1. Introduction
Breast cancer is the most commonly diagnosed cancer in women worldwide. With more than 1 million cases in 2012 [
1], it is one of the most common causes of cancer-related death in women [
2]. It is estimated that 1 woman in 8 will develop breast cancer during her life, but this estimation varies by country; in Italy for example, 50,000 new cases are discovered each year [
3,
4].
The incidence of breast cancer is strongly related to age with the highest incidence rates being in menopausal women. Breast cancer in women under 40 years is not a common condition; however, the increase of incidence in premenopausal women is particularly alarming. Premenopausal women are not commonly covered by national screening programs, and often a mammography cannot be performed because of the high density of their breast tissue [
5,
6,
7,
8]. Breast cancer can be suspected under different circumstances, such as a positive screening mammography or echography, discovery by palpation of a mass in the breast, or any morphological modification of the breast. Currently, the most widely used clinical diagnostic method is mammography. Even though mammography screening has been successful in reducing mortality, in particular in women over 50, some negative aspects related to cost, the availability of trained staff, the duration of the test, and the non-applicability to all women must be considered. In fact, despite being the gold standard of diagnostic techniques, mammography cannot be used on pregnant women because of ionizing radiation. This technique is also ineffective in young dense breasts [
7]. Moreover, as a routine diagnostic tool, mammography is costly, placing an economic load on already overburdened healthcare systems. There is, therefore, a need for low-cost, reliable early detection method for breast cancer, particularly in the younger population.
Currently, several products are available in the market in support of early detection. They are generally used for clinical breast examination (CBE) by physicians to perform their work and hence are not wearable [
9,
10]. These devices, through tactile imaging, take advantages of the different mechanical properties of the tissues in order to detect anomalies located inside the breast. Tactile imaging is a new emerging diagnostic technique based on visualizing the sense of touch. Biological tissues are characterized by specific mechanical properties, which change in the course of the lump’s development. Using these features, it is possible to create pressure maps in relation to the direction of tissue deformation. This is because the pressure response corresponding to areas with abnormalities is higher than the healthy tissue. Considering the effectiveness of this simple and risk-free methodology, several devices imitate the haptic process performed during palpation using different types of technologies, such as ultrasound or pressure sensors. Among the advantages of using tactile sensors instead of human touch are their greater reliability and repeatability [
10,
11,
12,
13].
According to a study performed by Egorov et al. [
10], tactile imaging showed a sensitivity of 91.4% and specificity of 86.1% on a sample of 32 malignant lesions and 147 benign ones. These results are comparable to the effectiveness of other diagnostic techniques but tactile imaging has several advantages, such as ease of use, portability, absence of radiation, and lower costs. The low cost is, above all, what makes the procedure interesting as a method for screening particularly in countries with limited resources where high technology techniques are not available.
