How Different Fermentation Type Affects Volatile Composition of Plum Jerkums
Abstract
:1. Introduction
2. Materials and Methods
2.1. Fermentation of Plum Mashes
2.2. Chemical Composition of Fresh and Fermented Plum Mashes
2.3. Determination of Sugar and Main Organic Acids Content by High-Performance Liquid Chromatography (HPLC)
2.4. Volatile Composition of Fresh and Fermented Plum Mashes Using Solid Phase Microextraction–Gas Chromatography–Mass Spectrometry (SPME–GC–MS) and Gas Chromatography–Flame Ionization Detector (GC–FID) Methods
2.5. Statistical Analysis
3. Results and Discussion
3.1. Chemical Composition of Fresh Plum Musts and Jerkums
3.2. Volatile Compounds of Fresh Plum Musts
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Januszek, M.; Satora, P.; Tarko, T. Oenological Characteristics of Fermented Apple Musts and Volatile Profile of Brandies Obtained from Different Apple Cultivars. Biomolecules 2020, 10, 853. [Google Scholar] [CrossRef] [PubMed]
- Pischl, J. Destylaty Alkoholowe. Wytwarzanie, Teoria i Praktyka; Borus & InterPolonus: Warszawa, Poland, 2010. [Google Scholar]
- López, F.; Rodríguez-Bencomo, J.J.; Orriols, I.; Pérez-Correa, J.R. Science and Technology of Fruit Wine Production; Academic Press: Cambridge, MA, USA, 2017; ISBN 9780128010341. [Google Scholar]
- Satora, P.; Kostrz, M.; Sroka, P.; Tarko, T. Chemical profile of spirits obtained by spontaneous fermentation of different varieties of plum fruits. Eur. Food Res. Technol. 2017, 243, 489–499. [Google Scholar] [CrossRef]
- Joshi, V.K.; Panesar, P.S.; Rana, V.S.; Kaur, S. Science and Technology of Fruit Wines: An Overview; Elsevier Inc.: Amsterdam, The Netherlands, 2017; ISBN 9780128010341. [Google Scholar]
- Januszek, M.; Satora, P.; Łukasz, W.; Tarko, T. Saccharomyces bayanus Enhances Volatile Profile of Apple Brandies. Molecules 2020, 25, 3127. [Google Scholar] [CrossRef]
- Du Plessis, H.; Du Toit, M.; Nieuwoudt, H.; Van der Rijst, M.; Hoff, J.; Jolly, N. Modulation of wine flavor using Hanseniaspora uvarum in combination with different Saccharomyces cerevisiae, lactic acid bacteria strains and malolactic fermentation strategies. Fermentation 2019, 5, 64. [Google Scholar] [CrossRef] [Green Version]
- Walkowiak-Tomczak, D. Wpływ stopnia dojrzałości na parametry fizyczno-chemiczne i zawartość związków polifenolowych w wybranych odmianach śliwek (Prunus domestica). Nauk. Przyr. Technol. 2009, 3, 149. [Google Scholar]
- Miljić, U.; Puškaš, V. Suitability of chosen plum cultivars (Prunus domestica L.) for fruit wine production. J. Process. Energy Agric. 2015, 19, 95–97. [Google Scholar]
- Skotniczny, M.; Satora, P.; Panczyszyn, K.; Cioch-Skoneczny, M. Growth dynamics and diversity of yeasts during spontaneous plum mash fermentation of different varieties. Foods 2020, 9, 1054. [Google Scholar] [CrossRef] [PubMed]
- OIV. International Organisation of Vine and Wine (OIV). Compendium of International Methods of Wine and Must Analysis; OIV: Paris, France, 2016. [Google Scholar]
- Abernathy, D.; Spedding, G.; Starcher, B. Analysis of protein and total usable nitrogen in beer and wine using a microwell ninhydrin assay. J. Inst. Brew. 2009, 115, 122–127. [Google Scholar] [CrossRef]
- Antalick, G.; Perello, M.-C.; de Revel, G. Development, validation and application of a specific method for the quantitative determination of wine esters by headspace-solid-phase microextraction-gas chromatography–mass spectrometry. Food Chem. 2010, 121, 1236–1245. [Google Scholar] [CrossRef]
- Pashova, S. Chemical composition of plum fruits. J. Mt. Agric. Balk. 2006, 9, 239–249. [Google Scholar]
