Examining the Effects of Induced Plant Defenses on Spodoptera frugiperda Performance
Abstract
:1. Introduction
Background and Study System
2. Materials and Methods
2.1. Plant Cultivars and Propagation
2.2. Experimental Procedure
2.3. Statistical Analysis
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bueno, R.C.O.F.; Bueno, A.F.; Moscardi, F.; Parra, J.R.P.; Hoffmann-Campo, C.B. Lepidopteran larva consumption of soybean foliage: Basis for developing multiple-species economic thresholds for pest management decisions. Pest Manag. Sci. 2011, 67, 170–174. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bueno, A.F.; Paula-Moraes, S.V.; Gazzoni, D.L.; Pomari, A.F. Economic thresholds in soybean-integrated pest management: Old concepts, current adoption, and adequacy. Neotrop. Entomol. 2013, 42, 439–447. [Google Scholar] [CrossRef] [PubMed]
- Bennett, R.N.; Wallsgrove, R.M. Secondary Metabolites in Plant Defense-Mechanisms. New Phytol. 1994, 127, 617–633. [Google Scholar] [CrossRef]
- Gatehouse, J.A. Plant resistance towards insect herbivores: A dynamic interaction. New Phytol. 2002, 156, 145–169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Awmack, C.S.; Leather, S.R. Host plant quality and fecundity in herbivorous insects. Annu. Rev. Entomol. 2002, 47, 817–844. [Google Scholar] [CrossRef]
- Gordy, J.W.; Leonard, B.R.; Blouin, D.; Davis, J.A.; Stout, M.J. Comparative effectiveness of potential elicitors of plant resistance against Spodoptera frugiperda (JE Smith) (Lepidoptera: Noctuidae) in four crop plants. PLoS ONE 2015, 10, e0136689. [Google Scholar] [CrossRef] [Green Version]
- Gaillard, M.D.; Glauser, G.; Robert, C.A.; Turlings, T.C. Fine-tuning the ‘plant domestication-reduced defense’ hypothesis: Specialist vs. generalist herbivores. New Phytol. 2018, 217, 355–366. [Google Scholar] [CrossRef] [Green Version]
- Cruz, I.; Figueiredo, M.L.C.; Oliveira, A.C.; Vasconcelos, C.A. Damage of Spodoptera frugiperda (Smith) in different maize genotypes cultivated in soil under three levels of aluminium saturation. Int. J. Pest Manag. 1999, 45, 293–296. [Google Scholar] [CrossRef]
- Morales, X.C.; Tamiru, A.; Sobhy, I.S.; Bruce, T.J.; Midega, C.A.; Khan, Z. Evaluation of African Maize Cultivars for Resistance to Fall Armyworm Spodoptera frugiperda (JE Smith) (Lepidoptera: Noctuidae) Larvae. Plants 2021, 10, 392. [Google Scholar] [CrossRef]
- Shikano, I.; McCarthy, E.; Hayes-Plazolles, N.; Slavicek, J.M.; Hoover, K. Jasmonic acid-induced plant defenses delay caterpillar developmental resistance to a baculovirus: Slow-growth, high-mortality hypothesis in plant–insect–pathogen interactions. J. Invertebr. Pathol. 2018, 158, 16–23. [Google Scholar] [CrossRef]
- Shikano, I.; McCarthy, E.M.; Elderd, B.D.; Hoover, K. Plant genotype and induced defenses affect the productivity of an insect-killing obligate viral pathogen. J. Invertebr. Pathol. 2017, 148, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Elderd, B.D. Bottom-up trait-mediated indirect effects decrease pathogen transmission in a tritrophic system. Ecology 2019, 100, e02551. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Escribano, A.; Williams, T.; Goulson, D.; Cave, R.D.; Chapman, J.W.; Caballero, P. Selection of a nucleopolyhedrovirus for control of Spodoptera frugiperda (Lepidoptera: Noctuidae): Structural, genetic, and biological comparison of four isolates from the Americas. J. Econ. Entomol 1999, 92, 1079–1085. [Google Scholar] [CrossRef] [PubMed]
- Moscardi, F. Assessment of the application of baculoviruses for control of Lepidoptera. Annu. Rev. Entomol. 1999, 44, 257–289. [Google Scholar] [CrossRef] [PubMed]
- Luginbill, P. The Fall Army Worm; US Department of Agriculture: Washington, DC, USA, 1928; Volume 34.
