1. Introduction
Substantial quantities of radioactive contaminants are generated each year from nuclear weapon tests, nuclear facilities, and reprocessing of spent fuels due to military and industrial activities. A significant amount of soil [
1] and water [
2] is contaminated by these contaminants in some districts, which poses great risks to the environment and human health. It is known that radioactive
137Cs and
90Sr, with half-lives of 30.1 and 28.9 years, respectively, are major contaminants.
137Cs is an analog of potassium that is easily incorporated in living organisms [
3], and
90Sr can be introduced into the food chain, which seriously threatens human health [
4]. They also have an adverse impact on ecology [
5,
6]. Thus, removal of radioactive contaminants from contaminated water has become a significant environmental matter in recent years. In addition, the removed
137Cs and
90Sr isotopes have potential application as gamma radiation sources for medical and industrial applications [
7,
8]. Accordingly, further research for feasible separation processes to recover Cs(I) and Sr(II) is required.
Solvent extraction has been proved to be one of the most effective partitioning and recovery techniques from the viewpoints of speed, simplicity and easiness of scaling up. This method has been used for the removal of Cs(I) and Sr(II) for long time. Thus, the selection of the extractants plays an important role in the extraction process, and can greatly affect the separation efficiency. In the past few decades, some conventional and newly developed extractants have been used to study solvent extraction of Cs(I) and Sr(II). Crown ethers, such as 4,5″-bis(
tert-octylbenzo)dibenzo-24-crown-8 [
9], dibenzo-21-crown-7 (DB21C7) [
10], and 4′,4′,(5′)-di-(
tert-butylcyclohexano)-18-crown-6 (DtBuCH18C6) [
11]; calixarene-crown ethers, such as 1,3-alternate calix[4]arene-crown-6 conformers [
12], calix[4]dibenzocrowns-6 [
13], calix[4]arene-bis(naphthocrown-6) [
14], 25,27-bis(isopropoxy) calix[4]-26,28-crown-6 (iPr-C[4]C-6) [
15], calix[4]arene-bis-(
tert-octylbenzo-crown-6) (BOBCalixC6) [
16,
17], and acryl-calix[4]-crown-6 [
18]; and amide agents, such as
N,
N,
N,
N-tetraoctyl-3-oxapentanediamide (TODGA) [
19], are potent extractants for separating Cs(I) or Sr(II) from acid solutions. Bis(2-propyloxy)calix[4]crown-6 (BPC6) [
20], calix[4]arene-bis(crown-6-ethers) [
21,
22], and 4-
tert-butyl-2-(α-methylbenzyl) phenol (
t-BAMBP) [
23] have shown excellent extraction ability for Cs(I) from aqueous solutions. In addition, di-2-ethyl hexyl phosphoric acid (D2EHPA) [
24] has been proved to be feasible for the separation of Sr(II) from alkaline solutions. The abovementioned studies showed that crown ethers are generally selective for Sr(II), and calixarene-crown ethers are mainly selective for Cs(I). Extractants that are selective for both Cs(I) and Sr(II) in an aqueous solution have not been reported yet. Using a single extractant with high selectivity for both Cs(I) and Sr(II) can simplify the treatment of wastewater. Therefore, an agent with the ability to extract them efficiently is highly desired.
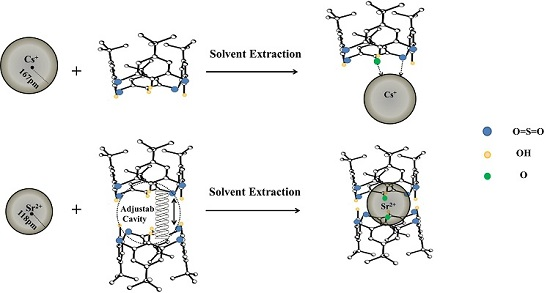
Calixarenes have been regarded as molecular platforms or building blocks for the construction of metal ion carriers, which is an attractive motif in coordination chemistry [
25,
26]. The extraction process of calixarenes for industrial applications is extremely important [
27]. Classical CH
2-bridged calixarenes were studied in cations extraction and transport [
28,
29], and calixarene amide ionophores for strontium extraction were also researched [
30]. In our present work, low-cost
p-
tert-butylsulfonylcalix[4]arene (SC4A), possessing the ability to extract Cs(I) and Sr(II) efficiently, was investigated. In order to research the selectivity of SC4A for the target ions and the combined form of the extracted complex, Na(I), K(I), Rb(I), Mg(II), Ca(II), Ba(II), Pb(II), Cd(II), Zn(II), Co(II), Ni(II), Ce(III), and Y(III) were also extracted with SC4A for comparison. Meanwhile,
p-
tert-butylthiacalix[4]arene (TC4A) and
p-
tert-butylsulfonylcalix[4]arene tetraacetate (SC4AOEt) were applied to extract Cs(I) or Sr(II) to examine the effects of the OH and SO
2 groups on the extraction. The composition of the complex, thermodynamic data for the extraction procedure, extraction mechanism, irradiation stability, and high-temperature resistance of SC4A were also researched.