- Sudar, R.; Jurković, Z.; Drugalić, K.; Tomać, I.; Jurković, V.; Viljevać, M. Sorbitol and sugar composition of plum fruit during ripening. In Proceedings of the 46th Croatian and 6th International Symposium on Agriculture, Opatija, Croatia, 14–18 February 2011. [Google Scholar]
- France Ethanol Red® Dry Alcohol Yeast; Lesaffre Advanced Fermentation Technologies; 2010. Available online: https://www.scribd.com/document/325756425/ER-EN (accessed on 5 April 2019).
- Tarko, T.; Kostrz, M.; Duda-Chodak, A.; Semik-Szczurak, D.; Sroka, P.; Senczyszyn, T. The effect of apple cultivars and yeast strains on selected quality parameters and antioxidant activity of fermented apple beverages. CYTA J. Food 2018, 16, 892–900. [Google Scholar] [CrossRef]
- Usenik, V.; Marn, M.V. Sugars and organic acids in Plum fruit affected by Plum pox virus. J. Sci. Food Agric. 2016, 97, 2154–2158. [Google Scholar] [CrossRef] [PubMed]
- Coulter, A.; Godden, P.; Pretorius, I. Succinic acid—How is it formed, what is its effect on titratable acidity, and what factors influence its concentration in wine? WINE Ind. J. 2004, 19, 16–25. [Google Scholar]
- Vilela-Moura, A.; Schuller, D.; Mendes-Faia, A.; Silva, R.D.; Chaves, S.R.; Sousa, M.J.; Côrte-Real, M. The impact of acetate metabolism on yeast fermentative performance and wine quality: Reduction of volatile acidity of grape musts and wines. Appl. Microbiol. Biotechnol. 2011, 89, 271–280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Inês, A.; Falco, V. Lactic Acid Bacteria Contribution to Wine Quality and Safety; Intech Open: London, UK, 2018; pp. 53–71. [Google Scholar]
- Valles, B.S.; Bedriñana, R.P.; Tascón, N.F.; Simón, A.Q.; Madrera, R.R. Yeast species associated with the spontaneous fermentation of cider Bele. Food Microbiol. 2007, 24, 25–31. [Google Scholar] [CrossRef]
- Mendes-Ferreira, A.; Barbosa, C.; Lage, P.; Mendes-Faia, A. The impact of nitrogen on yeast fermentation and wine quality. Ciênc. Téc. Vitivinic. 2011, 26, 17–32. [Google Scholar]
- Burdock, G.A. Fenaroli’s Handbook of Flavor Ingredients, 4th ed.; CRC Press: Boca Raton, FL, USA, 2005; ISBN 9781439863275. [Google Scholar]
- Pino, J.A.; Quijano, C.E. Study of the volatile compounds from plum (Prunus domestica L. cv. Horvin) and estimation of their contribution to the fruit aroma. Food Sci. Technol. 2012, 32, 76–83. [Google Scholar] [CrossRef] [Green Version]
- Verstrepen, K.J.; Van Laere, S.D.M.; Vanderhaegen, B.M.P.; Derdelinckx, G.; Dufour, J.P.; Pretorius, I.S.; Winderickx, J.; Thevelein, J.M.; Delvaux, F.R. Expression levels of the yeast alcohol acetyltransferase genes ATF1, Lg-ATF1, and ATF2 control the formation of a broad range of volatile esters. Appl. Environ. Microbiol. 2003, 69, 5228–5237. [Google Scholar] [CrossRef] [Green Version]
- Rojas, V.; Gil, J.V.; Piñaga, F.; Manzanares, P. Studies on acetate ester production by non-Saccharomyces wine yeasts. Int. J. Food Microbiol. 2001, 70, 283–289. [Google Scholar] [CrossRef]
- Tristezza, M.; Tufariello, M.; Capozzi, V.; Spano, G.; Mita, G.; Grieco, F. The oenological potential of hanseniaspora uvarum in simultaneous and sequential co-fermentation with Saccharomyces cerevisiae for industrial wine production. Front. Microbiol. 2016, 7, 670. [Google Scholar] [CrossRef] [Green Version]
- Gómez, E.; Ledbetter, C.A.; Hartsell, P.L. Volatile Compounds in Apricot, Plum, and Their Interspecific Hybrids. J. Agric. Food Chem. 1993, 41, 1669–1676. [Google Scholar] [CrossRef]
- Pietruszka, M.; Pielech-Przybylska, K.; Szopa, J.S. Synthesis of higher alcohols during alcoholic fermentation of rye mashes. Scientific bulletin of the Technical University of Lodz. Food Chem. Biotech. 2010, 2010, 74. [Google Scholar]
- Oliveira, J.M.; Faria, M.; Sá, F.; Barros, F.; Araújo, I.M. C6-alcohols as varietal markers for assessment of wine origin. Anal. Chim. Acta 2006, 563, 300–309. [Google Scholar] [CrossRef] [Green Version]
