Anti-Obesity Effect of Fresh and Browned Magnolia denudata Flowers in 3T3-L1 Adipocytes
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Preparation of Magnolia denudata Flower
2.3. Cell Culture Condition
2.4. Cell Toxicity
2.5. Cell Differentiation
2.6. Oil Red O Staining
2.7. Real Time-PCR
2.8. Western Blot
2.9. Statistical Analysis
3. Results
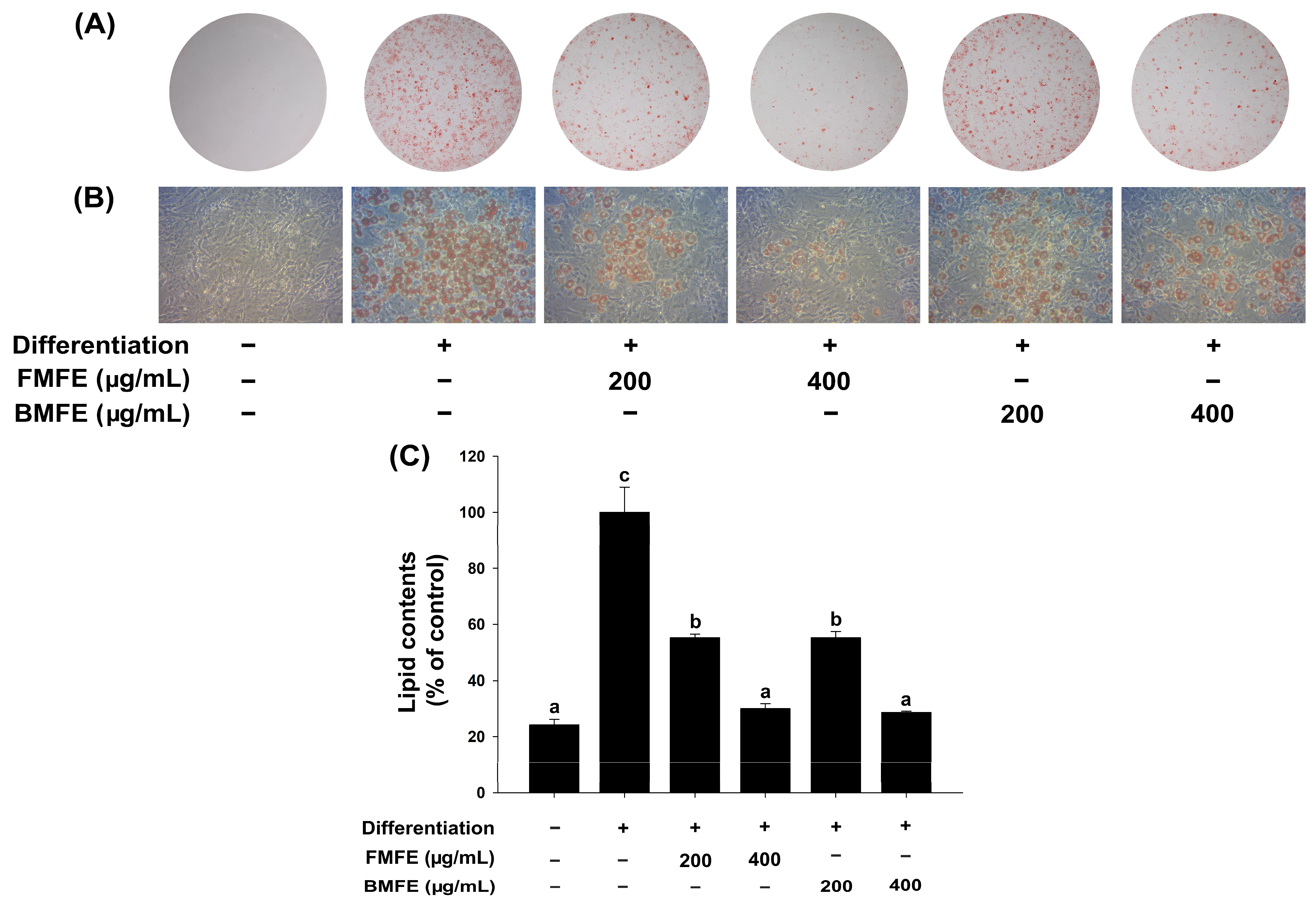
3.1. Effects of MFEs on Adipogenic Differentiation of 3T3-L1 Cells
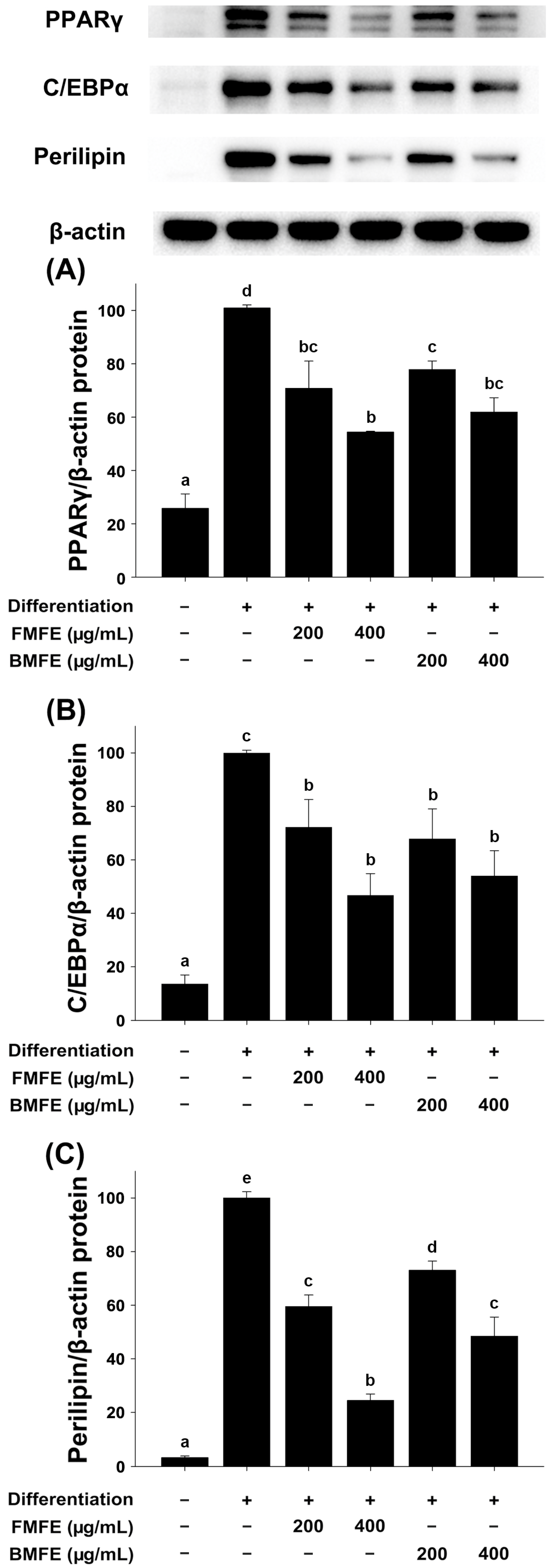
3.2. Effect of MFEs on the Expression Level of Adipogenic-Specific Genes in 3T3-L1 Cells
3.3. Effect of MFEs on the Expression Level of Adipogenic-Specific Markers in 3T3-L1 Cells
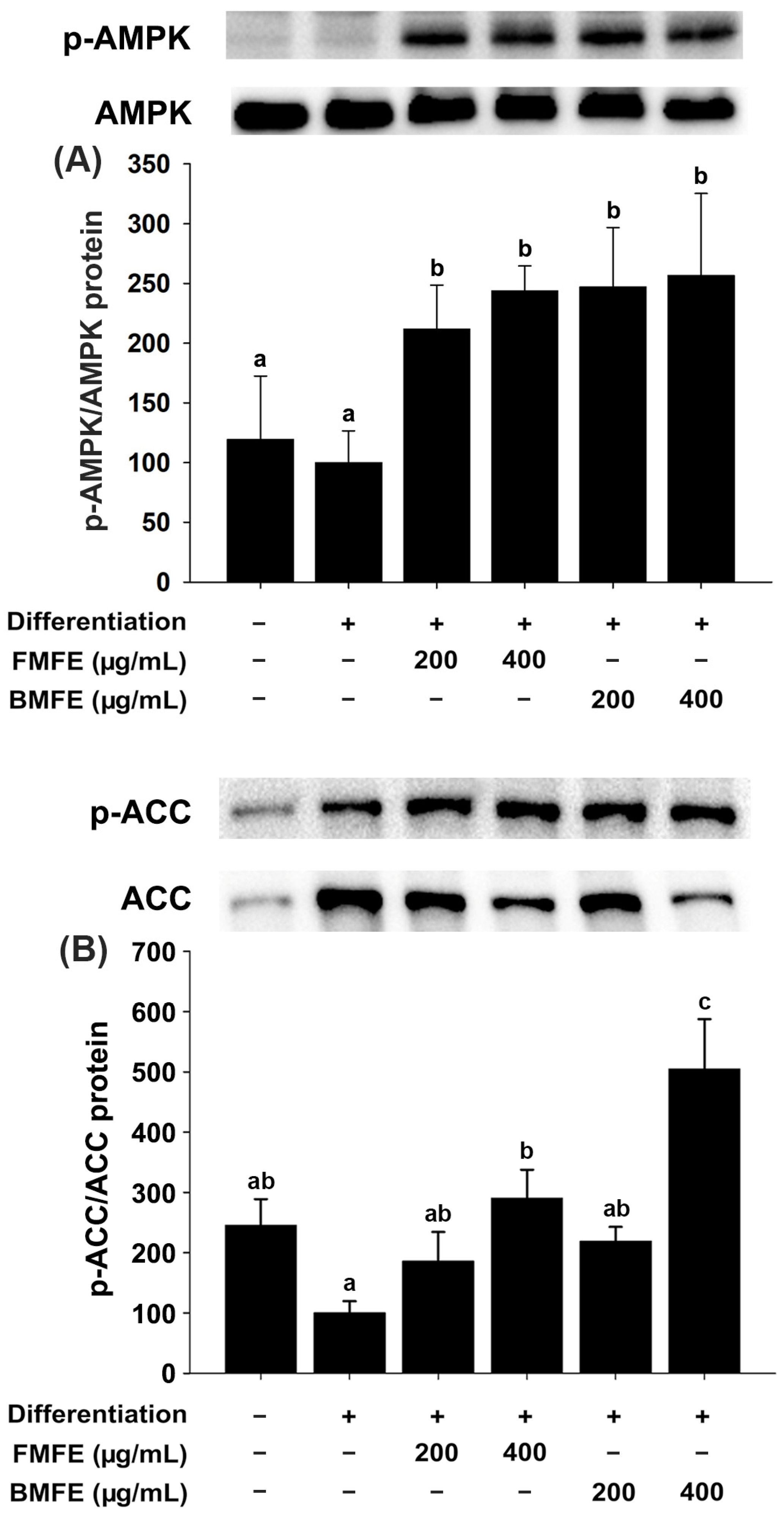
3.4. Effect of MFEs on AMPK Activation in 3T3-L1 Adipocytes
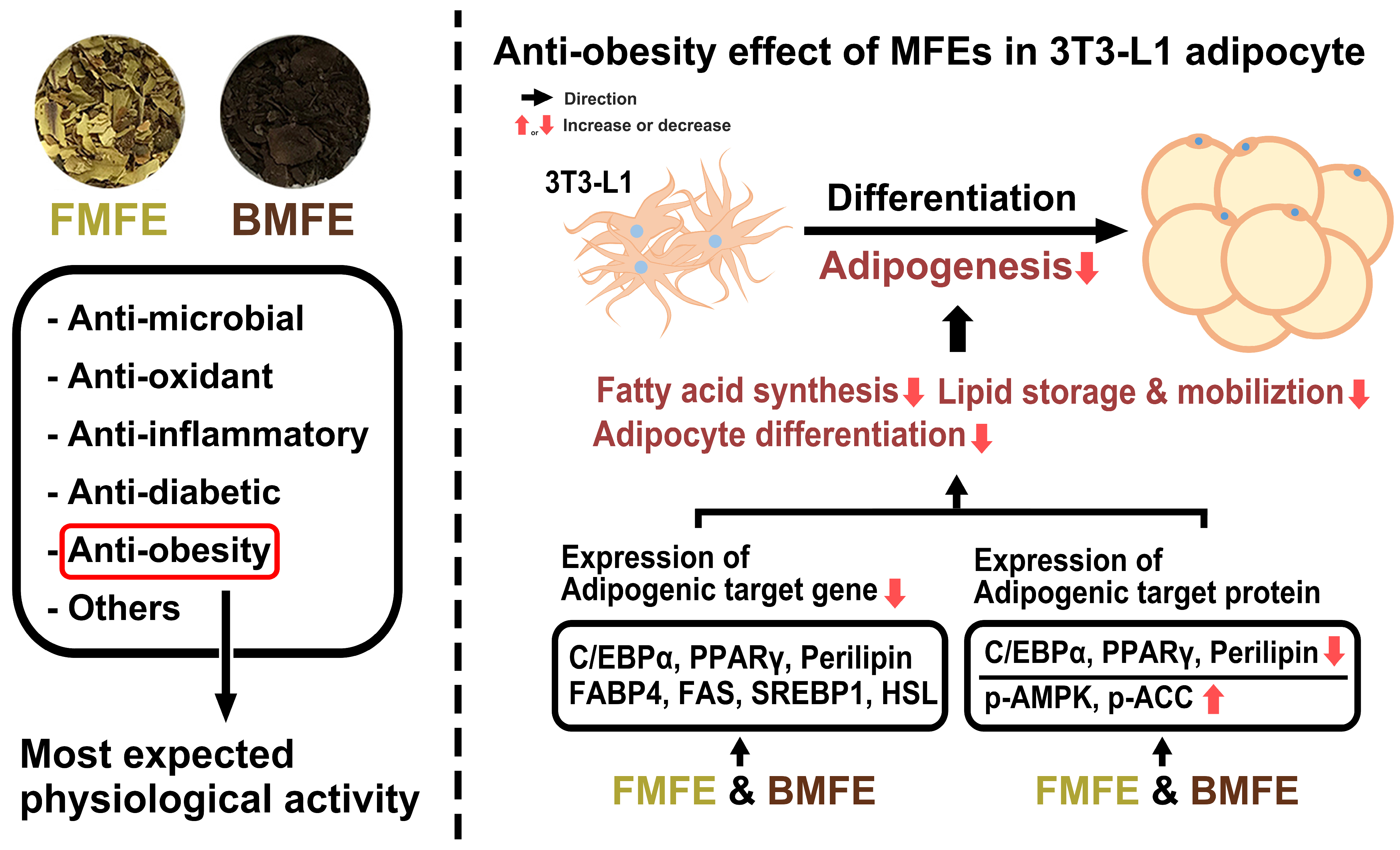
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Joo, Y.H.; Nam, M.H.; Chung, N.; Lee, Y.K. UPLC-QTOF-MS/MS screening and identification of bioactive compounds in fresh, aged, and browned Magnolia denudata flower extracts. Food Res. Int. 2020, 133, 109192. [Google Scholar] [CrossRef] [PubMed]
- Schühly, W.; Khan, I.; Fischer, N.H. The ethnomedicinal uses of magnoliaceae from the southeastern United States as leads in drug discovery. Pharm. Biol. 2001, 39, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Hyeon, H.; Hyun, H.B.; Go, B.; Kim, S.C.; Jung, Y.-H.; Ham, Y.-M. Profiles of essential oils and correlations with phenolic acids and primary metabolites in flower buds of Magnolia heptapeta and Magnolia denudata var. purpurascens. Molecules 2022, 27, 221. [Google Scholar] [CrossRef] [PubMed]
- Chung, I.-M.; Malathy, R.; Priyadharshini, R.; Hemapriya, V.; Kim, S.-H.; Prabakaran, M. Inhibition of mild steel corrosion using Magnolia kobus extract in sulphuric acid medium. Mater. Today Commun. 2020, 25, 101687. [Google Scholar] [CrossRef]