- Sparks, A.N. A review of the biology of the fall armyworm. Fla. Entomol. 1979, 62, 82–87. [Google Scholar] [CrossRef]
- Goergen, G.; Kumar, P.L.; Sankung, S.B.; Togola, A.; Tamò, M. First report of outbreaks of the fall armyworm Spodoptera frugiperda (J E Smith) (Lepidoptera, Noctuidae), a new alien invasive pest in west and central Africa. PLoS ONE 2016, 11, e0165632. [Google Scholar] [CrossRef] [Green Version]
- Wan, J.; Huang, C.; Li, C.-Y.; Zhou, H.-X.; Ren, Y.-L.; Li, Z.-Y.; Xing, L.-S.; Zhang, B.; Qiao, X.; Liu, B.; et al. Biology, invasion and management of the agricultural invader: Fall armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae). J. Integr. Agric. 2021, 20, 646–663. [Google Scholar] [CrossRef]
- Pitre, H.N.; Mulrooney, J.E.; Hogg, D.B. Fall armyworm (Lepidoptera: Noctuidae) oviposition: Crop preferences and egg distribution on plants. J. Econ. Entomol. 1983, 76, 463–466. [Google Scholar] [CrossRef]
- Levine, E.; Spencer, J.L.; Isard, S.A.; Onstad, D.W.; Gray, M.E. Adaptation of the western corn rootworm to crop rotation: Evolution of a new strain in response to management practice. Environ. Entomol. 2002, 48, 94–107. [Google Scholar] [CrossRef] [Green Version]
- Peruca, R.D.; Coelho, R.G.; Silva, G.G.; Pistori, H.; Ravaglia, L.M.; Roel, A.R.; Alcantara, G.B. Impacts of soybean-induced defenses on Spodoptera frugiperda (Lepidoptera: Noctuidae) development. Arthropod-Plant Interact. 2018, 12, 257–266. [Google Scholar] [CrossRef]
- Raffa, K.F. Effect of host plant on cannibalism rates by fall armyworm (Lepidoptera: Noctuidae) larvae. Environ. Entomol. 1987, 16, 672–675. [Google Scholar] [CrossRef]
- Chapman, J.W.; Williams, T.; Escribano, A.; Caballero, P.; Cave, R.D.; Goulson, D. Fitness consequences of cannibalism in the fall armyworm, Spodoptera frugiperda. Behav. Ecol. 1999, 10, 298–303. [Google Scholar] [CrossRef] [Green Version]
- Shikano, I.; Shumaker, K.L.; Peiffer, M.; Felton, G.W.; Hoover, K. Plant-mediated effects on an insect–pathogen interaction vary with intraspecific genetic variation in plant defences. Oecologia 2017, 183, 1121–1134. [Google Scholar] [CrossRef] [PubMed]
- Lanka, S.K.; Elderd, B.D.; Davis, J.A.; Stout, M.J. Jasmonic acid-induced resistance to fall armyworm in soybeans: Variation among genotypes and tradeoffs with constitutive resistance. Basic. Appl. Ecol. 2021, 56, 97–109. [Google Scholar] [CrossRef]
- Underwood, N.; Rausher, M.; Cook, W. Bioassay versus chemical assay: Measuring the impact of induced and constitutive resistance on herbivores in the field. Oecologia 2002, 131, 211–219. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013; Available online: http://www.R-project.org/ (accessed on 23 February 2022).
- Yu-Sung, S.; Yajima, M. R2jags: Using R to Run ‘JAGS’, R Package Version 0.6-1. 2020. Available online: https://CRAN.R-project.org/package=R2jags (accessed on 23 February 2022).
