Static Foot Hyperpronation Monitoring in Asymptomatic Young Individuals During Level and Sloped Gait Using an Instrumented Treadmill
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Sample
2.3. Testing Procedure
2.4. Instrumentation
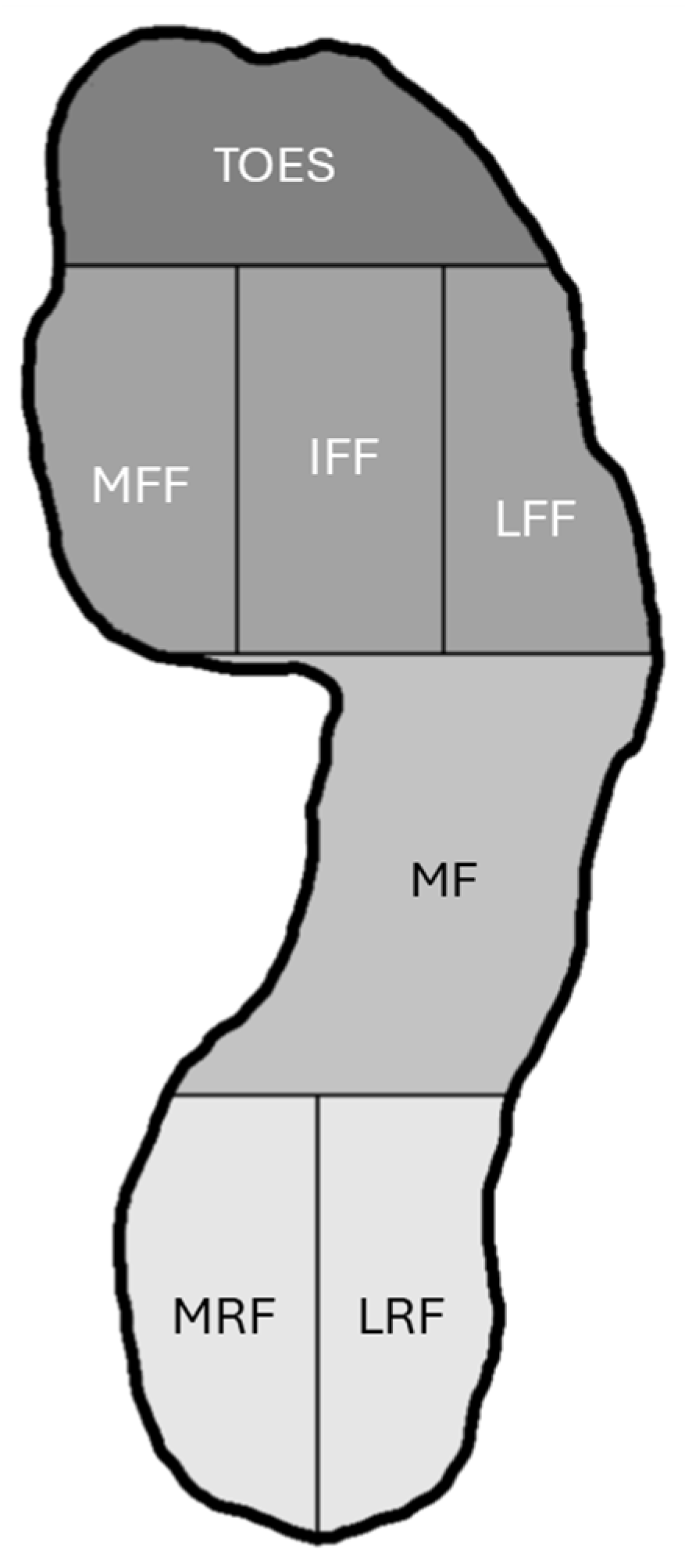
2.5. Measurement Outcomes
2.6. Reliability Study
2.7. Statistical Analysis
3. Results
3.1. Spatial Parameters
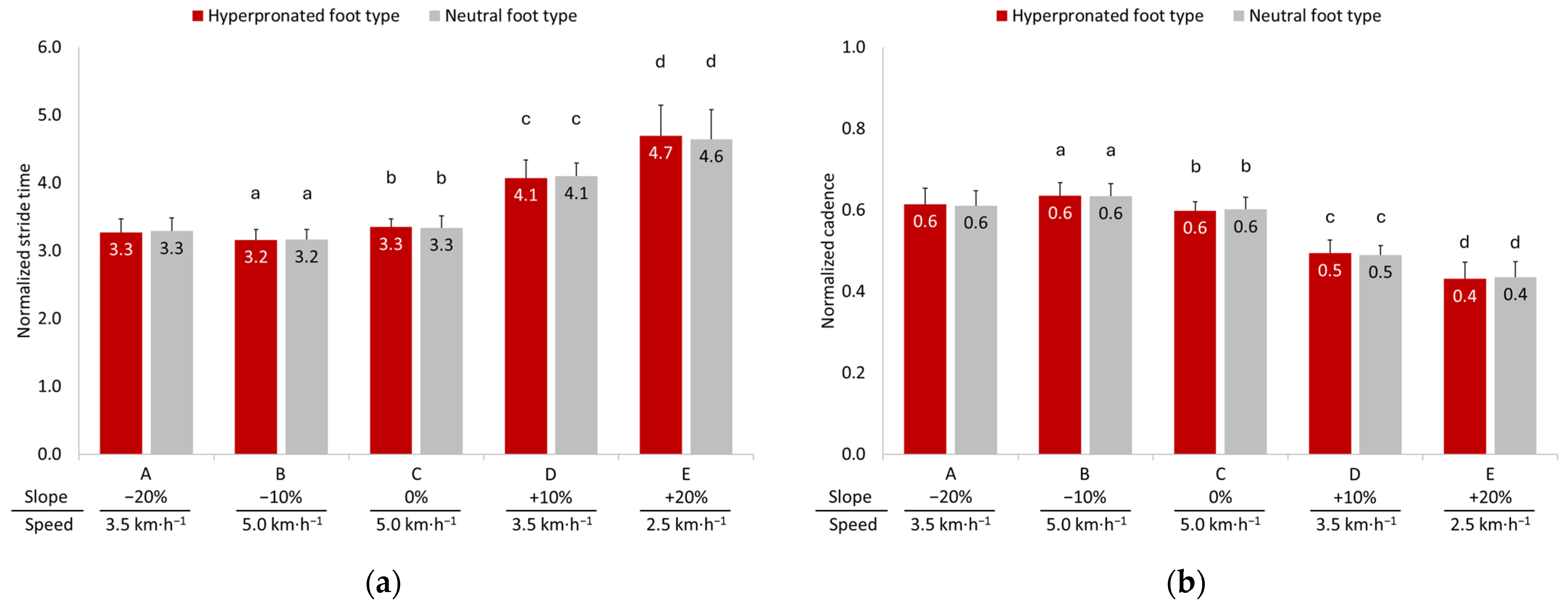
3.2. Temporal Parameters
3.3. Gait Phase Analysis
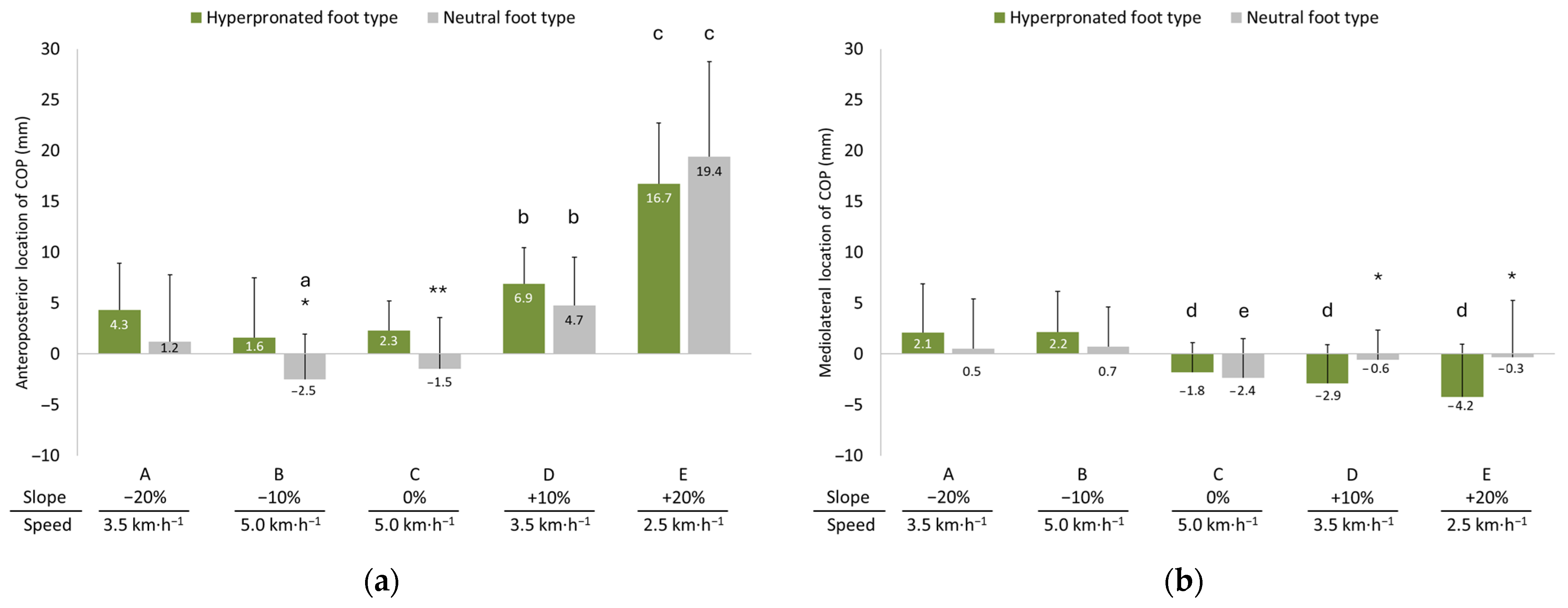
3.4. Anteroposterior and Mediolateral Locations of COP
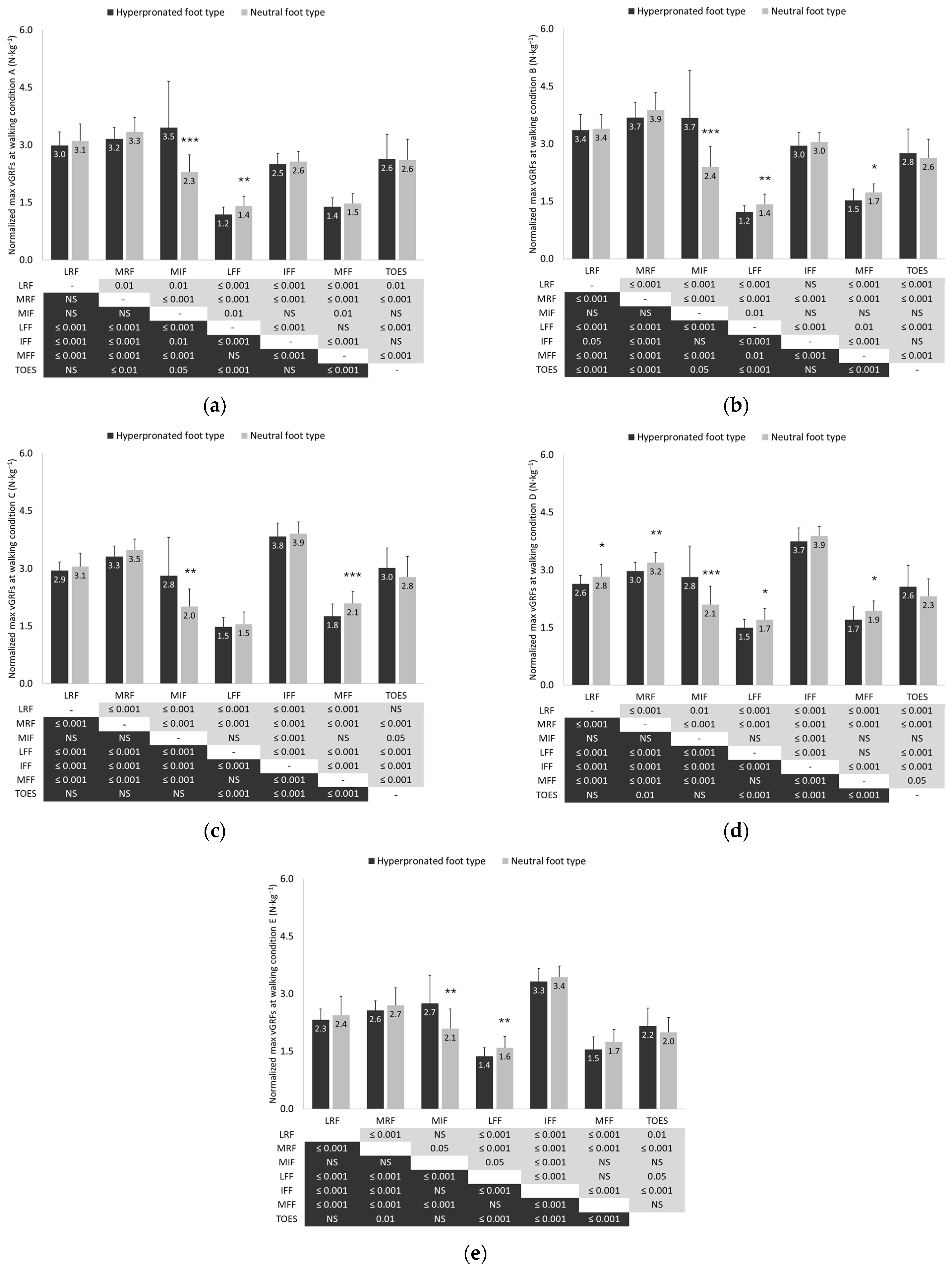
3.5. Vertical Ground Reaction Forces
4. Discussion
4.1. Spatiotemporal Parameters and Gait Phase Analysis
4.2. Anteroposterior and Mediolateral Locations of COP
4.3. Vertical Ground Reaction Forces
4.4. Limitations
4.5. Clinical Significance
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Horwood, A.M.; Chockalingam, N. Defining Excessive, over, or Hyper-Pronation: A Quandary. Foot 2017, 31, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Brosky, J.A.; Balazsy, J.E. Functional Anatomy of the Foot and Ankle. In Orthopaedic Physical Therapy Secrets, 3rd ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2017; pp. 581–586. ISBN 9780323286855. [Google Scholar]
- Sueki, D.G.; Cleland, J.A.; Wainner, R.S. A Regional Interdependence Model of Musculoskeletal Dysfunction: Research, Mechanisms, and Clinical Implications. J. Man. Manip. Ther. 2013, 21, 90–102. [Google Scholar] [CrossRef] [PubMed]
- Riegger-Krugh, C.; Keysor, J.J. Skeletal Malalignments of the Lower Quarter: Correlated and Compensatory Motions and Postures. J. Orthop. Sports Phys. Ther. 1996, 23, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Yazdani, F.; Razeghi, M.; Karimi, M.T.; Raeisi Shahraki, H.; Salimi Bani, M. The Influence of Foot Hyperpronation on Pelvic Biomechanics during Stance Phase of the Gait: A Biomechanical Simulation Study. Proc. Inst. Mech. Eng. H 2018, 232, 708–717. [Google Scholar] [CrossRef]
