Threatening Facial Expressions Impact Goal-Directed Actions Only if Task-Relevant
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Stimuli
2.3. Experimental Apparatus and Behavioral Tasks
2.4. Emotion Discrimination Task
2.5. Gender Discrimination Task
2.6. Emotion Recall
2.7. Data Analyses
3. Results
3.1. Analyses of RTs
3.2. Analyses of MTs
3.3. Analyses of Average Rates of Mistakes
3.4. Correlations between Behavioral Measures and the Recognition Scores
3.5. Emotion Recall Performance
4. Discussion
4.1. Task-Relevance Matters
4.2. The Effect of Angry, Fearful, and Happy Faces
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Crivelli, C.; Fridlund, A.J. Facial Displays Are Tools for Social Influence. Trends Cogn. Sci. 2018, 22, 388–399. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yiend, J. The effects of emotion on attention: A review of attentional processing of emotional information. Cogn. Emot. 2010, 24, 3–47. [Google Scholar] [CrossRef]
- Adolphs, R. Fear, faces, and the human amygdala. Curr. Opin. Neurobiol. 2008, 18, 166–172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davis, F.C.; Somerville, L.H.; Ruberry, E.J.; Berry, A.B.; Shin, L.M.; Whalen, P.J. A tale of two negatives: Differential memory modulation by threat-related facial expressions. Emotion 2011, 11, 647–655. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pool, E.; Brosch, T.; Delplanque, S.; Sander, D. Attentional bias for positive emotional stimuli: A meta-analytic investigation. Psychol. Bull. 2016, 142, 79–106. [Google Scholar] [CrossRef] [PubMed]
- Dawel, A.; O’Kearney, R.; McKone, E.; Palermo, R. Not just fear and sadness: Meta-analytic evidence of pervasive emotion recognition deficits for facial and vocal expressions in psychopathy. Neurosci. Biobehav. Rev. 2012, 36, 2288–2304. [Google Scholar] [CrossRef] [PubMed]
- Phillips, M.L.; David, A.S. Facial processing in schizophrenia and delusional misidentification: Cognitive neuropsychiatric approaches. Schizophr. Res. 1995, 17, 109–114. [Google Scholar] [CrossRef]
- Adolphs, R.; Gosselin, F.; Buchanan, T.W.; Tranel, D.; Schyns, P.; Damasio, A.R. A mechanism for impaired fear recognition after amygdala damage. Nature 2005, 433, 68–72. [Google Scholar] [CrossRef] [PubMed]
- Berkman, E.T.; Burklund, L.; Lieberman, M.D. Inhibitory spillover: Intentional motor inhibition produces incidental limbic inhibition via right inferior frontal cortex. Neuroimage 2009, 47, 705–712. [Google Scholar] [CrossRef] [Green Version]
- Schulz, K.P.; Clerkin, S.M.; Halperin, J.M.; Newcorn, J.H.; Tang, C.Y.; Fan, J. Dissociable neural effects of stimulus valence and preceding context during the inhibition of responses to emotional faces. Hum. Brain Mapp. 2009, 30, 2821–2833. [Google Scholar] [CrossRef] [Green Version]
- Schulz, K.P.; Fan, J.; Magidina, O.; Marks, D.J.; Hahn, B.; Halperin, J.M. Does the emotional go/no-go task really measure behavioral inhibition? Convergence with measures on a non-emotional analog. Arch. Clin. Neuropsychol. 2007, 22, 151–160. [Google Scholar] [CrossRef] [Green Version]
- Zhang, W.; Lu, J. Time course of automatic emotion regulation during a facial Go/Nogo task. Biol. Psychol. 2012, 89, 444–449. [Google Scholar] [CrossRef] [PubMed]
- Mirabella, G. Should I stay or should I go? Conceptual underpinnings of goal-directed actions. Front. Syst. Neurosci. 2014, 8, 206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Logan, G.D.; Cowan, W.B.; Davis, K.A. On the ability to inhibit simple and choice reaction time responses: A model and a method. J. Exp. Psychol. Hum. Percept. Perform. 1984, 10, 276–291. [Google Scholar] [CrossRef]