- Etschmann, M.; Bluemke, W.; Sell, D.; Schrader, J. Biotechnological production of 2-phenylethanol. Appl. Microbiol. Biotechnol. 2002, 59, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ohimain, E.I. Methanol contamination in traditionally fermented alcoholic beverages: The microbial dimension. Springerplus 2016, 5, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miljić, U.; Puškaš, V.S. Influence of fermentation conditions on production of plum (Prunus domestica L.) wine: A response surface methodology approach. Hem. Ind. 2014, 68, 199–206. [Google Scholar] [CrossRef]
- Gómez-Plaza, E.; Ledbetter, C. The Flavor of Plums. In Handbook of Fruit and Vegetable Flavors; Wiley: Hoboken, NJ, USA, 2010; pp. 415–430. ISBN 9780470227213. [Google Scholar]
- Miljić, U.; Puškaš, V.; Vučurović, V. Investigation of technological approaches for reduction of methanol formation in plum wines. J. Inst. Brew. 2016, 122, 635–643. [Google Scholar] [CrossRef] [Green Version]
- Nunes, C.; Coimbra, M.A.; Saraiva, J.; Rocha, S.M. Study of the volatile components of a candied plum and estimation of their contribution to the aroma. Food Chem. 2008, 111, 897–905. [Google Scholar] [CrossRef]
- Belitz, H.-D.; Grosch, W.; Schieberle, P. Food Chemistry; Springer: Berlin, Germany, 2004. [Google Scholar]
- Zhao, N.; Zhang, Y.; Liu, D.; Zhang, J.; Qi, Y.; Xu, J.; Wei, X.; Fan, M. Free and bound volatile compounds in ‘Hayward’ and ‘Hort16A’ kiwifruit and their wines. Eur. Food Res. Technol. 2020, 246, 875–890. [Google Scholar] [CrossRef]
- Blagoeva, N.; Bazhlekova, I.; Spasov, H.; Kostov, G. Influence of enzyme maceration and alcoholic fermentation temperature on the terpenes concentration in Muscat wine distillates. Bulg. J. Agric. Sci. 2020, 26, 1069–1075. [Google Scholar]
- Stork, G.; Grieco, P.A.; Gregson, M. Allylic Chlorides from Allylic Alcohols: Geranyl Chlo-ride. In Organic Syntheses; John Wiley and Sons, Inc.: Hoboken, NJ, USA, 1974; p. 638. [Google Scholar]
- Carballo, J. The Role of Fermentation Reactions in the Generation of Flavor and Aroma of Foods. In Fermentation. Effects on Food Properties; CRC Press: Boca Raton, FL, USA, 2016; p. 74. [Google Scholar]
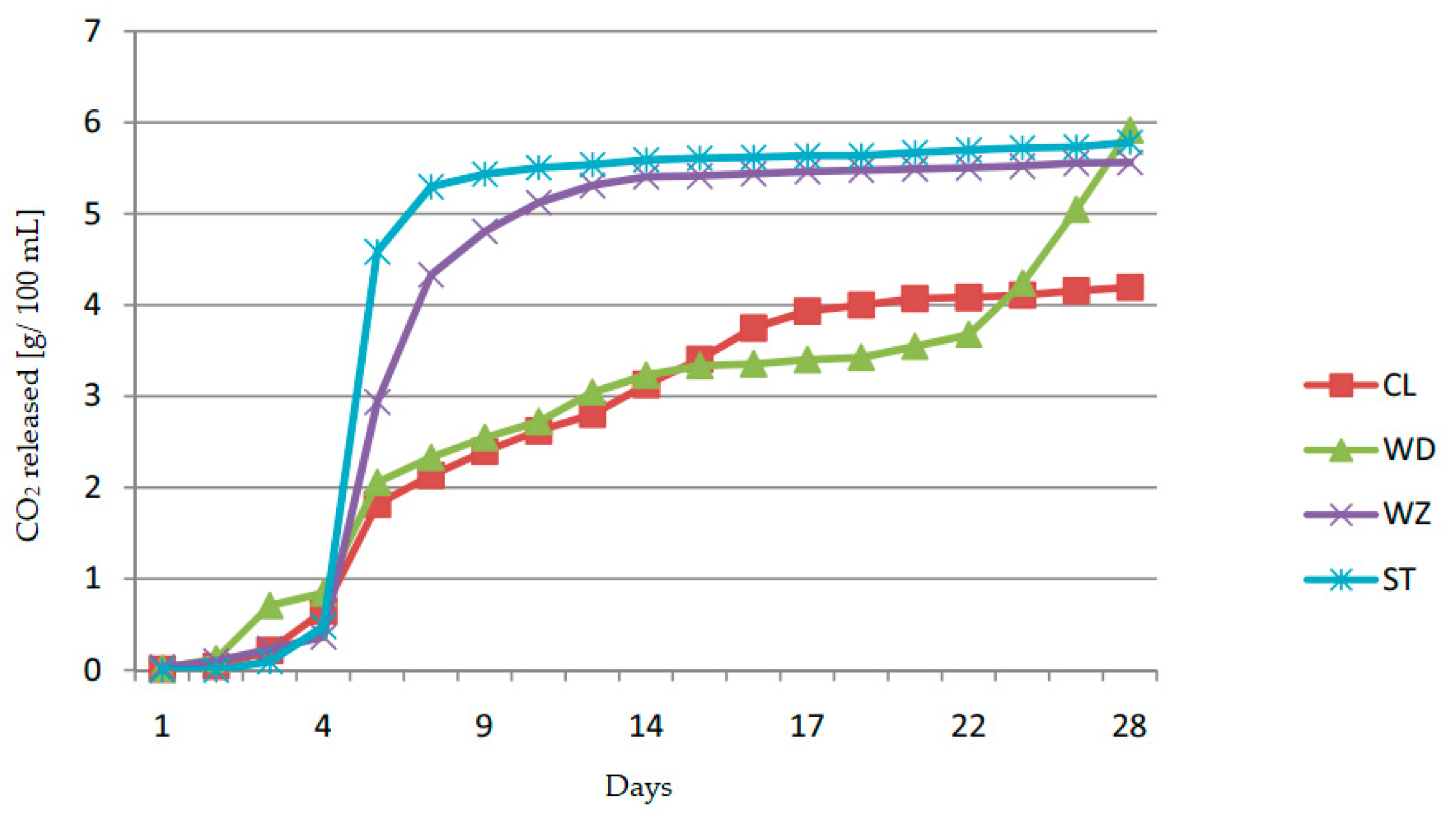

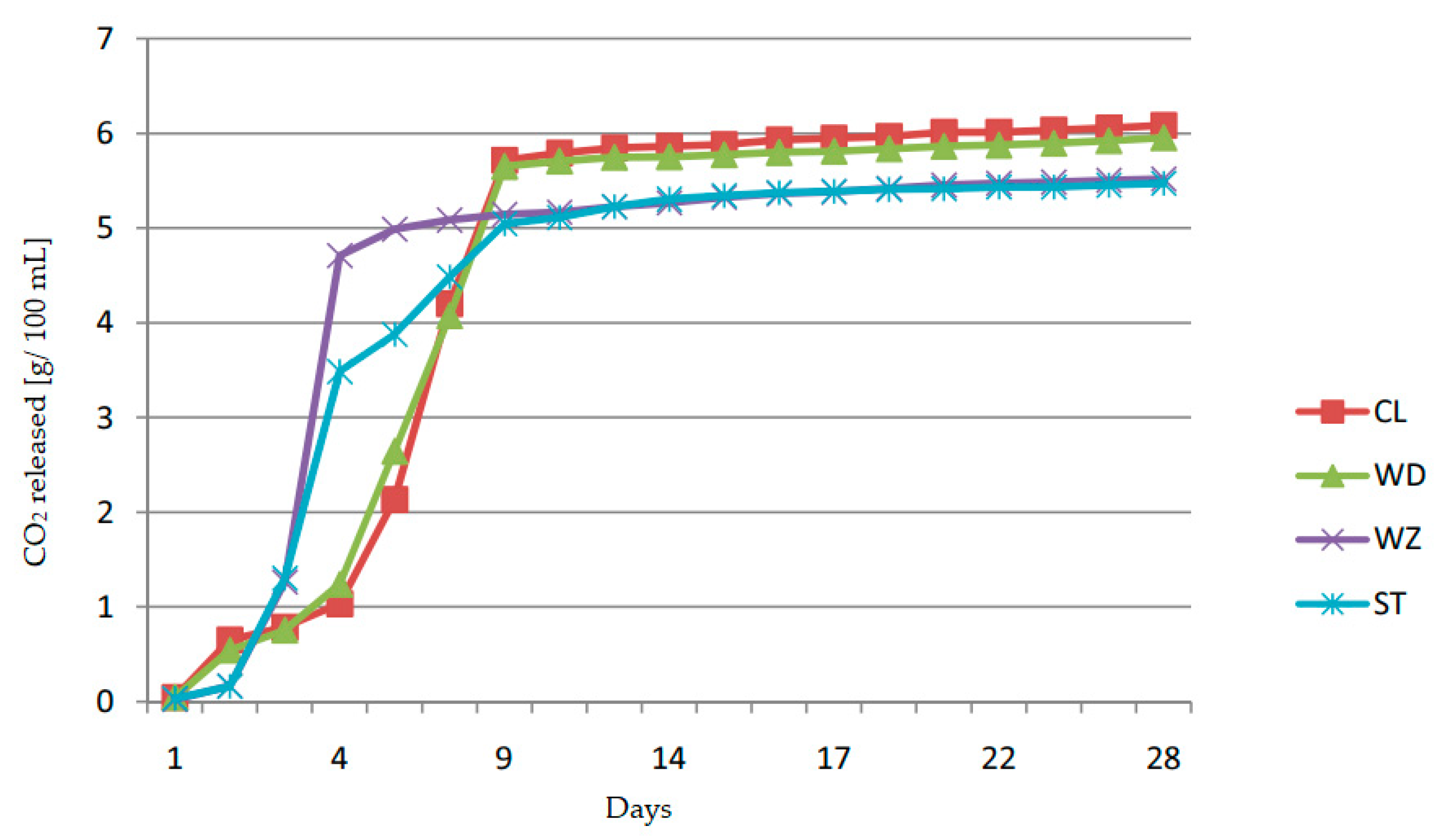
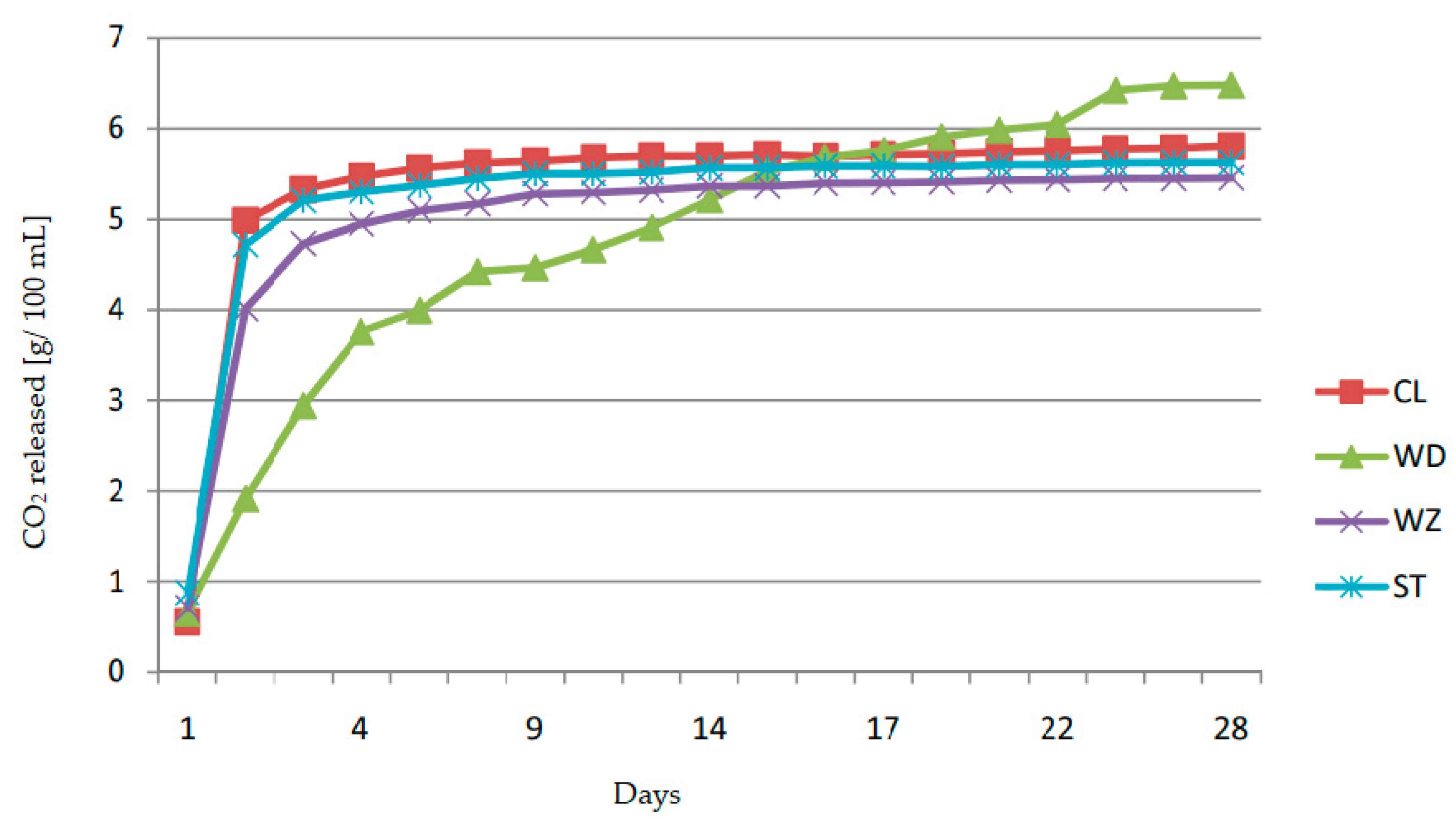
| Plum Cultivars | Total Extract | Total Sugars | Reducing Sugars | Sucrose | Sugar-Free Extract | Titratable Acidity | Citric Acid | Malic Acid | Succinic Acid | Free Amino Nitrogen |
|---|---|---|---|---|---|---|---|---|---|---|
| [g/L] | [mg/L] | |||||||||
| Węgierka Zwykła | 146.0 c (±2.0) | 113.2 b (±4.3) | 42.6 ab (±2.3) | 51.4 a (±2.0) | 26.7 b (±4.2) | 9.06 b (±0.39) | 1.12 b (±0.07) | 6.13 ab (±0.24) | 0.56 b (±0.11) | 119.6 c (±11.4) |
| Węgierka Dąbrowicka | 155.7 b (±1.5) | 123.8 a (±5.7) | 45.9 a (±2.6) | 47.9 ab (±2.9) | 37.8 a (±4.2) | 12.22 a (±0.23) | 1.00 b (±0.04) | 6.41 ab (±0.58) | 1.13 a (±0.14) | 135.6 bc (±4.6) |
| Stanley | 161.7 ab (±6.4) | 121.4 a (±2.6) | 40.3 ab (±4.3) | 56.7 a (±2.2) | 37.7 a (±9.0) | 9.81b (±0.47) | 1.50 a (±0.17) | 7.26 a (±0.29) | 0.82 a (±0.24) | 156.4 b (±6.6) |
| Čačanska Lepotica | 151.0 c (±1.0) | 119.4 ab (±2.6) | 32.1 b (±2.0) | 44.5 b (±1.6) | 41.4 a (±2.5) | 11.90 a (±0.53) | 1.04 b (±0.12) | 6.56 ab (±0.33) | 0.98 a (±0.05) | 212.1 a (±5.1) |
| Significance | ** | *** | *** | *** | *** | *** | *** | *** | *** | ** |
| Plum Cultivars | Type of Fermentation | Total Extract | Titratable Acidity | Ethanol | Free Amino Nitrogen | Fermentation Efficiency |
|---|---|---|---|---|---|---|
| [g/L] | [% vol.] | [mg/L] | [%] | |||
| Węgierka Zwykła | S. cerevisiae S1 | 42.0 ab (±2.2) | 3.72 e (±0.12) | 6.9 b (±0.2) | 46.29 d (±1.98) | 93.8 ab (±1.2) |