- Powers, C.M. The Influence of Altered Lower-Extremity Kinematics on Patellofemoral Joint Dysfunction: A Theoretical Perspective. J. Orthop. Sports Phys. Ther. 2003, 33, 639–646. [Google Scholar] [CrossRef]
- Neal, B.S.; Griffiths, I.B.; Dowling, G.J.; Murley, G.S.; Munteanu, S.E.; Franettovich Smith, M.M.; Collins, N.J.; Barton, C.J. Foot Posture as a Risk Factor for Lower Limb Overuse Injury: A Systematic Review and Meta-analysis. J. Foot Ankle Res. 2014, 7, 55. [Google Scholar] [CrossRef]
- Hintermann, B.; Nigg, B.M. Pronation in Runners. Sports Med. 1998, 26, 169–176. [Google Scholar] [CrossRef]
- Pérez-Morcillo, A.; Gómez-Bernal, A.; Gil-Guillen, V.F.; Alfaro-Santafé, J.; Alfaro-Santafé, J.V.; Quesada, J.A.; Lopez-Pineda, A.; Orozco-Beltran, D.; Carratalá-Munuera, C. Association between the Foot Posture Index and Running Related Injuries: A Case-Control Study. Clin. Biomech. 2019, 61, 217–221. [Google Scholar] [CrossRef]
- Kaufman, K.R.; Brodine, S.K.; Shaffer, R.A.; Johnson, C.W.; Cullison, T.R. The Effect of Foot Structure and Range of Motion on Musculoskeletal Overuse Injuries. Am. J. Sports Med. 1999, 27, 585–593. [Google Scholar] [CrossRef]
- Almeheyawi, R.N.; Bricca, A.; Riskowski, J.L.; Barn, R.; Steultjens, M. Foot Characteristics and Mechanics in Individuals with Knee Osteoarthritis: Systematic Review and Meta-analysis. J. Foot Ankle Res. 2021, 14, 24. [Google Scholar] [CrossRef]
- Razeghi, M.; Batt, M.E. Foot Type Classification: A Critical Review of Current Methods. Gait Posture 2002, 15, 282–291. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.S.; Lee, J.H.; Kim, E.J.; Kyung, M.G.; Yoo, H.J.; Lee, D.Y. Flatfoot Deformity Affected the Kinematics of the Foot and Ankle in Proportion to the Severity of Deformity. Gait Posture 2019, 72, 123–128. [Google Scholar] [CrossRef]
- Buldt, A.K.; Levinger, P.; Murley, G.S.; Menz, H.B.; Nester, C.J.; Landorf, K.B. Foot Posture Is Associated with Kinematics of the Foot during Gait: A Comparison of Normal, Planus and Cavus Feet. Gait Posture 2015, 42, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Marouvo, J.; Sousa, F.; Fernandes, O.; Castro, M.A.; Paszkiel, S. Gait Kinematics Analysis of Flatfoot Adults. Appl. Sci. 2021, 11, 7077. [Google Scholar] [CrossRef]
