1. Introduction
Water quality monitoring at remote field sites often entails the collection of water samples using automated water samplers and periodic retrieval of the water samples for laboratory analysis. Therefore, one of the most important issues for researchers dealing with water monitoring is how to preserve water samples while stored in the automated water sampler while awaiting retrieval. Water quality parameters that have the potential to change over time include nitrate-N and orthophosphate. The United States Environmental Protection Agency (US EPA) suggests methods to help obtain an accurate reading of concentrations in water samples. This includes preserving the sample immediately after on-site collection by placing samples in cool temperatures, adding chemical solutions, or altering the pH of the sample [
1]. The US EPA recommended methods for preserving water samples for nitrate-N and orthophosphate are listed in
Table 1.
These preservation methods slow factors that can lead to changes in water quality concentrations. By limiting biological activity, chemical hydrolysis, and absorption effects [
1], there can be a longer time delay between collecting the sample and analyzing it. Several chemical and physical interferences have been used as potential preservation methods. The most traditional forms include chemical addition, pH control, freezing, and refrigerating the sample [
1]. For example, chloroform has been explored as a preservation but produced erratic results for both nitrate-N and orthophosphate, with large increases in concentrations occurring between eight and 16 days after preservation was initiated [
2,
3].
One of the main challenges with preservation methods is length of time when the sample is stabilized, or preserved. There is often only a short period of time before concentrations within the sample begin to change. For example, in a study where water samples were not filtered immediately after collection (10-day delay) and then stored in refrigerated conditions for 107 days, both nitrate-N and orthophosphate showed significant increases, by as much as a 3.7-fold increase for nitrate-N and a 1.8-fold increase for orthophosphate [
4].
In other studies [
5,
6], it was determined that, while methods such as those listed in
Table 1 can be effective for one target parameter, it is difficult, if not impossible, to find a method that is effective for preserving multiple parameters. The variability in concentrations within a water sample over time can be caused by factors like location, chemical make-up of the water, and the microbiology of the water. As a result, the ultimate problem with a universal preservation method is there are too many variables that can influence changes. These variables lead to difficulty in finding a perfect preservation method that holds all water quality levels constant [
7]. The US EPA; therefore, recommends preservation methods for different parameters to make testing as reliable as possible. However, even with these techniques, there is still a possibility of fluctuating measurements.
Some studies have tested current preservation methods, like freezing, acidification by sulfuric acid, refrigeration, and regular storage-affected nitrogen and phosphorus levels, and then compared the abilities of these methods to maintain concentrations [
8,
9,
10,
11,
12,
13,
14,
15]. A study using tile drainage water, river water, and surface runoff water in Indiana found that freezing at subzero temperatures was the best overall method for preserving water samples for orthophosphate and nitrogen (nitrate-N and ammonium) [
8]. However, filtering of the water sample may be needed if the sample contains high levels of sediment. In contrast, a study found that while refrigeration and filtration were necessary, acidification was the most effective preservation method for nitrate-N [
9]. Sulfuric acid can serve as a sufficient preservative for maintaining nitrate-N and total N concentrations for up to seven days [
10], but may be unsuitable for samples containing high concentrations of nitrite because sulfuric acid can cause the chemical conversion to nitrate [
11]. Furthermore, a study in California, USA, found that long-term freezing can serve as a viable method, as it did not have a significant effect on concentration [
12]. A study looking at samples from Lake Ontario found that storage at 4 °C produced variable results, with orthophosphate concentrations decreasing by as much as 13% and nitrate-N by as much as 7% after eight days [
13]. In contrast, a study in New York found that soil water samples maintained stable nitrate-N concentrations for at least one week, with only slight changes after three to 16 weeks of storage at 4 °C [
14]. A study by Maher et al. [
15] looked at using slow freezing in addition to chemicals. This study determined that, while room temperature storage can be used for a short period of time, there needs to be an additional preservation method.
Sodium omadine is traditionally used as a preservative or additive in manufacturing materials and process fluids. In 1968, the chemical was registered originally as a biocide to reduce the growth of bacteria and fungi in various industrial products. Today, these products include lubricants, latex products, and various cleaners. It has not been used as a preservative for water-based chemicals, or for water concentrations [
16]. Since there has been no study focusing on sodium omadine in preserving nitrate-N and orthophosphate levels in water samples, this will be the first study to determine the impacts this chemical has.