| H. uvarum H2 | 44.7 ab (±1.2) | 3.63 e (±0.21) | 6.8 b (±0.1) | 51.67 cd (±2.18) | 92.3 ab (±2.1) | |
| Spontaneous fermentation | 39.0 ab (±0.8) | 3.73 e (±0.23) | 5.7 c (±0.1) | 52.46 c (±1.02) | 78.4 bc (±0.9) | |
| Ethanol RED (S. cerevisiae) | 45.3 ab (±0.5) | 4.43 d (±0.07) | 7.2 ab (±0.2) | 47.46 d (±1.06) | 97.8 a (±1.4) | |
| Węgierka Dąbrowicka | S. cerevisiae S1 | 36.3 b (±2.1) | 9.14 a (±0.03) | 7.9 a (±0.1) | 57.61 c (±2.61) | 98.8 a (±1.3) |
| H. uvarum H2 | 33.0 bc (±3.7) | 6.53 b (±1.73) | 7.8 a (±0.2) | 45.32 d (±1.51) | 98.6 a (±0.8) | |
| Spontaneous fermentation | 48.7 a (±5.8) | 8.55 a (±0.72) | 5.4 c (±0.3) | 44.91 d (±6.02) | 67.6 c (±2.1) | |
| Ethanol RED (S. cerevisiae) | 46.0 a (±0.8) | 8.33 a (±0.40) | 8.0 a (±0.3) | 54.41 c (±3.31) | 99.3 a (±0.3) | |
| Stanley | S. cerevisiae S1 | 44.7 ab (±0.5) | 5.81 c (±0.13) | 7.6 a (±0.3) | 66.33 b (±3.03) | 96.4 a (±0.8) |
| H. uvarum H2 | 43.0 ab (±0.8) | 6.10 b (±0.38) | 7.4 a (±0.1) | 59.11 c (±0.61) | 93.6 ab (±1.0) | |
| Spontaneous fermentation | 42.7 ab (±1.2) | 5.38 c (±0.32) | 6.7 b (±1.1) | 57.72 c (±0.83) | 84.8 b (±0.6) | |
| Ethanol RED (S. cerevisiae) | 39.7 ab (±1.6) | 7.19 ab (±0.41) | 7.8 a (±0.9) | 63.32 bc (±0.54) | 98.9 a (±1.2) | |
| Čačanska Lepotica | S. cerevisiae S1 | 28.0 c (±1.6) | 5.47 c (±0.41) | 7.4 a (±0.3) | 70.65 a (±1.44) | 97.6 a (±0.6) |
| H. uvarum H2 | 37.3 b (±2.1) | 5.65 c (±0.36) | 7.0 ab (±0.2) | 69.93 ab (±2.40) | 91.1 ab (±0.8) | |
| Spontaneous fermentation | 53.0 a (±7.4) | 4.93 cd (±0.18) | 5.1 c (±0.7) | 72.49 a (±1.43) | 66.6 c (±4.3) | |
| Ethanol RED (S. cerevisiae) | 46.0 a (±0.8) | 5.64 c (±0.12) | 7.5 a (±0.2) | 73.65 a (±3.89) | 97.7 a (±1.1) | |
| Significance | ** | * | *** | ** | ** | |
| Plum Cultivar | Type of Fermentation | Citric Acid | Malic Acid | Succinic Acid | Lactic Acid | Acetic Acid | Glycerol | Fructose | Sucrose | Glucose |
|---|---|---|---|---|---|---|---|---|---|---|
| Węgierka Zwykła | S. cerevisiae S1 | 0.29 a | 0.99 c | 2.01 a | 9.38 a | 0.28 c | 3.10 d | 0.47 d | 1.44 b | 2.79 bc |
| H. uvarum H2 | 0.00 d | 0.00 d | 1.39 b | 0.48 d | 0.59 bc | 2.54 d | 1.69 c | 0.42 d | 0.00 d | |
| Spontaneous fermentation | 0.00 d | 0.00 d | 0.12 d | 0.75 d | 0.47 c | 5.42 c | 1.01 c | 0.16 d | 0.00 d | |
| Ethanol RED | 0.00 d | 0.00 d | 0.17 d | 0.89 d | 0.69 b | 5.14 c | 0.22 d | 0.59 d | 2.16 b | |
| Węgierka Dąbrowicka | S. cerevisiae S1 | 0.19 b | 3.50 a | 2.15 a | 6.94 b | 0.84 b | 6.92 bc | 0.61 d | 0.32 d | 2.63 bc |
| H. uvarum H2 | 0.24 b | 3.21 a | 2.26 a | 5.39 bc | 0.93 ab | 6.25 bc | 0.00 e | 2.46 a | 4.14 b | |
| Spontaneous fermentation | 0.34 a | 1.07 c | 1.29 b | 4.62 c | 0.34 c | 5.01 c | 6.58 b | 1.16 c | 1.45 c | |
| Ethanol RED | 0.29 a | 3.32 a | 1.91 ab | 3.19 d | 0.57 bc | 8.88 ab | 2.35 c | 0.00 d | 9.85 a | |
| Stanley | S. cerevisiae S1 | 0.14 c | 1.23 c | 1.57 b | 6.38 b | 0.25 c | 7.93 b | 11.28 a | 0.85 cd | 0.65 cd |
| H. uvarum H2 | 0.07 d | 0.00 d | 1.16 b | 2.99 c | 1.07 a | 6.65 bc | 12.66 a | 1.53 b | 0.84 c | |
| Spontaneous fermentation | 0.15 c | 0.10 d | 0.73 c | 3.32 c | 0.67 b | 6.67 bc | 10.72 a | 2.08 ab | 0.91 c | |
| Ethanol RED | 0.23 b | 2.97 a | 2.39 a | 6.39 b | 1.29 a | 10.02 a | 14.05 a | 1.52 b | 0.00 d | |
| Čačanska Lepotica | S. cerevisiae S1 | 0.20 b | 2.00 b | 2.38 a | 8.45 a | 0.25 c | 5.50 c | 2.50 c | 1.33 c | 0.00 d |
| H. uvarum H2 | 0.16 c | 2.36 ab | 2.58 a | 6.51 b | 0.18 c | 5.73 c | 3.32 c | 0.00 d | 0.01 d | |
| Spontaneous fermentation | 0.28 a | 0.35 d | 1.43 b | 5.98 b | 1.33 a | 5.70 c | 0.00 e | 0.21 d | 0.84 c | |
| Ethanol RED | 0.25 ab | 3.56 a | 2.33 a | 5.50 bc | 0.36 c | 6.41 bc | 0.33 d | 0.00 d | 5.91 b | |
| SD pooled | 0.06 | 0.92 | 0.57 | 1.35 | 0.27 | 1.94 | 1.37 | 0.53 | 0.35 | |