- Okamura, K.; Egawa, K.; Ikeda, T.; Fukuda, K.; Kanai, S. Relationship between Foot Muscle Morphology and Severity of Pronated Foot Deformity and Foot Kinematics during Gait: A Preliminary Study. Gait Posture 2021, 86, 273–277. [Google Scholar] [CrossRef]
- Zhang, X.; Aeles, J.; Vanwanseele, B. Comparison of Foot Muscle Morphology and Foot Kinematics between Recreational Runners with Normal Feet and with Asymptomatic Over-Pronated Feet. Gait Posture 2017, 54, 290–294. [Google Scholar] [CrossRef] [PubMed]
- Abdul Razak, A.H.; Zayegh, A.; Begg, R.K.; Wahab, Y. Foot Plantar Pressure Measurement System: A Review. Sensors 2012, 12, 9884–9912. [Google Scholar] [CrossRef]
- Burnie, L.; Chockalingam, N.; Holder, A.; Claypole, T.; Kilduff, L.; Bezodis, N. Commercially Available Pressure Sensors for Sport and Health Applications: A Comparative Review. Foot 2023, 56, 102046. [Google Scholar] [CrossRef]
- Arzehgar, A.; Nia, R.G.N.N.; Hoseinkhani, M.; Masoumi, F.; Sayyed-Hosseinian, S.-H.; Eslami, S. An Overview of Plantar Pressure Distribution Measurements and Its Applications in Health and Medicine. Gait Posture 2025, 117, 235–244. [Google Scholar] [CrossRef]
- Fallahtafti, F.; Wurdeman, S.R.; Yentes, J.M. Sampling Rate Influences the Regularity Analysis of Temporal Domain Measures of Walking More than Spatial Domain Measures. Gait Posture 2021, 88, 216–220. [Google Scholar] [CrossRef]
- Veilleux, L.N.; Raison, M.; Rauch, F.; Robert, M.; Ballaz, L. Agreement of Spatio-Temporal Gait Parameters between a Vertical Ground Reaction Force Decomposition Algorithm and a Motion Capture System. Gait Posture 2016, 43, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Fujishita, H.; Ikuta, Y.; Maeda, N.; Komiya, M.; Morikawa, M.; Arima, S.; Sakamitsu, T.; Obayashi, H.; Fukuhara, K.; Ushio, K.; et al. Effects of Rearfoot Eversion on Foot Plantar Pressure and Spatiotemporal Gait Parameters in Adolescent Athletes. Healthcare 2023, 11, 1842. [Google Scholar] [CrossRef] [PubMed]