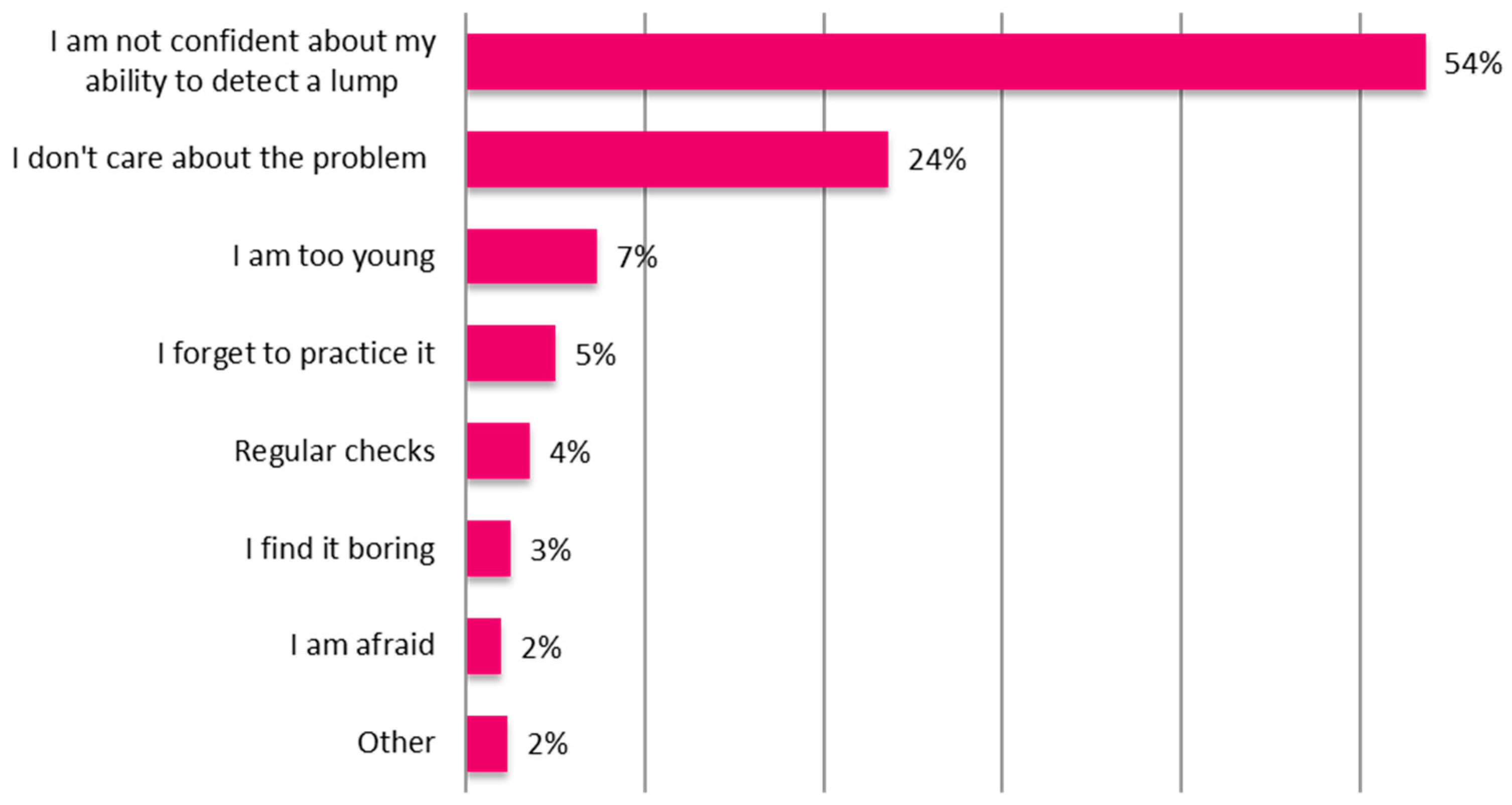
Breast self-examination (BSE) [
14] is the most common method of early-stage breast cancer detection. The sensitivity of BSE is related to significant changes in mechanical properties of tissue in the course of cancer development. BSE, or regularly examining one’s own breasts, could be used to find early signs of breast cancer, when it is more likely to be treated successfully. It is a simple non-invasive procedure that can be performed by all women, allowing them to become comfortable with their own bodies [
15]. Self-palpation should be performed once a month, several days after the beginning of menstruation when the breasts are softer. A woman palps her breast with the pads of her finger to detect either superficial or deeper lumps. The breast is assumed to be divided into four quadrants, each of which is checked separately. Several common patterns are designed to ensure complete coverage of the breast also including “the axillary tail” (the piece of breast tissue which extends under the armpit) [
14].
A number of wearable systems in support of BSE are already patented and commercialized. Among these, thermal technology, which estimates thermal maps of the breast using infrared sensors, is the most common. Other examples of intelligent bras show additional parameters such as the differences between oxygenated and non-oxygenated hemoglobin, and the evaluation of the levels of oxygenation, which have a different distribution in healthy and diseased tissues [
16,
17,
18,
19]. Diseased tissue is in fact characterized by a higher metabolism and therefore develops areas with higher temperature.