- Vehtari, A.; Gabry, J.; Magnusson, M.; Yao, Y.; Bürkner, P.; Paananen, T.; Gelman, A. loo: Efficient Leave-One-Out Cross-Validation and WAIC for Bayesian Models, R Package Version 2.4.1. 2020. Available online: https://mc-stan.org/loo/ (accessed on 23 February 2022).
- Adler, D.; Kelly, S.T. vioplot: Violin Plot, R Package Version 0.3.6. 2021. Available online: https://github.com/TomKellyGenetics/vioplot (accessed on 23 February 2022).
- Yamane, H.; Konno, K.; Sabelis, M.; Takabayashi, J.; Sassa, T.; Oikawa, H. Chemical defence and toxins of plants. In Comprehensive Natural Products II; Liu, H.W., Mander, L., Eds.; Elsevier: Amsterdam, The Netherlands, 2010; Volume 4, pp. 339–385. [Google Scholar]
- Levin, D.A. The role of trichomes in plant defense. Q. Rev. Biol. 1973, 48, 3–15. [Google Scholar] [CrossRef]
- Broadway, R.M.; Duffey, S.S. Plant proteinase inhibitors: Mechanism of action and effect on the growth and digestive physiology of larval Heliothis zea and Spodoptera exiqua. J. Insect Physiol. 1986, 32, 827–833. [Google Scholar] [CrossRef]
