Photographs of Actions: What Makes Them Special Cues to Social Perception
Abstract
:1. Photographs of Actions: What Makes Them Special Cues to Social Perception
2. Definitions of Key Terms and Concepts
2.1. Neural Representations of Actions
2.2. Knowledge of Actions
- Internal models for the control of the muscle activities that generate the movements. Various scholars have described the control of actions by signals from the brain using motor programs [93] or on the basis of models in which individuals select motor commands for a specific context, depending on multiple internal and external factors [92]. Motor programs are representations of rules for the execution of movements, according to which the spatial and temporal activity patterns of certain muscles are organized and controlled [93]. These programs are supposed to be stored in motor brain structures in a generalized or abstract format. “Models for motor control” [94], on the other hand, describe the control of actions more in connection with adjustments to specific action contexts and courses. Individuals transform sensory information into motor commands. The resulting movements produce sensory outcomes that provide feedback for further motor control.
- Somatosensory processes or sensations, for example, in relation to proprioception, the processing of haptic or tactile information, heat, cold, or pain [9,10,74,92,95,96]. A crucial property of the knowledge about movements, internal models, and somatosensory processes and sensations is that this knowledge includes information about changes over time and outcomes of these changes [10,56,92,94]. Individuals can use this change-related information to anticipate the immediate further course of an action that they are performing or observing [56,62,73,97]. Somatosensory anticipation plays a particularly important role in performing actions [10,79,96]. It conveys information about the immediate somatosensory consequences of movements, for example, proprioceptive or tactile stimulation.
- Knowledge of the desired outcomes of movements, that is, of action goals [91]. Knowledge of goals includes goals at different hierarchical levels. The overarching goal of an action is often referred to in the literature as the “intention” [3,50]. Goals of actions are related to motives, needs, or desires and have a certain importance or value. For this reason, mental representations of motor actions fundamentally include an emotional component [11,55,57].
3. Cognitive Products of Processing Observed Actions in the AON
3.1. Action Understanding
3.1.1. Generalization and Categorization
3.1.2. Conceptual and Semantic Processes in Action Categorization
3.2. Knowledge of Specific Properties of Observed Actions
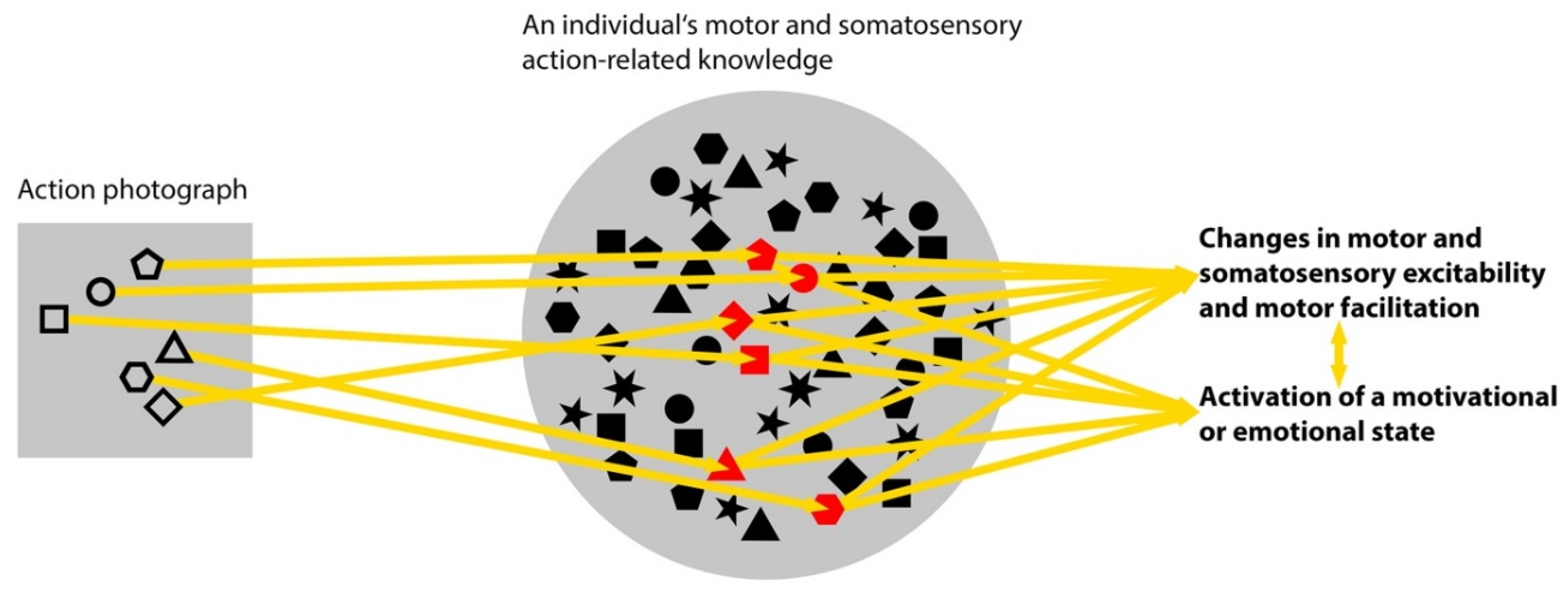
3.3. Changes in Motor and Somatosensory Excitability
3.4. Activation of a Motivational or Emotional State
3.5. Experiences That Are Accompanied by Conscious Awareness
4. Cognitive Products of Neural Responses to Action Photos in the AON
5. Specific Representational Characteristics of Action Photos
5.1. Clarity of the Pictured Movements
5.2. Visibility of Muscle Activities and Skin
5.3. Visibility of Somatosensory Operations or Sensations
5.4. Clarity of the Involved Object or Context
5.5. Clarity of the Action Goal
5.6. Emotional Value of the Action or Sensation
5.7. Questions for Future Research
6. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Cross, E.S.; Kraemer, D.J.M.; Hamilton, A.; Kelley, W.M.; Grafton, S.T. Sensitivity of the Action Observation Network to Physical and Observational Learning. Cereb. Cortex 2009, 19, 315–326. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feurra, M.; Blagovechtchenski, E.; Nikulin, V.V.; Nazarova, M.; Lebedeva, A.; Pozdeeva, D.; Yurevich, M.; Rossi, S. State-Dependent Effects of Transcranial Oscillatory Currents on the Motor System during Action Observation. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Rizzolatti, G.; Cattaneo, L.; Destro, M.F.; Rozzi, S. Cortical Mechanisms Underlying the Organization of Goal-Directed Actions and Mirror Neuron-Based Action Understanding. Physiol. Rev. 2014, 94, 655–706. [Google Scholar] [CrossRef] [PubMed]
- Di Pellegrino, G.; Fadiga, L.; Fogassi, L.; Gallese, V.; Rizzolatti, G. Understanding motor events: A neurophysiological study. Exp. Brain Res. 1992, 91, 176–180. [Google Scholar] [CrossRef]
- Gallese, V.; Fadiga, L.; Fogassi, L.; Rizzolatti, G. Action recognition in the premotor cortex. Brain 1996, 119, 593–610. [Google Scholar] [CrossRef] [Green Version]
- Mukamel, R.; Ekstrom, A.; Kaplan, J.; Iacoboni, M.; Fried, I. Single-Neuron Responses in Humans during Execution and Observation of Actions. Curr. Biol. 2010, 20, 750–756. [Google Scholar] [CrossRef] [Green Version]
- Naish, K.R.; Houston-Price, C.; Bremner, A.J.; Holmes, N.P. Effects of action observation on corticospinal excitability: Muscle specificity, direction, and timing of the mirror response. Neuropsychologia 2014, 64, 331–348. [Google Scholar] [CrossRef]
- Keysers, C.; Kaas, J.H.; Gazzola, V. Somatosensation in social perception. Nat. Rev. Neurosci. 2010, 11, 417–428. [Google Scholar] [CrossRef]
