Abstract
In the past, complex rehabilitations, such as the rehabilitation of an entire arch with a prosthesis on implants, were reserved for the few patients who presented an optimal state of health as the interventions were long and traumatic. Nowadays, the use of devices such as the Magnetic Mallet and the laser allows us to perform the same interventions in less time and in a minimally invasive way. The case report we present shows how a fragile patient, subjected to the insertion of eight implants on the same day, had a positive response, thanks to the use of a Magnetic Mallet to prepare the implant sites, the application of the photodynamic therapy without dye (diode laser + hydrogen peroxide) to decontaminate the post-extraction alveoli and the use of an erbium laser to induce more bone bleeding and promote healing. The implants were then loaded in 48 h with a Toronto-type temporary total prosthesis. The patient had a pain-free and complication-free outcome. It is interesting to note how technological development, aimed at reducing the morbidity of surgery, makes it possible to perform almost all surgical therapies, even the most advanced, on any patient, regardless of general health conditions.
1. Introduction
Over the past decade, the study and use of minimally invasive procedures in medicine and dentistry have increased exponentially. In particular, minimally invasive surgery allows for a reduction in post-operative complications [1,2,3], a lower consumption of analgesic and anti-inflammatory drugs [4], a shorter hospital stay and therefore, in general, a reduction in the time required to carry out a therapy. All this is even more relevant when it is necessary to treat more fragile patients due to past and/or present systemic conditions, and who therefore may also have to take drugs that are contraindicated to the therapeutic act that must be performed. The MID World Congress defines minimally invasive dentistry as that discipline in which techniques are applied to respect the health, function and aesthetics of the oral tissue, preventing the onset of disease or intercepting its progress with minimal tissue loss [5].
On the other hand, the use of innovative technologies has increased the cost of therapy. The use of advanced technologies, although it can increase the cost of the surgical procedure, allows the patient to return to normal life earlier, generating benefits that make the procedure more cost effective than conventional surgical techniques [6]. Among these new technologies, great successes in oral surgery were the Magnetic Mallet, the diode lasers combined with solutions with a high oxygen content (photodynamic therapy without dye) and the erbium laser.
1.1. Magnetic Mallet
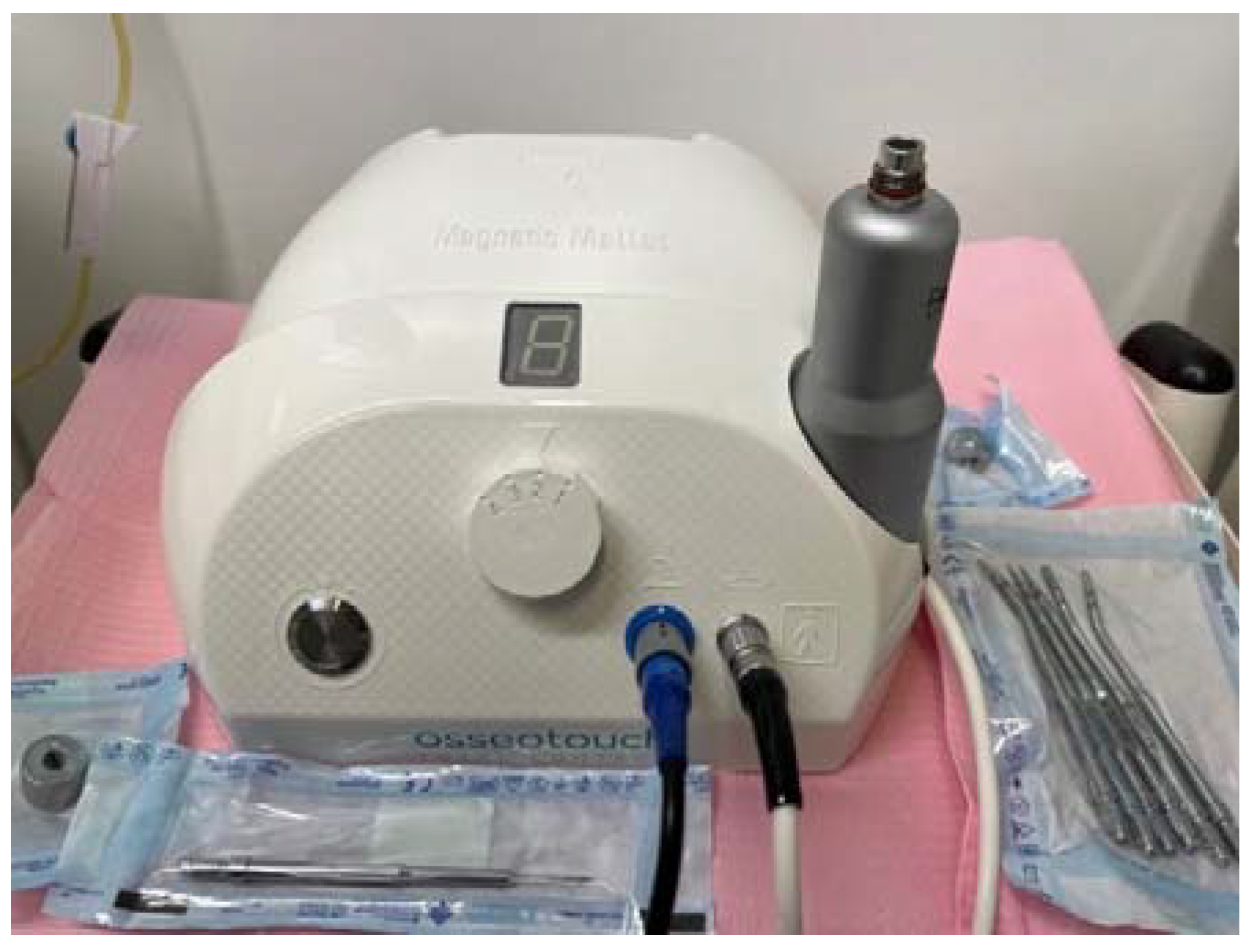
The Magnetic Mallet (Osseotouch, Gallarate, Italy) (Figure 1) is a surgical medical device equipped with different inserts and has replaced many surgical instruments during the operations of the extraction of the dental elements, of implant surgery, of regenerative surgery and of the removal of incongruous prosthetic artifacts. The device exploits the electromagnetic impact, which allows it to have an impact force of a high intensity and a very short duration such as to obtain the plastic deformation of the bone, but without propagating throughout the bone component, as usually occurs with blows of the surgical hammer [7].
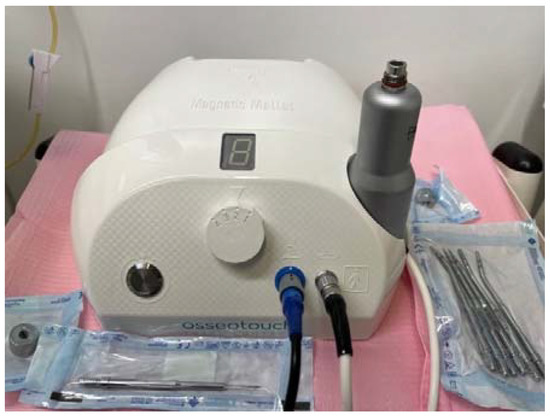
Figure 1.
Magnetic Mallet with inserts for implant surgery.
For this reason, the use of the Magnetic Mallet prevents the patient from developing the so-called benign paroxysmal vertiginous syndrome, that is, vertiginous post-operative symptoms [8]. The impact time is 50 microseconds and can generate a force of up to 300 kg. The magneto-dynamic propulsion applies much more force, in a short time, than manual instruments. The forces are focused on the treated area, limiting their dispersion in the craniofacial mass as much as possible. This means more patient comfort and total accuracy during the procedure. There is more precise control of the osteotome and the direction of entry of the tip into the bone. This is very important as bones are generally made up of parts with different densities and therefore the instrument tends to be deflected when moving from one specific density to another.
The Instruments operate with a longitudinal movement without the risk of deviations due to the different bone consistencies. The use of the handpiece is also very simple; in fact, it can be handled with one hand. For whatever procedure is used (from extraction to split crest), [9,10,11,12,13] the forces are applied with great precision and in a predictable manner, ensuring total control by the operator during surgery.
Another very important aspect of using the Magnetic Mallet is its ability to condense the bone, resulting in bone densification, which is useful for minimizing tissue trauma and increasing the level of bone. A study by Crespi et al. [14] compared electrical versus hand mallets in osteotome-assisted surgery for maxillary bone condensing in the immediately loaded implant procedure. These results demonstrated stable marginal bone levels over time and a significant increase in bone height between 6 and 12 months with the osteotome technique. The use of an electrical mallet provided some essential clinical advantages for the patients during the surgical procedure in comparison with the hand mallet.