- Sagaspe, P.; Schwartz, S.; Vuilleumier, P. Fear and stop: A role for the amygdala in motor inhibition by emotional signals. Neuroimage 2011, 55, 1825–1835. [Google Scholar] [CrossRef]
- Rebetez, M.M.L.; Rochat, L.; Billieux, J.; Gay, P.; Van der Linden, M. Do emotional stimuli interfere with two distinct components of inhibition? Cogn. Emot. 2015, 29, 559–567. [Google Scholar] [CrossRef] [PubMed]
- Derntl, B.; Habel, U. Angry but not neutral faces facilitate response inhibition in schizophrenia patients. Eur. Arch. Psychiatry Clin. Neurosci. 2017, 267, 621–627. [Google Scholar] [CrossRef] [PubMed]
- De Valk, J.M.; Wijnen, J.G.; Kret, M.E. Anger fosters action. Fast responses in a motor task involving approach movements toward angry faces and bodies. Front. Psychol. 2015, 6, 1240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lundqvist, D.; Juth, P.; Öhman, A. Using facial emotional stimuli in visual search experiments: The arousal factor explains contradictory results. Cogn. Emot. 2014, 28, 1012–1029. [Google Scholar] [CrossRef]
- Soloff, P.H.; Abraham, K.; Ramaseshan, K.; Burgess, A.; Diwadkar, V.A. Hyper-modulation of brain networks by the amygdala among women with Borderline Personality Disorder: Network signatures of affective interference during cognitive processing. J. Psychiatr. Res. 2017, 88, 56–63. [Google Scholar] [CrossRef] [Green Version]
- Tottenham, N.; Hare, T.A.; Quinn, B.T.; McCarry, T.W.; Nurse, M.; Gilhooly, T.; Millner, A.; Galvan, A.; Davidson, M.C.; Eigsti, I.M.; et al. Prolonged institutional rearing is associated with atypically large amygdala volume and difficulties in emotion regulation. Dev. Sci 2010, 13, 46–61. [Google Scholar] [CrossRef] [Green Version]
- Brown, B.K.; Murrell, J.; Karne, H.; Anand, A. The effects of DAT1 genotype on fMRI activation in an emotional go/no-go task. Brain Imaging Behav. 2017, 11, 185–193. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mirabella, G. The Weight of Emotions in Decision-Making: How Fearful and Happy Facial Stimuli Modulate Action Readiness of Goal-Directed Actions. Front. Psychol. 2018, 9, 1334. [Google Scholar] [CrossRef]
- Engen, H.G.; Smallwood, J.; Singer, T. Differential impact of emotional task relevance on three indices of prioritised processing for fearful and angry facial expressions. Cogn. Emot. 2017, 31, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Wang, Y.; Xu, S.; Wang, Y.; Zhang, R.; Li, T. Aggression differentially modulates brain responses to fearful and angry faces: An exploratory study. Neuroreport 2015, 26, 663–668. [Google Scholar] [CrossRef] [PubMed]
- Ashley, V.; Swick, D. Angry and Fearful Face Conflict Effects in Post-traumatic Stress Disorder. Front. Psychol. 2019, 10, 136. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oldfield, R.C. The assessment and analysis of handedness: The Edinburgh inventory. Neuropsychologia 1971, 9, 97–113. [Google Scholar] [CrossRef]
- Open Science Framework Platform. Available online: https://osf.io/m8fex/ (accessed on 29 October 2020).
- Ekman, P.; Friesen, W.V. Pictures of Facial Affect; Consulting Psychologists Press: Palo Alto, CA, USA, 1976. [Google Scholar]
- Dodich, A.; Cerami, C.; Canessa, N.; Crespi, C.; Marcone, A.; Arpone, M.; Realmuto, S.; Cappa, S.F. Emotion recognition from facial expressions: A normative study of the Ekman 60-Faces Test in the Italian population. Neurol. Sci. 2014, 35, 1015–1021. [Google Scholar] [CrossRef]
- Cortex and Cortex Explorer: Real-Time Software and Data Analysis Tools. Available online: https://www.nimh.nih.gov/research/research-conducted-at-nimh/research-areas/clinics-and-labs/ln/shn/software-projects.shtml (accessed on 29 October 2020).