- Gazzola, V.; Keysers, C. The Observation and Execution of Actions Share Motor and Somatosensory Voxels in all Tested Subjects: Single-Subject Analyses of Unsmoothed fMRI Data. Cereb. Cortex 2009, 19, 1239–1255. [Google Scholar] [CrossRef] [Green Version]
- Valchev, N.; Gazzola, V.; Avenanti, A.; Keysers, C. Primary somatosensory contribution to action observation brain activity—combining fMRI and cTBS. Soc. Cogn. Affect. Neurosci. 2016, 11, 1205–1217. [Google Scholar] [CrossRef]
- Keysers, C.; Paracampo, R.; Gazzola, V. What neuromodulation and lesion studies tell us about the function of the mirror neuron system and embodied cognition. Curr. Opin. Psychol. 2018, 24, 35–40. [Google Scholar] [CrossRef] [Green Version]
- Jacquet, P.O.; Avenanti, A. Perturbing the Action Observation Network During Perception and Categorization of Actions’ Goals and Grips: State-Dependency and Virtual Lesion TMS Effects. Cereb. Cortex 2015, 25, 598–608. [Google Scholar] [CrossRef] [Green Version]
- Neal, A.; Kilner, J.M. What is simulated in the action observation network when we observe actions? Eur. J. Neurosci. 2010, 32, 1765–1770. [Google Scholar] [CrossRef] [Green Version]
- Downing, P.E.; Peelen, M.; Wiggett, A.J.; Tew, B.D. The role of the extrastriate body area in action perception. Soc. Neurosci. 2006, 1, 52–62. [Google Scholar] [CrossRef]
- Hafri, A.; Trueswell, J.C.; Epstein, R.A. Neural Representations of Observed Actions Generalize across Static and Dynamic Visual Input. J. Neurosci. 2017, 37, 3056–3071. [Google Scholar] [CrossRef]
- Lu, Z.; Li, X.; Meng, M. Encodings of implied motion for animate and inanimate object categories in the two visual pathways. NeuroImage 2016, 125, 668–680. [Google Scholar] [CrossRef]
- O’Toole, A.J.; Natu, V.; An, X.; Rice, A.; Ryland, J.; Phillips, P.J. The neural representation of faces and bodies in motion and at rest. NeuroImage 2014, 91, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Proverbio, A.M.; Riva, F.; Zani, A. Observation of Static Pictures of Dynamic Actions Enhances the Activity of Movement-Related Brain Areas. PLoS ONE 2009, 4, e5389. [Google Scholar] [CrossRef] [PubMed]
- Thierry, G.; Pegna, A.; Dodds, C.; Roberts, M.; Basan, S.; Downing, P. An event-related potential component sensitive to images of the human body. NeuroImage 2006, 32, 871–879. [Google Scholar] [CrossRef] [PubMed]
- Hermsdörfer, J.; Goldenberg, G.; Wachsmuth, C.; Conrad, B.; Baumannb, A.-; Bartenstein, P.; Schwaiger, M.; Boecker, H. Cortical Correlates of Gesture Processing: Clues to the Cerebral Mechanisms Underlying Apraxia during the Imitation of Meaningless Gestures. NeuroImage 2001, 14, 149–161. [Google Scholar] [CrossRef]
- Kolesar, T.A.; Kornelsen, J.; Smith, S.D. Separating neural activity associated with emotion and implied motion: An fMRI study. Emotion 2017, 17, 131–140. [Google Scholar] [CrossRef]
- Kourtzi, Z.; Kanwisher, N. Activation in Human MT/MST by Static Images with Implied Motion. J. Cogn. Neurosci. 2000, 12, 48–55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arioli, M.; Perani, D.; Cappa, S.; Proverbio, A.M.; Zani, A.; Falini, A.; Canessa, N. Affective and cooperative social interactions modulate effective connectivity within and between the mirror and mentalizing systems. Hum. Brain Mapp. 2018, 39, 1412–1427. [Google Scholar] [CrossRef] [PubMed]
- Canessa, N.; Alemanno, F.; Riva, F.; Zani, A.; Proverbio, A.M.; Mannara, N.; Perani, D.; Cappa, S. The Neural Bases of Social Intention Understanding: The Role of Interaction Goals. PLoS ONE 2012, 7, e42347. [Google Scholar] [CrossRef] [PubMed]
- Pierno, A.C.; Becchio, C.; Turella, L.; Tubaldi, F.; Castiello, U. Observing social interactions: The effect of gaze. Soc. Neurosci. 2008, 3, 51–59. [Google Scholar] [CrossRef]
- Proverbio, A.M.; Riva, F.; Paganelli, L.; Cappa, S.; Canessa, N.; Perani, D.; Zani, A. Neural Coding of Cooperative vs. Affective Human Interactions: 150 ms to Code the Action’s Purpose. PLoS ONE 2011, 6, e22026. [Google Scholar] [CrossRef] [PubMed]
- Bühler, M.; Vollstädt-Klein, S.; Klemen, J.; Smolka, M.N. Does erotic stimulus presentation design affect brain activation patterns? Event-related vs. blocked fMRI designs. Behav. Brain Funct. 2008, 4, 30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferretti, A.; Caulo, M.; Del Gratta, C.; Di Matteo, R.; Merla, A.; Montorsi, F.; Pizzella, V.; Pompa, P.; Rigatti, P.; Rossini, P.M.; et al. Dynamics of male sexual arousal: Distinct components of brain activation revealed by fMRI. NeuroImage 2005, 26, 1086–1096. [Google Scholar] [CrossRef]
- Gu, X.; Han, S. Attention and reality constraints on the neural processes of empathy for pain. NeuroImage 2007, 36, 256–267. [Google Scholar] [CrossRef]
- Ogawa, K.; Inui, T. Neural representation of observed actions in the parietal and premotor cortex. NeuroImage 2011, 56, 728–735. [Google Scholar] [CrossRef]
- Redouté, J.; Stoléru, S.; Grégoire, M.-C.; Costes, N.; Cinotti, L.; Lavenne, F.; Le Bars, D.; Forest, M.G.; Pujol, J.-F. Brain processing of visual sexual stimuli in human males. Hum. Brain Mapp. 2000, 11, 162–177. [Google Scholar] [CrossRef]