The Magnetic Mallet substantially reduces the generation of heat, so the use of irrigation is not necessary. Since irrigation is not necessary, the visibility of the operator is increased and he is able to control the progressive penetration of the instrument. The mechanical frictions that develop over millionths of a second are not sufficient to increase the temperature of the bone. The ergonomics of the handpiece allow it to be used with one hand and facilitates its positioning in the area to be treated.
1.2. Lasers in Dentistry
The laser is light and is characterized by its specific wavelength. The laser (Light Amplification by Stimulated Emission of Radiation) is an electromedical device which, after the input of energy, determines the emission by specific chemical elements of an electromagnetic radiation, light, at a high intensity, with monochromatic characteristics, coherence and unidirectionality.
There are many lasers used in dentistry [15] and they differ from each other mainly in the active medium that feeds them (erbium, CO2, diodes...). Each active medium (source) emits its own wave-like radiant energy with its own wavelength. The various wavelengths have different affinities with the various elements of the organism.
Most medical lasers are in the invisible field. Below 375 nm there is the range of UV rays, above 775 there are infrared rays. Both ultraviolet and infrared are invisible as the human eye perceives light between 375 and 775 nm.
Depending on the target and wavelength, the laser will have different effects.
Er: the YAG laser and CO2 laser are very much absorbed by water and hydroxyapatite and therefore by target tissues (soft and hard tissues); consequently, the penetration into the tissues is very low (2 microns for the Er: YAG laser, 20 microns for the CO2 laser) and therefore does not cause any effect in depth. On the contrary, diodes and Nd: YAG lasers penetrate very deeply, so in addition to having a visible effect on the surface, they can also have effects that are not visible in depth. It is precisely on these effects that laser therapy in periodontology and oral surgery are based.
Based on the concept that the laser transforms light energy into thermal energy, it is evident that the effects of the lasers on tissues are due to the same increase in temperature due to the transfer of thermal energy by the laser sources.
Precisely because of important and localized increases in temperature, it is possible to obtain, with lasers, the first and most evident effect, as it is visible: tissue ablation.
In fact, the volatilization of the tissues occurs when the temperature exceeds 100 degrees centigrade.
If the localized temperature increases significantly, it will be possible to have protein denaturation effects with the dissolution of collagen (between 50 °C and 75 °C). This determines coagulation effects, however irreversible the denaturation of the tissues. If, on the other hand, it is possible to keep the localized temperature increase below 50 °C, there will be only a slight vasodilation, without irreversible changes in the tissues affected by the laser effect.
The differences in the effects on the target surface of poorly penetrating (erbium, CO2) and very penetrating (diodes, Nd: YAG, Helio-Neon) lasers are important:
- Both laser families have photoablative effects;
- Low penetrating lasers do not go deep, and they have thermal effects but do not have great photodynamic and decontaminating effects. They are ideal for their ablative abilities on hard tissues (Er: YAG) and on soft tissues (CO2 and Er: YAG);
- Highly penetrating lasers have ablative, thermal (strong, medium, weak), photodynamic (or decontaminating) and biostimulating effects. The deeper we go into the fabric, the more the decontaminating and biostimulating effects increase and the thermal ones are reduced.
1.3. Photodynamic Therapy without Dye
In the case of a bacterial infection, the infectious process must be eliminated and then decontaminated. If the bacterial infection has resulted in a loss of substance, the goal is to regenerate the loss of substance thanks to laser biostimulation, which determines a sort of “biological doping” for the patient’s tissues, promoting their regeneration.
The photochemical effect (or photodynamic therapy) [16] consists of the activation of a hyper-oxygenated tissue with a monochromatic beam; the interaction between oxygenated tissues and the laser beam produces photochemical reactions in which the energy acceptor is oxygen. Therefore, the photosensitizing substance used must always contain oxygen since the presence of oxygen allows the reactions of photo-activation and the production of singlet oxygen. Singlet oxygen is a free radical of oxygen (a free radical is a highly reactive chemical species that has unpaired electrons in the outer layer); free oxygen radicals are also produced in our body.
Neutrophils stimulated by antibodies release singlet oxygen which, through various mechanisms, destroys bacterial cells (the destruction of the plasma membrane, degradation of the lysosomal membrane, alteration of mitochondrial functions, denaturation of DNA molecules); however, if the bacteria are numerically too many or too aggressive, the body is unable to limit the aggression and the systemic administration of antibiotics becomes necessary.
Research in periodontology [15] has developed protocols that on the one hand use periodontal lasers with a low energy index, with the aim of not overheating the periodontal tissues, and on the other hand use dyes (for example, phenothiazine chloride, characteristic of the Helbo system), which are able to be injected into the pocket to color the bacteria present there. The laser energy, absorbed by the dye for which it was related, determines the release of free radicals and singlet oxygen and is able to denature the cell membrane and lead to death of the bacteria, without damaging the host tissues. However, the literature agrees that there is no statistically significant difference in terms of the eradication of pathogenic bacteria between a conventional non-surgical protocol and a conventional non-surgical protocol followed by photodynamic therapy with a low-power laser and dyes.
From in vitro and in vivo studies [17,18,19,20] it has been shown that the decontaminating action is adequate only if the penetrating laser (diode) is set at values above 2 Watts (the energy necessary to be able to penetrate deeply, overcoming all phenomena of reflection, refraction and transmission that reduce the absorption capacity of the beam in the tissues) and if carried by a liquid, defined as a photo-sensitizer, preferably transparent and with a high oxygenating power, so as not to reduce the depth of penetration of the laser beam into the tissues. This substance has been identified in hydrogen peroxide (hydrogen peroxide H2O2, 10 volumes 3%, non-toxic to tissues).
The periodontal tissue, both soft and hard and previously hyper-oxygenated by hydrogen peroxide irrigations, after waiting for at least two minutes and once stimulated by the laser light first releases essential oxygen and then, for the passage of an electron to the superior orbital, releases singlet oxygen, a highly unstable molecule with a very short half life (in the order of 0.0045 microseconds) capable of binding the fatty acids of the bacterial membrane and dissolving the latter, causing bacteriolysis.
As already mentioned, a crest power greater than 2 W is required to be able to destroy the red and orange Socransky bacterial complexes and to be able to adequately penetrate in depth. However, if the diode laser constantly emits such power levels, an excessive thermal overheating would result which would lead, from an initial vasodilation effect (45 °C), to a denaturation of the molecules up to the carbonization of the tissues, with transformations due to the progressive increase in tissue temperature.
To overcome this problem, the possibility of interrupting the emission of laser pulses has been introduced with a software that regulates the emission intervals (Ton) with tissue cooling times (Toff).
In order to remain below the predetermined threshold of 45 °C, the software must work in microseconds (and therefore with high frequencies, up to 10,000 Hz). This allows you to work with high peak powers, up to 3.5 W but without exceeding the average power threshold of 0.8 W. In addition, high frequencies, up to 10,000 Hz, allow for the release of singlet oxygen molecules within the periodontal tissues up to 10,000 times per second.
The scientific works of Rey and Caccianiga [21,22,23,24] have shown that hydrogen peroxide is bactericidal only for Prevotella intermedia and for Actinobacillus Actinomicetemcomitans, which the laser without irrigants (at 2 Watts of regulation) is effective only on Actinobacillus Actinomicetemcomitans (therefore, the laser is less effective than hydrogen peroxide). All other bacteria in Sokransky’s red and orange complexes are resistant.
The combination of laser and hydrogen peroxide, on the other hand, has shown efficacy on all periodontal pathogenic bacteria and, from the latest in vitro tests carried out, also on Staphylococcus aureus resistant to antibiotics and responsible for skin infections in defected patients.
Therefore, the protocol devised by Dr. Rey in 1992, now called “Photodynamic Therapy without dye”, appears, in the light of the literature, to be effective for treating periodontitis refractory when compared with traditional methods [16,17,23,24].