- Park, C.H.; Park, S.-Y.; Lee, S.Y.; Kim, J.K.; Park, S.U. Analysis of metabolites in white flowers of Magnolia denudata Desr. and violet flowers of Magnolia liliiflora Desr. Molecules 2018, 23, 1558. [Google Scholar] [CrossRef]
- Yoon, H. Effects of aging on the phenolic content and antioxidant activities of magnolia (Magnolia denudata) flower extracts. Food Sci. Biotechnol. 2014, 23, 1715–1718. [Google Scholar] [CrossRef]
- Nho, J.-W.; Hwang, I.-G.; Joung, E.-M.; Jeong, H.-S. Biological activities of Magnolia denudata Desr. flower extracts. J. Korean Soc. Food Sci. Nutr. 2009, 38, 1478–1484. [Google Scholar] [CrossRef][Green Version]
- Lv, H.-P.; Zhang, Y.-J.; Lin, Z.; Lianga, Y.-R. Processing and chemical constituents of Pu-erh tea: A review. Food Res. Int. 2013, 53, 608–618. [Google Scholar] [CrossRef]
- Choi, I.; Park, Y.; Choi, H.; Lee, E.H. Anti-adipogenic activity of rutin in 3T3-L1 cells and mice fed with high-fat diet. Biofactors 2006, 26, 273–281. [Google Scholar] [CrossRef]
- Cheng, L.; Shi, L.; He, C.; Wang, C.; Lv, Y.; Li, H.; An, Y.; Dai, H.; Duan, Y.; Zhang, H.; et al. Rutin-activated adipose tissue thermogenesis is correlated with increased intestinal short-chain fatty acid levels. Phytother. Res. 2022, 36, 2495–2510. [Google Scholar] [CrossRef]
- Muvhulawa, N.; Dludla, P.V.; Mthembu, S.X.H.; Ziqubu, K.; Tiano, L.; Mazibuko-Mbeje, S.E. Rutin attenuates tumor necrosis factor-α-induced inflammation and initiates fat browning in 3T3-L1 adipocytes: Potential therapeutic implications for anti-obesity therapy. S. Afr. J. Bot. 2023, 160, 697–704. [Google Scholar] [CrossRef]