- Chuckpaiwong, B.; Nunley, J.A.; Mall, N.A.; Queen, R.M. The Effect of Foot Type on In-Shoe Plantar Pressure during Walking and Running. Gait Posture 2008, 28, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Mei, Q.; Gu, Y.; Xiang, L.; Yu, P.; Gao, Z.; Shim, V.; Fernandez, J. Foot Shape and Plantar Pressure Relationships in Shod and Barefoot Populations. Biomech. Model. Mechanobiol. 2020, 19, 1211–1224. [Google Scholar] [CrossRef]
- Buldt, A.K.; Forghany, S.; Landorf, K.B.; Levinger, P.; Murley, G.S.; Menz, H.B. Foot Posture Is Associated with Plantar Pressure during Gait: A Comparison of Normal, Planus and Cavus Feet. Gait Posture 2018, 62, 235–240. [Google Scholar] [CrossRef]
- Han, J.T.; Koo, H.M.; Jung, J.M.; Kim, Y.J.; Lee, J.H. Differences in Plantar Foot Pressure and COP between Flat and Normal Feet During Walking. J. Phys. Ther. Sci. 2011, 23, 683–685. [Google Scholar] [CrossRef]
- Ledoux, W.R.; Hillstrom, H.J. The Distributed Plantar Vertical Force of Neutrally Aligned and Pes Planus Feet. Gait Posture 2002, 15, 1–9. [Google Scholar] [CrossRef]
- Wong, Y.S. Influence of the Abductor Hallucis Muscle on the Medial Arch of the Foot: A Kinematic and Anatomical Cadaver Study. Foot Ankle Int. 2007, 28, 617–620. [Google Scholar] [CrossRef]
- Semaan, M.B.; Wallard, L.; Ruiz, V.; Gillet, C.; Leteneur, S.; Simoneau-Buessinger, E. Is Treadmill Walking Biomechanically Comparable to Overground Walking? A Systematic Review. Gait Posture 2022, 92, 249–257. [Google Scholar] [CrossRef]
- Kim, M.-K. Foot Pressure Analysis of Adults with Flat and Normal Feet at Different Gait Speeds on an Ascending Slope. J. Phys. Ther. Sci. 2015, 27, 3767–3769. [Google Scholar] [CrossRef]
- Kim, M.-K.; Lee, Y.-S. A Three-Dimensional Gait Analysis of People with Flat Arched Feet on an Ascending Slope. J. Phys. Ther. Sci. 2014, 26, 1437–1440. [Google Scholar] [CrossRef] [PubMed]
- Resende, R.A.; Pinheiro, L.S.P.; Ocarino, J.M. Effects of Foot Pronation on the Lower Limb Sagittal Plane Biomechanics during Gait. Gait Posture 2019, 68, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Requelo-Rodríguez, I.; Castro-Méndez, A.; Jiménez-Cebrián, A.M.; González-Elena, M.L.; Palomo-Toucedo, I.C.; Pabón-Carrasco, M. Assessment of Selected Spatio-Temporal Gait Parameters on Subjects with Pronated Foot Posture on the Basis of Measurements Using Optogait. A Case-Control Study. Sensors 2021, 21, 2805. [Google Scholar] [CrossRef]