| Significance | *** | ** | *** | ** | *** | ** | ** | *** | ** | |
| Compounds [µg/L] | LRI 2 | Węgierka Zwykła | Węgierka Dąbrowicka | Stanley | Čačanska Lepotica | OT 4 | Sig. | Characteristic Aroma |
|---|---|---|---|---|---|---|---|---|
| Esters | ||||||||
| Ethyl acetate | 868 | 6667 b | 5966 b | 6540 b | 10118 a | 5000 | * | sweet, solvent |
| Isobutyl acetate | 1011 | 5.5 b | 10.6 ab | 5.4 b | 16.4 a | 66 | ** | fig-like, banana |
| Ethyl butanoate 3 | 1033 | 112 b | 112 b | 119 b | 229 a | 1 | *** | pineapple |
| Ethyl 2-methylbutyrate | 1048 | 7.6 a | 8.0 a | 10.9 a | 12.3 a | 0.3 | ns | berry, tropical |
| Butyl butanoate | 1215 | 151 a | 0 b | 0 b | 0 b | 100 | *** | fruity banana pineapple sweet |
| Ethyl hexanoate | 1230 | 23 a | 15 a | 21 a | 24 a | 1 | ns | apple peel, pineapple |
| Hexyl butanoate | 1416 | 10.5 a | 0 b | 10.7 a | 2.2 b | 250 | *** | green, fruity, estry vegetative |
| Ethyl octanoate | 1438 | 7.8 ab | 6.7 b | 9.2 ab | 12.4 a | 15 | * | Fruity, winey, sweet |
| 3-hexenyl butanoate | 1464 | 0.5 bc | 0 c | 1.8 b | 10.1 a | 20,000 | *** | Fresh, green apple, fruity |
| Hexyl hexanoate | 1585 | 0.6 a | 0 b | 0.6 a | 0 b | 6.4 | ** | Herbaceous |
| Ethyl decanoate | 1612 | 311 b | 382 ab | 311 b | 474 a | 510 | * | Sweet, fatty, nut-like, winey |
| 1-methylethyl dodecanoate | 1800 | 2.4 d | 24.6 a | 9.3 b | 5.3 c | - | *** | - |
| Ethyl dodecanoate | 1812 | 210 a | 261 a | 242 a | 208 a | 2000 | ns | Oily, fatty, floral |
| Ethyl tridecanoate | 1994 | 0 b | 34 a | 0 b | 60 a | - | *** | - |
| Ethyl tetradecanoate | 2093 | 37 b | 44 b | 35 b | 85 a | 4000 | *** | Mild, waxy, soapy |
| Alcohols | ||||||||
| 1-butanol | 1144 | 18.6 b | 0 b | 83.7 a | 0 b | 500 | *** | banana harsh alcoholic sweet |
| 1-hexanol | 1348 | 2088 bc | 3001 b | 8691 a | 1112 c | 2500 | *** | herbal ethereal alcoholic green |
| 3-hexen-1-ol | 1385 | 6.0 b | 8.9 b | 4.5 b | 23.5 a | 70 | *** | grassy-green freshly cut grass |
| 2-hexen-1-ol | 1409 | 10.6 a | 4.9 b | 4.6 b | 2.2 b | 400 | *** | Sharp green leafy |
| 2-ethyl-1-hexanol | 1487 | 2.8 a | 0 c | 2.2 ab | 1.5 b | 138 | *** | citrus fresh floral oily sweet |
| 1-nonanol | 1642 | 10.2 a | 18.4 a | 12.0 a | 10.3 a | 50 | ns | citrus |
| Benzyl alcohol | 1858 | 4.7 a | 2.5 a | 3.7 a | 4.0 a | 10,000 | ns | floral rose phenolic balsamic |
| Phenol | 1972 | 0.1 b | 0.5 ab | 0.5 ab | 0.9 a | 5900 | * | phenolic plastic rubber |
| 2-phenoxyethanol | 2114 | 0.5 a | 0 b | 0.6 a | 0.4 ab | - | * | mild rose balsam cinnamyl |
| Carbonyl compounds | ||||||||
| 5-methyl-3-hexanone | 1069 | 2205 b | 3140 a | 1753 b | 3365 a | - | *** | pleasant fruity |
| Hexanal | 1076 | 1122 b | 7783 a | 7118 a | 8562 a | 4.5–5 | *** | grassy |
| 4-methyl-2-hexanone | 1113 | 0 b | 1.5 a | 2.8 a | 2.4 a | - | *** | pleasant fruity |
| 2-hexenal | 1199 | 291 bc | 467 ab | 549 a | 134 c | 17 | ** | green |
| Nonanal 3 | 1392 | 4.0 ab | 2.5 b | 5.9 a | 2.3 b | 1 | ** | aldehydic rose orange peel |
| 2,4-hexadienal | 1406 | 0.3 a | 0.6 a | 0.8 a | 0.6 a | 60 | ns | Green, fruity, aldehydic, citrus |
| Decanal | 1491 | 7.7 a | 0 b | 2.0 b | 0 b | 2 | ** | aldehydic orange peel citrus |
| Benzaldehyde | 1513 | 2.1 b | 0.7 b | 2.2 b | 6.1 a | 350 | ** | spicy bitter-almond |
| Acetophenone | 1640 | 10.8 a | 4.0 bc | 0 c | 7.7 ab | 65 | *** | pungent hawthorn almond |
| Terpenoids | ||||||||
| p-Cymene | 1259 | 1.2 bc | 0.9 c | 1.6 b | 2.6 a | - | *** | solvent, gasoline, citrus |
| Bornylene | 1506 | 0.7 a | 0 b | 0 b | 0.3 ab | - | ** | - |
| Linalool | 1536 | 13.7 a | 1.4 c | 4.3 b | 5.0 b | 6 | *** | Floral, woody, lavender |
| Calamenene | 1567 | 0.5 b | 3.2 a | 1.3 ab | 0.5 b | - | *** | Herb spice |