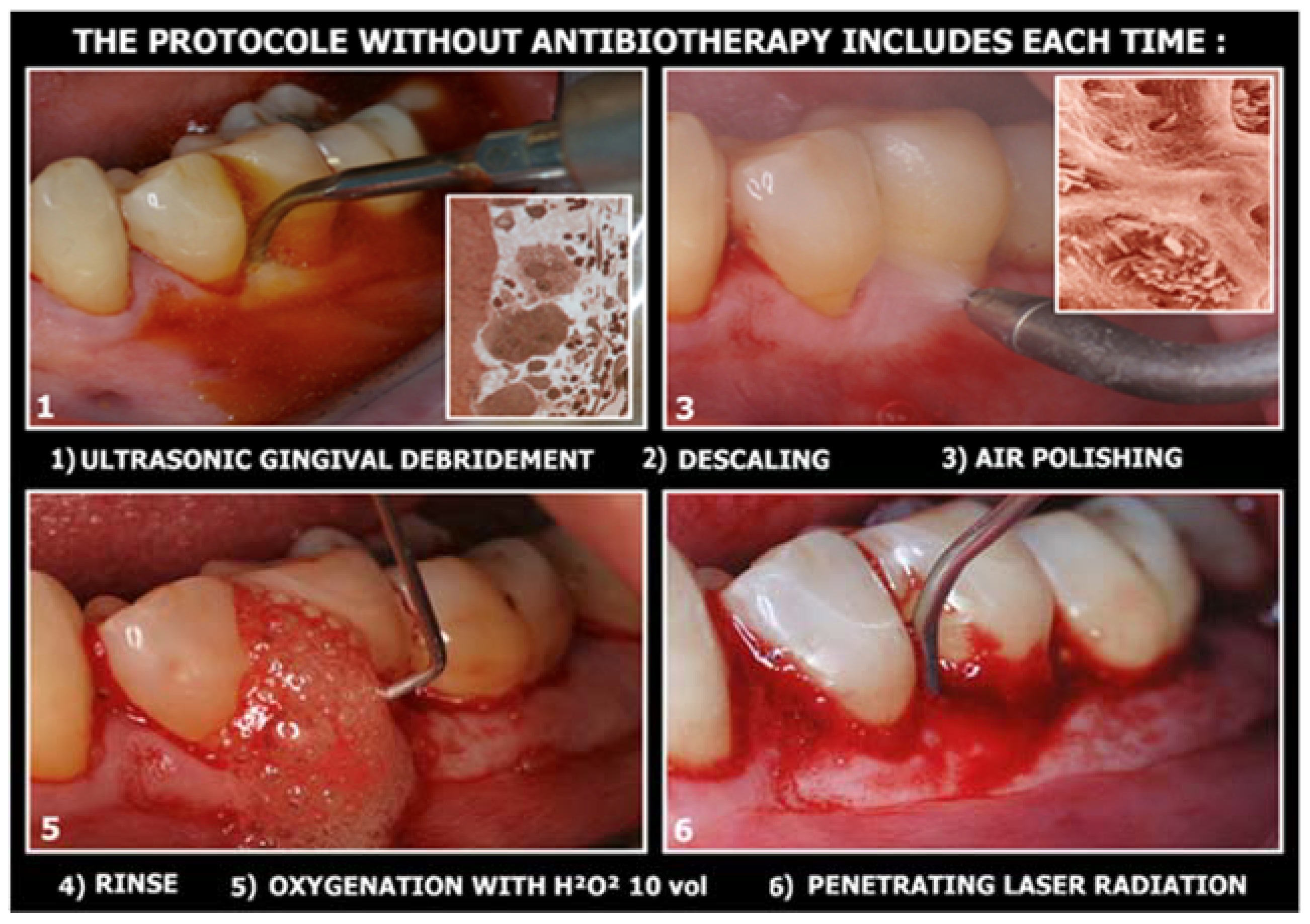
The non-surgical operative protocol can be summarized as follows (Figure 2):
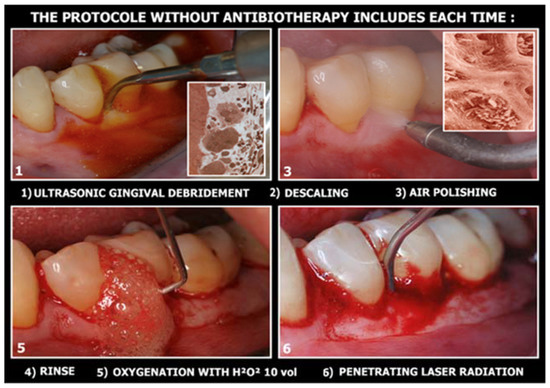
Figure 2.
The 6 clinical steps for dye-free photodynamic therapy.
- Use of ultrasonic instruments and irrigation with polyvinyl jodate-based solutions (Betadine), with complete debridement of the periodontal/peri-implant pocket;
- Use of air flow with glycine or bicarbonate with low abrasiveness on dental surfaces (on exposed implant surfaces it is preferable to use powders with high abrasive power, as the goal is not to preserve cellularity on contaminated surfaces, but to perfectly decontaminate the implant surfaces infected);
- Insert the 3% 10-volume hydrogen peroxide or Sioxyl solution (Doctor Smile, Vicenza, Italy) into the periodontal/peri-implant pocket and leave to act for at least 2 min, removing the excess with a surgical aspirator or dabbing with a sterile gauze pad;
- After the adequate oxygenation of the periodontal/peri-implant bone defect and of the corresponding soft tissues, the diode laser (980 nm) with 400 micron fiber, peak power 2.5 W but with the zeroing of the thermal effect (Ton by 20/30% and lowering the average power to 0.8 W) is used for 60 s;
- At the end of the treatment, advise the patient not to rinse to keep the bleeding resulting from the thermal effect of vasodilation in place, to refrain from oral hygiene maneuvers for at least 6 h and then resume normal home hygiene protocols.
From the studies conducted using these protocols and from in vivo and in vitro tests [16,17,18,19,20], it has proved useful to include laser-assisted protocols within the non-surgical periodontal therapy, since it is a “pain free” treatment for the patient; it allows for management and maintenance complex defects, reducing by 90% the need for surgical treatment; and frames the operative protocol in a minimally invasive perspective and limits the need to administer antibiotics only for patients at risk and thus reduces the risk of antibiotic-resistance phenomena.
1.4. OHLLT (Oxygen High Level Laser Therapy) Technology
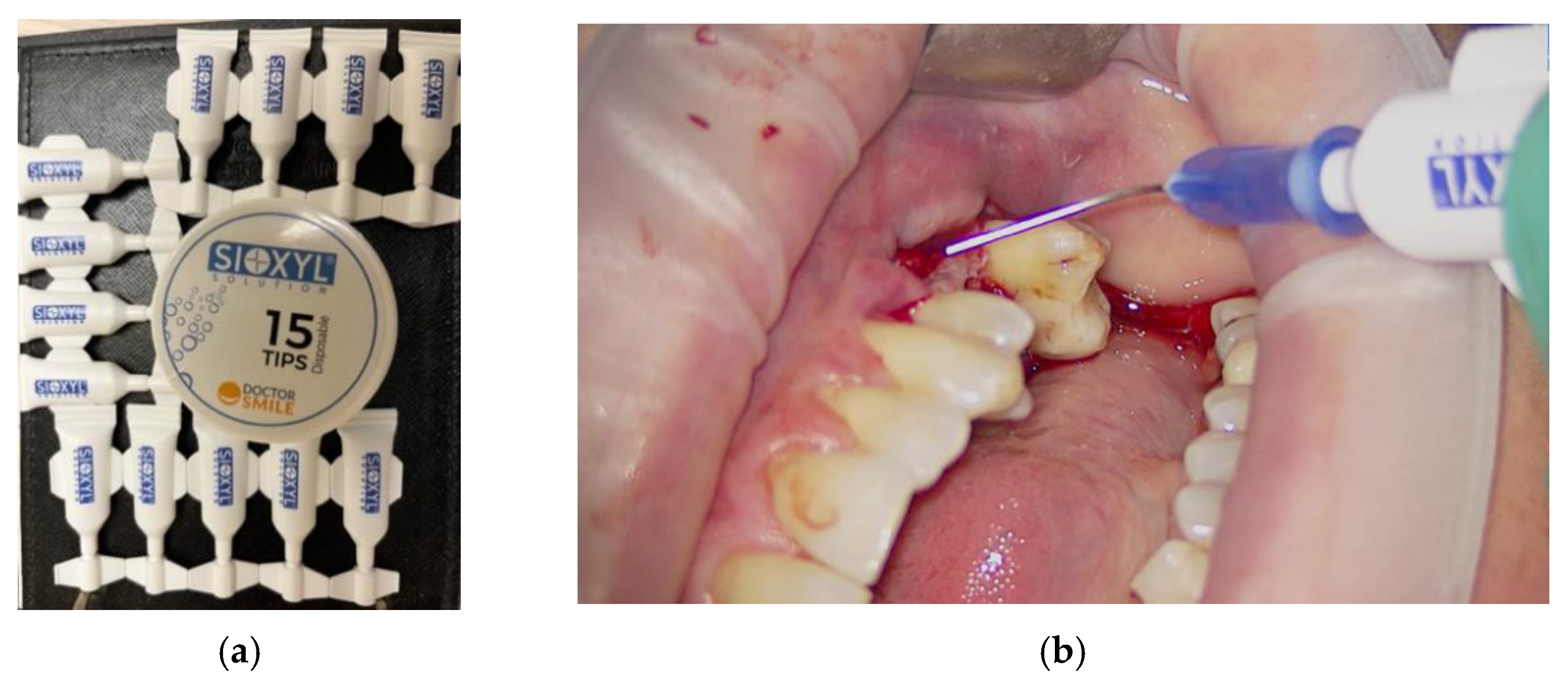
OHLLT (Oxygen High Level Laser Therapy) Technology is a dye-free photodynamic therapy that includes the association of a penetrating laser with a modified and stabilized H2O2 solution with glycerol-phosphate, called a Sioxyl solution (derived from H2O2 10 volumes 3%, therefore without cytotoxic actions) (Figure 3) [21,22,23,24].
