Red Blood Cell Aging as a Homeostatic Response to Exercise-Induced Stress
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Red blood Cell Fractionation
2.3. Deformability, Osmotic Fragility, and Aggregation
2.4. Flow Cytometry
2.5. Isolation and Characterization of Microvesicles
2.6. Statistical Analysis
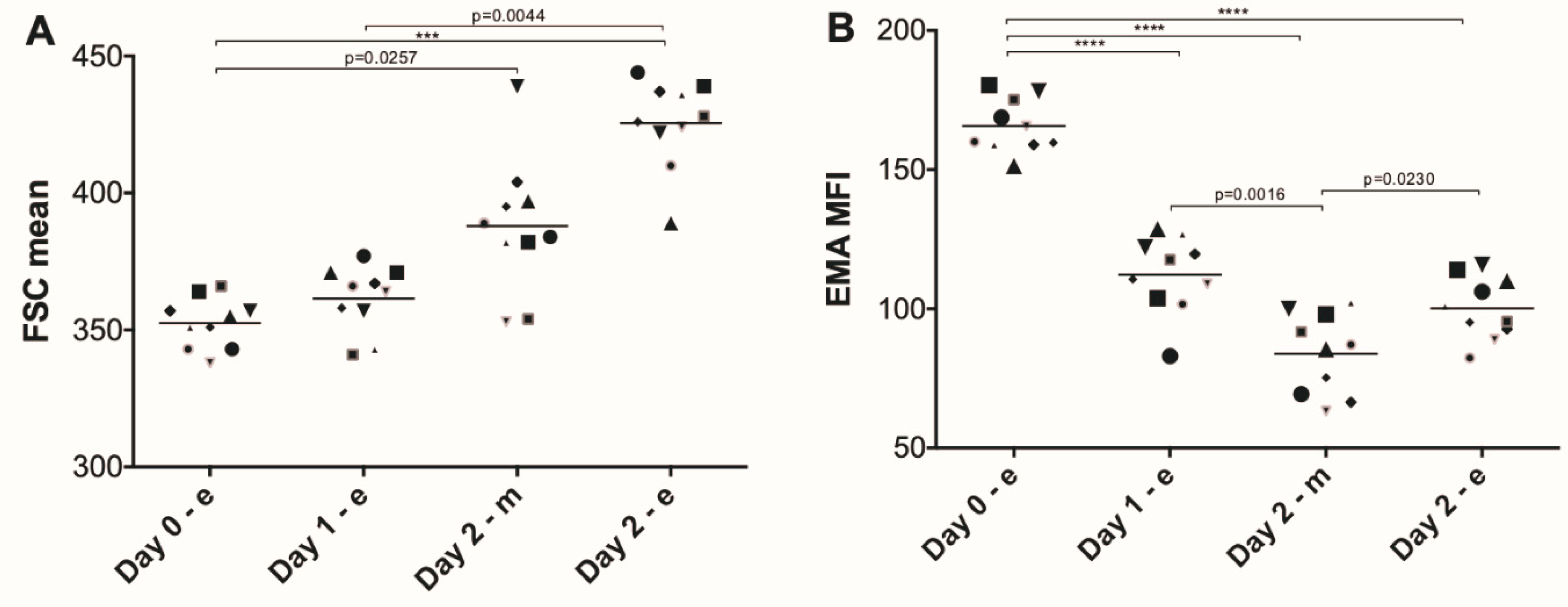
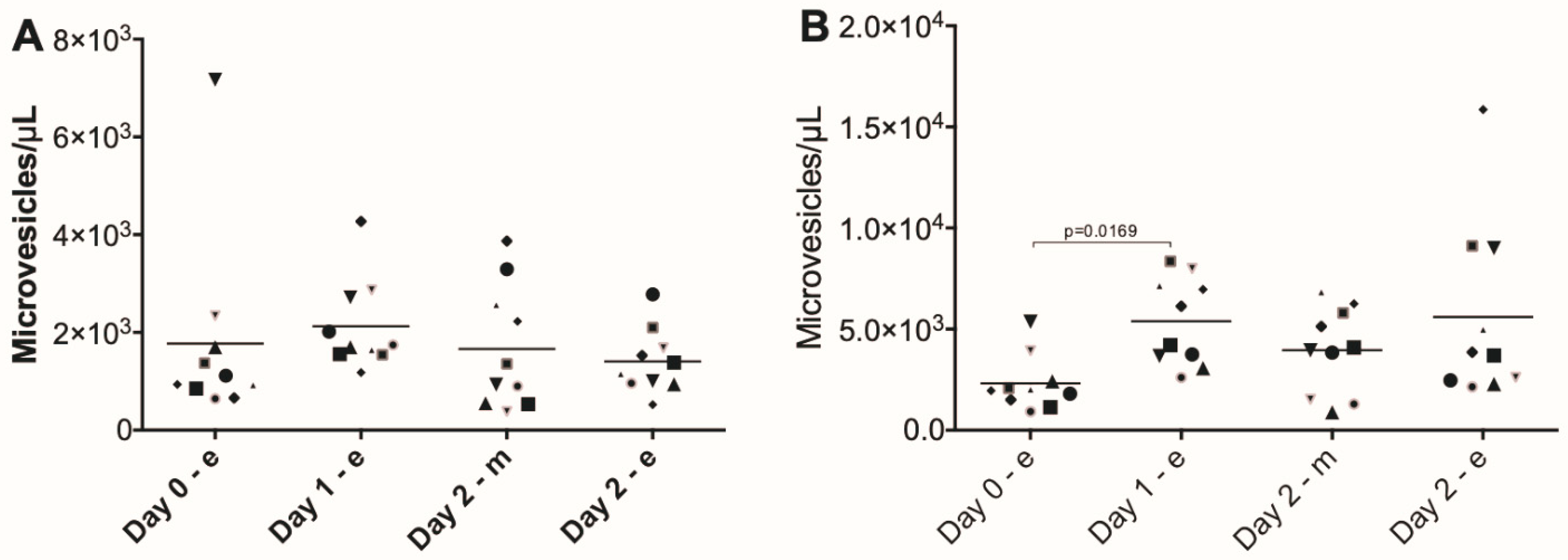
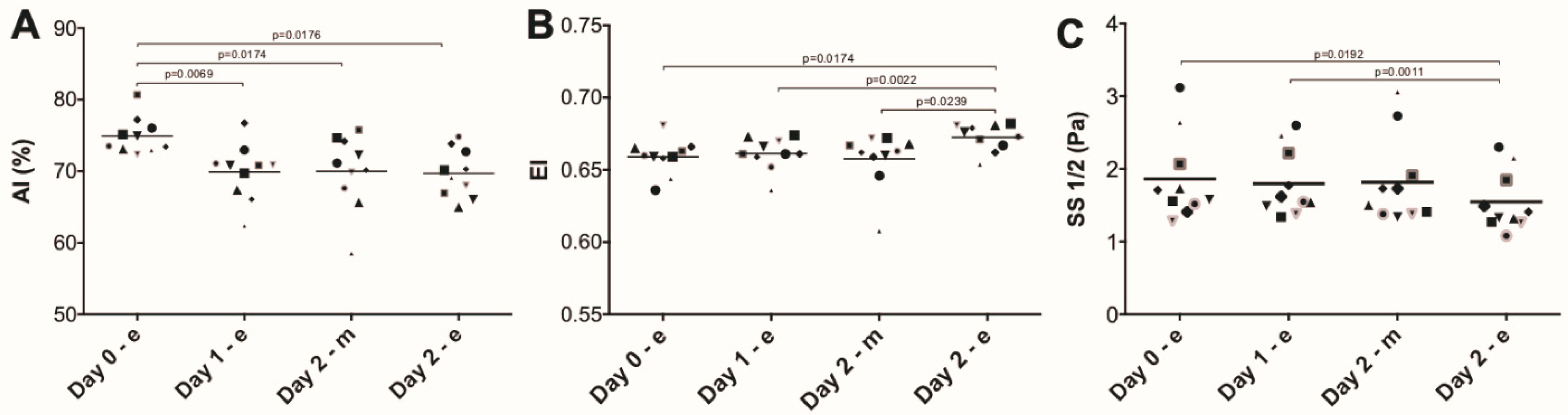
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bosman, G.J.; Cluitmans, J.C.A.; Groenen, Y.A.M.; Werre, J.M.; Willekens, F.L.A.; Novotný, V.M.J. Susceptibility to hyperosmotic stress-induced phosphatidylserine exposure increases during red blood cell storage. Transfusion 2011, 51, 1072–1078. [Google Scholar] [CrossRef] [PubMed]
- Gwozdzinski, K.; Pieniazek, A.; Tabaczar, S.; Jegier, A.; Brzeszczynska, J. Investigation of oxidative stress parameters in different lifespan erythrocyte fractions in young untrained men after acute exercise. Exp. Physiol. 2017, 102, 190–201. [Google Scholar] [CrossRef] [PubMed]
- Ghashghaeinia, M.; Cluitmans, J.C.A.; Akel, A.; Dreischer, P.; Toulany, M.; Köberle, M.; Skabytska, Y.; Saki, M.; Biedermann, T.; Duszenko, M.; et al. The impact of erythrocyte age on eryptosis. Br. J. Haematol. 2012, 157, 606–614. [Google Scholar] [CrossRef] [PubMed]
- Dinkla, S.; Wessels, K.; Verdurmen, W.P.R.; Tomelleri, C.; Cluitmans, J.C.A.; Fransen, J.; Fuchs, B.; Schiller, J.; Joosten, I.; Brock, R.; et al. Functional consequences of sphingomyelinase-induced changes in erythrocyte membrane structure. Cell Death Dis. 2012, 3, e410–e412. [Google Scholar] [CrossRef] [PubMed]
- Dinkla, S.; van Eijk, L.T.; Fuchs, B.; Schiller, J.; Joosten, I.; Brock, R.; Pickkers, P.; Bosman, G.J.C.G.M. Inflammation-associated changes in lipid composition and the organization of the erythrocyte membrane. BBA Clin. 2016, 5, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Bosman, G.J. The involvement of erythrocyte metabolism in organismal homeostasis in health and disease. Proteom. Clin. Appl. 2016, 10, 774–777. [Google Scholar] [CrossRef] [PubMed]
- Bosman, G.J. Disturbed red blood cell structure and function: An exploration of the role of red blood cells in neurodegeneration. Front. Med. 2018, 5, 198. [Google Scholar] [CrossRef] [PubMed]