- Xu, H.-M.; Wu, H.-W. Rutin inhibits 3T3-L1 preadipocytes differentiation and promotes beige adipogenesis. Chin. J. Public Health 2021, 37, 845–848. [Google Scholar] [CrossRef]
- Choi, C.G.; Lee, D.J.; Chung, N.; Joo, Y.H. Anti-obesity effects of isoacteoside on 3T3-L1 adipocytes. Appl. Biol. Chem. 2022, 65, 33. [Google Scholar] [CrossRef]
- Liu, S.-L.; Peng, B.-J.; Zhong, Y.-L.; Liu, Y.-L.; Song, Z.; Wang, Z. Effect of 5-caffeoylquinic acid on the NF-κB signaling pathway, peroxisome proliferator-activated receptor gamma 2, and macrophage infiltration in high-fat diet-fed Sprague–Dawley rat adipose tissue. Food Funct. 2015, 6, 2779–2786. [Google Scholar] [CrossRef]
- Mili, N.; Paschou, S.A.; Goulis, D.G.; Dimopoulos, M.-A.; Lambrinoudaki, I.; Psaltopoulou, T. Obesity, metabolic syndrome, and cancer: Pathophysiological and therapeutic associations. Endocrine 2021, 74, 478–497. [Google Scholar] [CrossRef]
- Scully, T.; Ettela, A.; LeRoith, D.; Gallagher, E.J. Obesity, type 2 diabetes, and cancer risk. Front Oncol. 2021, 10, 615375. [Google Scholar] [CrossRef]
- Jin, X.; Qiu, T.; Li, L.; Yu, R.; Chen, X.; Li, C.; Proud, C.G.; Jiang, T. Pathophysiology of obesity and its associated diseases. Acta. Pharm. Sin. B 2023, 13, 2403–2424. [Google Scholar] [CrossRef]
- Wen, X.; Zhang, B.; Wu, B.; Xiao, H.; Li, Z.; Li, R.; Xu, X.; Li, T. Signaling pathways in obesity: Mechanisms and therapeutic interventions. Signal Transduct. Target Ther. 2022, 7, 298. [Google Scholar] [CrossRef]
- Yang, M.; Liu, S.; Zhang, C. The related metabolic diseases and treatments of obesity. Healthcare 2022, 10, 1616. [Google Scholar] [CrossRef]
- Luo, L.; Liu, M. Adipose tissue in control of metabolism. J. Endocrinol. 2016, 231, R77–R99. [Google Scholar] [CrossRef]