- Federico, P.; Mirabella, G. Effects of probability bias in response readiness and response inhibition on reaching movements. Exp. Brain Res. 2014, 232, 1293–1307. [Google Scholar] [CrossRef]
- Lakens, D. Calculating and reporting effect sizes to facilitate cumulative science: A practical primer for t-tests and ANOVAs. Front. Psychol. 2013, 4, 863. [Google Scholar] [CrossRef] [Green Version]
- Rouder, J.N.; Speckman, P.L.; Sun, D.; Morey, R.D.; Iverson, G. Bayesian t tests for accepting and rejecting the null hypothesis. Psychon. Bull. Rev. 2009, 16, 225–237. [Google Scholar] [CrossRef]
- Donald, S. Chi-Square Test is Statistically Significant: Now What? Pract. Assess. Res. Evaluation. 2015, 20. [Google Scholar] [CrossRef]
- R Core Team. The R Project for Statistical Computing. Available online: http://www.R-project.org/ (accessed on 29 October 2020).
- Berger, N.; Richards, A.; Davelaar, E.J. When Emotions Matter: Focusing on Emotion Improves Working Memory Updating in Older Adults. Front. Psychol. 2017, 8, 1565. [Google Scholar] [CrossRef] [Green Version]
- Schutter, D.J.; Hofman, D.; Van Honk, J. Fearful faces selectively increase corticospinal motor tract excitability: A transcranial magnetic stimulation study. Psychophysiology 2008, 45, 345–348. [Google Scholar] [CrossRef]
- Becker, D.V.; Anderson, U.S.; Mortensen, C.R.; Neufeld, S.L.; Neel, R. The face in the crowd effect unconfounded: Happy faces, not angry faces, are more efficiently detected in single- and multiple-target visual search tasks. J. Exp. Psychol. Gen. 2011, 140, 637–659. [Google Scholar] [CrossRef]
- Goren, D.; Wilson, H.R. Quantifying facial expression recognition across viewing conditions. Vision Res. 2006, 46, 1253–1262. [Google Scholar] [CrossRef] [Green Version]
- Becker, D.V.; Neel, R.; Srinivasan, N.; Neufeld, S.; Kumar, D.; Fouse, S. The vividness of happiness in dynamic facial displays of emotion. PLoS ONE 2012, 7, e26551. [Google Scholar] [CrossRef]
- Lang, P.J. The emotion probe: Studies of motivation and attention. Am. Psychol. 1995, 50, 372–385. [Google Scholar] [CrossRef]
- Fox, E.; Russo, R.; Dutton, K. Attentional Bias for Threat: Evidence for Delayed Disengagement from Emotional Faces. Cogn. Emot. 2002, 16, 355–379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vuilleumier, P.; Huang, Y.-M. Emotional Attention: Uncovering the Mechanisms of Affective Biases in Perception. Curr. Dir. Psychol. Sci. 2009, 18, 148–152. [Google Scholar] [CrossRef] [Green Version]