- Peeling, P.; Dawson, B.; Goodman, C.; Landers, G.; Wiegerinck, E.T.; Swinkels, D.W.; Trinder, D. Cumulative effects of consecutive running sessions on hemolysis, inflammation and hepcidin activity. Eur. J. Appl. Physiol. 2009, 106, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.; Dawson, B.; Peeling, P. Hemolytic effects of a football-specific training session in elite female players. Int. J. Sports Physiol. Perform. 2012, 7, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Tomschi, F.; Niemann, D.; Bloch, W.; Predel, H.-G.; Grau, M. Ischemic preconditioning enhances performance and erythrocyte deformability of responders. Int. J. Sports Med. 2018, 39, 596–603. [Google Scholar] [CrossRef] [PubMed]
- Bongers, C.C.W.G.; Alsady, M.; Nijenhuis, T.; Hartman, Y.A.W.; Eijsvogels, T.M.H.; Deen, P.M.T.; Hopman, M.T.E. Impact of acute versus repetitive moderate intensity endurance exercise on kidney injury markers. Physiol. Rep. 2017, 5, e13544. [Google Scholar] [CrossRef] [PubMed]
- Wouters, H.; Aalbers, T.; Maessen, M.F.H.; Verbeek, A.L.M.; Rikkert, M.G.M.O.; Kessels, R.P.C.; Hopman, M.T.E.; Eijsvogels, T.M.H. Physical activity and cognitive function of long-distance walkers: Studying Four Days Marches participants. Rejuvenation Res. 2017, 20, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Terink, R.; Ten Haaf, D.; Bongers, C.W.G.; Balvers, M.G.J.; Witkamp, R.F.; Mensink, M.; Eijsvogels, T.M.H.; Gunnewiek, J.M.T.K.; Hopman, M.T.E. Changes in iron metabolism during prolonged repeated walking exercise in middle-aged men and women. Eur. J. Appl. Physiol. 2018, 118, 2349–2357. [Google Scholar] [CrossRef] [PubMed]
- Verheggen, R.J.H.M.; Eijsvogels, T.M.H.; Catoire, M.; Terink, R.; Ramakers, R.; Bongers, C.C.W.G.; Mensink, M.; Hermus, A.R.M.M.; Thijssen, D.H.J.; Hopman, M.T.E. Cytokine responses to repeated, prolonged walking in lean versus overweight/obese individuals. J. Sci. Med. Sport 2019, 22, 196–200. [Google Scholar] [CrossRef] [PubMed]
- Freitas Leal, J.K.; Adjobo-Hermans, M.J.W.; Brock, R.; Bosman, G.J. Acetylcholinesterase provides new insights into red blood cell ageing in vivo and invitro. Blood Transfus. 2017, 15, 232–238. [Google Scholar] [PubMed]