- An, S.M.; Cho, S.H.; Yoon, J.C. Adipose tissue and metabolic health. Diabetes Metab. J. 2023, 47, 595–611. [Google Scholar] [CrossRef]
- Liu, Y.; Qian, S.-W.; Tang, Y.; Tang, Q.Q. The secretory function of adipose tissues in metabolic regulation. Life Metab. 2024, 3, loae003. [Google Scholar] [CrossRef]
- Bahmad, H.F.; Daouk, R.; Azar, J.; Sapudom, J.; Teo, J.C.; Abou-Kheir, W.; Al-Sayegh, M. Modeling adipogenesis: Current and future perspective. Cells 2020, 9, 2326. [Google Scholar] [CrossRef] [PubMed]
- Etesami, B.; Ghaseminezhad, S.; Nowrouzi, A.; Rashidipour, M.; Yazdanparast, R. Investigation of 3T3-L1 cell differentiation to adipocyte, affected by aqueous seed extract of Phoenix dactylifera L. Rep. Biochem. Mol. Biol. 2020, 9, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Mubtasim, N.; Gollahon, L. Characterizing 3T3-L1 MBX adipocyte cell differentiation maintained with fatty acids as an in vitro model to study the effects of obesity. Life 2023, 13, 1712. [Google Scholar] [CrossRef] [PubMed]
- Khan, F.; Khan, H.; Khan, A.; Yamasaki, M.; Moustaid-Moussa, N.; Al-Harrasi, A.; Rahman, S.M. Autophagy in adipogenesis: Molecular mechanisms and regulation by bioactive compounds. Biomed. Pharmacother. 2022, 155, 113715. [Google Scholar] [CrossRef]
- Joo, Y.H.; Chung, N.; Lee, Y.K. Anti-obesity effect of fresh and browned Magnolia denudata flowers in a high fat diet murine model. J. Funct. Foods 2020, 75, 104227. [Google Scholar] [CrossRef]
- Salman, S.; Öz, G.; Felek, R.; Haznedar, A.; Turna, T.; Özdemir, F. Effects of fermentation time on phenolic composition, antioxidant and antimicrobial activities of green, oolong, and black teas. Food Biosc. 2022, 49, 101884. [Google Scholar] [CrossRef]