2. Materials and Methods
2.1. Reagents
Alkali metals nitrates MINO3 (MI = Na, K, Rb, and Cs); alkaline earth nitrates MII(NO3)2 (MII = Sr, Ba), Ca(NO3)2·4H2O, and Mg(NO3)2·6H2O; rare earth metal nitrates MIII(NO3)3·6H2O (MIII = Ce and Y); and heavy metals nitrates Cd(NO3)2·4H2O, Zn(NO3)2·6H2O, Co(NO3)2·6H2O, Ni(NO3)2·6H2O and Pb(NO3)2, were of analytical grade. Salt solutions in nitric acid medium were prepared by dissolving the required weights of the above nitrates in nitric acid solutions. Ultrapure water was used for preparation of nitrate solutions. The desired pH was achieved by injecting negligible volumes of HNO3 or NaOH. An IS128 pH meter was used to measure the adjusted pH.
SC4A and its raw material, TC4A (
Figure 1), were synthesized in our laboratory according to a previously reported procedure [
31] with modification. In the general synthesis procedure of SC4A, 25 mL of acetic acid and 2.57 g (16.71 mmol) of NaBO
3·4H
2O were added to a solution of 1.8 g (2.48 mmol) of TC4A in 30 mL of chloroform. After the mixture was stirred at 55 °C for four days, the product was extracted with chloroform (100 mL × 3). The extract liquor was dried with anhydrous magnesium sulfate for 2 h, and then filtered and evaporated. Then, the white SC4A powder was recrystallized from ethanol and dried under vacuum. SC4AOEt (
Figure 1) was prepared following a reported procedure [
32]. Detail synthesis procedure of TC4A (
Figure S1), SC4A (
Figure S2), and SC4AOEt (
Figure S3), and characterization data of them are described in
Supplementary Materials. Analytical grade chloroform was used as a diluent to dissolve SC4A, TC4A, and SC4AOEt for the extraction process.
2.2. Extraction Procedure
Equal volumes (5 mL) of organic phase and aqueous phase containing the elements were mixed in a conical flask and horizontally shaken at 150 rpm for 60 min in a thermostated water bath maintained at 298 ± 0.5 K unless otherwise stated. Sequentially, the two phases were centrifuged at 4000 rpm for 5 min. Then, the concentrations of the ions were measured using a Nexion 300Q model inorganic mass spectrometer (ICP-MS) (PerkinElmer, Boston, MA, USA). The distribution ratio (
DM) of element M was calculated using the following equation:
where
C(o) and
C(a) represent the equilibrium concentrations of the tested ions in the organic and aqueous phase, respectively.
The separation factors of Cs(I) and Sr(II) (
SFCs and
SFSr) to other ions were, respectively, calculated as follows:
2.3. Irradiation Procedure and Thermogravimetric Analysis
Irradiations of SC4A and SC4A/CHCl3 were carried out at 25 ± 0.2 °C in air using a 60Co radiation source (China institute of atomic energy, Beijing, China) with a total absorbed dose ranging from 1.0 × 104 to 1.1 × 106 Gy.
Weighed quantities of SC4A were subjected to thermogravimetric analysis (TGA) in a nitrogen atmosphere using a TG/DTA6200 instrument (Seiko instruments inc, Tokyo, Japan) at a heating rate of 10 °C·min−1.
The structure of SC4A was analyzed using a Nicolet iS10 infrared (IR) spectrometer (Thermo Fisher, Waltham, MA, USA), Bruker DPX-400 nuclear magnetic resonance (1H-NMR) spectrometer (Bruker, Karlsruhe, Germany), and Trace DSQ electrospray ionization mass spectrometry (ESI-MS) instrument (Thermo Fisher, Waltham, MA, USA).