- Willekens, F.L.A.; Werre, J.M.; Groenen-Döpp, Y.A.M.; de Pauw, B.R.-S.B.; Bosman, G.J.C.G.M. Erythrocyte vesiculation: A self-protective mechanism? Br. J. Haematol. 2008, 141, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Cluitmans, J.C.A.; Hardeman, M.R.; Dinkla, S.; Brock, R.; Bosman, G.J. Red blood cell deformability during storage: Towards functional proteomics and metabolomics in the blood bank. Blood Transfus. 2012, 10, 12–18. [Google Scholar]
- Dinkla, S.; Peppelman, M.; De Raadt, J.; Atsma, F.; Novotny, V.M.; Van Kraaij, M.G.; Joosten, I.; Bosman, G.J. Phosphatidylserine exposure on stored red blood cells as a parameter for donor-dependent variation in product quality. Blood Transfus. 2014, 12, 204–209. [Google Scholar] [PubMed]
- Bergmann-Leitner, E.S.; Leitner, W.W.; Tsokos, G.C. Complement 3d: From molecular adjuvant to target of immune escape mechanisms. Clin. Immunol. 2006, 121, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Ovchynnikova, E.; Aglialoro, F.; von Lindern, M.; van den Akker, E. The shape shifting story of reticulocyte maturation. Front. Physiol. 2018, 9, 829. [Google Scholar] [CrossRef] [PubMed]
- Cobb, C.E.; Beth, A.H. Identification of the eosinyl-5-maleimide reaction site on the human erythrocyte anion-exchange protein: Overlap with the reaction sites of other chemical probes. Biochemistry 1990, 29, 8283–8290. [Google Scholar] [CrossRef] [PubMed]
- Crisp, R.L.; Solari, L.; Vota, D.; García, E.; Miguez, G.; Chamorro, M.E.; Schvartzman, G.A.; Alfonso, G.; Gammella, D.; Caldarola, S.; et al. A prospective study to assess the predictive value for hereditary spherocytosis using five laboratory tests (cryohemolysis test, eosin-5′-maleimide flow cytometry, osmotic fragility test, autohemolysis test, and SDS PAGE) on 50 hereditary spherocytosis families in Argentina. Ann. Hematol. 2011, 90, 625–634. [Google Scholar] [PubMed]
- Dinkla, S.; Brock, R.; Joosten, I.; Bosman, G.J. Gateway to understanding microparticles: Standardized isolation and identification of plasma membrane-derived vesicles. Nanomedicine 2013, 8, 1657–1668. [Google Scholar] [CrossRef] [PubMed]