- Jongsma, M.A.; Bakker, P.L.; Peters, J.; Bosch, D.; Stiekema, W.J. Adaptation of Spodoptera exigua larvae to plant proteinase inhibitors by induction of gut proteinase activity insensitive to inhibition. Proc. Natl. Acad. Sci. USA 1995, 92, 8041–8045. [Google Scholar] [CrossRef] [Green Version]
- Mohamed, H.I.; Mohammed, A.H.M.; Mohamed, N.M.; Ashry, N.A.; Zaky, L.M.; Mogazy, A.M. Comparative Effectiveness of Potential Elicitors of Soybean Plant Resistance against Spodoptera Littoralis and their Effects on Secondary Metabolites and Antioxidant Defense System. Gesunde Pflanz. 2021, 73, 273–285. [Google Scholar] [CrossRef]
- Bi, J.L.; Felton, G.W.; Mueller, A.J. Induced resistance in soybean to Helicoverpa zea: Role of plant protein quality. J. Chem. Ecol. 1994, 20, 183–198. [Google Scholar] [CrossRef]
- Nijhout, H.F.; Riddiford, L.M.; Mirth, C.; Shingleton, A.W.; Suzuki, Y.; Callier, V. The developmental control of size in insects. Wiley Interdiscip. Rev. Dev. Biol. 2014, 3, 113–134. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.L.; Xue, F.S.; Chen, C.; Guo, X.; Tang, J.J.; Zhong, L.; He, H.M. Effects of temperature on life-history traits of the newly invasive fall armyworm, Spodoptera frugiperda in Southeast China. Ecol. Evol. 2021, 11, 5255–5264. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.W.; Chen, Y. Slow-growth high-mortality: A meta-analysis for insects. Insect Sci. 2018, 25, 337–351. [Google Scholar] [CrossRef] [PubMed]
- Cory, J.S.; Hoover, K. Plant-mediated effects in insect–pathogen interactions. Trends Ecol. Evol. 2006, 21, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Issa, P.P.; Garvey, M.; Grimmell, S.; Pantha, P.; Dassanayake, M.; Elderd, B.D. Hitching a Ride: Examining the Ability of a Specialist Baculovirus to Translocate through Its Insect Host’s Food Plant. Pathogens 2021, 10, 1500. [Google Scholar] [CrossRef]