- Kang, S.; Lee, S.J.; Kwon, Y.H.; Moon, D.-G.; Sun, J.H.; Hwang, K.-W.; Moon, J.-K. Characteristic of phenotype, amino acids and volatile compounds for fresh tea leaves of Korean tea cultivars (Camellia sinensis (L.) O. Kuntze). Appl. Biol. Chem. 2024, 67, 67. [Google Scholar] [CrossRef]
- Tang, G.Y.; Meng, X.; Gan, R.Y.; Zhao, C.N.; Liu, Q.; Feng, Y.B.; Li, S.; Wei, X.L.; Atanasov, A.G.; Corke, H.; et al. Health functions and related molecular mechanisms of tea components: An update review. Int. J. Mol. Sci. 2019, 20, 6196. [Google Scholar] [CrossRef]
- Weerawatanakorn, M.; Hung, W.-L.; Pan, M.-H.; Li, S.; Li, D.; Wan, X.; Ho, C.-T. Chemistry and health beneficial effects of oolong tea and theasinensins. Food Sci. Hum. Well. 2015, 4, 133–146. [Google Scholar] [CrossRef]
- Shang, A.; Li, J.; Zhou, D.-D.; Gan, R.-Y.; Li, H.-B. Molecular mechanisms underlying health benefits of tea compounds. Free Radic. Biol. Med. 2021, 172, 181–200. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Anand, S.K.; Singh, N.; Dwivedi, U.N.; Kakkar, P. AMP-activated protein kinase: An energy sensor and survival mechanism in the reinstatement of metabolic homeostasis. Exp. Cell Res. 2023, 428, 113614. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Lee, D.Y.; Yang, H.R.; Im, K.-H.; Park, S.U.; Kim, J.G.; Kim, J.K. Evaluation of obesity prevention effect of black ginseng on serum, liver, and hypothalamus of mice on a high-fat diet using a metabolomics approach. Appl. Biol. Chem. 2023, 66, 73. [Google Scholar] [CrossRef]
- Jakab, J.; Miškić, B.; Mikšić, Š.; Juranić, B.; Ćosić, V.; Schwarz, D.; Včev, A. Adipogenesis as a potential anti-obesity target: A review of pharmacological treatment and natural products. Diabetes Metab. Syndr. Obes. 2021, 14, 67–83. [Google Scholar] [CrossRef]