- Wehrum, S.; Klucken, T.; Kagerer, S.; Walter, B.; Hermann, A.; Vaitl, D.; Stark, R. Gender Commonalities and Differences in the Neural Processing of Visual Sexual Stimuli. J. Sex. Med. 2013, 10, 1328–1342. [Google Scholar] [CrossRef] [PubMed]
- Johnson-Frey, S.H.; Maloof, F.R.; Newman-Norlund, R.; Farrer, C.; Inati, S.; Grafton, S.T. Actions or Hand-Object Interactions? Human Inferior Frontal Cortex and Action Observation. Neuron 2003, 39, 1053–1058. [Google Scholar] [CrossRef] [Green Version]
- Mazzarella, E.; Ramsey, R.; Conson, M.; Hamilton, A. Brain systems for visual perspective taking and action perception. Soc. Neurosci. 2013, 8, 248–267. [Google Scholar] [CrossRef] [PubMed]
- Watson, C.E.; Cardillo, E.R.; Bromberger, B.; Chatterjee, A. The specificity of action knowledge in sensory and motor systems. Front. Psychol. 2014, 5, 494. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bolognini, N.; Rossetti, A.; Convento, S.; Vallar, G. Understanding Others’ Feelings: The Role of the Right Primary Somatosensory Cortex in Encoding the Affective Valence of Others’ Touch. J. Neurosci. 2013, 33, 4201–4205. [Google Scholar] [CrossRef] [Green Version]
- Cheng, Y.; Yang, C.-Y.; Lin, C.-P.; Lee, P.-L.; Decety, J. The perception of pain in others suppresses somatosensory oscillations: A magnetoencephalography study. NeuroImage 2008, 40, 1833–1840. [Google Scholar] [CrossRef]
- Deuse, L.; Rademacher, L.; Winkler, L.; Schultz, R.; Gründer, G.; Lammertz, S. Neural correlates of naturalistic social cognition: Brain-behavior relationships in healthy adults. Soc. Cogn. Affect. Neurosci. 2016, 11, 1741–1751. [Google Scholar] [CrossRef] [Green Version]
- Jackson, P.L.; Meltzoff, A.N.; Decety, J. How do we perceive the pain of others? A window into the neural processes involved in empathy. NeuroImage 2005, 24, 771–779. [Google Scholar] [CrossRef] [Green Version]
- Hadjikhani, N.; de Gelder, B. Seeing Fearful Body Expressions Activates the Fusiform Cortex and Amygdala. Curr. Biol. 2003, 13, 2201–2205. [Google Scholar] [CrossRef]
- Poyo Solanas, M.P.; Zhan, M.; Vaessen, M.; Hortensius, R.; Engelen, T.; De Gelder, B. Looking at the face and seeing the whole body. Neural basis of combined face and body expressions. Soc. Cogn. Affect. Neurosci. 2018, 13, 135–144. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Keysers, C.; Perrett, D. Demystifying social cognition: A Hebbian perspective. Trends Cogn. Sci. 2004, 8, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Pitcher, D.; Ungerleider, L.G. Evidence for a Third Visual Pathway Specialized for Social Perception. Trends Cogn. Sci. 2021, 25, 100–110. [Google Scholar] [CrossRef] [PubMed]
- Amoruso, L.; Finisguerra, A.; Urgesi, C. Spatial frequency tuning of motor responses reveals differential contribution of dorsal and ventral systems to action comprehension. Proc. Natl. Acad. Sci. USA 2020, 117, 13151–13161. [Google Scholar] [CrossRef] [PubMed]
- Bar, M.; Kassam, K.S.; Ghuman, A.; Boshyan, J.; Schmid, A.M.; Dale, A.M.; Hamalainen, M.S.; Marinkovic, K.; Schacter, D.; Rosen, B.R.; et al. Top-down facilitation of visual recognition. Proc. Natl. Acad. Sci. USA 2006, 103, 449–454. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Decroix, J.; Roger, C.; Kalénine, S. Neural dynamics of grip and goal integration during the processing of others’ actions with objects: An ERP study. Sci. Rep. 2020, 10, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Hickok, G. Eight Problems for the Mirror Neuron Theory of Action Understanding in Monkeys and Humans. J. Cogn. Neurosci. 2009, 21, 1229–1243. [Google Scholar] [CrossRef] [Green Version]
- Hutchison, R.M.; Gallivan, J.P. Functional coupling between frontoparietal and occipitotemporal pathways during action and perception. Cortex 2018, 98, 8–27. [Google Scholar] [CrossRef]
- Wurm, M.F.; Caramazza, A. Distinct roles of temporal and frontoparietal cortex in representing actions across vision and language. Nat. Commun. 2019, 10, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Kilner, J.M. More than one pathway to action understanding. Trends Cogn. Sci. 2011, 15, 352–357. [Google Scholar] [CrossRef] [Green Version]
- Kislinger, L. Photographs Beyond Concepts: Access to Actions and Sensations. Rev. Gen. Psychol. 2021, 25, 44–59. [Google Scholar] [CrossRef]
- Mc Cabe, S.I.; Villalta, J.I.; Saunier, G.; Grafton, S.T.; Della-Maggiore, V. The Relative Influence of Goal and Kinematics on Corticospinal Excitability Depends on the Information Provided to the Observer. Cereb. Cortex 2015, 25, 2229–2237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Angelini, M.; Fabbri-Destro, M.; Lopomo, N.F.; Gobbo, M.; Rizzolatti, G.; Avanzini, P. Perspective-dependent reactivity of sensorimotor mu rhythm in alpha and beta ranges during action observation: An EEG study. Sci. Rep. 2018, 8, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Bolognini, N.; Rossetti, A.; Maravita, A.; Miniussi, C. Seeing touch in the somatosensory cortex: A TMS study of the visual perception of touch. Hum. Brain Mapp. 2011, 32, 2104–2114. [Google Scholar] [CrossRef] [Green Version]
- Borgomaneri, S.; Vitale, F.; Avenanti, A. Early changes in corticospinal excitability when seeing fearful body expressions. Sci. Rep. 2015, 5, srep14122. [Google Scholar] [CrossRef]