- Knutson, G.A. Anatomic and Functional Leg-Length Inequality: A Review and Recommendation for Clinical Decision-Making. Part I, Anatomic Leg-Length Inequality: Prevalence, Magnitude, Effects and Clinical Significance. Chiropr. Osteopat. 2005, 13, 11. [Google Scholar] [CrossRef] [PubMed]
- Bunnell, W.P. An Objective Criterion for Scoliosis Screening. J. Bone Jt. Surg. Am. 1984, 66, 1381–1387. [Google Scholar] [CrossRef]
- Farahmand, B.; Ebrahimi Takamjani, E.; Yazdi, H.R.; Saeedi, H.; Kamali, M.; Bagherzadeh Cham, M. A Systematic Review on the Validity and Reliability of Tape Measurement Method in Leg Length Discrepancy. Med. J. Islam Repub. Iran. 2019, 33, 46. [Google Scholar] [CrossRef]
- Powden, C.J.; Hoch, J.M.; Hoch, M.C. Reliability and Minimal Detectable Change of the Weight-Bearing Lunge Test: A Systematic Review. Man Ther. 2015, 20, 524–532. [Google Scholar] [CrossRef] [PubMed]
- Tobler, W. Three Presentations on Geographical Analysis and Modeling: Non- Isotropic Geographic Modeling; Speculations on the Geometry of Geography and Global Spatial Analysis. Tech. Rep. 1993, 93, 1–25. [Google Scholar]
- Pingel, T.J. Modeling Slope as a Contributor to Route Selection in Mountainous Areas. Cartogr. Geogr. Inf. Sci. 2010, 37, 137–148. [Google Scholar] [CrossRef]
- Meyer, C.; Killeen, T.; Easthope, C.S.; Curt, A.; Bolliger, M.; Linnebank, M.; Zörner, B.; Filli, L. Familiarization with Treadmill Walking: How Much Is Enough? Sci. Rep. 2019, 9, 5232. [Google Scholar] [CrossRef]
- Paillard, T. Effects of General and Local Fatigue on Postural Control: A Review. Neurosci. Biobehav. Rev. 2012, 36, 162–176. [Google Scholar] [CrossRef] [PubMed]
- Hof, A.L. Scaling Gait Data to Body Size. Gait Posture 1996, 4, 222–223. [Google Scholar] [CrossRef]
- Zebris Medical GmbH. Software Manual Zebris FDM, version 2.0.x; Zebris Medical GmbH: Isny im Allgäu, Germany, 2022.