- Weierich, M.R.; Treat, T.A.; Hollingworth, A. Theories and measurement of visual attentional processing in anxiety. Cogn. Emot. 2008, 22, 985–1018. [Google Scholar] [CrossRef]
- Mirabella, G.; Pani, P.; Ferraina, S. Context influences on the preparation and execution of reaching movements. Cogn. Neuropsychol. 2008, 25, 996–1010. [Google Scholar] [CrossRef]
- Halbig, T.D.; Borod, J.C.; Frisina, P.G.; Tse, W.; Voustianiouk, A.; Olanow, C.W.; Gracies, J.M. Emotional processing affects movement speed. J. Neural Transm. Vienna 2011, 118, 1319–1322. [Google Scholar] [CrossRef] [PubMed]
- Esteves, P.O.; Oliveira, L.A.; Nogueira-Campos, A.A.; Saunier, G.; Pozzo, T.; Oliveira, J.M.; Rodrigues, E.C.; Volchan, E.; Vargas, C.D. Motor planning of goal-directed action is tuned by the emotional valence of the stimulus: A kinematic study. Sci. Rep. 2016, 6, 28780. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Jaquess, K.J.; Hatfield, B.D.; Zhou, C.; Li, H. Valence and arousal of emotional stimuli impact cognitive-motor performance in an oddball task. Biol. Psychol. 2017, 125, 105–114. [Google Scholar] [CrossRef] [PubMed]
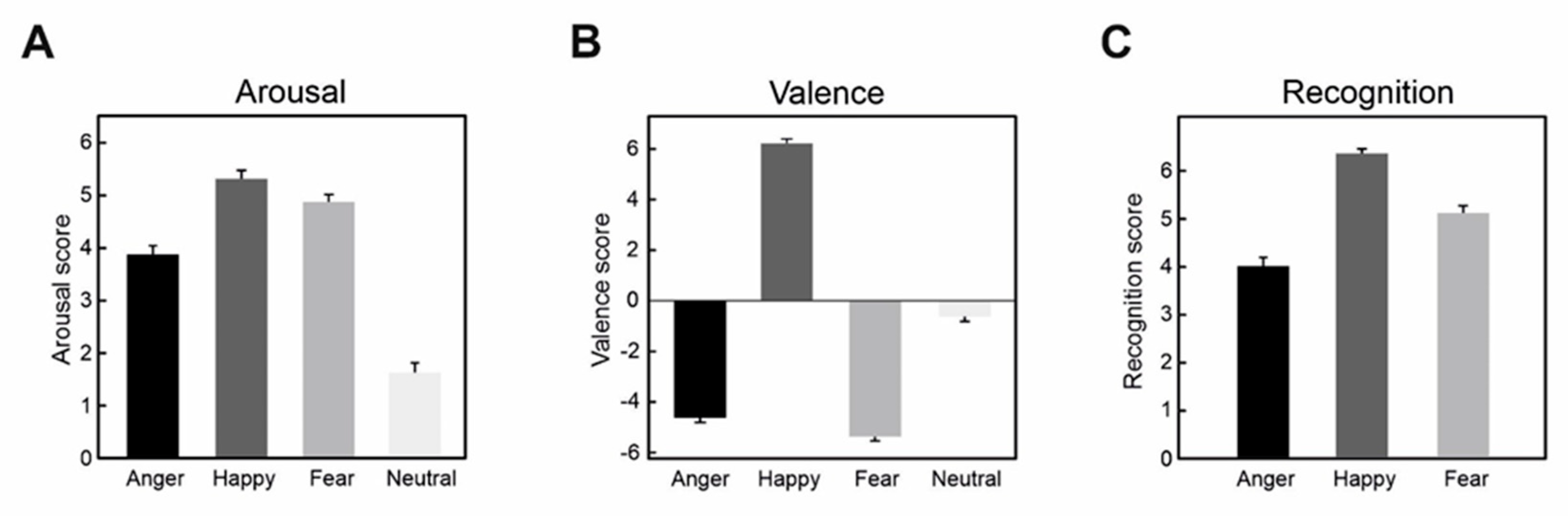



| One-Way ANOVA on Arousal: Emotion (Anger, Happiness, Fear) | ||||
|---|---|---|---|---|
| Value of Parameters | p values | Effect Size | BF10 | |
| Main effect: Emotion | F(2,110) = 44.8 | p < 0.0001 | ηp2 = 0.45 | 9.46 × 106 |
| Post hoc tests: | ||||
| Anger vs. Happiness | t(55) = 8.28 | p < 0.0001 | d = 1.19 | 1.49 × 106 |
| Anger vs. Fear | t(55) = 8.81 | p < 0.0001 | d = 0.94 | 5934 |
| Happiness vs. Fear | t(55) = 2.48 | p = 0.049 | d = 0.38 | 1.16 |
| One-way ANOVA on Valence: Emotion (Anger, Happiness, Fear) | ||||
| Value of Parameters | p values | Effect Size | BF10 | |
| Main effect: Emotion | F(2,110) = 1558.0 | p < 0.0001 | ηp2 = 0.97 | 1.90 × 1099 |
| Post hoc tests: | ||||
| Anger vs. Happiness | t(55) = 38.4 | p < 0.0001 | d = 8.01 | 9.19 × 1065 |
| Anger vs. Fear | t(55) = −4.38 | p = 0.00016 | d = 0.50 | 4.27 |
| Happiness vs. Fear | t(55) = 49.4 | p < 0.0001 | d = 10.5 | 1.63 × 1078 |
| One-way ANOVA of Recognition scores: Emotion (Anger, Happiness, Fear) | ||||
| Value of Parameters | p values | Effect Size | BF10 | |
| Main effect: Emotion | F(2,110) = 96.6 | p < 0.0001 | ηp2 = 0.64 | 1.67 × 1017 |
| Post hoc tests: | ||||
| Anger vs. Happiness | t(55) = 12.5 | p < 0.0001 | d = 2.07 | 1.27 × 1016 |