- Urgesi, C.; Maieron, M.; Avenanti, A.; Tidoni, E.; Fabbro, F.; Aglioti, S.M. Simulating the Future of Actions in the Human Corticospinal System. Cereb. Cortex 2010, 20, 2511–2521. [Google Scholar] [CrossRef] [Green Version]
- Borgomaneri, S.; Gazzola, V.; Avenanti, A. Motor mapping of implied actions during perception of emotional body language. Brain Stimul. 2012, 5, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Borgomaneri, S.; Gazzola, V.; Avenanti, A. Temporal dynamics of motor cortex excitability during perception of natural emotional scenes. Soc. Cogn. Affect. Neurosci. 2014, 9, 1451–1457. [Google Scholar] [CrossRef] [Green Version]
- Borgomaneri, S.; Gazzola, V.; Avenanti, A. Transcranial magnetic stimulation reveals two functionally distinct stages of motor cortex involvement during perception of emotional body language. Brain Struct. Funct. 2015, 220, 2765–2781. [Google Scholar] [CrossRef] [Green Version]
- Catmur, C.; Mars, R.B.; Rushworth, M.F.; Heyes, C. Making Mirrors: Premotor Cortex Stimulation Enhances Mirror and Counter-mirror Motor Facilitation. J. Cogn. Neurosci. 2011, 23, 2352–2362. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hajcak, G.; Molnar, C.; George, M.S.; Bolger, K.; Koola, J.; Nahas, Z. Emotion facilitates action: A transcranial magnetic stimulation study of motor cortex excitability during picture viewing. Psychophysiology 2007, 44, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Urgesi, C.; Moro, V.; Candidi, M.; Aglioti, S.M. Mapping Implied Body Actions in the Human Motor System. J. Neurosci. 2006, 26, 7942–7949. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Urgesi, C.; Candidi, M.; Ionta, S.; Aglioti, S.M. Representation of body identity and body actions in extrastriate body area and ventral premotor cortex. Nat. Neurosci. 2007, 10, 30–31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Avanzini, P.; Destro, M.F.; Campi, C.; Pascarella, A.; Barchiesi, G.; Cattaneo, L.; Rizzolatti, G. Spatiotemporal dynamics in understanding hand--object interactions. Proc. Natl. Acad. Sci. USA 2013, 110, 15878–15885. [Google Scholar] [CrossRef] [Green Version]
- Hauk, O.; Shtyrov, Y.; Pulvermuller, F. The time course of action and action-word comprehension in the human brain as revealed by neurophysiology. J. Physiol. 2008, 102, 50–58. [Google Scholar] [CrossRef] [Green Version]
- Ubaldi, S.; Barchiesi, G.; Cattaneo, L. Bottom-Up and Top-Down Visuomotor Responses to Action Observation. Cereb. Cortex 2015, 25, 1032–1041. [Google Scholar] [CrossRef] [Green Version]
- Catmur, C. Understanding intentions from actions: Direct perception, inference, and the roles of mirror and mentalizing systems. Conscious. Cogn. 2015, 36, 426–433. [Google Scholar] [CrossRef] [Green Version]
- Fazekas, P.; Nanay, B.; Pearson, J. Offline perception: An introduction. Philos. Trans. R. Soc. B Biol. Sci. 2021, 376, 20190686. [Google Scholar] [CrossRef]
- Koenig-Robert, R.; Pearson, J. Why do imagery and perception look and feel so different? Philos. Trans. R. Soc. B Biol. Sci. 2021, 376, 20190703. [Google Scholar] [CrossRef]
- Orlandi, A.; Arno, E.; Proverbio, A.M. The Effect of Expertise on Kinesthetic Motor Imagery of Complex Actions. Brain Topogr. 2020, 33, 238–254. [Google Scholar] [CrossRef] [PubMed]
- Urgesi, C.; Candidi, M.; Fabbro, F.; Romani, M.; Aglioti, S.M. Motor facilitation during action observation: Topographic mapping of the target muscle and influence of the onlooker’s posture. Eur. J. Neurosci. 2006, 23, 2522–2530. [Google Scholar] [CrossRef]
- Fadiga, L.; Fogassi, L.; Pavesi, G.; Rizzolatti, G. Motor facilitation during action observation: A magnetic stimulation study. J. Neurophysiol. 1995, 73, 2608–2611. [Google Scholar] [CrossRef] [PubMed]
- Avenanti, A.; Paracampo, R.; Annella, L.; Tidoni, E.; Aglioti, S.M. Boosting and Decreasing Action Prediction Abilities Through Excitatory and Inhibitory tDCS of Inferior Frontal Cortex. Cereb. Cortex 2018, 28, 1282–1296. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valchev, N.; Tidoni, E.; Hamilton, A.F.D.C.; Gazzola, V.; Avenanti, A. Primary somatosensory cortex necessary for the perception of weight from other people’s action: A continuous theta-burst TMS experiment. NeuroImage 2017, 152, 195–206. [Google Scholar] [CrossRef]
- Avikainen, S.; Forss, N.; Hari, R. Modulated Activation of the Human SI and SII Cortices during Observation of Hand Actions. NeuroImage 2002, 15, 640–646. [Google Scholar] [CrossRef]
- Barchiesi, G.; Cattaneo, L. Early and late motor responses to action observation. Soc. Cogn. Affect. Neurosci. 2013, 8, 711–719. [Google Scholar] [CrossRef] [Green Version]
- Van Loon, A.M.; van den Wildenberg, W.P.; Van Stegeren, A.H.; Ridderinkhof, K.R.; Hajcak, G. Emotional stimuli modulate readiness for action: A transcranial magnetic stimulation study. Cogn. Affect. Behav. Neurosci. 2010, 10, 174–181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berlot, E.; Popp, N.J.; Diedrichsen, J. In search of the engram, 2017. Curr. Opin. Behav. Sci. 2018, 20, 56–60. [Google Scholar] [CrossRef]