- Ho, I.-J.; Hou, Y.-Y.; Yang, C.-H.; Wu, W.-L.; Chen, S.-K.; Guo, L.-Y. Comparison of Plantar Pressure Distribution between Different Speed and Incline During Treadmill Jogging. J. Sports Sci. Med. 2010, 9, 154–160. [Google Scholar]
- Cao, Y.; Zhuang, H.; Zhang, X.; Guo, R.; Pang, H.; Zheng, P.; Xu, H. Impact of Foot Progression Angle on Spatiotemporal and Plantar Loading Pattern in Intoeing Children during Gait. Sci. Rep. 2024, 14, 14402. [Google Scholar] [CrossRef] [PubMed]
- Koo, T.K.; Li, M.Y. A Guideline of Selecting and Reporting Intraclass Correlation Coefficients for Reliability Research. J. Chiropr. Med. 2016, 15, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Hillstrom, H.J.; Song, J.; Kraszewski, A.P.; Hafer, J.F.; Mootanah, R.; Dufour, A.B.; Chow, B.S.; Deland, J.T. Foot Type Biomechanics Part 1: Structure and Function of the Asymptomatic Foot. Gait Posture 2013, 37, 445–451. [Google Scholar] [CrossRef]
- Farahpour, N.; Jafarnezhad, A.A.; Damavandi, M.; Bakhtiari, A.; Allard, P. Gait Ground Reaction Force Characteristics of Low Back Pain Patients with Pronated Foot and Able-Bodied Individuals with and without Foot Pronation. J. Biomech. 2016, 49, 1705–1710. [Google Scholar] [CrossRef]
- Barton, C.J.; Levinger, P.; Webster, K.E.; Menz, H.B. Kinematics Associated with Foot Pronation in Individuals with Patellofemoral Pain Syndrome: A Case-control Study. J. Foot Ankle Res. 2011, 4, 1. [Google Scholar] [CrossRef]
- Dodelin, D.; Tourny, C.; L’Hermette, M. The Biomechanical Effects of Pronated Foot Function on Gait. An Experimental Study. Scand. J. Med. Sci. Sports 2020, 30, 2167–2177. [Google Scholar] [CrossRef]
- Wang, Y.; Qi, Y.; Ma, B.; Wu, H.; Wang, Y.; Wei, B.; Wei, X.; Xu, Y. Three-Dimensional Gait Analysis of Orthopaedic Common Foot and Ankle Joint Diseases. Front. Bioeng. Biotechnol. 2024, 12, 1303035. [Google Scholar] [CrossRef]
- Sakamoto, K.; Kudo, S. Morphological Characteristics of Intrinsic Foot Muscles among Flat Foot and Normal Foot Using Ultrasonography. Acta Bioeng. Biomech. 2020, 22, 161–166. [Google Scholar] [CrossRef]
- Gray, E.G.; Basmajian, J.V. Electromyography and Cinematography of Leg and Foot (“Normal” and Flat) during Walking. Anat. Rec. 1968, 161, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Murley, G.S.; Menz, H.B.; Landorf, K.B. Foot Posture Influences the Electromyographic Activity of Selected Lower Limb Muscles during Gait. J. Foot Ankle Res. 2009, 2, 35. [Google Scholar] [CrossRef] [PubMed]
- Hunt, A.E.; Smith, R.M. Mechanics and Control of the Flat versus Normal Foot during the Stance Phase of Walking. Clin. Biomech. 2004, 19, 391–397. [Google Scholar] [CrossRef]
- Kwon, Y.; Shin, G. Foot Kinematics and Leg Muscle Activation Patterns Are Altered in Those with Limited Ankle Dorsiflexion Range of Motion during Incline Walking. Gait Posture 2022, 92, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, R.; Phadke, C.P. Effects of Treadmill Incline and Speed on Peroneus Longus Muscle Activity in Persons with Chronic Stroke and Healthy Subjects. Gait Posture 2017, 54, 221–228. [Google Scholar] [CrossRef]
- Duchateau, J.; Enoka, R.M. Neural Control of Lengthening Contractions. J. Exp. Biol. 2016, 219, 197–204. [Google Scholar] [CrossRef]
- Jalaleddini, K.; Nagamori, A.; Laine, C.M.; Golkar, M.A.; Kearney, R.E.; Valero-Cuevas, F.J. Physiological Tremor Increases When Skeletal Muscle Is Shortened: Implications for Fusimotor Control. J. Physiol. 2017, 595, 7331–7346. [Google Scholar] [CrossRef]
- Hiemstra, L.A.; Lo, I.K.Y.; Fowler, P. Effect of Fatigue on Knee Proprioception: Implications for Dynamic Stabilization. J. Orthop. Sports Phys. Ther. 2001, 31, 59–605. [Google Scholar] [CrossRef]
- Strutzenberger, G.; Leutgeb, L.; Claußen, L.; Schwameder, H. Gait on Slopes: Differences in Temporo-Spatial, Kinematic and Kinetic Gait Parameters between Walking on a Ramp and on a Treadmill. Gait Posture 2022, 91, 73–78. [Google Scholar] [CrossRef]
- Leroux, A.; Fung, J.; Barbeau, H. Postural Adaptation to Walking on Inclined Surfaces: I. Normal Strategies. Gait Posture 2002, 15, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Kafetzakis, I.; Konstantinou, I.; Mandalidis, D. Effects of Hiking-Dependent Walking Speeds and Slopes on Spatiotemporal Gait Parameters and Ground Reaction Forces: A Treadmill-Based Analysis in Healthy Young Adults. Appl. Sci. 2024, 14, 4383. [Google Scholar] [CrossRef]