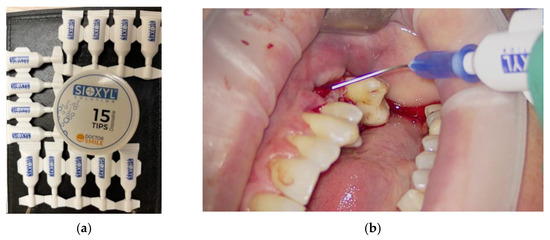
Figure 3.
Sioxyl solution, solution based on hydrogen peroxide 10 volumes 3%, stabilized with glycerophosphates: samples (a); use in clinical practice (b).
As in the dye-free photodynamic therapy conceived by Gérard Rey, the laser energy activates the modified H2O2 solution, releasing free radicals and singlet oxygen which have antibacterial activities on the Gram+ and Gram− groups specific to periodontal diseases. The laser is used with high power peaks and a low average power (<0.8 Watts).
The elimination of silver residues from H2O2 and stabilization with glycerol-phosphates resulted in, with the same bactericidal effect, an increase in biostimulatory activity, which results in the faster healing of the irradiated tissues.
The scheme provides:
- Stabilized H2O2 laser irradiation (Sioxyl solution)
- Liberation of molecules of H2O and fundamental oxygen (O2 has two unpaired electrons with parallel spin)
- New laser irradiation allows the passage of an electron to a higher orbital with spin inversion that allows the release of singlet oxygen
- Singlet oxygen causes the oxidation of the lipid membranes of bacteria with consequent bactericidal action.
The mechanisms of cellular destruction during photodynamic therapy are put in place by the singlet oxygen of free radicals causing:
- DNA denaturation of microorganisms
- Destruction of the lysosomal membrane
- Alteration of mitochondrial functions
1.5. Erbium Lasers
The erbium laser emits a wavelength that is positioned in the mid-infrared spectrum, at 2940 nm. Thanks to the affinity that its wavelength has both with water and with hydroxyapatite, the erbium laser can interact with both soft and hard tissues. The water contained in the target tissues hit by the laser beam absorbs the thermal energy that causes vaporization, causing an increase in pressure that causes microexplosions capable of removing the tissue (the photoacoustic effect) [25].
The erbium laser can intervene on the hard tissues of the oral cavity in a simple way and with less trauma, creating both a gingival access flap and a rapid and precise bone cut, characterized by more accurate margins of the bone breach than those obtained with rotating instruments. Its air and water spray keep the temperature of the target tissue unchanged, and the bactericidal effect of the laser light decreases the risk of infection.
This operating mechanism guarantees respect for the biological properties of the bone, hindering the creation of necrosis and the denaturation of the protein matrix.
1.6. Minimally Invasive Implant Surgery Protocols
The characteristics of the Magnetic Mallet and the lasers can be combined when performing oral surgery, especially in the regenerative one.
In fact, it is possible to exploit the photoablative potential of the erbium laser on the bone tissue to create the invitation to the inserts of the Magnetic Mallet with which to practice the expansion of a residual bone crest lacking in thickness and/or the preparation of the implant sites, as well as the properties decontaminants and photo-bio-modulators of photodynamic therapy without dye (OHLLT or dye-free photodynamic therapy) [21,22,23,24].
2. Materials, Methods and Results
An 81-year-old patient undergoing treatment at the practice showed mobility of the upper elements remaining to support a removable resin prosthesis. The patient requested to rehabilitate the upper arch with a fixed type of prosthesis. The patient had a previous history of hepatitis C still under treatment, which degenerated into liver cancer in 2016 and was treated and resolved in the three-year period of 2017–2020. She also reported that she is an anxious person, with current back and neck problems, while in the past she had suffered from sinusitis. Therefore, given the advanced age of the patient, the patient’s general health conditions and the stability of her blood values, she can be considered a fragile subject (ASA-3).
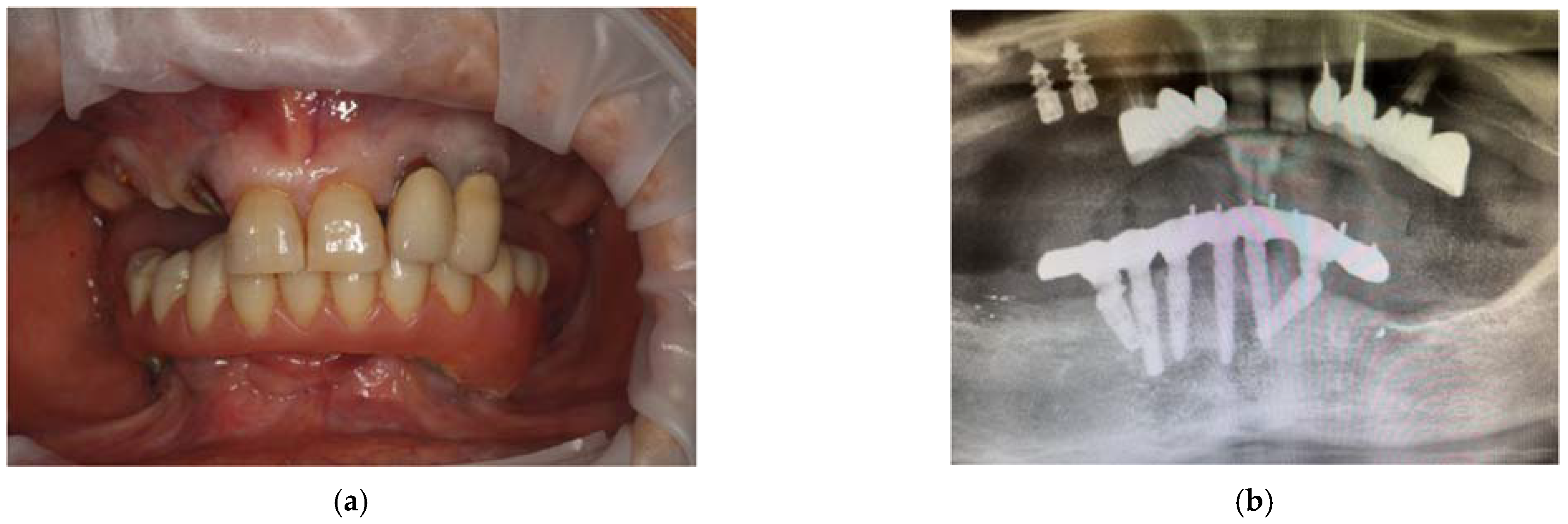
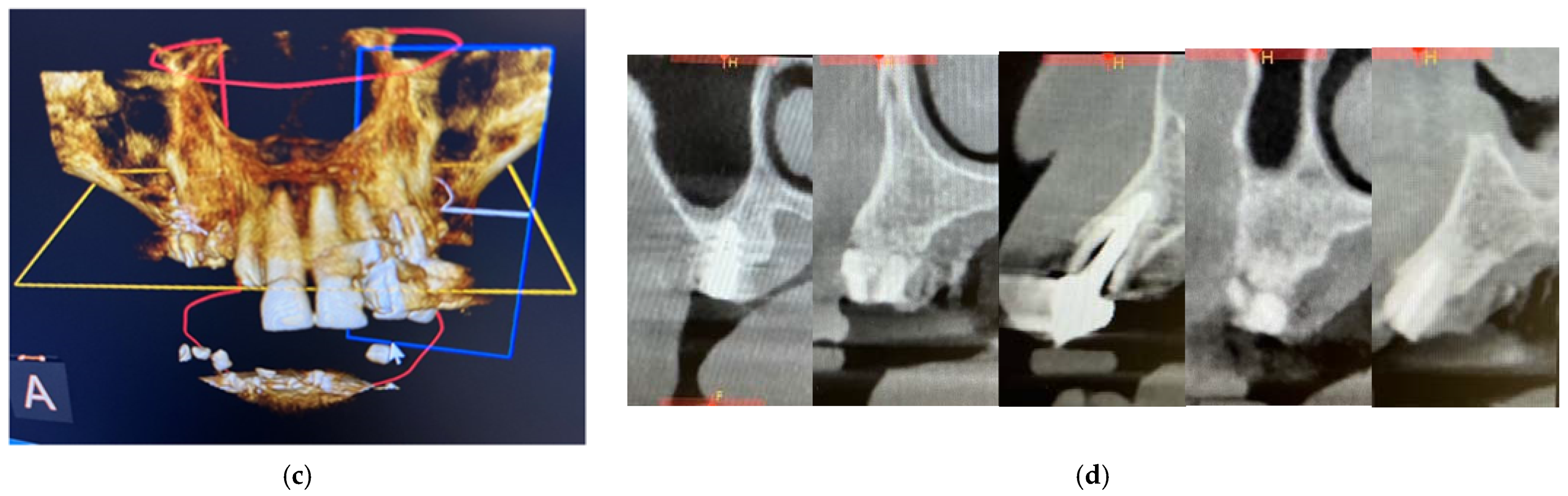
From the dental point of view, the patient had partial edentulism in the upper arch (first class of Kennedy) with the permanence of the elements 11, 21, 22 and 23 and the root residues of the elements 12, 13 and 14 and of partially osseointegrated implants in the area 15 and 16. (Figure 4a) In the lower arch, there was a fixed rehabilitation on the Toronto-Bridge-type implants. Orthopantomography and CBCT (Figure 4b–d) showed the remaining periodontally compromised dental elements, root residues and implants not supported by prosthetic crowns that had lost the vestibular cortex. There was also an inadequate thickness of the bone tissue in some areas of the arch; thus, we had to design and apply regenerative techniques to insert implants to support a fixed type of rehabilitation. The bone density was D3, according to the Misch classification, in almost all the areas where the implants were placed.