- Flick, A.J.; Acevedo, M.A.; Elderd, B.D. The negative effects of pathogen-infected prey on predators: A meta-analysis. Oikos 2016, 125, 1554–1560. [Google Scholar] [CrossRef]
- Flick, A.J.; Coudron, T.A.; Elderd, B.D. Intraguild predation decreases predator fitness with potentially varying effects on pathogen transmission in a herbivore host. Oecologia 2020, 193, 789–799. [Google Scholar] [CrossRef]
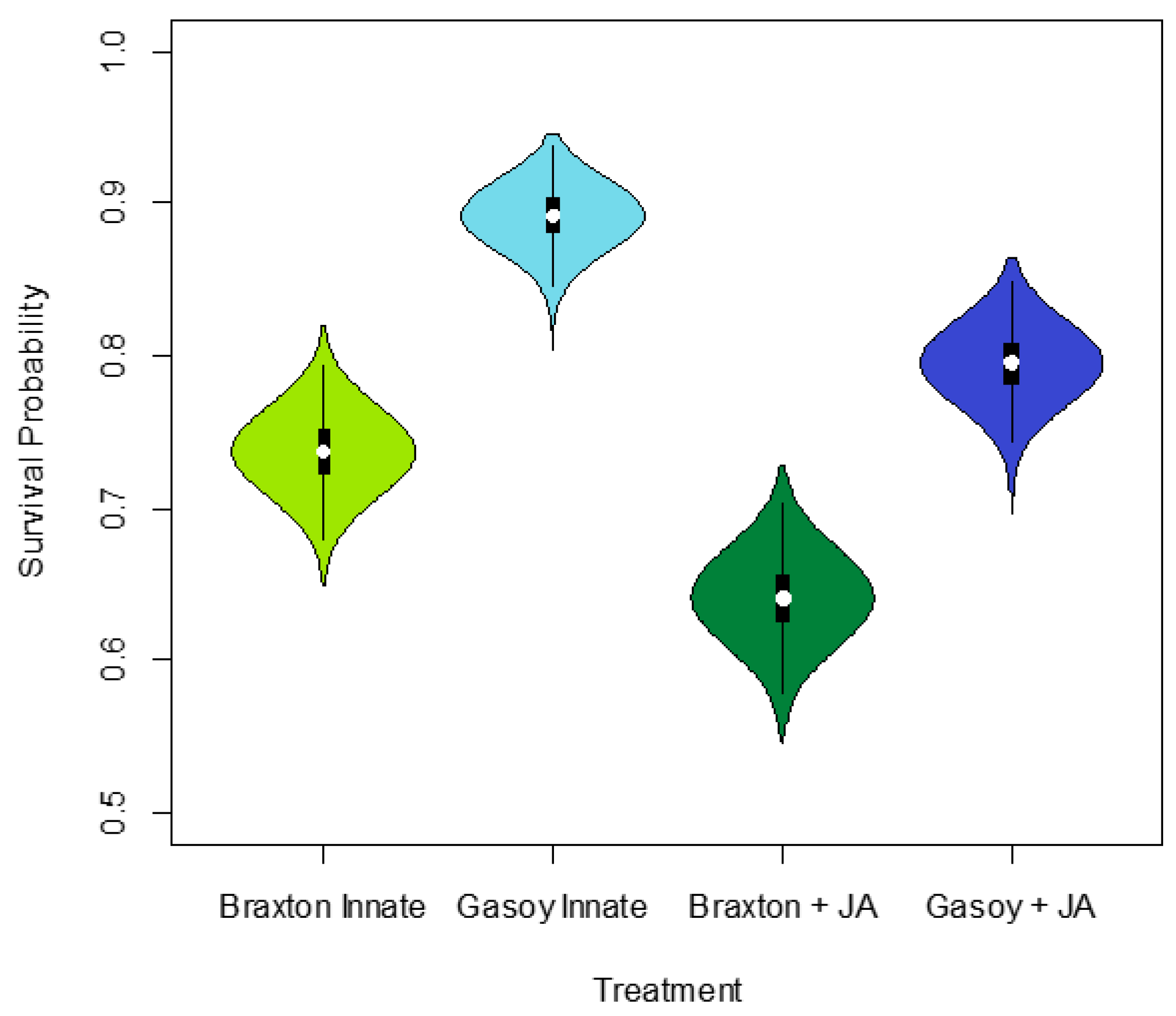
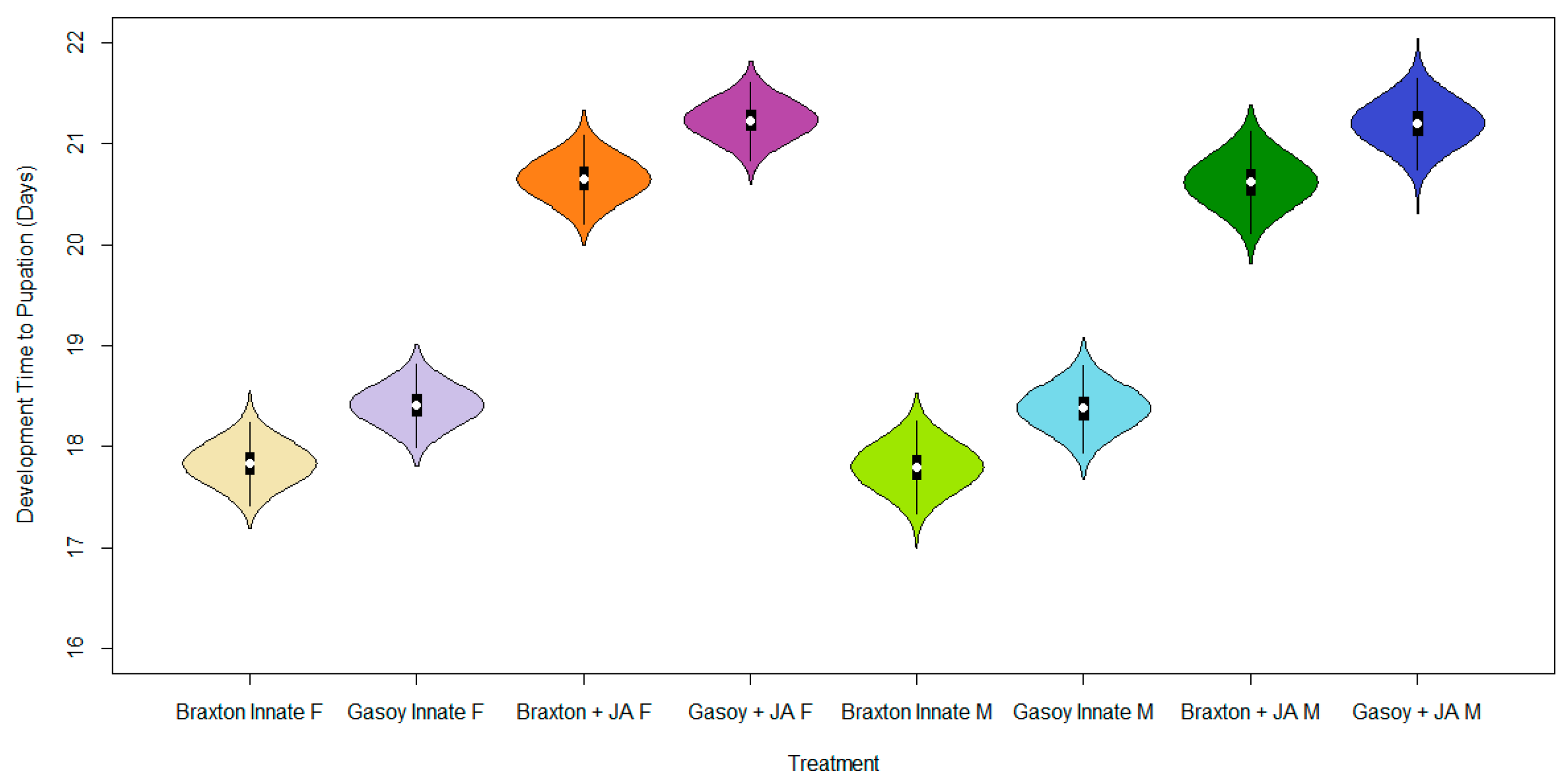
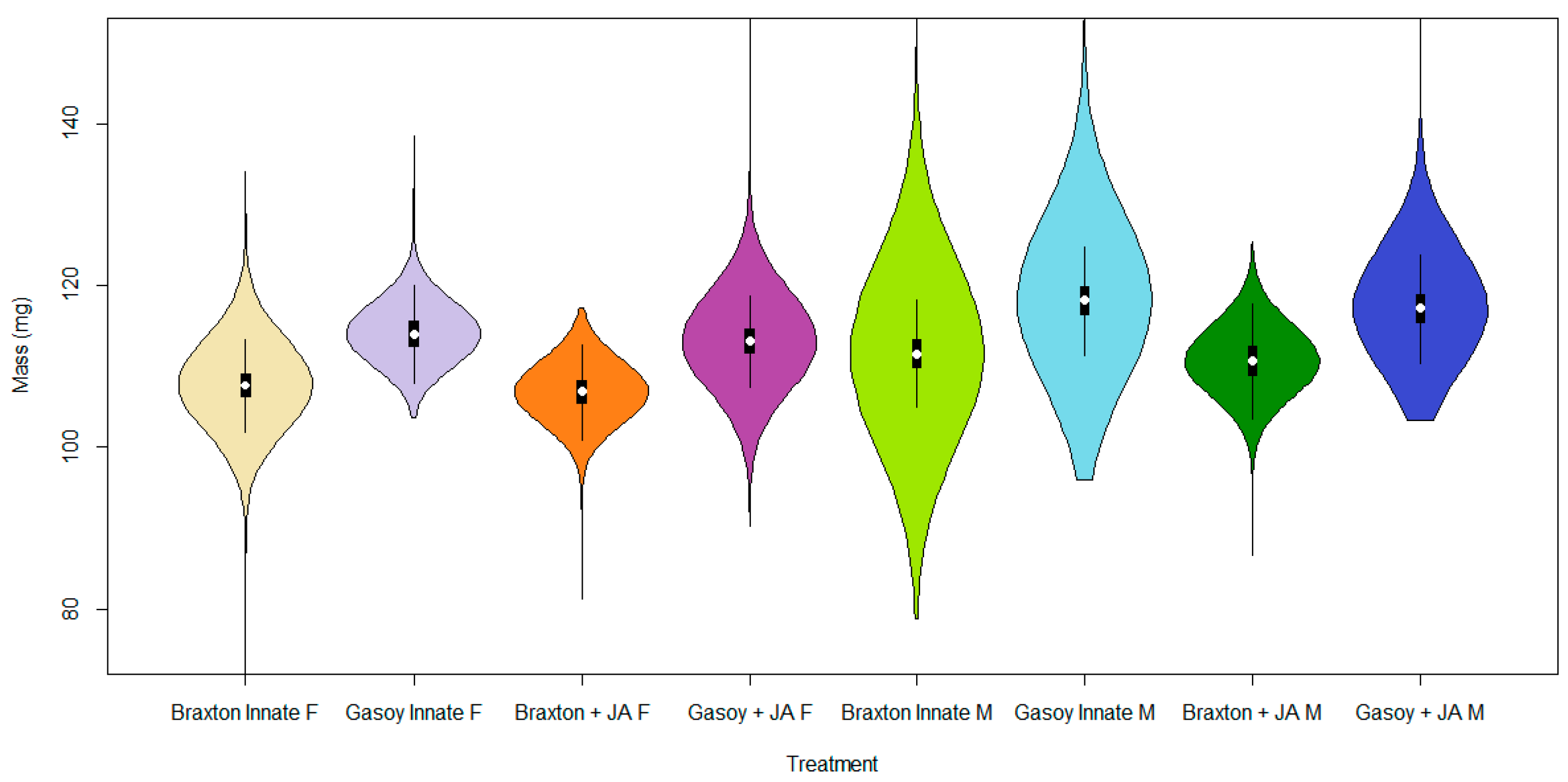
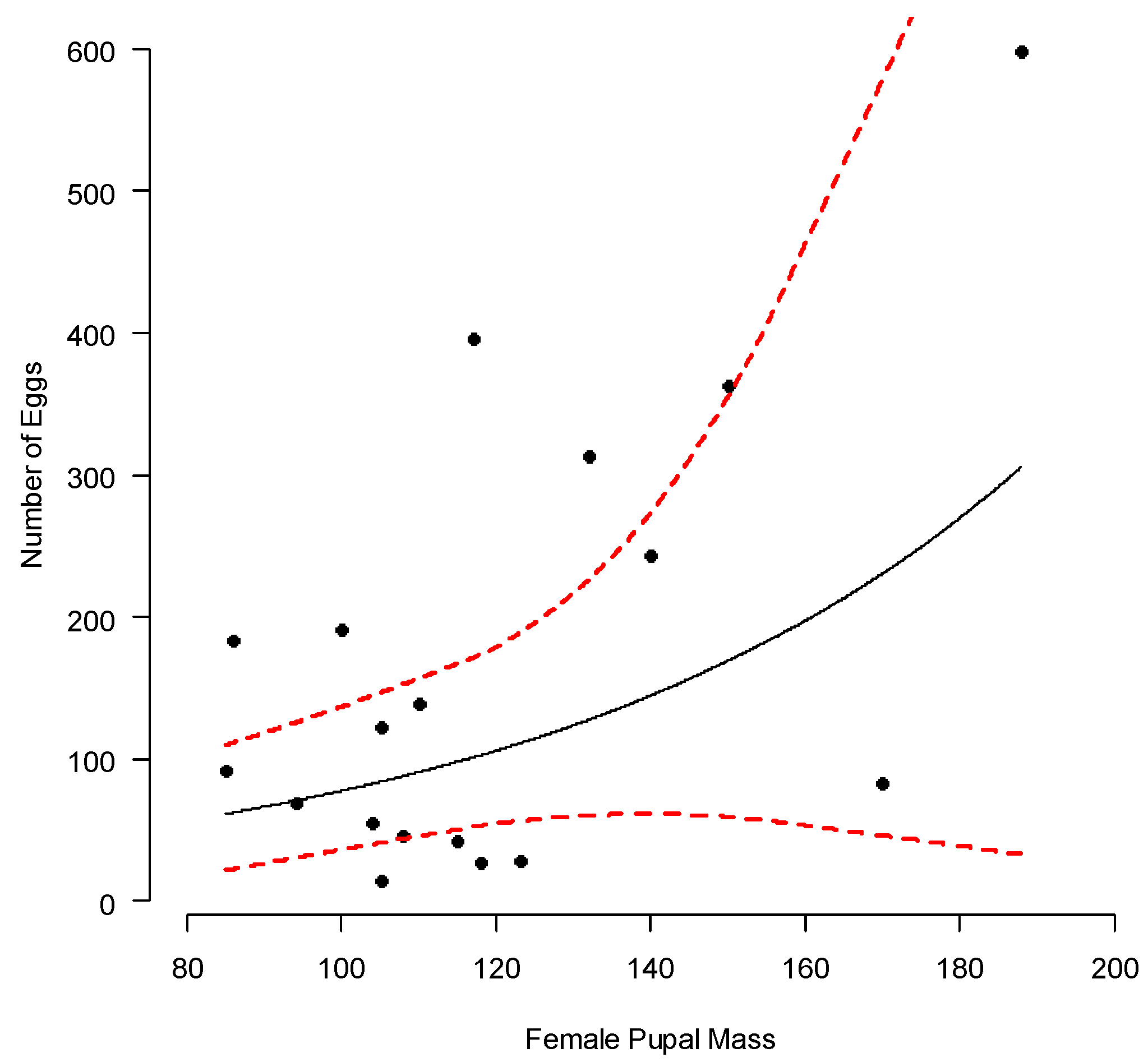
| Treatment Group | Mean | S.D. | 2.5% C.I. | 25% C.I. | 50% C.I. | 75% C.I. | 97.5% C.I. | N. eff | |
|---|---|---|---|---|---|---|---|---|---|
| Braxton Innate | 1.034 | 0.107 | 0.827 | 0.962 | 1.032 | 1.105 | 1.249 | 1.001 | 27,000 |
| Gasoy Innate | 2.117 | 0.175 | 1.783 | 1.997 | 2.114 | 2.233 | 2.471 | 1.001 | 27,000 |
| Braxton + JA | 0.581 | 0.102 | 0.383 | 0.512 | 0.581 | 0.650 | 0.782 | 1.001 | 13,000 |
| Gasoy | 1.358 | 0.119 | 1.127 | 1.276 | 1.356 | 1.437 | 1.598 | 1.002 | 3200 |