- Solomonow, M. Ligaments: A Source of Musculoskeletal Disorders. J. Bodyw. Mov. Ther. 2009, 13, 136–154. [Google Scholar] [CrossRef]
- Pereira, B.S.; Andrade, R.; Espregueira-Mendes, J.; Marano, R.P.C.; Oliva, X.M.; Karlsson, J. Current Concepts on Subtalar Instability. Orthop. J. Sports Med. 2021, 9, 23259671211021352. [Google Scholar] [CrossRef]
- Bednarczyk, E.; Sikora, S.; Kossobudzka-Górska, A.; Jankowski, K.; Hernandez-Rodriguez, Y. Understanding Flat Feet: An in-Depth Analysis of Orthotic Solutions. J. Orthop. Rep. 2024, 3, 100250. [Google Scholar] [CrossRef]
- Buldt, A.K.; Allan, J.J.; Landorf, K.B.; Menz, H.B. The Relationship between Foot Posture and Plantar Pressure during Walking in Adults: A Systematic Review. Gait Posture 2018, 62, 56–67. [Google Scholar] [CrossRef]
- Jonely, H.; Brismée, J.-M.; Sizer, P.S.; James, C.R. Relationships between Clinical Measures of Static Foot Posture and Plantar Pressure during Static Standing and Walking. Clin. Biomech. 2011, 26, 873–879. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.; Song, J.; Kraszewski, A.; Backus, S.; Ellis, S.J.; Md, J.T.D.; Hillstrom, H.J. The Effect of Foot Structure on 1st Metatarsophalangeal Joint Flexibility and Hallucal Loading. Gait Posture 2011, 34, 131–137. [Google Scholar] [CrossRef]
- Cornwall, M.W.; McPoil, T.G. Relative Movement of the Navicular Bone During Normal Walking. Foot Ankle Int. 1999, 20, 507–512. [Google Scholar] [CrossRef]
- Bolgla, L.A.; Malone, T.R. Plantar Fasciitis and the Windlass Mechanism: A Biomechanical Link to Clinical Practice. J. Athl. Train. 2004, 39, 77–82. [Google Scholar]
- Zwirner, J.; Zhang, M.; Ondruschka, B.; Akita, K.; Hammer, N. An Ossifying Bridge—On the Structural Continuity between the Achilles Tendon and the Plantar Fascia. Sci. Rep. 2020, 10, 14523. [Google Scholar] [CrossRef] [PubMed]
- Cheung, J.T.M.; Zhang, M.; An, K.N. Effect of Achilles Tendon Loading on Plantar Fascia Tension in the Standing Foot. Clin. Biomech. 2006, 21, 194–203. [Google Scholar] [CrossRef] [PubMed]
- Taunton, J.E.; Ryan, M.B.; Clement, D.B.; McKenzie, D.C.; Lloyd-Smith, D.R. Plantar Fasciitis: A Retrospective Analysis of 267 Cases. Phys. Ther. Sport 2002, 3, 57–65. [Google Scholar] [CrossRef]
- Mickle, K.J.; Angin, S.; Crofts, G.; Nester, C.J. Effects of Age on Strength and Morphology of Toe Flexor Muscles. J. Orthop. Sports Phys. Ther. 2016, 46, 1065–1070. [Google Scholar] [CrossRef]
- Tanaka, T.; Suga, T.; Imai, Y.; Ueno, H.; Misaki, J.; Miyake, Y.; Otsuka, M.; Nagano, A.; Isaka, T. Characteristics of Lower Leg and Foot Muscle Thicknesses in Sprinters: Does Greater Foot Muscles Contribute to Sprint Performance? Eur. J. Sport Sci. 2019, 19, 442–450. [Google Scholar] [CrossRef]
- Kaneda, K.; Maeda, N.; Ikuta, Y.; Tashiro, T.; Tsutsumi, S.; Arima, S.; Sasadai, J.; Suzuki, Y.; Morikawa, M.; Komiya, M.; et al. The Features of Foot Morphology and Intrinsic Foot Muscle Property in Adolescent Swimmers: An Ultrasound-Based Study. J. Hum. Kinet. 2023, 88, 95–103. [Google Scholar] [CrossRef]
- Langley, B.; Cramp, M.; Morrison, S.C. Clinical Measures of Static Foot Posture Do Not Agree. J. Foot Ankle Res. 2016, 9, 45. [Google Scholar] [CrossRef]
- Chevalier, T.L.; Hodgins, H.; Chockalingam, N. Plantar Pressure Measurements Using an In-Shoe System and a Pressure Platform: A Comparison. Gait Posture 2010, 31, 397–399. [Google Scholar] [CrossRef]
- Sánchez-Rodríguez, R.; Valle-Estévez, S.; Fraile-García, P.A.; Martínez-Nova, A.; Gómez-Martín, B.; Escamilla-Martínez, E. Modification of Pronated Foot Posture after a Program of Therapeutic Exercises. Int. J. Environ. Res. Public Health 2020, 17, 8406. [Google Scholar] [CrossRef]
- Mousavi, S.H.; Khorramroo, F.; Jafarnezhadgero, A. Gait Retraining Targeting Foot Pronation: A Systematic Review and Meta-Analysis. PLoS ONE 2024, 19, e0298646. [Google Scholar] [CrossRef]