| Anger vs. Fear | t(55) = 6.88 | p < 0.0001 | d = 0.897 | 2645 |
| Happiness vs. Fear | t(55) = 7.95 | p < 0.0001 | d = 1.22 | 3.30 × 106 |
| Four-way ANOVA of RTs: Emotion (Anger, Happiness, Fear); Sex (F, M); AL (High, Medium, Low); | ||||||
|---|---|---|---|---|---|---|
| Task (Emotion Discrimination Task, Gender Discrimination Task) | ||||||
| Value of Parameters | p values | Mdiff | 95% CI | Effect Size | BF10 | |
| Main effect: Emotion | F(1.54,76.8) = 51.8 | p < 0.001 | ηp2 = 0.51 | 3.1 × 107 | ||
| Post hoc Tests: | ||||||
| Anger vs. Happiness | t(111) = 8.46 | p < 0.001 | 19.27 | (13.63, 24.92) | d = 0.80 | 4.4 × 108 |
| Anger vs. Fear | t(111) = 6.80 | p < 0.001 | 15.07 | (9.58, 20.55) | d = 0.64 | 1.2 × 106 |
| Happiness vs. Fear | t(111) = -3.14 | p = 0.008 | −4.21 | (−7.53, −0.89) | d = 0.30 | 12.20 |
| Main effect: Task | F(1,50) = 5.15 | p = 0.028 | 8.86 | (1.02, 16.70) | ηp2 = 0.09 | 305 |
| Interaction: Emotion*Task | F(1.77,88.5) = 47.10 | p < 0.001 | ηp2 = 0.49 | 5.7 × 107 | ||
| Post hoc Tests: | ||||||
| Emotion Task-Anger vs. Happiness | t(55) = 10.08 | p < 0.001 | 37.02 | (27.92, 46.12) | d = 1.35 | 1.9 × 1012 |
| Emotion Task-Anger vs. Fear | t(55) = 7.35 | p < 0.001 | 27.26 | (18.07, 36.45) | d = 0.98 | 9.3 × 107 |
| Emotion Task-Happiness vs. Fear | t(55) = −5.31 | p < 0.001 | −9.76 | (−14.31, −5.21) | d = 0.71 | 1.8 × 104 |
| Gender Task-Anger vs. Happiness | t(55) = 0.76 | p = 1 | 1.53 | (−3.48, 6.53) | d = 0.10 | 0.20 |
| Gender Task-Anger vs. Fear | t(55) = 1.26 | p = 0.640 | 2.87 | (−2.75, 8.48) | d = 0.17 | 0.21 |
| Gender Task-Happiness vs. Fear | t(55) = 0.60 | p = 1 | 1.34 | (−4.16, 6.85) | d = 0.08 | 0.15 |
| Emotion Task-Anger vs. Gender Task-Anger | t(55) = 5.92 | p < 0.001 | 28.83 | (19.05, 38.61) | d = 0.79 | 2.6 × 105 |
| Four-way ANOVA of MTs: Emotion (Anger, Happiness, Fear), Sex (F, M), AL (High, Medium, Low), | ||||||
|---|---|---|---|---|---|---|
| Task (Emotion Discrimination Task, Gender Discrimination Task) | ||||||
| Value of Parameters | p values | Mdiff | 95% CI | Effect Size | BF10 | |
| Main effect: Emotion | F(1.55,77.3) = 16.64 | p < 0.001 | ηp2 = 0.25 | 2.82 | ||
| Post hoc Tests: | ||||||
| Anger vs. Happiness | t(111) = 4.18 | p < 0.001 | 8.10 | (3.31, 12.90) | d = 0.40 | 431 |
| Anger vs. Fear | t(111) = 4.75 | p < 0.001 | 8.31 | (3.98, 12.65) | d = 0.45 | 3120 |
| Happiness vs. Fear | t(111) = 0.19 | p = 1 | 0.21 | (−2.61, 3.03) | d = 0.02 | 0.12 |