In this study, we present a non-invasive wearable device named Palpreast to simulate breast self-examination using pressure sensing textiles. As far as we know, the use of the tactile of technology on a wearable device to provide a tool for home screening has not been proposed, as all current devices are based on different techniques (heat and oxygen levels). Palpreast is not conceived to be a diagnostic tool but as a support that, in combination with other screening methods, can increase the odds of early detection for better prognosis.
In the following sections, we outline the results of a survey conducted to better understand the reasons why women do not practice BSE. The device concept is introduced, and then the finite element (FE) analysis of the mechanics underlying the working principle is presented. The physical implementation of the device and its characterization are discussed. Finally, the proposed solution is validated using a breast phantom with an embedded inclusion.
3. Device Concept
Palpreast is a wearable device similar to a stretchy bra (
Figure 3a), with an internal pocket, adaptable to breasts with different shapes and sizes. A pressure sensing textile responsible for nodule detection is located under the stretchy top, in contact with the skin (
Figure 3b). An inflation system, composed of four independent air compartments centered on the breast, is located in the inner part of the top (see
Figure 3c).
The working principle of the device is based on a pressure sensing textile able to distinguish tissue stiffness, thus differentiating between healthy and abnormal tissue. The textile covers the breast and the inflator system separately inflates and deflates each of the four compartments allowing the sensing textile to adhere sequentially to the breast, simulating the process of self-examination. An intuitive graphical interface presents the result of the difference in stiffness region-by-region between the left and right breast, which can be an indicator of the presence of a malignant nodule (considering that it is highly improbable to have two identical nodules in the left and right breast, with the same stiffness and in the same position). With this strategy, the contralateral breast constitutes the internal control, without the need for further calibration. According to this conceptual design, the proposed wearable device, as explained in
Section 1, is not intended to be a diagnostic tool, but is conceived as a support for BSE promoting the detection of the early stage of disease for improving its prognosis. The device was developed according to the approach proposed by the EU funded UBORA project [
20,
21], which aimed at developing open source medical devices compliant to European Medical Device Regulation 2017/745.
4. Finite Element Analysis
A finite element (FE) analysis of a breast model was performed to investigate its response to a compression load, in order to demonstrate the feasibility of the idea and determine the technical specifications to design the prototype of Palpreast, e.g., the spatial resolution of the pressure sensing textile necessary to identify the presence of a nodule.
Mechanical properties of tissues are highly sensitive to the structural changes of various physiological and pathological processes. Thus, the characterization of embedded lesions in terms of mechanical properties (such as stiffness and size) provides a means for distinguishing them from diseased tissues. Although women have different breast thickness, shape, and stiffness, the FE analysis is based on a simplified hemispherical breast model characterized by average mechanical properties and a reduced number of details (e.g., heterogeneous tissues). This choice derives from the device concept, which is based on the evaluation of the differences in stiffness between the right and left breast—healthy heterogeneous tissues present in both breasts should have the same behavior, whereas there may be substantial differences between healthy and diseased tissues of the same person.
In order to define the Young’s modulus of the breast and the nodule, we took into account the study of Egorov et al. [
12], which was based on the application of a similar technology and on in vivo studies on patients. According to this study, the Young’s modulus of healthy breast tissue was 7 kPa, calculated as the mean value of clinical data of six patients involved in the study (6.9 ± 1.4 kPa). Tumors are in general stiffer than healthy tissue, and it is well known that the Young’s modulus increases with the malignancy of a tumor [
22,
23]. In this research, we performed a parametric analysis with respect to the Young’s modulus of the inclusion E
nod, in the range 50 to 125 kPa with a step of 25 kPa, according to the in vivo study performed in [
12], where the elastic modulus of inclusions increased from 50 kPa (benign fibrocystic tissue) to 123 kPa (ductal carcinoma).