| ß-Cyclocitral | 1595 | 0.5 b | 0 c | 0 c | 0.9 a | 5 | *** | Minty, citrus |
| Menthol | 1617 | 0.8 a | 0.2 b | 0 b | 0.2 ab | - | * | Cooling, fresh, sweet, minty |
| Damascenone | 1804 | 3.6 b | 17.9 a | 6.8 b | 4.0 b | 0.002 | ** | Apple, rose honeys |
| Geraniol | 1816 | 0.3 a | 1.4 a | 1.4 a | 1.4 a | 40 | ns | floral fruity rose waxy citrus |
| Geranyl acetone | 1828 | 3.3 a | 1.3 a | 1.4 a | 3.1 a | 60 | ns | rose leaf magnolia aldehydic |
| ß-ionone | 1918 | 0.6 b | 0.9 ab | 2.2 a | 2.1 a | 7 | * | violet raspberry woody fruity |
| p-cresol | 1967 | 0.3 a | 0.2 a | 0.2 a | 0.1 a | 55 | ns | phenolic narcissus |
| Eugenol | 2136 | 0.4 a | 1.6 a | 1.0 a | 1.4 a | 6 | ns | sweet spicy clove woody |
| Lactones | ||||||||
| Γ-nonanolactone | 2128 | 1.1 ab | 2.0 a | 1.2 ab | 0.5 b | 65 | * | coconut creamy waxy buttery waxy peach coconut buttery |
| Γ-decanolactone | 2328 | 3.2 a | 2.9 a | 2.6 a | 0.2 b | 11 | ** | |
| Hydrocarbons | ||||||||
| Acenaphthene | 2154 | 1.5 a | 0.8 a | 1.5 a | 1.4 a | 80 | * | Pungent |
| Other compounds | ||||||||
| Dimethyl sulfoxide | 1204 | 2.0 a | 2.3 a | 4.3 a | 2.6 a | - | ns | Garlic-like |
| Benzothiazole 3 | 1952 | 8.8 a | 7.8 a | 11.0 a | 9.3 a | 80 | ns | Sulphurous, rubbery, burnt |
| LRI | Węgierka Zwykła | Węgierka Dąbrowicka | Stanley | Čačanska Lepotica | Methods | Significance | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| S. cerevisiae S1 | H. uvarum H2 | Spontaneous Fermentation | Ethanol RED | S. cerevisiae S1 | H. uvarum H2 | Spontaneous Fermentation | Ethanol RED | S. cerevisiae S1 | H. uvarum H2 | Spontaneous Fermentation | Ethanol RED | S. cerevisiae S1 | H. uvarum H2 | Spontaneous Fermentation | Ethanol RED | ||||
| Esters | |||||||||||||||||||
| Ethyl acetate | 599 | 76,600 e | 78,513 e | 104,256 cd | 74,717 e | 104,351 cd | 193,967 a | 202,212 a | 75,380 e | 45,933 f | 51,830 f | 94,536 d | 117,636 c | 24,323 g | 25,456 g | 154,846 b | 16,920 g | FID | *** |
| Isobutyl acetate | 771 | 8.1 a | 0.8 f | 4.9 bc | 2.5 cdef | 1.5 ef | 3.9 bcde | 5.6 ab | 1.7 def | 1.4 ef | 0.8 f | 4.6 bc | 1.3 ef | 1.3 ef | 0.6 f | 4.4 bcd | 2.98 b–f | MS | *** |
| Ethyl butanoate | 789 | 37.6 a | 8.1 bcd | 9.1 bcd | 1.9 d | 1.9 d | 14.1 bc | 1.6 d | 2.0 d | 8.3 bcd | 10.1 bcd | 7.8 bcd | 3.7 cd | 16.4 b | 16.3 b | 8.7 bcd | 9.7 bcd | MS | *** |
| Ethyl 2-hydroxypropanoate | 798 | 1118.5 bc | 946.9 bc | 762.3 cd | 170.3 d | 3187.2 a | 1061.5 bc | 1186.6 bc | 140.9 d | 1637.7 b | 966.0 bc | 1066.3 bc | 122.3 d | 736.1 cd | 1050.4 bc | 619.4 cd | 67.9 d | MS | *** |
| 3-Methylbutyl acetate | 872 | 4.7 def | 3.5 ef | 9.3 bcd | 10.3 bc | 3.6 ef | 6.3 cdef | 8.9 bcd | 1.2 f | 5.2 cdef | 7.7 bcde | 8.7 bcde | 1.6 f | 7.1 bcde | 12.4 b | 9.5 bcd | 18.6 a | MS | *** |
| Methyl hexanoate | 915 | 4.4 c | 0.7 efg | 1.4 defg | 0.2 g | 2.1 de | 11.0 a | 0.9 efg | 0.2 g | 4.6 c | 8.0 b | 0.9 efg | 0.5 fg | 1.8 def | 2.6 d | 1.9 def | 0.5 fg | MS | *** |
| (s)-i-butyl lactate | 960 | 4.8 bcde | 5.8 bcd | 2.9 defg | 0.5 g | 13.5 a | 3.6 cdef | 2.6 efg | 0.2 g | 7.6 b | 5.7 bcd | 3.5 cdef | 0.2 g | 1.9 efg | 1.2 fg | 3.7 cdef | 0.0 g | MS | *** |
| Ethyl hexanoate | 986 | 77.7 bcd | 11.6 h | 38.7 efgh | 40.9 defg | 25.9 fgh | 179.1 a | 18.9 gh | 13.7 h | 80.9 bc | 95.2 b | 54.0 c–g | 33.7 e–h | 63.5 b–f | 65.4 bcde | 41.1 d–h | 69.4 bcde | MS | *** |
| Isoamyl lactate | 1047 | 18.4 ab | 21.6 a | 2.6 cd | 0.1 d | 24.6 a | 8.9 cd | 3.6 cd | 0.1 d | 24.1 a | 18.2 ab | 3.8 cd | 0.1 d | 11.2 bc | 10.0 bc | 2.2 cd | 0.1 d | MS | *** |
| Ethyl methyl succinate | 1070 | 18.4 ab | 25.1 a | 7.5 cdef | 2.7 ef | 10.7 bcde | 11.5 bcd | 0.4 f | 0.4 f | 14.6 bc | 18.6 ab | 5.7 def | 1.8 f | 4.1 def | 5.9 def | 6.2 def | 2.1 f | MS | *** |