This research is significant because it (1) compares existing recommended preservation methods for water samples containing both nitrate-N and orthophosphate, and (2) looks at a new preservation option that has not been previously used for this purpose. This study mimics water monitoring that includes collecting samples from a field site, using an automated water sampler and retrieving the samples up to 14 days after initial collection.
The objective of this study is to determine the impact of three different preservation methods on nitrate-N and orthophosphate concentrations: (1) Filtration, as it is the US EPA recommended method for preserving water samples for orthophosphate; (2) addition of sulfuric acid to the water sample, as this is the US EPA recommended method for preserving water samples for nitrate-N; and (3) addition of sodium omadine to the water sample, as a possible alternative to currently recommended methods.
2. Materials and Methods
Since water types vary by source and location, three different source waters were collected and used in this study: (1) Tile water from an agricultural production field in Crawford County, IL, USA; (2) pond water collected from a retention basin on near the intersection of Windsor Road and First Street in Champaign, IL, USA; and (3) stream water from the Embarras River just south of Urbana, IL, USA. A five-gallon bucket was used to collect water from each location. The source water samples were refrigerated upon collection.
Additional measurements were taken to determine the pH, turbidity, and conductivity of each source water. To test the pH of the water samples, an Oakton pH/mV/°C/°F meter (Oakton Instruments, Vernon Hills, IL, USA) was used after standard calibration was completed using pH standards. For turbidity testing, the Hach DR 900 colorimeter (Hach, Loveland, CO, USA) was used. To measure turbidity, 10 mL of the sample was transferred to a smaller vile where it was then shaken and placed into the colorimeter. Conductivity was measured using the Hach Pocket Pro Tester (Hach, Loveland, CO, USA). The device was calibrated using the Singlet Conductivity Solution, which consisted of KCI 12.88 mS/cm Standard Solution at 25 °C.
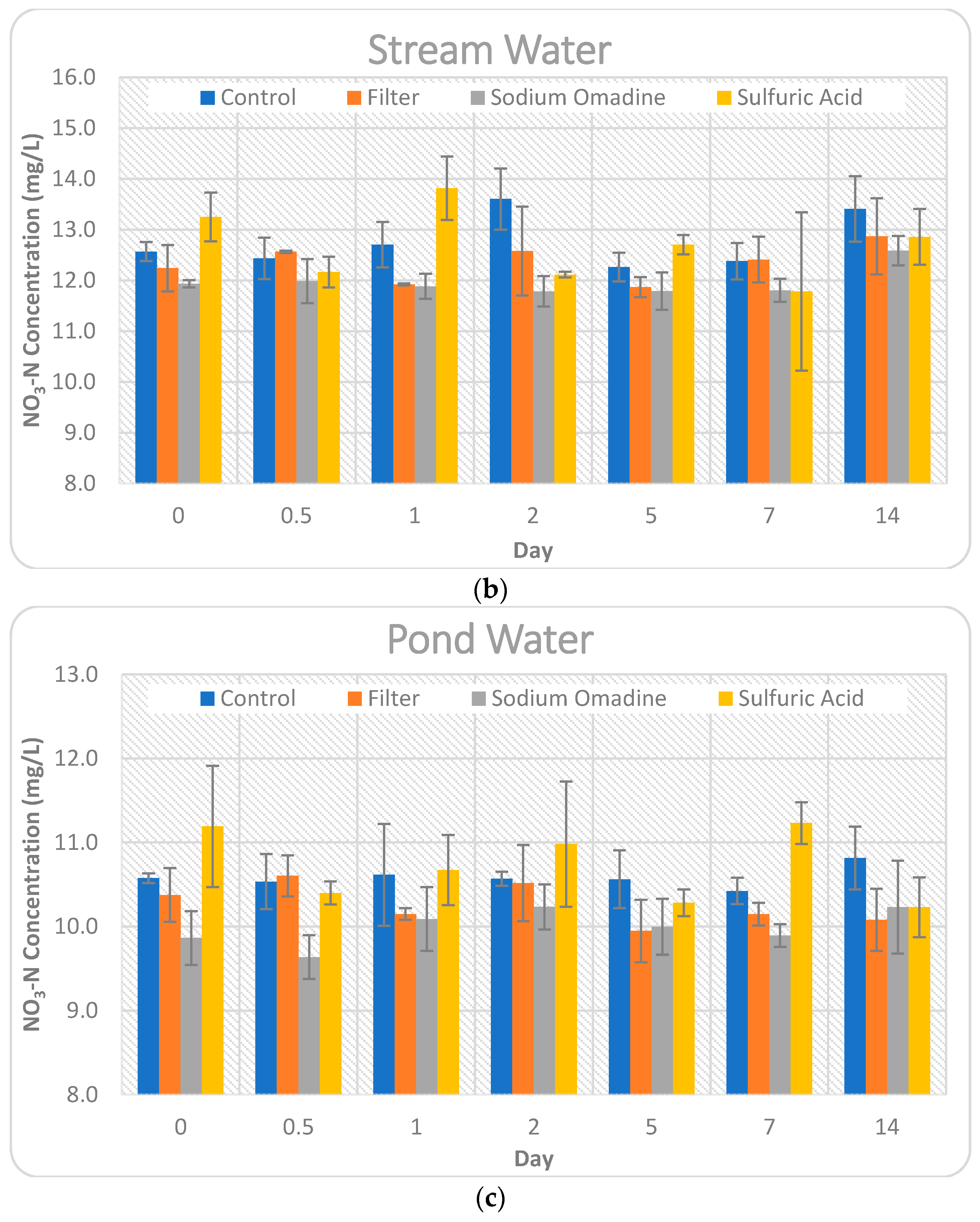
To ensure detectable concentrations of nitrate-N and orthophosphate, all source water was spiked with 10 ppm nitrate-N and 1 ppm orthophosphate. A 100 mL volume of the spiked source water was distributed into a 125 mL Nalgene bottle, labelled with corresponding names for the source water, time-period to be tested, and which preservation method was used.