| Treatment Group | Mean | S.D. | 2.5% C.I. | 25% C.I. | 50% C.I. | 75% C.I. | 97.5% C.I. | N. eff | |
|---|---|---|---|---|---|---|---|---|---|
| Intercept | 17.827 | 0.157 | 17.519 | 17.722 | 17.826 | 17.932 | 18.136 | 1.001 | 45,000 |
| Cultivar | 0.580 | 0.164 | 0.261 | 0.467 | 0.579 | 0.691 | 0.901 | 1.001 | 45,000 |
| +JA | 2.824 | 0.164 | 2.503 | 2.714 | 2.823 | 2.935 | 3.144 | 1.001 | 45,000 |
| Sex | −0.030 | 0.166 | −0.355 | −0.143 | −0.030 | 0.082 | 0.294 | 1.001 | 45,000 |
| Treatment Group | Mean | S.D. | 2.5% C.I. | 25% C.I. | 50% C.I. | 75% C.I. | 97.5% C.I. | N. eff | |
|---|---|---|---|---|---|---|---|---|---|
| Intercept | 2.032 | 0.009 | 2.015 | 2.026 | 2.032 | 2.038 | 2.050 | 1.001 | 110,000 |
| Cultivar | 0.025 | 0.009 | 0.007 | 0.019 | 0.025 | 0.013 | 0.043 | 1.001 | 140,000 |
| +JA | −0.004 | 0.009 | −0.022 | −0.010 | −0.004 | 0.003 | 0.014 | 1.001 | 43,000 |
| Sex | 0.015 | 0.009 | −0.003 | 0.009 | 0.015 | 0.022 | 0.034 | 1.001 | 100,000 |