| Interaction: Emotion vs. Task | F(1.99,99.5) = 16.54 | p < 0.001 | ηp2 = 0.25 | 1.49 | ||
| Post hoc Tests: | ||||||
| Emotion Task-Anger vs. Happiness | t(55) = 4.84 | p < 0.001 | 14.59 | (7.12, 22.05) | d = 0.65 | 3208 |
| Emotion Task-Anger vs. Fear | t(55) = 5.63 | p < 0.001 | 15.82 | (8.85, 22.78) | d = 0.75 | 2.0 × 104 |
| Gender Task-Anger vs. Happiness | t(55) = 0.98 | p = 0.99 | 1.62 | (−2.47, 5.71) | d = 0.13 | 0.16 |
| Gender Task-Anger vs. Fear | t(55) = 0.55 | p = 1 | 0.81 | (−2.80, 4.42) | d = 0.07 | 0.17 |
| Gender Task-Happiness vs. Fear | t(55) = −0.44 | p = 1 | -0.81 | (−5.38, 3.76) | d = 0.06 | 0.15 |
| Emotion Task-Anger vs. Gender Task - Anger | t(55) = 3.17 | p = 0.003 | 18.51 | (6.79, 30.23) | d = 0.42 | 5.67 |
| Four-way ANOVA of Mistakes: Emotion (Anger, Happiness, Fear, Sex (F, M), AL (High, Medium, Low), | ||||||
|---|---|---|---|---|---|---|
| Task (Emotion Discrimination Task, Gender Discrimination Task) | ||||||
| Value of Parameters | p values | Mdiff | 95% CI | Effect Size | BF10 | |
| Main effect: Emotion | F(1.69, 84.8) = 37.44 | p < 0.001 | ηp2 = 0.43 | 9.9 × 105 | ||
| Post hoc Tests: | ||||||
| Anger vs. Happiness | t(111) = 7.01 | p < 0.001 | 3.54 | (2.29, 4.79) | d = 0.66 | 4.9 × 104 |
| Anger vs. Fear | t(111) = 6.56 | p < 0.001 | 3.27 | (2.03, 4.50) | d = 0.62 | 1.0 × 105 |
| Happiness vs. Fear | t(111) = −0.79 | p = 1 | −0.27 | (−1.13, 0.58) | d = 0.07 | 0.11 |
| Main effect: Task | F(1, 50) = 44.54 | p < 0.001 | 3.81 | (2.66, 4.96) | ηp2 = 0.47 | 5.8 × 1013 |
| Main effect: Sex | F(1, 50) = 5.27 | p = 0.026 | 1.60 | (0.20, 3.01) | ηp2 = 0.10 | 0.44 |
| Main effect: AL | F(2, 50) = 3.97 | p = 0.025 | ηp2 = 0.14 | 0.19 | ||
| Post hoc test: High vs. Low | t(34) = 2.81 | p = 0.021 | 2.45 | (0.29, 4.61) | d = 0.47 | 1.42 |
| Interaction: Emotion*Task | F(1.56, 78.21) = 47.64 | p < 0.001 | ηp2 = 0.49 | 8.3 × 1013 | ||
| Post hoc Tests: | ||||||
| Emotion Task-Anger vs. Happiness | t(55) = 7.58 | p < 0.001 | 7.55 | (5.09, 10.02) | d = 0.72 | 9.7 × 107 |
| Emotion Task-Anger vs. Fear | t(55) = 7.71 | p < 0.001 | 7.03 | (4.77, 9.29) | d = 0.73 | 2.6 × 108 |
| Gender Task-Anger vs. Happiness | t(55) = 1.43 | p = 0.477 | −0.47 | (1.30, 0.35) | d = 0.19 | 0.65 |
| Gender Task-Anger vs. Fear | t(55) = 1.38 | p = 0.521 | −0.49 | (1.38, 0.39) | d = 0.18 | 0.32 |
| Gender Task-Happiness vs. Fear | t(55) = 0.05 | p = 1 | −0.02 | (−1.0, 0.96) | d = 0.01 | 0.16 |
| Emotion Task-Anger vs. Gender Task-Anger | t(55) = 9.80 | p < 0.001 | 9.00 | (7.15, 10.84) | d = 1.31 | 5.8 × 1011 |