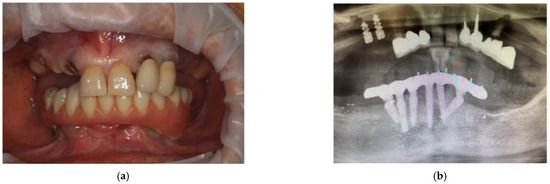
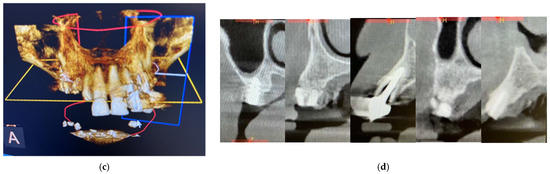
Figure 4.
Intraoral photos of the patient (a) and orthopantomography (b) and CBCT 3D reconstruction (c) and cross sections (d).
Considering the initial clinical situation, the patient’s request and her general health conditions, the extraction of the remaining dental elements, root residues and implants was proposed in the same operation, as was the subsequent use of the laser to erbium and the Magnetic Mallet for the preparation of the new implant sites and the insertion of eight implant fixtures. Within the next two days, the implants were loaded with a screwed-in temporary total prosthesis with an immediate load of the Toronto Bridge type. The patient, correctly informed, accepted and gave consent to the execution of the therapy.
Considering the age of the patient and the complexity of the operation, it was agreed with her to perform the operation in a conscious sedation regime, requiring the intervention of an anesthetist specialist.
Surgical Procedure
On the day of the surgery, the patient took an antibiotic prophylaxis based on Amoxicillin and clavulanic acid (875 mg + 125 mg) one hour before the start of the surgery.
Initially, complete anesthesia of the upper arch was performed on both the buccal and palatal sides, using, as the anesthetizing molecule, articaine with the addition of adrenaline at a concentration of 1: 100,000. A full-thickness flap was initially performed to clinically highlight the total residual bone volume of the superior alveolar process, root residues and implants.
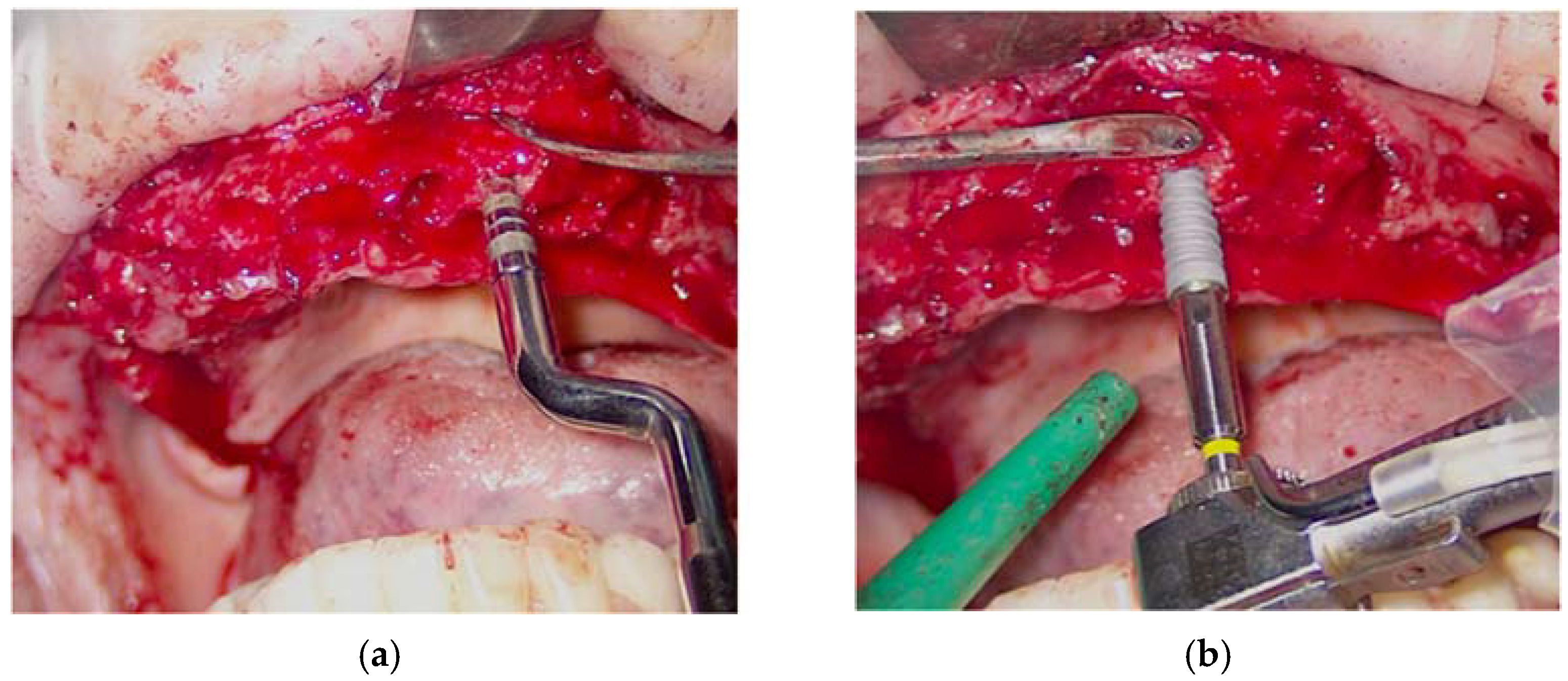
The simple extraction of the remaining dental elements was then performed. Instead, for the removal of root residues and implants, the Magnetic Mallet was used with the inserts for the extractions. This made it possible to avoid osteotomy and/or odontotomy maneuvers in order to save precious bone tissue for the subsequent insertion of the implants. Given the presence of infection and inflammatory tissue, photodynamic therapy (OHLLT protocol) (Figure 5a,b) was practiced in the post-extraction sockets by irrigation with a Sioxyl solution activated by a diode laser (parameters), to decontaminate in depth.
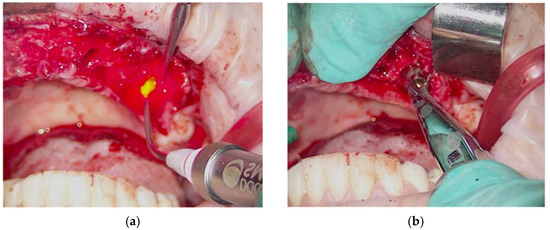
Figure 5.
Oxygen high level laser therapy, OHLLT (a); measurement of the post-extraction site (b).
Subsequently, for each of the eight implant sites, a series of steps were performed in order to insert the implants with sufficient primary stability and in the best prosthetic position to make the intervention as predictable but also as minimally invasive as possible. Initially, the erbium laser (160 mj, 20 Hz, with an average power of 3.2 W) was used to bleed the bone tissue and thus increase the regenerative potential. At the same time, the ablative capacity of the erbium laser allowed the creation of an access for the osteotomy inserts of the Magnetic Mallet. The implant sites, in fact, were prepared using only the Magnetic Mallet with osteotomic inserts of an increasing diameter, from 100 (apical diameter of 1 mm) to 300 (apical diameter of 3 mm) (Figure 6a). Therefore, no bone tissue was removed, but it was only compacted to increase its density and obtain greater primary stability. Before inserting the implants, the implant sites were in turn decontaminated by photodynamic therapy (OHLLT protocol). The eight implants were then inserted (Figure 6b) with a torque of 35 N in zone 17 and 24 (diameter 4, length 10), zone 14 (diameter 4, length 11), zone 21 and 23 (diameter 4, length 13), zone 11 and 13 (diameter 3, 7, length 13) and zone 26 (diameter 4.7, length 10). Finally, the healing screws were screwed to the implants and the flaps were sutured with a 4-0 silk thread after placing the PRF previously taken and they were centrifuged both in the extraction sites and to protect the flaps.
