Numerous studies on the functionality of RES in poultry and pigs have confirmed its efficacy as a feed additive for improving animal production and health [
12,
29]. RES facilitates the inactivation and subsequent elimination of oxide precursors and mobilizes the expression of antioxidation-related proteins [
53,
54]. In our present study, the activities of the CAT, T-SOD, and GSH-Px enzymes are increased by RES and PGZ+RES supplementation, and ROS is reduced in the cells of thigh muscle. The mechanism underlying this effect may involve increased mRNA expression of
NRF2,
SOD1, and
GPX4.
NRF2 is a redox-sensitive transcription factor that can initiate the transcription of antioxidant genes, and the synthesis of CAT, SOD, and GSH-Px is regulated by
NRF2 [
55,
56]. The above results of antioxidant enzyme activities are confirmed through an analysis of the antioxidant gene expression. Indeed, a strong positive correlation is found between antioxidant enzyme activities and the corresponding mRNA levels, which suggests that activities of antioxidant enzymes may be mainly regulated by their transcriptional levels [
43].
From the present and some previous studies [
13,
14,
15,
16,
17,
18,
19,
20,
21,
22], we can conclude that dietary PGZ can increase muscle fat deposition and improve the meat quality of food animals. It has potential as a new functional feed additive. However, in many countries PGZ is a human drug and it is not allowed to be used in feeds or foods. Dietary supplementation with PGZ has the potential to generate residues in animal-derived products. There is limited information on the magnitude of PGZ residue worldwide. Therefore, extensive work has to be carried out to investigate the effects of dietary supplementation with PGZ on food-producing animals, and to prevent the occurrence of PGZ residues.