| Emotion Task-Fear vs. Gender Task-Fear | t(55) = 2.23 | p = 0.030 | 1.48 | (0.15, 2.80) | d = 0.30 | 3.41 |
| Interaction: Sex*AL | F(2, 50) = 5.85 | p = 0.005 | ηp2 = 0.19 | 5.97 | ||
| Post hoc Tests: | ||||||
| Female-High vs. Male-High | t(50) = 3.83 | p < 0.001 | 4.84 | (2.30, 7.39) | d = 0.51 | 406 |
| Male-High vs. Medium | t(50) = 3.41 | p = 0.004 | 4.31 | (1.17, 7.45) | d = 0.45 | 47.10 |
| Male-High vs. Low | t(50) = 3.38 | p = 0.004 | 4.42 | (1.18, 7.65) | d = 0.45 | 80.30 |
| Recalled Emotions after Emotion Discrimination Task | Recalled Emotions after Gender Discrimination Task | |||
|---|---|---|---|---|
| Value of Parameters | p Values | Value of Parameters | p Values | |
| Total recalled emotions: | χ2 (2, N = 62) = 4.29 | p = 0.117 | χ2 (2, N = 63) = 3.52 | p = 0.17 |
| First recalled emotion: | χ2 (2, N = 25) = 6.32 | p = 0.042 | χ2 (2, N = 24) = 27.0 | p < 0.0001 |
| Post hoc: Happiness | SR = 2.40 | p = 0.049 | SR = 5.20 | p < 0.0001 |
| Post hoc: Anger | SR = -0.57 | p = 1 | SR = −2.60 | p = 0.028 |
| Post hoc: Fear | SR = −1.84 | p = 0.20 | SR = −2.60 | p = 0.028 |
| Second recalled emotion: | χ2 (2, N = 22) = 0.09 | p = 0.956 | χ2 (2, N = 24) = 0.75 | p = 0.687 |
| Third recalled emotion: | χ2 (2, N = 15) = 7.60 | p = 0.022 | χ2 (2, N = 15) = 2.80 | p = 0.247 |
| Post hoc: Happiness | SR = −1.10 | p = 0.82 | - | - |
| Post hoc: Anger | SR = 2.74 | p = 0.019 | - | - |
| Post hoc: Fear | SR = −1.64 | p = 0.30 | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mancini, C.; Falciati, L.; Maioli, C.; Mirabella, G. Threatening Facial Expressions Impact Goal-Directed Actions Only if Task-Relevant. Brain Sci. 2020, 10, 794. https://doi.org/10.3390/brainsci10110794
Mancini C, Falciati L, Maioli C, Mirabella G. Threatening Facial Expressions Impact Goal-Directed Actions Only if Task-Relevant. Brain Sciences. 2020; 10(11):794. https://doi.org/10.3390/brainsci10110794
Chicago/Turabian StyleMancini, Christian, Luca Falciati, Claudio Maioli, and Giovanni Mirabella. 2020. "Threatening Facial Expressions Impact Goal-Directed Actions Only if Task-Relevant" Brain Sciences 10, no. 11: 794. https://doi.org/10.3390/brainsci10110794
APA StyleMancini, C., Falciati, L., Maioli, C., & Mirabella, G. (2020). Threatening Facial Expressions Impact Goal-Directed Actions Only if Task-Relevant. Brain Sciences, 10(11), 794. https://doi.org/10.3390/brainsci10110794