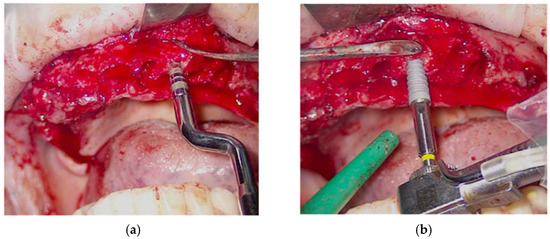
Figure 6.
Use of the Magnetic Mallet to prepare the implant site (a); implant insertion (b).
After the insertion of all the implants, the primary stability was measured with an implant dynamometer ratchet and it was found to be good, with values of about 35 N.
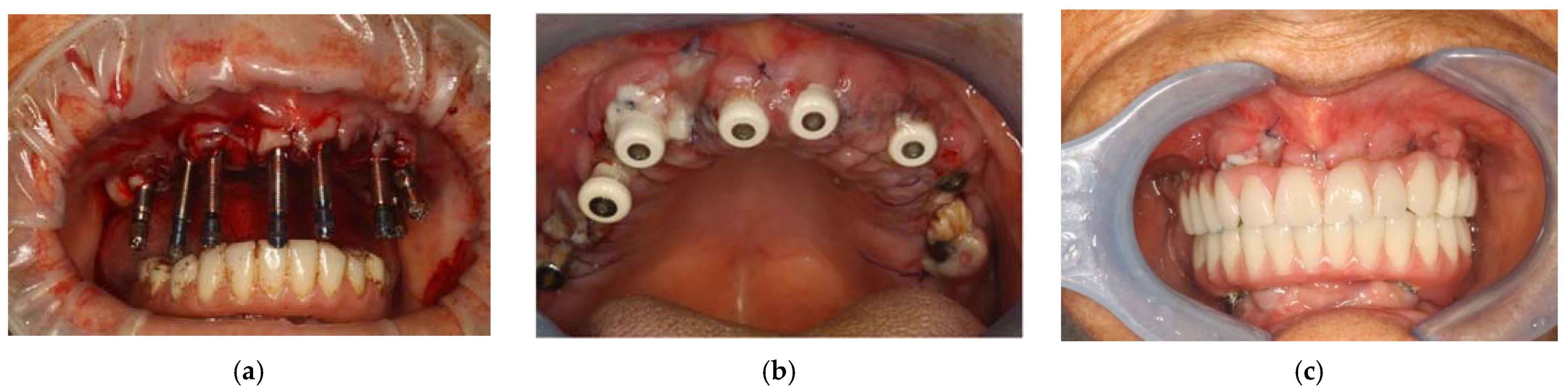
For the realization of the prosthetic product, the impressions were made. Above, by implant transfers, an impression was made with plaster mixed with fluid silicone, while at the bottom an alginate impression was made. (Figure 7a) Once the surgery was completed, the patient was given an anti-inflammatory drug based on naproxen sodium 550 mg (Synflex) to avoid the onset of pain. Before being discharged, the patient underwent a photobiomodulation session using the ATP38 (Biotech Dental, Salon-de-Provence, France) for 12 min.
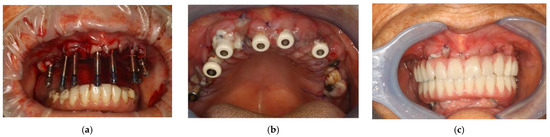
Figure 7.
Transfer on implants to make the impression (a); healing caps on implants (b); prosthesis with immediate loading after 48 h (c).
The following day, the patient returned to the office to perform a checkup, which included a further photobiomodulation session using ATP38 and a test of the new upper prosthesis, in which the passive fit of the rigid bar with the implants was checked with orthopantomography, as were the aesthetics and occlusion. (Figure 7b) The next day, the provisional upper prosthesis screwed with immediate loading was delivered. (Figure 7c)
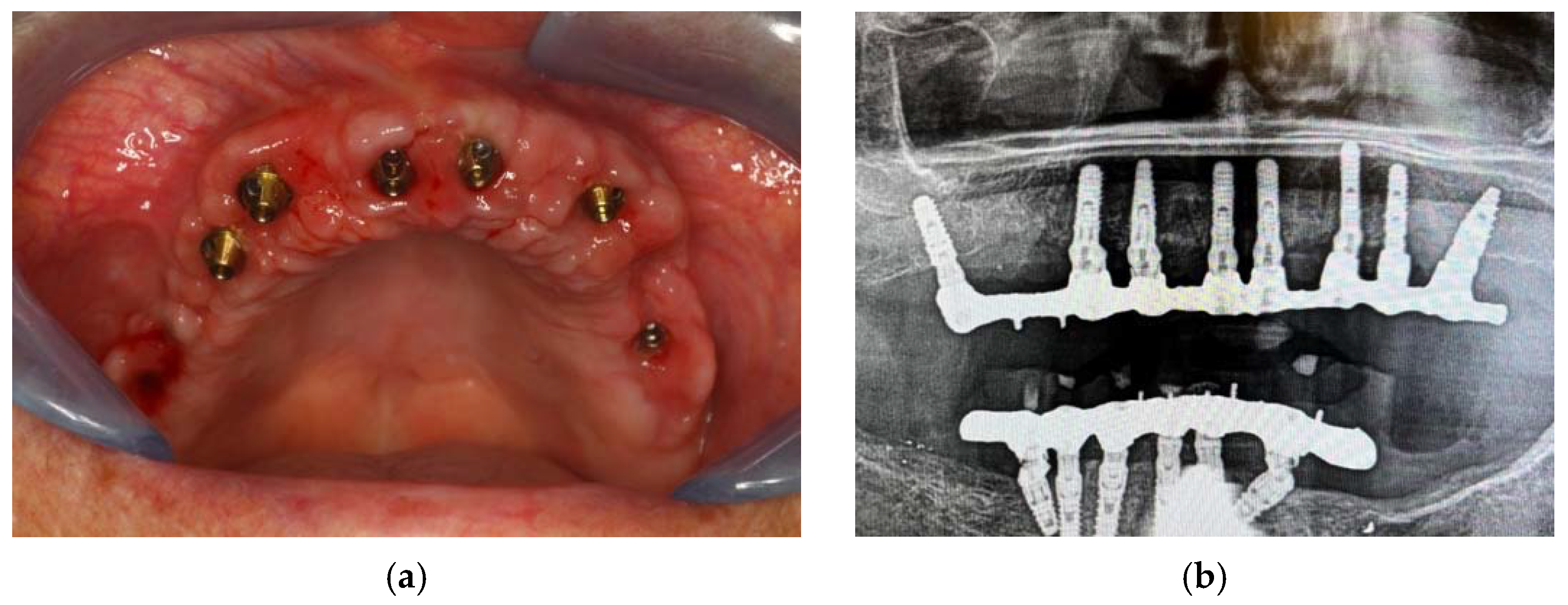
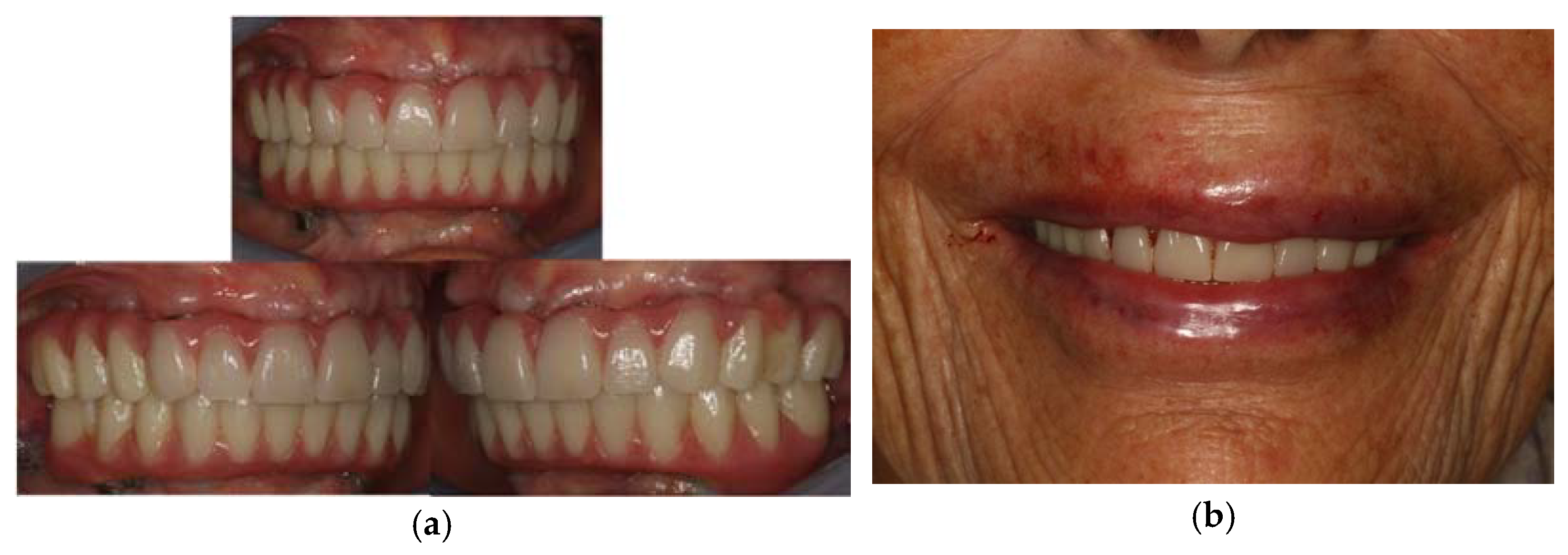
Finally, three weeks after the surgery, the sutures were removed, verifying that healing took place correctly. (Figure 8) The patient reported that she did not have any particular post-operative problems and complications; in particular, she reported that she did not take any anti-inflammatory drugs after surgery and that she was satisfied with the result obtained. For a further improvement from an aesthetic point of view, the patient also decided to perform a lip filler based on hyaluronic acid one month after the delivery of the prosthesis. (Figure 9)
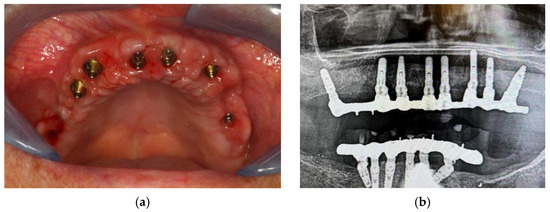
Figure 8.