- Hamano, Y.H.; Sugawara, S.K.; Yoshimoto, T.; Sadato, N. The motor engram as a dynamic change of the cortical network during early sequence learning: An fMRI study. Neurosci. Res. 2020, 153, 27–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huber, D.; Gutnisky, D.; Peron, S.; O’Connor, D.H.; Wiegert, J.S.; Tian, L.; Oertner, T.; Looger, L.L.; Svoboda, K. Multiple dynamic representations in the motor cortex during sensorimotor learning. Nature 2012, 484, 473–478. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schellekens, W.; Petridou, N.; Ramsey, N.F. Detailed somatotopy in primary motor and somatosensory cortex revealed by Gaussian population receptive fields. NeuroImage 2018, 179, 337–347. [Google Scholar] [CrossRef]
- Rogerson, T.; Cai, D.; Frank, A.; Sano, Y.; Shobe, J.; Lopez-Aranda, M.F.; Silva, A.J. Synaptic tagging during memory allocation. Nat. Rev. Neurosci. 2014, 15, 157–169. [Google Scholar] [CrossRef]
- Tonegawa, S.; Liu, X.; Ramirez, S.; Redondo, R. Memory Engram Cells Have Come of Age. Neuron 2015, 87, 918–931. [Google Scholar] [CrossRef] [Green Version]
- Josselyn, S.A.; Kohler, S.; Frankland, P.W. Finding the engram. Nat. Rev. Neurosci. 2015, 16, 521–534. [Google Scholar] [CrossRef]
- Rashid, A.J.; Yan, C.; Mercaldo, V.; Hsiang, H.-L.; Park, S.; Cole, C.J.; De Cristofaro, A.; Yu, J.; Ramakrishnan, C.; Lee, S.Y.; et al. Competition between engrams influences fear memory formation and recall. Science 2016, 353, 383–387. [Google Scholar] [CrossRef] [Green Version]
- Sun, X.; Bernstein, M.J.; Meng, M.; Rao, S.; Sorensen, A.; Yao, L.; Zhang, X.; Anikeeva, P.O.; Lin, Y. Functionally Distinct Neuronal Ensembles within the Memory Engram. Cell 2020, 181, 410–423.e17. [Google Scholar] [CrossRef] [PubMed]
- Bennett, M.R.; Hacker, P.M. Philosophical Foundations of Neuroscience; Blackwell Publishing: Oxford, UK, 2003. [Google Scholar]
- Damasio, A.R. Self Comes to Mind: Constructing the Conscious Brain; William Heinemann: London, UK, 2010. [Google Scholar]
- Calvo-Merino, B.; Glaser, D.; Grèzes, J.; Passingham, R.; Haggard, P. Action Observation and Acquired Motor Skills: An fMRI Study with Expert Dancers. Cereb. Cortex 2005, 15, 1243–1249. [Google Scholar] [CrossRef] [Green Version]
- Byrne, R.W. Imitation as behaviour parsing. Philos. Trans. R. Soc. B Biol. Sci. 2003, 358, 529–536. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grafton, S.T.; Hamilton, A. Evidence for a distributed hierarchy of action representation in the brain. Hum. Mov. Sci. 2007, 26, 590–616. [Google Scholar] [CrossRef] [Green Version]
- Wolpert, D.M.; Ghahramani, Z. Computational principles of movement neuroscience. Nat. Neurosci. 2000, 3, 1212–1217. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, R.A. A schema theory of discrete motor skill learning. Psychol. Rev. 1975, 82, 225–260. [Google Scholar] [CrossRef]
- Wolpert, D.; Kawato, M. Multiple paired forward and inverse models for motor control. Neural Netw. 1998, 11, 1317–1329. [Google Scholar] [CrossRef]
- Del Vecchio, M.; Caruana, F.; Sartori, I.; Pelliccia, V.; Zauli, F.M.; Russo, G.L.; Rizzolatti, G.; Avanzini, P. Action execution and action observation elicit mirror responses with the same temporal profile in human SII. Commun. Biol. 2020, 3, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Hantman, A.W.; Jessell, T.M. Clarke’s column neurons as the focus of a corticospinal corollary circuit. Nat. Neurosci. 2010, 13, 1233–1239. [Google Scholar] [CrossRef]
- Kilner, J.M.; Friston, K.; Frith, C. Predictive coding: An account of the mirror neuron system. Cogn. Process. 2007, 8, 159–166. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.; Snow, J.C.; Culham, J.C.; Goodale, A.M. What Role Does “Elongation” Play in “Tool-Specific” Activation and Connectivity in the Dorsal and Ventral Visual Streams? Cereb. Cortex 2018, 28, 1117–1131. [Google Scholar] [CrossRef]
- Hebart, M.N.; Hesselmann, G. What Visual Information Is Processed in the Human Dorsal Stream? J. Neurosci. 2012, 32, 8107–8109. [Google Scholar] [CrossRef] [PubMed]
- Sakuraba, S.; Sakai, S.; Yamanaka, M.; Yokosawa, K.; Hirayama, K. Does the Human Dorsal Stream Really Process a Category for Tools? J. Neurosci. 2012, 32, 3949–3953. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bradley, M.M.; Codispoti, M.; Cuthbert, B.N.; Lang, P.J. Emotion and motivation I: Defensive and appetitive reactions in picture processing. Emotion 2001, 1, 276–298. [Google Scholar] [CrossRef] [PubMed]
- Kislinger, L.; Kotrschal, K. Hunters and Gatherers of Pictures: Why Photography Has Become a Human Universal. Front. Psychol. 2021, 12, 654474. [Google Scholar] [CrossRef]
- Nelissen, K.; Borra, E.; Gerbella, M.; Rozzi, S.; Luppino, G.; Vanduffel, W.; Rizzolatti, G.; Orban, G. Action Observation Circuits in the Macaque Monkey Cortex. J. Neurosci. 2011, 31, 3743–3756. [Google Scholar] [CrossRef] [PubMed]