After the source water was distributed into the bottles, the respective preservation method was applied to each sample. The control for each water source received no preservation. The samples receiving sulfuric acid received 0.2 mL of sulfuric acid in each 100 mL sample. The samples receiving sodium omadine received 0.25 mL of sodium omadine in each 100 mL sample. The bottles were capped and distributed into three automated water samplers, separated based on water source, in the shaded area adjacent to the Agricultural Engineering and Sciences Building at the University of Illinois at Urbana-Champaign (
Figure 1), to mimic outdoor field conditions without refrigeration. Nitrate-N and orthophosphate concentrations were measured on the following time points: Day 0 (when the respective preservation method was applied to the samples), Day 0.5, Day 1, Day 2, Day 5, Day 7, and Day 14 after preservation, resulting in seven time points. Each sample was prepared and analyzed in triplicate.
At each time point, the corresponding samples were taken from the autosamplers into the lab. The control samples as well as those receiving sulfuric acid and sodium omadine were immediately placed in the freezer. The filtered samples from each water source were filtered by pouring the water sample through a 0.45 µm filter on top of an Erlenmeyer flask (
Figure 2). A vacuum was used to expedite the filtration process. Upon completion of filtration, samples were placed in the freezer. At the completion of the 14 days, all samples were thawed and analyzed for nitrate-N and orthophosphate.
2.1. Nutrient Analyses
Nutrient analyses were completed by the Water Quality Laboratory in the Department of Agricultural and Biological Engineering at the University of Illinois, Urbana-Champaign, USA. The acidity of each sample was first tested and adjusted accordingly. This type of adjustment was only performed on the sulfuric acid preservation samples. Samples were then analyzed for nitrate-N using the automated hydrazine reduction method (Standard Methods 4500-NO3-H, National Environmental Methods Index) and for orthophosphate using the ascorbic acid reduction method (Standard Methods 4500-P-F, National Environmental Methods Index). Spiked samples, duplicate samples, and standard solutions were incorporated into nutrient analyses for quality control.
2.2. Statistical Analysis
Differences between the treatments were analyzed for significance using a one-way analysis of variance (ANOVA) with post-hoc Tukey honestly significant difference (HSD) for comparing multiple treatments. When sample sizes were unequal, the Tukey–Kramer method was used.