Two-dimensional (2D) plane strain and three-dimensional (3D) brick models were implemented under the following assumptions:
The biological tissue and inclusions were homogeneous, linear elastic, and isotropic;
The Poisson’s ratio of each material is 0.45, i.e., the breast could be considered as an almost incompressible material;
The breast was assumed to be placed on a non-deformable hard surface (identifiable with the rib cage), with a no-displacement constraint as boundary condition;
The density of each material was set to 1000 kg/m3 (close to the density of water);
Two different breast sizes were investigated—large breast (LB) and medium breast (MB);
The ideal breast tissue has a hemispherical shape, whereas the nodule has a spherical shape, whose dimensions are indicated in
Figure 4 and
Table 1 for 2D model, and
Figure 5 and
Table 2 for the 3D model;
A pressure of 10 kPa was considered to simulate breast palpation.
All the FE simulations were modeled using COMSOL Multiphysics 3.5.
In order to simulate the behavior of breast tissue with better accuracy, a FE analysis of an idealized 3D breast model (
Figure 5) was performed considering the parameters described in
Table 2. The dimensions were the same as the 2D models described in
Table 1.
Figure 6 represents the total displacement in response to an external pressure on the 2D breast model—according to the model, the maximum difference in displacement between healthy and abnormal breast tissue ranges from 2.5 mm up to 3 mm depending on the Young’s modulus of the inclusion.
Figure 7 highlights the total displacement of boundary #1 of the 2D breast model for different positions of the inclusion—the position of the maximum difference in displacement between a healthy and abnormal breast changes with the position of the inclusion, and reaches a higher value when the inclusion is closer to the surface of the breast.
The 3D FE model was used to evaluate the spatial resolution necessary to clearly identify the presence of the inclusion (
Figure 8).
According to the 3D FE model, differences in the Von Mises stress between the healthy and abnormal tissue (
Figure 8b,c) indicated that a sensor matrix able to detect a change in pressure of 600 Pa over a distance of 5 mm was sufficient to detect the presence of a stiffer inclusion of 10-mm diameter located inside a breast. The analysis also suggested that as the size of breast increased, the pressure gradient due to inclusion decreased (lower variation), i.e., detecting an inclusion in a smaller breast was easier than in a larger one.
8. Conclusions
We present a wearable device—Palpreast—for breast self-examination based on tactile imaging. The device was modelled using FE analysis, and then designed, assembled, and tested using breast phantoms. It consists of a stretchy top which covers both breasts, with internal layers consisting of (a) wearable sensors (n.b., although the prototype is constructed using piezo resistive fabric, other pressure or displacement sensing materials (e.g., piezo capacitive) could also be used in the device); (b) a compartment containing sub-compartments which can be selectively inflated by applying a precise pressure to different zones of the breast. Alternate inflation of each subcompartment mimics the action of breast palpation and the resulting haptic feedback detected by the wearable sensors gives an indication of the difference in stiffness between the left and right breast, which are correlated to the size and location of nodules; (c) an intelligent interface for analysis of differences in stiffness between the left and right breast giving appropriate alerts to users to consult a doctor.
Results from the calibration of the system indicate that it is highly reproducible although we did observe some crosstalk between taxels. More sophisticated data analysis, using, for example, a neural network, could overcome this limitation and even take advantage of this crosstalk [
25]. Despite this limit in resolution, the device in its current form is able detect the presence of a 2 cm nodule in a breast phantom.
As Palpreast can be classified as a Class I medical device according to Medical Device Regulation 2017/745 (low risk, not being a diagnostic tool), it does need to undergo clinical trials. However, further improvements on the FE analysis using reconstructed 3D images of real breasts would be useful to understand how non-symmetrical shapes of the breast or irregular contours of the nodule may influence the resolution of the system, and thus if a sensor pad with smaller and more densely packed taxels is required. In addition, miniaturization of the pneumatic system as well as preliminary tests on a number of volunteers is necessary for the device to be marketed as a support for personal screening in women.