- Rizzolatti, G.; Craighero, L. The mirror-neuron system. Annu. Rev. Neurosci. 2004, 27, 169–192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amoruso, L.; Gelormini, C.; Aboitiz, F.A.; González, M.A.; Manes, F.; Cardona, J.F.; Ibanez, A. N400 ERPs for actions: Building meaning in context. Front. Hum. Neurosci. 2013, 7, 57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Espírito Santo, M.G.E.; Maxim, O.S.; Schürmann, M. N1 responses to images of hands in occipito-temporal event-related potentials. Neuropsychologia 2017, 106, 83–89. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moreau, Q.; Parrotta, E.; Era, V.; Martelli, M.L.; Candidi, M. Role of the occipito-temporal theta rhythm in hand visual identification. J. Neurophysiol. 2020, 123, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Peelen, M.; Downing, P. The neural basis of visual body perception. Nat. Rev. Neurosci. 2007, 8, 636–648. [Google Scholar] [CrossRef] [PubMed]
- Hafri, A.; Papafragou, A.; Trueswell, J.C. Getting the gist of events: Recognition of two-participant actions from brief displays. J. Exp. Psychol. Gen. 2013, 142, 880–905. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Geuzebroek, A.C.; van den Berg, A.V. Eccentricity scale independence for scene perception in the first tens of milliseconds. J. Vis. 2018, 18, 9. [Google Scholar] [CrossRef] [Green Version]
- Lee, B.B.; Martin, P.; Grünert, U. Retinal connectivity and primate vision. Prog. Retin. Eye Res. 2010, 29, 622–639. [Google Scholar] [CrossRef] [Green Version]
- Musel, B.; Bordier, C.; Dojat, M.; Pichat, C.; Chokron, S.; Le Bas, J.-F.; Peyrin, C. Retinotopic and Lateralized Processing of Spatial Frequencies in Human Visual Cortex during Scene Categorization. J. Cogn. Neurosci. 2013, 25, 1315–1331. [Google Scholar] [CrossRef] [Green Version]
- Cohen, M.A.; Rubenstein, J. How much color do we see in the blink of an eye? Cognition 2020, 200, 104268. [Google Scholar] [CrossRef]
- Edwards, M.; Goodhew, S.C.; Badcock, D.R. Using perceptual tasks to selectively measure magnocellular and parvocellular performance: Rationale and a user’s guide. Psychon. Bull. Rev. 2021, 28, 1029–1050. [Google Scholar] [CrossRef] [PubMed]
- Masri, R.A.; Grünert, U.; Martin, P.R. Analysis of Parvocellular and Magnocellular Visual Pathways in Human Retina. J. Neurosci. 2020, 40, 8132–8148. [Google Scholar] [CrossRef] [PubMed]
- Carretié, L.; Kessel, D.; García-Rubio, M.J.; Giménez-Fernández, T.; Hoyos, S.; Hernández-Lorca, M. Magnocellular Bias in Exogenous Attention to Biologically Salient Stimuli as Revealed by Manipulating Their Luminosity and Color. J. Cogn. Neurosci. 2017, 29, 1699–1711. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, A.; Eysel, U.T.; Vidyasagar, T.R. The role of the magnocellular pathway in serial deployment of visual attention. Eur. J. Neurosci. 2004, 20, 2188–2192. [Google Scholar] [CrossRef]
- Graziano, M.S. New Insights into Motor Cortex. Neuron 2011, 71, 387–388. [Google Scholar] [CrossRef] [Green Version]
- Binder, J.R.; Desai, R.H. The neurobiology of semantic memory. Trends Cogn. Sci. 2011, 15, 527–536. [Google Scholar] [CrossRef] [Green Version]
- Gallese, V.; Fadiga, L.; Fogassi, L.; Rizzolatti, G. Action representation and the inferior parietal lobule. In Common Mechanisms in Perception and Action: Attention and Performance XIX; Prinz, W., Hommel, B., Eds.; Oxford University Press: New York, NY, USA, 2002; pp. 334–355. [Google Scholar]
- Goodale, M.A.; Milner, A. Separate visual pathways for perception and action. Trends Neurosci. 1992, 15, 20–25. [Google Scholar] [CrossRef]
- Urgesi, C.; Candidi, M.; Avenanti, A. Neuroanatomical substrates of action perception and understanding: An anatomic likelihood estimation meta-analysis of lesion-symptom mapping studies in brain injured patients. Front. Hum. Neurosci. 2014, 8, 344. [Google Scholar] [CrossRef] [Green Version]
- Bradley, M.M.; Lang, P.J. The International Affective Picture System (IAPS) in the study of emotion and attention. In Handbook of Emotion Elicitation and Assessment; Coan, J.A., Allen, J.J.B., Eds.; Oxford University Press: New York, NY, USA, 2007; pp. 29–46. [Google Scholar]
- Mouras, H.; Stoléru, S.; Moulier, V.; Pélégrini-Issac, M.; Rouxel, R.; Grandjean, B.; Glutron, D.; Bittoun, J. Activation of mirror-neuron system by erotic video clips predicts degree of induced erection: An fMRI study. NeuroImage 2008, 42, 1142–1150. [Google Scholar] [CrossRef] [PubMed]
- Codispoti, M.; Bradley, M.M.; Lang, P.J. Affective reactions to briefly presented pictures. Psychophysiology 2001, 38, 474–478. [Google Scholar] [CrossRef]
- Lang, P.J.; Greenwald, M.K.; Bradley, M.M.; Hamm, A.O. Looking at pictures: Affective, facial, visceral, and behavioral reactions. Psychophysiology 1993, 30, 261–273. [Google Scholar] [CrossRef]
- Strigo, I.A.; Craig, A.D. Interoception, homeostatic emotions and sympathovagal balance. Philos. Trans. R. Soc. B Biol. Sci. 2016, 371, 20160010. [Google Scholar] [CrossRef] [Green Version]
- Bolliet, O.; Collet, C.; Dittmar, A. Observation of Action and Autonomic Nervous System Responses. Percept. Mot. Ski. 2005, 101, 195–202. [Google Scholar] [CrossRef]
- Chivers, M.L.; Seto, M.; Lalumière, M.L.; Laan, E.; Grimbos, T. Agreement of Self-Reported and Genital Measures of Sexual Arousal in Men and Women: A Meta-Analysis. Arch. Sex. Behav. 2010, 39, 5–56. [Google Scholar] [CrossRef] [Green Version]