- Boozari, S.; Jamshidi, A.A.; Sanjari, M.A.; Jafari, H. Effect of Functional Fatigue on Vertical Ground-Reaction Force in Individuals with Flat Feet. J. Sport Rehabil. 2013, 22, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Christina, K.A.; White, S.C.; Gilchrist, L.A. Effect of Localized Muscle Fatigue on Vertical Ground Reaction Forces and Ankle Joint Motion during Running. Hum. Mov. Sci. 2001, 20, 257–276. [Google Scholar] [CrossRef] [PubMed]
- Tartaruga, M.P.; Brisswalter, J.; Peyré-Tartaruga, L.A.; Alberton, C.L.; Cadore, E.L.; Tiggemann, C.L.; Silva, E.M.; Kruel, L.F.M.; Ávila, A.O.V.; Coertjens, M. The Relationship between Running Economy and Biomechanical Variables in Distance Runners. Res. Q Exerc. Sport 2012, 83, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Esmaeili, A.; Hosseininejad, S.E.; Jafarnezhadgero, A.A.; Dionisio, V.C. The Interaction Effect of Different Footwear Types and Static Navicular Drop or Dynamic Ankle Pronation on the Joint Stiffness of the Lower Limb during Running. Gait Posture 2024, 108, 28–34. [Google Scholar] [CrossRef]
- Genova, J.M.; Gross, M.T. Effect of Foot Orthotics on Calcaneal Eversion During Standing and Treadmill Walking for Subjects with Abnormal Pronation. J. Orthop. Sports Phys. Ther. 2000, 30, 664–675. [Google Scholar] [CrossRef]


| Side | Groups | |||
|---|---|---|---|---|
| HFT (n = 21) | NFT (n = 21) | Total (n = 42) | ||
| Sex (M/F) | 11/10 | 11/10 | 22/20 | |
| Age (yrs) | 24.4 ± 2.7 | 23.8 ± 3.1 | 24.1 ± 2.9 | |
| Body Mass (kg) | 71.0 ± 14.3 | 68.5 ± 9.7 | 69.8 ± 12.1 | |
| Height (m) | 1.7 ± 0.1 | 1.7 ± 0.1 | 1.7 ± 0.1 | |
| Body Mass Index (kg·m−1) | 20.5 ± 3.3 | 19.9 ± 2.3 | 20.2 ± 2.9 | |
| FPI-6 score | R | 10.6 ± 1.1 | 2.4 ± 1.5 ** | 6.5 ± 4.3 |
| L | 10.5 ± 0.9 | 2.6 ± 1.3 ** | 6.5 ± 4.2 | |
| Leg length | R | 88.7± 6.1 | 87.0 ± 4.0 | 87.8 ± 5.1 |
| L | 88.7± 6.1 | 87.0 ± 4.1 | 87.8 ± 5.2 | |
| Ankle dorsi flexion (°) | R | 53.8 ± 5.1 | 50.0 ± 6.0 * | 51.9 ± 5.8 |
| L | 53.6 ± 5.5 | 50.0 ± 5.8 * | 51.8 ± 5.9 | |
| Gait Phases | Group | Walking Conditions (Slope/Speed) | ||||
|---|---|---|---|---|---|---|
| A | B | C | D | E | ||
| −20% | −10% | 0% | +10% | +20% | ||
| 3.5 km·h−1 | 5.0 km·h−1 | 5.0 km·h−1 | 3.5 km·h−1 | 2.5 km·h−1 | ||
| Stance phase (%LL) | HFT | 62.4 ± 0.9 | 60.8 ± 0.6 a | 61.8 ± 0.9 b | 64.6 ± 0.9 d | 66.6 ± 1.0 e |
| NFT | 62.9 ± 1.1 | 60.9 ± 1.1 a | 61.7 ± 1.1 c | 64.7 ± 1.3 d | 67.5 ± 1.6 e | |
| Load response (%LL) | HFT | 12.4 ± 0.9 | 10.8 ± 0.6 a | 11.8 ± 0.9 b | 14.6 ± 0.9 d | 16.6 ± 1.0 e |
| NFT | 12.9 ± 1.1 | 10.9 ± 1.1 a | 11.7 ± 1.1 c | 14.7 ± 1.3 d | 17.5 ± 1.6 e | |
| Mid-stance (%LL) | HFT | 37.6 ± 0.9 | 39.2 ± 0.6 a | 38.2 ± 0.9 b | 35.4 ± 0.9 d | 33.4 ± 1.0 e |
| NFT | 37.1 ± 1.1 | 39.1 ± 1.1 a | 38.3 ± 1.1 c | 35.3 ± 1.3 d | 32.5 ± 1.6 e | |
| Pre-swing (%LL) | HFT | 12.4 ± 0.9 | 10.8 ± 0.6 a | 11.8 ± 0.9 b | 14.6 ± 0.9 d | 16.6 ± 1.0 e |
| NFT | 12.9 ± 1.1 | 10.9 ± 1.1 a | 11.7 ± 1.1 c | 14.7 ± 1.3 d | 17.5 ± 1.6 e | |
| Swing phase (%LL) | HFT | 37.6 ± 0.9 | 39.2 ± 0.6 a | 38.2 ± 0.9 b | 35.4 ± 0.9 d | 33.4 ± 1.0 e |
| NFT | 37.1 ± 1.1 | 39.1 ± 1.1 a | 38.3 ± 1.1 c | 35.3 ± 1.3 d | 32.5 ± 1.6 e | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kamitsou, N.; Kafetzakis, I.; Mandalidis, D. Static Foot Hyperpronation Monitoring in Asymptomatic Young Individuals During Level and Sloped Gait Using an Instrumented Treadmill. Appl. Sci. 2025, 15, 3209. https://doi.org/10.3390/app15063209
Kamitsou N, Kafetzakis I, Mandalidis D. Static Foot Hyperpronation Monitoring in Asymptomatic Young Individuals During Level and Sloped Gait Using an Instrumented Treadmill. Applied Sciences. 2025; 15(6):3209. https://doi.org/10.3390/app15063209
Chicago/Turabian StyleKamitsou, Natalia, Ioannis Kafetzakis, and Dimitris Mandalidis. 2025. "Static Foot Hyperpronation Monitoring in Asymptomatic Young Individuals During Level and Sloped Gait Using an Instrumented Treadmill" Applied Sciences 15, no. 6: 3209. https://doi.org/10.3390/app15063209
APA StyleKamitsou, N., Kafetzakis, I., & Mandalidis, D. (2025). Static Foot Hyperpronation Monitoring in Asymptomatic Young Individuals During Level and Sloped Gait Using an Instrumented Treadmill. Applied Sciences, 15(6), 3209. https://doi.org/10.3390/app15063209