Clinical (a) and radiographic (b) healing.
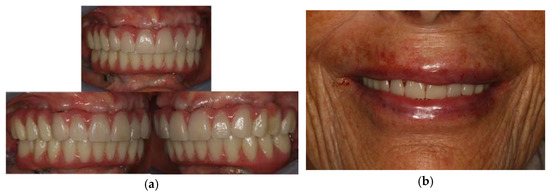
Figure 9.
Final result of the prosthesis: (a) intra-oral view; (b) extra-oral view.
3. Discussion
The rehabilitation of an arch through the extraction of compromised elements, bone regeneration and the insertion of implants is generally a procedure that requires surgery lasting several hours with a considerable level of trauma for the patient. It is therefore essential to be able to take advantage of new technological tools that lead to a simplification of the maneuvers performed during the surgery, making the latter faster and less traumatic, reducing the risk of intra- and post-operative complications. In this specific case, using the Magnetic Mallet and the laser made it possible to face the clinical case of a fragile patient in complete serenity with a reduction in risks and morbidity.
As for the Magnetic Mallet, having carried out the preparation of the implant sites by using only the osteotomic inserts allowed us to save bone tissue; in fact, it was only compacted to increase its density, and thus we did not have to use preparatory drills that could have created necrosis areas and a consequent, more severe post-operative procedure [26,27,28], with a higher risk of short- and long-term adverse post-operative consequences, namely peri-implantitis [29,30,31,32,33].
The use of the erbium laser allowed for more bone bleeding, resulting in better and faster healing, and the diode laser, on the other hand, allowed us to decontaminate the infected areas, due to the presence of compromised elements, and the implant sites in order to avoid any opportunistic infection that could have led to poor osseointegration [16,17,18,19,20,21,22,23,24,25]. Furthermore, the biostimulatory capacity of the diode laser and the photobiomodulation obtained with the use of ATP38 made it possible to obtain a biological “doping” for the tissues in order to accelerate healing and reduce post-operative pain [34,35,36]. The erbium laser, on the other hand, together with the combination of the Magnetic mallet made it possible to create a more precise bone breach than that created with rotating instruments, keeping the temperature of the bone tissue low thanks to the air–water spray.
4. Conclusions
The use of tools such as the Magnetic Mallet and the laser resulted in the implementation of a therapy, normally considered traumatic, in a minimally invasive manner. The patient had no pain or other complications during the surgery. She did not suffer from any episodes of agitation, although she herself stated that she had had it in the past after previous dental surgery. In the following days, she confirmed that she had not developed any edematous areas and that she had not had post-operative pain. She stated that she therefore did not need to take pain-relieving or anti-inflammatory drugs. In addition, it can be said that she did not have any infectious complications.
It can therefore be concluded that the combined use of the laser and the Magnetic Mallet made it possible to perform complex therapies even in this patient who is admittedly fragile due to clinical history and a present systemic condition. To the detriment of the past, in which these therapeutic paths were reserved for a narrow circle of people, this could allow clinicians to be able to guarantee optimal functional and aesthetic results for many fragile patients.
Author Contributions
Conceptualization, G.C.; data curation, P.C.; formal analysis, A.A.B.; investigation, G.C.; methodology, G.C.; project administration, M.B.; software, P.C.; supervision, M.B.; validation, P.C.; visualization, A.A.B.; writing—original draft, L.F.; writing—review and editing, P.C. and A.A.B. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
This study was conducted according to the guidelines of the Declaration of Helsinki and was approved by the Istituto Superiore di Sanità (Prot. 30/07/2007-0040488).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Cortellini, P.; Tonetti, M.S. Improved wound stability with a modified minimally invasive surgical technique in the regenerative treatment of isolated interdental intrabony defects. J. Clin. Periodontol. 2009, 36, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Aimetti, M.; Ferrarotti, F.; Mariani, G.M.; Romano, F. A novel flapless approach versus minimally invasive surgery in periodontal regeneration with enamel matrix derivative proteins: A 24-month randomized controlled clinical trial. Clin. Oral Investig. 2017, 21, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Aimetti, M.; Ferrarotti, F.; Bergandi, L.; Saksing, L.; Parducci, F.; Romano, F. Increase in periodontal interleukin-1β gene expression following osseous resective surgery using conventional rotary instruments compared with piezosurgery: A split-mouth randomized clinical trial. Int. J. Periodontics Restor. Dent. 2016, 36, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Schneider, L.F.; Barr, J.; Aarabi, S.; Chibbaro, P.; Grayson, B. Comparison of minimally invasive versus conventional open harvesting techniques for iliac bone graft in secondary alveolar cleft patients. Plast. Reconstr. Surg. 2011, 128, 485–491. [Google Scholar] [CrossRef]
- Novy, B.B.; Fuller, C.E. The material science of minimally invasive esthetic restorations. Compend. Contin. Educ. Dent. 2008, 29, 338–346. [Google Scholar]
- Vertuani, S.; Nilsson, J.; Borgman, B.; Buseghin, G.; Leonard, C.; Assietti, R.; Quraishi, N.A. A cost-effectiveness analysis of minimally invasive versus open surgery techniques for lumbar spinal fusion in Italy and the United Kingdom. Value Health 2015, 18, 810–816. [Google Scholar] [CrossRef]
- Bennardo, F.; Barone, S.; Vocaturo, C.; Nucci, L.; Antonelli, A.; Giudice, A. Usefulness of Magnetic Mallet in Oral Surgery and Implantology: A Systematic Review. Oral Maxillofac. Surg. 2014, 18, 59–64. [Google Scholar] [CrossRef]
- Menchini-Fabris, G.B.; Toti, P.; Crespi, G.; Covani, U.; Crespi, R. Distal Displacement of Maxillary Sinus Anterior Wall Versus Conventional Sinus Lift with Lateral Access: A 3-Year Retrospective Computerized Tomography Study. Int. J. Environ. Res. Public Health 2020, 17, 7199. [Google Scholar] [CrossRef]
- Schierano, G.; Baldi, D.; Peirone, B.; Mauthe von Degerfeld, M.; Navone, R.; Bragoni, A.; Colombo, J.; Autelli, R.; Muzio, G. Biomolecular, Histological, Clinical, and Radiological Analyses of Dental Implant Bone Sites Prepared Using Magnetic Mallet Technology: A Pilot Study in Animals. Materials 2021, 14, 6945. [Google Scholar] [CrossRef]
- Crespi, R.; Capparè, P.; Gherlone, E. Sinus floor elevation by osteotome: Hand mallet versus electric mallet. A prospective clinical study. Int. J. Oral Maxillofac. Implant. 2012, 27, 1144–1150. [Google Scholar] [PubMed]