- Freitas Leal, J.K.; Adjobo-Hermans, M.J.W.; Bosman, G.J. Red blood cell homeostasis: Mechanisms and effects of microvesicle generation in health and disease. Front. Physiol. 2018, 9, 703. [Google Scholar] [CrossRef] [PubMed]
- Weisel, J.W.; Litvinov, R.I. Red blood cells: The forgotten player in hemostasis and thrombosis. J. Thromb. Haemost. 2019, 17, 271–282. [Google Scholar] [CrossRef] [PubMed]
- Filar-Mierzwa, K.; Marchewka, A.; Dąbrowski, Z.; Bac, A.; Marchewka, J. Effects of dance movement therapy on the rheological properties of blood in elderly women. Clin. Hemorheol. Microcirc. 2019, 67, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-C.; Hsu, C.-C.; Wang, J.-S. High-Intensity Interval Training Improves Erythrocyte Osmotic Deformability. Med. Sci. Sports Exerc. 2019, 51, 1404–1412. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.A.; Martin, D.T.; Telford, R.D.; Ballas, S.K. Greater erythrocyte deformability in world-class endurance athletes. Am. J. Physiol. 1999, 276, H2188–H2193. [Google Scholar] [CrossRef] [PubMed]
- Suhr, F.; Brenig, J.; Müller, R.; Behrens, H.; Bloch, W.; Grau, M. Moderate exercise promotes human RBC-NOS activity, NO production and deformability through Akt kinase pathway. PLoS ONE 2012, 7, e45982. [Google Scholar] [CrossRef] [PubMed]
- Simmonds, M.J.; Meiselman, H.J.; Baskurt, O.K. Blood rheology and aging. J. Geriatr. Cardiol. 2013, 10, 291–301. [Google Scholar] [PubMed]
- Lutz, H.U.; Bogdanova, A. Mechanisms tagging senescent red blood cells for clearance in healthy humans. Front. Physiol. 2013, 4, 387. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, J.G.; Nagababu, E.; Rifkind, J.M. Red blood cell oxidative stress impairs oxygen delivery and induces red blood cell aging. Front. Physiol. 2014, 5, 84. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Willekens, F.L.A.; Werre, J.M.; Kruijt, J.K.; Roerdinkholder-Stoelwinder, B.; Groenen-Döpp, Y.A.; van den Bos, A.G.; Bosman, G.J.; van Berkel, T.J. Liver Kupffer cells rapidly remove red blood cell-derived vesicles from the circulation by scavenger receptors. Blood 2005, 105, 2141–2145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Konig, D.; Berg, A.; Weinstock, C.; Keul, J.; Northoff, H. Essential fatty acids, immune function, and exercise. Exerc. Immunol. Rev. 1997, 3, 1–31. [Google Scholar] [PubMed]