- Bastiaansen, J.; Thioux, M.; Keysers, C. Evidence for mirror systems in emotions. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 2391–2404. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borhani, K.; Ladavas, E.; Maier, M.E.; Avenanti, A.; Bertini, C. Emotional and movement-related body postures modulate visual processing. Soc. Cogn. Affect. Neurosci. 2015, 10, 1092–1101. [Google Scholar] [CrossRef] [Green Version]
- Goldberg, H.; Preminger, S.; Malach, R. The emotion–action link? Naturalistic emotional stimuli preferentially activate the human dorsal visual stream. NeuroImage 2014, 84, 254–264. [Google Scholar] [CrossRef] [PubMed]
- Fazekas, P.; Overgaard, M. A Multi-Factor Account of Degrees of Awareness. Cogn. Sci. 2018, 42, 1833–1859. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rudrauf, D.; Lachaux, J.-P.; Damasio, A.; Baillet, S.; Hugueville, L.; Martinerie, J.; Damasio, H.; Renault, B. Enter feelings: Somatosensory responses following early stages of visual induction of emotion. Int. J. Psychophysiol. 2009, 72, 13–23. [Google Scholar] [CrossRef]
- Barrett, L.F.; Bar, M. See it with feeling: Affective predictions during object perception. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 1325–1334. [Google Scholar] [CrossRef] [PubMed]
- Pourtois, G.; Schettino, A.; Vuilleumier, P. Brain mechanisms for emotional influences on perception and attention: What is magic and what is not. Biol. Psychol. 2013, 92, 492–512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dijkerman, H.C.; de Haan, E. Somatosensory processes subserving perception and action. Behav. Brain Sci. 2007, 30, 189–201. [Google Scholar] [CrossRef] [PubMed]
- Gombrich, E.H. Moment and Movement in Art. J. Warbg. Court. Inst. 1964, 27, 293. [Google Scholar] [CrossRef]
- Brysbaert, M. How many words do we read per minute? A review and meta-analysis of reading rate. J. Mem. Lang. 2019, 109, 104047. [Google Scholar] [CrossRef] [Green Version]
- Hayes, T.R.; Henderson, J.M. Center bias outperforms image salience but not semantics in accounting for attention during scene viewing. Atten. Percept. Psychophys. 2020, 82, 985–994. [Google Scholar] [CrossRef]
- Fan, Y.; Han, S. Temporal dynamic of neural mechanisms involved in empathy for pain: An event-related brain potential study. Neuropsychologia 2008, 46, 160–173. [Google Scholar] [CrossRef]
- Zeki, S.; Watson, J.; Frackowiak, R. Going beyond the information given: The relation of illusory visual motion to brain activity. Proc. R. Soc. B Boil. Sci. 1993, 252, 215–222. [Google Scholar] [CrossRef]
- Cutting, J.E. Representing Motion in a Static Image: Constraints and Parallels in Art, Science, and Popular Culture. Perception 2002, 31, 1165–1193. [Google Scholar] [CrossRef]
- Winawer, J.; Huk, A.C.; Boroditsky, L. A Motion Aftereffect From Still Photographs Depicting Motion. Psychol. Sci. 2008, 19, 276–283. [Google Scholar] [CrossRef]
- Chao, L.; Martin, A. Representation of Manipulable Man-Made Objects in the Dorsal Stream. NeuroImage 2000, 12, 478–484. [Google Scholar] [CrossRef] [Green Version]
- Iacoboni, M.; Molnar-Szakacs, I.; Gallese, V.; Buccino, G.; Mazziotta, J.C.; Rizzolatti, G. Grasping the Intentions of Others with One’s Own Mirror Neuron System. PLoS Biol. 2005, 3, e79. [Google Scholar] [CrossRef] [Green Version]
- Ortigue, S.; Sinigaglia, C.; Rizzolatti, G.; Grafton, S.T. Understanding Actions of Others: The Electrodynamics of the Left and Right Hemispheres. A High-Density EEG Neuroimaging Study. PLoS ONE 2010, 5, e12160. [Google Scholar] [CrossRef] [PubMed]
- Amoruso, L.; Finisguerra, A. Low or High-Level Motor Coding? The Role of Stimulus Complexity. Front. Hum. Neurosci. 2019, 13, 332. [Google Scholar] [CrossRef]
- Liu, L.; Ioannides, A.A. Emotion Separation Is Completed Early and It Depends on Visual Field Presentation. PLoS ONE 2010, 5, e9790. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wood, A.; Rychlowska, M.; Korb, S.; Niedenthal, P. Fashioning the Face: Sensorimotor Simulation Contributes to Facial Expression Recognition. Trends Cogn. Sci. 2016, 20, 227–240. [Google Scholar] [CrossRef] [PubMed]
- Hietanen, J.K.; Nummenmaa, L. The Naked Truth: The Face and Body Sensitive N170 Response Is Enhanced for Nude Bodies. PLoS ONE 2011, 6, e24408. [Google Scholar] [CrossRef]
- De Gelder, B.; De Borst, A.; Watson, R. The perception of emotion in body expressions. Wiley Interdiscip. Rev. Cogn. Sci. 2015, 6, 149–158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brosch, T.; Sander, D.; Pourtois, G.; Scherer, K.R. Beyond Fear. Psychol. Sci. 2008, 19, 362–370. [Google Scholar] [CrossRef]
- Fiave, P.A.; Nelissen, K. Motor resonance in monkey parietal and premotor cortex during action observation: Influence of viewing perspective and effector identity. NeuroImage 2021, 224, 117398. [Google Scholar] [CrossRef]
- Ge, S.; Liu, H.; Lin, P.; Gao, J.; Xiao, C.; Li, Z. Neural Basis of Action Observation and Understanding from First- and Third-Person Perspectives: An fMRI Study. Front. Behav. Neurosci. 2018, 12, 283. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vogt, S.; Taylor, P.; Hopkins, B. Visuomotor priming by pictures of hand postures: Perspective matters. Neuropsychologia 2003, 41, 941–951. [Google Scholar] [CrossRef]

| Brain Areas | Studies |