| Methyl octanoate | 1107 | 4.4 bc | 0.4 e | 1.7 de | 1.6 de | 2.2 cde | 10.5 a | 1.3 e | 1.7 de | 3.8 cd | 6.3 b | 1.4 de | 1.6 de | 1.3 e | 1.8 de | 1.8 de | 1.9 de | MS | *** |
| Ethyl benzoate | 1144 | 281.0 a | 166.1 b–f | 247.0 ab | 156.0 b–f | 85.3 fg | 91.4 efg | 105.1 defg | 30.7 g | 213.7 abc | 207.7 abcd | 174.0 b–f | 152.3 b–f | 126.2 c–g | 144.5 b–f | 191.5 a–e | 119.7 c–g | MS | *** |
| Diethyl succinate | 1149 | 1955.0 b | 3202.7 a | 1593.7 bcde | 1620.7 bcd | 1390.8 bcde | 1709.0 bcd | 64.9 g | 338.3 fg | 1558.0 bcde | 1730.3 bc | 1209.7 bcde | 1398.7 bcde | 865.9 defg | 1070.2 cdef | 743.2 efg | 1213.9 bcde | MS | *** |
| Ethyl octanoate | 1180 | 74.0 b | 8.7 d | 68.7 bc | 31.3 bcd | 57.2 bcd | 330.4 a | 48.8 bcd | 16.8 cd | 75.0 b | 78.6 b | 70.8 b | 52.6 bcd | 84.3 b | 81.9 b | 70.2 b | 74.0 b | MS | *** |
| Ethyl decanoate | 1397 | 41.1 bcd | 16.9 cd | 52.6 bc | 20.8 cd | 52.5 bc | 117.1 a | 38.5 bcd | 14.5 d | 47.0 bcd | 65.5 b | 67.9 b | 17.1 cd | 52.4 bcd | 62.7 b | 52.6 bc | 72.6 b | MS | *** |
| Ethyl isopentyl succinate | 1421 | 4.8 a | 5.2 a | 2.3 c | 2.2 c | 5.0 a | 5.1 a | 0.3 d | 1.0 cd | 5.9 a | 5.6 a | 2.1 cd | 2.3 c | 2.6 bc | 2.6 bc | 2.1 cd | 4.4 ab | MS | *** |
| Ethyl dodecanoate | 1581 | 34.9 bcd | 46.5 bc | 34.0 bcd | 15.9 cd | 84.5 a | 42.6 bcd | 31.0 bcd | 13.5 d | 45.4 bc | 43.9 bcd | 49.9 b | 18.1 cd | 44.4 bcd | 33.5 bcd | 61.5 ab | 61.2 ab | MS | *** |
| Ethyl tetradecanoate | 1790 | 7.9 bcd | 13.1 b | 8.2 bcd | 8.1 bcd | 35.0 a | 12.1 bc | 7.6 bcd | 2.8 d | 14.0 b | 12.5 b | 7.8 bcd | 5.3 cd | 8.1 bcd | 5.4 cd | 8.6 bcd | 12.2 bc | MS | *** |
| Ethyl hexadecanoate | 1990 | 41.5 c–g | 58.3 bcde | 34.9 defg | 55.2 b–f | 146.6 a | 85.9 b | 22.4 g | 45.9 c–g | 70.5 bc | 65.7 bcd | 30.5 efg | 62.6 bcd | 15.5 g | 24.7 fg | 33.9 defg | 65.2 bcd | MS | *** |
| Alcohols | |||||||||||||||||||
| Methanol | 382 | 1,763,141 a | 1,604,822 a | 908,463 bc | 892,061 bc | 289,671 ef | 981,453 b | 1,031,741 b | 435,692 de | 653,663 cd | 981,323 b | 770,277 bc | 663,322 cd | 245,341 ef | 964,122 b | 112,285 f | 101,331 f | FID | *** |
| Propanol | 543 | 331,832 c | 283,571 e | 130,221 g | 125,921 gh | 509,675 a | 240,771 f | 250,788 g | 292,321 de | 381,141 b | 382,263 b | 321,697 cd | 406,823 b | 234,256 f | 316,467 cd | 96,651 h | 119,851 gh | FID | *** |
| Isobutanol | 629 | 187,873 c | 157,952 d | 97,726 fg | 198,263 bc | 244,759 a | 204,076 bc | 212,523 b | 130,967 e | 100,969 fg | 99,567 fg | 832,967 g | 112,063 ef | 46,673 h | 99,713 fg | 25,600 h | 33,013 h | FID | *** |
| Butanol | 653 | 11,171 cd | 22,277 a | 1834 ef | 3356 ef | 2043 ef | 1687 ef | 0 f | 1810 ef | 15,913 b | 14,843 bc | 5233 e | 9960 d | 213 f | 0 f | 647 f | 0 f | FID | *** |
| Amyl alcohols | 740 | 83,167 g | 455,136 b | 217,710 d | 621,077 a | 71,897 gh | 45,643 hi | 272,343 c | 46,077 hi | 167,463 ef | 145,333 f | 38,873 i | 266,590 c | 80,873 g | 185,700 e | 24,574 i | 86,897 g | FID | *** |
| Pentanol | 757 | 241 c | 92 c | 2161 a | 220 c | 0 c | 0 c | 0 c | 0 c | 0 c | 321 c | 1632 b | 0 c | 0 c | 0 c | 121 c | 0 c | FID | *** |
| 2,3-Butanediol | 768 | 119.9 de | 778.3 b | 284.3 de | 138.3 de | 737.1 bc | 1156.4 a | 383.6 cd | 80.6 de | 192.7 de | 973.0 ab | 362.7 d | 146.7 de | 131.3 de | 0.00 e | 230.0 de | 166.5 de | MS | *** |
| (Z)-3-Hexen-1-ol | 858 | 1.1 e | 1.1 e | 5.8 ab c | 1.9 de | 7.8 a | 2.6 de | 5.7 ab c | 1.2 de | 2.6 de | 1.2 e | 6.1 ab | 2.1 de | 3.1 bcde | 0.8 e | 4.6 bcd | 2.8 cde | MS | *** |
| Hexanol | 862 | 18,096 bcd | 19,480 bcd | 18,076 bcd | 5761 ef | 22,333 bc | 23,310 b | 23,697 b | 7557 e | 15,581 d | 17,526 cd | 29,541 a | 65,901 ef | 19,787 bcd | 20,200 bcd | 3757 ef | 1747 f | FID | *** |
| Benzyl alcohol | 1006 | 28.8 a | 28.2 a | 17.6 a–e | 18.1 abcd | 13.3 cde | 8.3 de | 5.4 e | 13.9 cde | 24.7 abc | 20.6 abcd | 17.1 a–e | 27.3 ab | 13.3 cde | 15.6 bcde | 23.8 abc | 23.4 abc | MS | *** |