- Crespi, R.; Capparè, P.; Gherlone, E.F. Electrical mallet in implants placed in fresh extraction sockets with simultaneous osteotome sinus floor elevation. Int. J. Oral Maxillofac. Implant. 2013, 28, 869–874. [Google Scholar] [CrossRef] [PubMed]
- Crespi, R.; Capparè, P.; Gherlone, E. Electrical Mallet Provides Essential Advantages in Maxillary Bone Condensing. A Prospective Clinical Study. Clin. Implant. Dent. Relat. Res. 2012, 15, 874–882. [Google Scholar] [CrossRef] [PubMed]
- Desai, A.; Patil, S.; Mitra, D.; Shah, R. Magnetic Mallet-Feel the Future. JIDA J. Indian Dent. Assoc. 2020, 14, 26–30. [Google Scholar] [CrossRef]
- Crespi, R.; Capparè, P.; Gherlone, E. A comparison of manual and electrical mallet in maxillary bone condensing for immediately loaded implants: A randomized study. Clin. Implant. Dent. Relat. Res. 2014, 16, 374–382. [Google Scholar] [CrossRef] [PubMed]
- Maggioni, M.; Attanasio, T.; Scarpelli, F. I Laser in Odontoiatria; Piccin: Padova, Italy, 2009. [Google Scholar]
- Caccianiga, G.; Rey, G.; Baldoni, M.; Paiusco, A. Clinical, Radiographic and Microbiological Evaluation of High Level Laser Therapy, a New Photodynamic Therapy Protocol, in Peri-Implantitis Treatment; A Pilot Experience. Biomed Res. Int. 2016, 2016, 6321906. [Google Scholar] [CrossRef]
- Caccianiga, G.; Baldoni, M.; Ghisalberti, C.A.; Paiusco, A. A Preliminary In Vitro Study on the Efficacy of High-Power Photodynamic Therapy (HLLT): Comparison between Pulsed Diode Lasers and Superpulsed Diode Lasers and Impact of Hydrogen Peroxide with Controlled Stabilization. Biomed Res. Int. 2016, 2016, 1386158. [Google Scholar] [CrossRef]
- Caccianiga, G.; Rey, G.; Paiusco, A.; Lauritano, D.; Cura, F.; Ormianer, Z.; Carinci, F. Oxygen high level laser therapy is efficient in treatment of chronic periodontitis: A clinical and microbiological study using PCR analysis. J. Biol. Regul. Homeost. Agents 2016, 30, 87–97. [Google Scholar] [PubMed]
- Caccianiga, G.; Cambini, A.; Donzelli, E.; Baldoni, M.; Rey, G.; Paiusco, A. Effects of laser biostimulation on the epithelial tissue for keratinized layer differentiation: An in vitro study. J. Biol. Regul. Homeost. Agents 2016, 30, 99–105. [Google Scholar] [PubMed]
- Leonida, A.; Paiusco, A.; Rossi, G.; Carini, F.; Baldoni, M.G.; Caccianiga, G. Effects of low-level laser irradiation on proliferation and osteoblastic differentiation of human mesenchymal stem cells seeded on a three-dimensional biomatrix: In vitro pilot study. Lasers Med. Sci. 2013, 28, 125–132. [Google Scholar] [CrossRef]
- Caccianiga, G.; Rey, G.; Baldoni, M.; Caccianiga, P.; Baldoni, A.; Ceraulo, S. Periodontal Decontamination Induced by Light and Not by Heat: Comparison between Oxygen High Level Laser Therapy (OHLLT) and LANAP. Appl. Sci. 2021, 11, 4629. [Google Scholar] [CrossRef]
- Caccianiga, G.; Rey, G.; Caccianiga, P.; Leonida, A.; Baldoni, M.; Baldoni, A.; Ceraulo, S. Rough Dental Implant Surfaces and Peri-Implantitis: Role of Phase- Contrast Microscopy, Laser Protocols, and Modified Home Oral Hygiene in Maintenance. A 10-Year Retrospective Study. Appl. Sci. 2021, 11, 4985. [Google Scholar] [CrossRef]
- Caccianiga, G.; Rey, G.; Caccianiga, P.; Leonida, A.; Baldoni, M.; Baldoni, A.; Ceraulo, S. Peri-Implantitis Management: Surgical versus Non-Surgical Approach Using Photodynamic Therapy Combined with Hydrogen Peroxide (OHLLT—Oxygen High Level Laser Therapy). A Retrospective Controlled Study. Appl. Sci. 2021, 11, 5073. [Google Scholar] [CrossRef]
- Caccianiga, G.; Rey, G.; Caccianiga, P.; Leonida, A.; Baldoni, M.; Baldoni, A.; Ceraulo, S. Laser Management of Peri-Implantitis: A Comparison between Photodynamic Therapy Combined with Hydrogen Peroxide (OHLLT) and OHLLT + Er:YAG Laser. A Retrospective Controlled Study. Appl. Sci. 2021, 11, 6771. [Google Scholar] [CrossRef]
- Seka, W.; Featherstone, J.D.B.; Fried, D.; Visuri, S.R.; Walsh, J.T. Laser ablation of dental hard tissues from explosive ablation to plasma mediate ablation. In Lasers in Dentistry II.; SPIE: Bellingham, WA, USA, 1996; Volume 2672, pp. 144–158. [Google Scholar]
- Adell, R.; Lekholm, U.; Rockler, B.R.; Brånemark, P.I. A study of osseointegrated implants in the treatment of the edentulous jaws. Int. J. Oral Surg. 1981, 6, 387–399. [Google Scholar] [CrossRef]
- Buser, D.; Martin, W.; Belser, U.C. Optimizing esthetics for implant restorations in the anterior maxilla: Anatomic and surgical considerations. Int. J. Oral Maxillofac. Implant. 2004, 19, 43–61. [Google Scholar]
- Evans, C.D.; Chen, S.T. Esthetic outcomes of immediate implant placements. Clin. Oral Implant. Res. 2008, 19, 73–80. [Google Scholar] [CrossRef]
- Alsaadi, G.; Quirynen, M.; Komárek, A.; Van Steenberghe, D. Impact of local and systemic factors on the incidence of late oral implant loss. Clin. Oral Implant. Res. 2008, 19, 670–676. [Google Scholar]
- Berglundh, T.; Lindhe, J. Dimension of the periimplant mucosa. Biological width revisited. J Clin Periodontol. 1996, 23, 971–973. [Google Scholar] [CrossRef] [PubMed]
- Brägger, U.; Karoussis, I.; Persson, R.; Pjetursson, B.; Salvi, G.; Lang, N. Technical and biological complications/failures with single crowns and fixed partial dentures on implants: A 10-year prospective cohort study. Clin. Oral Implants Res. 2005, 16, 326. [Google Scholar] [CrossRef] [PubMed]
- Etter, T.H.; Håkanson, I.; Lang, N.P.; Trejo, P.M.; Caffesse, R.G. Healing after standardized clinical probing of the perlimplant soft tissue seal: A histomorphometric study in dogs. Clin Oral Implants Res. 2002, 13, 571–580. [Google Scholar] [CrossRef] [PubMed]
- Evian, C.I.; Emling, R.; Rosenberg, E.S.; Waasdorp, J.A.; Halpern, W.; Shah, S.; Garcia, M. Retrospective analysis of implant survival and the influence of periodontal disease and immediate placement on long-term results. Int. J. Oral Maxillofac. Implant. 2004, 19, 393–398. [Google Scholar]
- Caccianiga, G.; Caccianiga, P.; Baldoni, M.; Lo Giudice, A.; Perillo, L.; Moretti, N.; Ceraulo, S. Pain Reduction during Rapid Palatal Expansion Due to LED Photobiomodulation Irradiation: A Randomized Clinical Trial. Life 2022, 12, 37. [Google Scholar] [CrossRef]
- Lo Giudice, A.; Nucera, R.; Leonardi, R.; Paiusco, A.; Baldoni, M.; Caccianiga, G. A Comparative Assessment of the Efficiency of Orthodontic Treatment With and Without Photobiomodulation During Mandibular Decrowding in Young Subjects: A Single-Center, Single-Blind Randomized Controlled Trial. Photobiomodulation Photomed. Laser Surg. 2020, 38, 272–279. [Google Scholar] [CrossRef]
- Caccianiga, G.; Rey, G.; Baldoni, M.; Caccianiga, P.; Porcaro, G.; Baldoni, A.; Ceraulo, S. Laser Decontamination and LED Photobiomodulation Promote Bone Regeneration and Wound Healing by Secondary Intention, in Alveolar Ridge Preservation-Clinical and Radiographic Evaluation: A Pilot Experience. Photobiomodulation Photomed. Laser Surg. 2022, 40, 343–354. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).