|---|---|
| Extrastriate body area (EBA) | Downing et al., 2006 [14]; Hafri et al., 2017 [15]; Lu et al., 2016 [16]; O’Toole et al., 2014 [17]; Proverbio et al., 2009 [18]; Thierry et al., 2006 [19] |
| Middle temporal area (MT) | Hafri et al., 2017 [15]; Hermsdörfer et al., 2001 [20]; Kolesar et al., 2017 [21]; Kourtzi & Kanwisher, 2000 [22]; Lu et al., 2016 [16]; O’Toole et al., 2014 [17]; Proverbio et al., 2009 [18] |
| Additional regions of the posterior superior temporal sulcus (pSTS) | Arioli et al., 2018 [23]; Canessa et al., 2012 [24]; Hafri et al., 2017 [15]; Hermsdörfer et al., 2001 [20]; Kourtzi & Kanwisher, 2000 [22]; O’Toole et al., 2014 [17]; Pierno et al., 2008 [25]; Proverbio et al., 2011 [26] |
| Inferior parietal lobule (IPL) and/or intraparietal sulus (IPS) | Bühler et al., 2008 [27]; Canessa et al., 2012 [24]; Ferretti et al., 2005 [28]; Gu & Han, 2007 [29]; Hafri et al., 2017 [15]; Hermsdörfer et al., 2001 [20]; Kolesar et al., 2017 [21]; Ogawa & Inui, 2011 [30]; Proverbio et al., 2009 [18]; Redouté et al., 2000 [31]; Wehrum et al., 2013 [32] |
| Premotor cortex (PMC) and/or inferior frontal gyrus (IFG) | Arioli et al., 2018 [23]; Canessa et al., 2012 [24]; Hafri et al., 2017 [15]; Johnson-Frey et al., 2003 [33]; Kolesar et al., 2017 [21]; Mazzarella et al., 2013 [34]; Ogawa & Inui, 2011 [30]; Pierno et al., 2008 [25]; Proverbio et al., 2009 [18]; Watson et al., 2014 [35] |
| Primary and/or secondary somatosensory cortex (S1, S2) | Bolognini et al., 2013 [36]; Bühler et al., 2008 [27]; Cheng et al., 2008 [37]; Gu & Han, 2007 [29]; Proverbio et al., 2011 [26] |
| Insula | Arioli et al., 2018 [23]; Bühler et al., 2008 [27]; Deuse et al., 2016 [38]; Gu & Han, 2007 [29]; Jackson et al., 2005 [39]; Kolesar et al., 2017 [21]; Wehrum et al., 2013 [32] |
| Anterior cingulate cortex (ACC) | Bühler et al., 2008 [27]; Gu & Han, 2007 [29]; Jackson et al., 2005 [39]; Kolesar et al., 2017 [21]; Proverbio et al., 2009 [18], 2011 [26]; Redouté et al., 2000 [31]; Wehrum et al., 2013 [32] |
| Orbitofrontal cortex (OFC) | Bühler et al., 2008 [27]; Deuse et al., 2016 [38]; Redouté et al., 2000 [31]; Wehrum et al., 2013 [32] |
| Amygdala | Deuse et al., 2016 [38]; Ferretti et al., 2005 [28]; Hadjikhani & de Gelder, 2003 [40]; Pierno et al., 2008 [25]; Poyo Solanas et al., 2018 [41] |
| Changes in Corticospinal Excitability, Motor Facilitation, and/or Downstream Modulation | |
|---|---|
| Photos as stimuli | Amoruso et al., 2020 [44]; Borgomaneri et al., 2014 [58], 2015 [59], 2015 [55]; Catmur et al., 2011 [60]; Hajcak et al., 2007 [61]; Urgesi et al., 2006 [62], 2010 [56] |
| Video clips (or apparent motion cues) as stimuli | Mc Cabe et al., 2015 [52]; Ubaldi et al., 2015 [66]; Urgesi et al., 2006 [71] |
| Real actions as stimuli | Fadiga et al., 1995 [72]; Feurra et al., 2019 [2] |
| Muscle specificity in changed motor excitability and facilitation | |
| Photos as stimuli | Amoruso et al., 2020 [44]; Borgomaneri et al., 2015 [55]; Catmur et al., 2011 [60]; Urgesi et al., 2006 [62], 2010 [56] |
| Real actions as stimuli | Fadiga et al., 1995 [72] |
| Activations in the AON form a neural simulation of a pictured action | |
| Photos as stimuli | Bolognini et al., 2013 [36]; Borgomaneri et al., 2012 [57], 2015 [59]; Urgesi et al., 2006 [62], 2010 [56] |
| Video clips as stimuli | Bolognini et al., 2011 [54]; Avenanti et al., 2018 [73] a; Jacquet & Avenanti, 2015 [12] |
| Specific causal contributions from certain brain areas in action perception | |
| Photos as stimuli | Bolognini et al., 2013 [36]; Catmur et al., 2011 [60]; Urgesi et al., 2007 [63] |
| Video clips as stimuli | Avenanti et al., 2018 [73] a; Bolognini et al., 2011 [54]; Jacquet & Avenanti, 2015 [12]; Valchev et al., 2016 [10], 2017 [74] |
| Involvement of somatosensory activations in action perception | |
| Photos as stimuli | Bolognini et al., 2013 [36] |
| Video clips as stimuli | Bolognini et al., 2011 [54]; Jacquet & Avenanti, 2015 [12]; Valchev et al., 2016 [10], 2017 [74] |
| Real actions as stimuli | Avikainen et al., 2002 [75] b |
| Time courses of neural processing stages of pictured actions | |
| Photos as stimuli | Avanzini et al., 2013 [64]; Borgomaneri et al., 2014 [58], 2015 [59], 2015 [55] |
| Video clips (or apparent motion cues) as stimuli | Barchiesi & Cattaneo, 2013 [76]; Ubaldi et al., 2015 [66] |
| Close connections between the processing of body postures and emotional value or behavioral relevance | |
| Photos as stimuli | Borgomaneri et al., 2012 [57], 2014 [58], 2015 [59], 2015 [55]; Hajcak et al., 2007 [61]; van Loon et al., 2010 [77] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kislinger, L. Photographs of Actions: What Makes Them Special Cues to Social Perception. Brain Sci. 2021, 11, 1382. https://doi.org/10.3390/brainsci11111382
Kislinger L. Photographs of Actions: What Makes Them Special Cues to Social Perception. Brain Sciences. 2021; 11(11):1382. https://doi.org/10.3390/brainsci11111382
Chicago/Turabian StyleKislinger, Leopold. 2021. "Photographs of Actions: What Makes Them Special Cues to Social Perception" Brain Sciences 11, no. 11: 1382. https://doi.org/10.3390/brainsci11111382