- Batchuluun, B.; Pinkosky, S.L.; Steinberg, G.R. Lipogenesis inhibitors: Therapeutic opportunities and challenges. Nat. Rev. Drug Discov. 2022, 21, 283–305. [Google Scholar] [CrossRef]
- Lodhi, I.J.; Wei, X.; Semenkovich, C.F. Lipoexpediency: De novo lipogenesis as a metabolic signal transmitter. Trends Endocrinol. Metab. 2011, 22, 1–8. [Google Scholar] [CrossRef]
- Rowland, L.A.; Guilherme, A.; Henriques, F.; DiMarzio, C.; Munroe, S.; Wetoska, N.; Kelly, M.; Reddig, K.; Hendricks, G.; Pan, M.; et al. De novo lipogenesis fuels adipocyte autophagosome and lysosome membrane dynamics. Nat. Commun. 2023, 14, 1362. [Google Scholar] [CrossRef]
- Griseti, E.; Bello, A.A.; Bieth, E.; Sabbagh, B.; Iacovoni, J.S.; Bigay, J.; Laurell, H.; Čopič, A. Molecular mechanisms of perilipin protein function in lipid droplet metabolism. FEBS Lett. 2024, 598, 1170–1198. [Google Scholar] [CrossRef]
- Sztalryd, C.; Kimmel, A.R. Perilipins: Lipid droplet coat proteins adapted for tissue-specific energy storage and utilization, and lipid cytoprotection. Biochimie 2014, 96, 96–101. [Google Scholar] [CrossRef]
- Beller, M.; Bulankina, A.V.; Hsiao, H.-H.; Urlaub, H.; Jäckle, H.; Kühnlein, R.P. Perilipin-dependent control of lipid droplet structure and fat storage in Drosophila. Cell Metab. 2010, 12, 521–532. [Google Scholar] [CrossRef] [PubMed]
- Itabe, H.; Yamaguchi, T.; Nimura, S.; Sasabe, N. Perilipins: A diversity of intracellular lipid droplet proteins. Lipids Health Dis. 2017, 16, 83. [Google Scholar] [CrossRef] [PubMed]
- Bartholomew, S.R.; Bell, E.H.; Summerfield, T.; Newman, L.C.; Miller, E.L.; Patterson, B.; Niday, Z.P.; Ackerman, W.E., 4th; Tansey, J.T. Distinct cellular pools of perilipin 5 point to roles in lipid trafficking. Biochim. Biophys. Acta. 2012, 1821, 268–278. [Google Scholar] [CrossRef] [PubMed]