| Parameter | Mean | S.D. | 2.5% C.I. | 25% C.I. | 50% C.I. | 75% C.I. | 97.5% C.I. | N. eff | |
|---|---|---|---|---|---|---|---|---|---|
| Intercept | 2.793 | 1.175 | 0.452 | 2.036 | 2.794 | 3.539 | 5.126 | 1.001 | 13,000 |
| Slope | 0.016 | 0.010 | −0.004 | 0.009 | 0.016 | 0.022 | 0.035 | 1.001 | 12,000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garvey, M.; Costanza, K.; Grimmell, S.; Elderd, B.D. Examining the Effects of Induced Plant Defenses on Spodoptera frugiperda Performance. Appl. Sci. 2022, 12, 3907. https://doi.org/10.3390/app12083907
Garvey M, Costanza K, Grimmell S, Elderd BD. Examining the Effects of Induced Plant Defenses on Spodoptera frugiperda Performance. Applied Sciences. 2022; 12(8):3907. https://doi.org/10.3390/app12083907
Chicago/Turabian StyleGarvey, Michael, Kale Costanza, Scott Grimmell, and Bret D. Elderd. 2022. "Examining the Effects of Induced Plant Defenses on Spodoptera frugiperda Performance" Applied Sciences 12, no. 8: 3907. https://doi.org/10.3390/app12083907