- Nieman, D.C.; Groen, A.J.; Pugachev, A.; Vacca, G. Detection of functional overreaching in endurance athletes using proteomics. Proteomes 2018, 6, 33. [Google Scholar] [CrossRef] [PubMed]
- Miron, V.V.; Baldissarelli, J.; Pranke, G.; Mânica, A.; da Silva Rosa Bonadiman, B.; Manfredi, L.H.; Moesch, V.M.; Schetinger, M.R.C.; Cardoso, A.M. High-intensity intermittent exercise increases adenosine hydrolysis in platelets and lymphocytes and promotes platelet aggregation in futsal athletes. Platelets 2019, 30, 878–885. [Google Scholar] [CrossRef] [PubMed]
- Anz, A.W.; Parsa, R.S.; Romero-Creel, M.F.; Nabors, A.; Tucker, M.S.; Harrison, R.M.; Matuska, A.M. Exercise-mobilized platelet-rich plasma: Short-term exercise increases stem cell and platelet concentrations in platelet-rich plasma. Arthroscopy 2019, 35, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Weiss, G.; Ganz, T.; Goodnough, L.T. Anemia of inflammation. Blood 2019, 33, 40–50. [Google Scholar] [CrossRef] [PubMed] [Green Version]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Freitas Leal, J.K.; Lazari, D.; Bongers, C.C.W.G.; Hopman, M.T.E.; Brock, R.; Bosman, G.J.C.G.M. Red Blood Cell Aging as a Homeostatic Response to Exercise-Induced Stress. Appl. Sci. 2019, 9, 4827. https://doi.org/10.3390/app9224827
Freitas Leal JK, Lazari D, Bongers CCWG, Hopman MTE, Brock R, Bosman GJCGM. Red Blood Cell Aging as a Homeostatic Response to Exercise-Induced Stress. Applied Sciences. 2019; 9(22):4827. https://doi.org/10.3390/app9224827
Chicago/Turabian StyleFreitas Leal, Joames K., Dan Lazari, Coen C.W.G. Bongers, Maria T.E. Hopman, Roland Brock, and Giel J.C.G.M. Bosman. 2019. "Red Blood Cell Aging as a Homeostatic Response to Exercise-Induced Stress" Applied Sciences 9, no. 22: 4827. https://doi.org/10.3390/app9224827
APA StyleFreitas Leal, J. K., Lazari, D., Bongers, C. C. W. G., Hopman, M. T. E., Brock, R., & Bosman, G. J. C. G. M. (2019). Red Blood Cell Aging as a Homeostatic Response to Exercise-Induced Stress. Applied Sciences, 9(22), 4827. https://doi.org/10.3390/app9224827