- Michler, S.; Schöffmann, F.A.; Robaa, D.; Volmer, J.; Hinderberger, D. Fatty acid binding to the human transport proteins FABP3, FABP4, and FABP5 from a Ligand’s perspective. J. Biol. Chem. 2024, 300, 107396. [Google Scholar] [CrossRef] [PubMed]
- Furuhashi, M.; Saitoh, S.; Shimamoto, K.; Miura, T. Fatty acid-binding protein 4 (FABP4): Pathophysiological insights and potent clinical biomarker of metabolic and cardiovascular diseases. Clin. Med. Insights Cardiol. 2015, 8, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Wu, D.; Fan, Z.; Yang, J.; Li, Y.; Meng, Y.; Gao, C.; Zhan, H. FABP4 in obesity-associated carcinogenesis: Novel insights into mechanisms and therapeutic implications. Front. Mol. Biosci. 2022, 19, 973955. [Google Scholar] [CrossRef]
- Althaher, A.R. An overview of hormone-sensitive lipase (HSL). Sci. World J. 2022, 2022, 1964684. [Google Scholar] [CrossRef]
- Duncan, R.E.; Ahmadian, M.; Jaworski, K.; Sarkadi-Nagy, E.; Sul, H.S. Regulation of lipolysis in adipocytes. Annu. Rev. Nutr. 2007, 27, 79–101. [Google Scholar] [CrossRef]
- Lampidonis, A.D.; Rogdakis, E.; Voutsinas, G.E.; Stravopodis, D.J. The resurgence of hormone-sensitive lipase (HSL) in mammalian lipolysis. Gene 2011, 477, 1–11. [Google Scholar] [CrossRef]
- Steinberg, G.R.; Hardie, D.G. New insights into activation and function of the AMPK. Nat. Rev. Mol. Cell Biol. 2023, 24, 255–272. [Google Scholar] [CrossRef]
- Garcia, D.; Shaw, R.J. AMPK: Mechanisms of cellular energy sensing and restoration of metabolic balance. Mol. Cell. 2017, 66, 789–800. [Google Scholar] [CrossRef] [PubMed]
- Hardie, D.G. AMP-activated protein kinase: An energy sensor that regulates all aspects of cell function. Genes Dev. 2011, 25, 1895–1908. [Google Scholar] [CrossRef] [PubMed]