| Phenylethanol | 1114 | 65,111 a | 39,700 fgh | 42,221 e–h | 44,515 c–h | 59,463 ab | 33,640 hi | 38,444 gh | 55,211 abcd | 56,100 abc | 57,063 ab | 43,931 d–h | 52,081 b–e | 48,973 b–g | 50,587 b–f | 23,541 i | 37,526 gh | FID | *** |
| 1-Nonanol | 1166 | 3.2 de | 2.8 de | 3.3 de | 2.7 de | 4.1 bcde | 5.7 bc | 4.8 bcd | 1.9 e | 3.3 de | 3.8 bcde | 2.8 de | 3.4 cde | 5.9 b | 3.9 bcde | 8.9 a | 4.8 bcd | MS | *** |
| Terpenoids | |||||||||||||||||||
| cis-Linalol oxide | 1066 | 2.4 a | 2.3 a | 2.0 ab | 1.5 b | 0.3 c | 0.4 c | 0.3 c | 0.1 c | 0.5 c | 0.6 c | 0.6 c | 0.5 c | 0.1 c | 0.2 c | 0.4 c | 0.3 c | MS | *** |
| à-Terpineol | 1176 | 3.9 bc | 5.4 a | 2.5 c–g | 1.4 fg | 2.9 bcde | 3.3 bcd | 2.9 b–f | 1.8 defg | 3.3 bcd | 4.2 ab | 2.6 c–g | 1.8 efg | 1.3 g | 1.3 g | 2.4 defg | 1.5 efg | MS | *** |
| Eugenol | 1339 | 2.2 bc | 3.5 a | 2.6 b | 2.2 bc | 0.5 fg | 0.5 fg | 0.1 fg | 0.1 fg | 0.7 fg | 0.6 fg | 0.1 g | 0.1 g | 1.7 cd | 1.5 cde | 0.8 efg | 0.9 def | MS | *** |
| (E)-β-Damascenone | 1384 | 1.2 a–e | 1.2 a–e | 1.6 ab | 0.3 de | 0.4 cde | 1.2 a–e | 1.7 a | 0.2 e | 1.2 a–e | 1.3 abcd | 1.6 ab | 0.3 de | 1.1 a–e | 0.8 a–e | 1.5 abc | 0.5 b–e | MS | *** |
| Geranyl acetone | 1434 | 2.7 ab | 2.3 bc | 2.9 ab | 0.7 d | 2.8 ab | 3.6 a | 3.6 a | 0.5 d | 2.7 ab | 3.1 ab | 3.1 ab | 0.9 ab | 1.0 d | 1.2 cd | 2.9 ab | 0.9 d | MS | *** |
| Carbonyl compounds | |||||||||||||||||||
| Acetaldehyde | 360 | 6490 de | 24,700 b | 5310 ef | 6440 de | 770 g | 4530 efg | 4440 efg | 10,340 cd | 6710 de | 12,990 c | 6690 de | 43,000 a | 5460 e | 13,580 c | 13,860 c | 1150 fg | FID | *** |
| Acetone | 469 | 2886 b | 2800 b | 2133 b | 2233 b | 2367 bc | 1267 c | 2113 bc | 2656 b | 1244 c | 1233 c | 9172 a | 952 c | 2213 bc | 3444 b | 2786 b | 3133 b | FID | *** |
| Hexanal | 780 | 4.22 d | 4.00 d | 7.94 bc | 1.08 fg | 3.41 def | 1.40 efg | 12.74 a | 0.00 g | 4.28 d | 3.54 de | 9.94 b | 0.79 g | 3.26 def | 0.00 g | 7.04 c | 1.53 efg | MS | *** |
| Benzaldehyde | 930 | 2.7 fg | 3.5 ef | 6.6 cd | 4.9 de | 3.3 ef | 2.3 fg | 6.5 cd | 9.6 a | 3.5 ef | 3.4 ef | 7.4 bc | 8.8 ab | 1.5 g | 1.3 g | 7.4 bc | 3.7 ef | MS | *** |
| 2-Heptenal | 936 | 0.6 bc | 0.8 a | 0.7 ab | 0.2 d | 0.2 d | 0.0 e | 0.5 c | 0.1 de | 0.0 e | 0.0 e | 0.0 e | 0.0 e | 0.0 e | 0.0 e | 0.0 e | 0.0 e | MS | *** |
| Lactones | |||||||||||||||||||
| Butyrolactone | 908 | 2.6 b | 4.6 b | 6.3 b | 5.8 b | 24.7 ab | 197.9 a | 177.6 ab | 5.9 b | 17.5 ab | 6.7 b | 8.8 b | 6.4 b | 10.9 b | 2.9 b | 3.7 b | 7.3 b | MS | ** |
| (R)-γ-decalactone | 1428 | 1.7 c | 1.7 c | 1.7 c | 1.2 c | 34.0 ab | 24.9 b | 43.9 a | 6.9 c | 1.6 c | 1.4 c | 1.5 c | 1.2 c | 0.5 c | 1.2 c | 1.0 c | 0.6 c | MS | *** |
| ç-Dodecalactone | 1655 | 1.6 d | 1.8 d | 1.9 d | 1.6 d | 13.4 a | 5.4 c | 9.4 b | 2.5 cd | 2.5 cd | 2.1 cd | 2.4 cd | 2.4 cd | 0.2 d | 0.1 d | 0.2 d | 0.2 d | MS | *** |

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Januszek, M.; Satora, P. How Different Fermentation Type Affects Volatile Composition of Plum Jerkums. Appl. Sci. 2021, 11, 4658. https://doi.org/10.3390/app11104658
Januszek M, Satora P. How Different Fermentation Type Affects Volatile Composition of Plum Jerkums. Applied Sciences. 2021; 11(10):4658. https://doi.org/10.3390/app11104658
Chicago/Turabian StyleJanuszek, Magdalena, and Paweł Satora. 2021. "How Different Fermentation Type Affects Volatile Composition of Plum Jerkums" Applied Sciences 11, no. 10: 4658. https://doi.org/10.3390/app11104658
APA StyleJanuszek, M., & Satora, P. (2021). How Different Fermentation Type Affects Volatile Composition of Plum Jerkums. Applied Sciences, 11(10), 4658. https://doi.org/10.3390/app11104658