- Lage, R.; Diéguez, C.; Vidal-Puig, A.; López, M. AMPK: A metabolic gauge regulating whole-body energy homeostasis. Trends Mol. Med. 2008, 14, 539–549. [Google Scholar] [CrossRef] [PubMed]


| Genes | Forward Primer (5′-3′) | Reverse Primer (5′-3′) |
|---|---|---|
| C/EBP-α | TGCTGGAGTTGACCAGTGAC | AAACCATCCTCTGGGTCTCC |
| PPAR-γ | TTTTCAAGGGTGCCAGTTTCAATCC | AATCCTTGGCCCTCTGAGAT |
| Perilipin | CTCTGGGAAGCATCGAGAAG | GCATGGTGTGTCGAGAAAGA |
| FABP4 | AGACGACAGGAAGGTGAAGAG | TCATAACACATTCCACCACCAG |
| FAS | TGCTGTTGGAAGTCAGCTATGAA | GATGCCTCTGAACCACTCACAC |
| SREBP1 | CCGAGATGTGCGAACTGGA | GAAGTCACTGTCTTGGTTGTTGATG |
| HSL | TCCTGGAACTAAGTGGACGCAAG | CAGACACACTCCTGCGCATAGAC |
| β-actin | AGCCATGTACGTAGCCATCC | TCCCTCTCAGCTGTGGTGGTGAA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, D.J.; Yeom, J.H.; Lee, Y.K.; Joo, Y.H.; Chung, N. Anti-Obesity Effect of Fresh and Browned Magnolia denudata Flowers in 3T3-L1 Adipocytes. Appl. Sci. 2024, 14, 9254. https://doi.org/10.3390/app14209254
Lee DJ, Yeom JH, Lee YK, Joo YH, Chung N. Anti-Obesity Effect of Fresh and Browned Magnolia denudata Flowers in 3T3-L1 Adipocytes. Applied Sciences. 2024; 14(20):9254. https://doi.org/10.3390/app14209254
Chicago/Turabian StyleLee, Deok Jae, Jae Ho Yeom, Yong Kwon Lee, Yong Hoon Joo, and Namhyun Chung. 2024. "Anti-Obesity Effect of Fresh and Browned Magnolia denudata Flowers in 3T3-L1 Adipocytes" Applied Sciences 14, no. 20: 9254. https://doi.org/10.3390/app14209254
APA StyleLee, D. J., Yeom, J. H., Lee, Y. K., Joo, Y. H., & Chung, N. (2024). Anti-Obesity Effect of Fresh and Browned Magnolia denudata Flowers in 3T3-L1 Adipocytes. Applied Sciences, 14(20), 9254. https://doi.org/10.3390/app14209254







