Auditory Brainstem Response Wave I Amplitude Has Limited Clinical Utility in Diagnosing Tinnitus in Humans
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
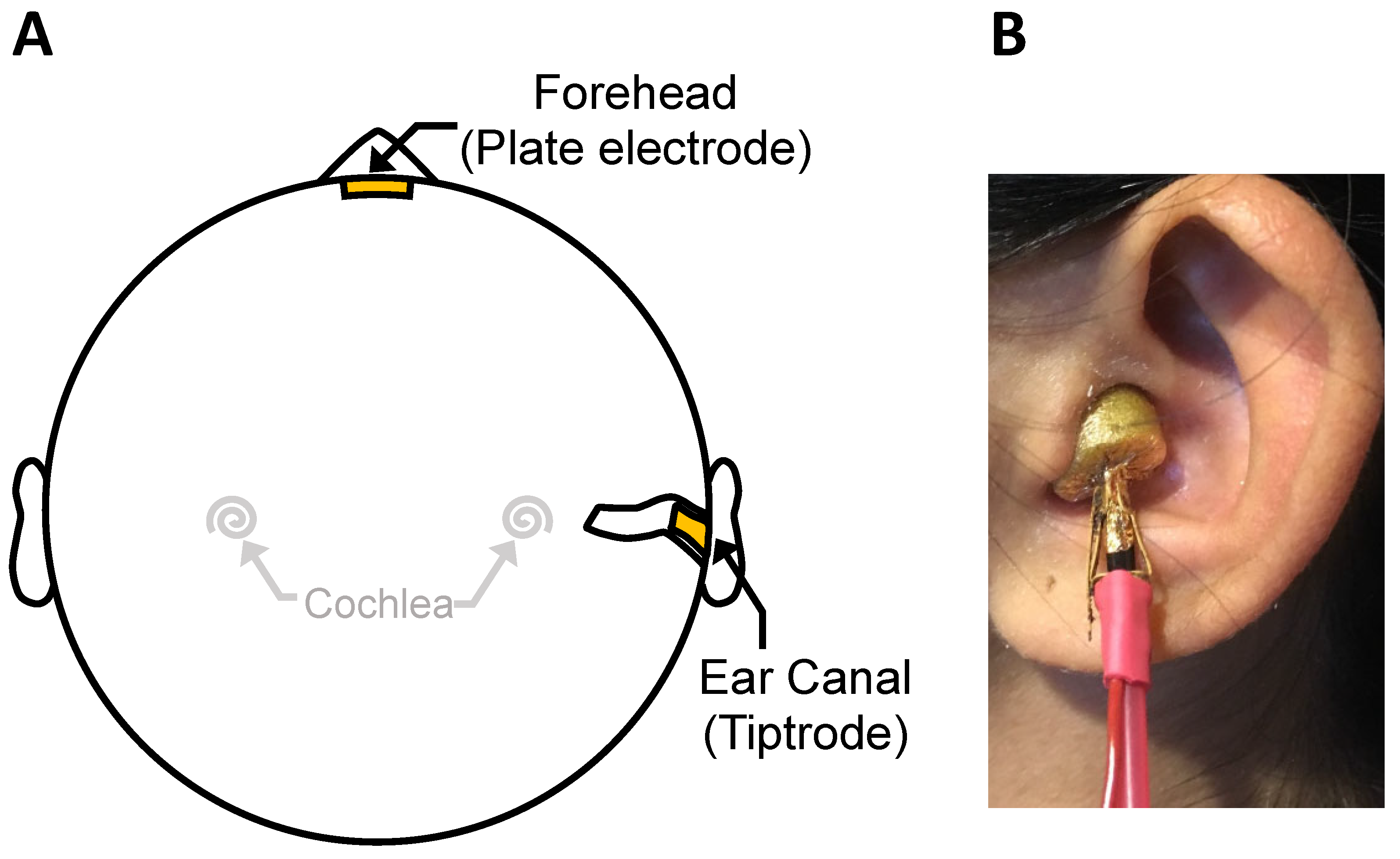
2.2. Auditory Brainstem Responses
2.3. Data Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kujawa, S.G.; Liberman, M.C. Adding Insult to Injury: Cochlear Nerve Degeneration after "Temporary" Noise-Induced Hearing Loss. J. Neurosci. 2009, 29, 14077–14085. [Google Scholar] [CrossRef] [Green Version]
- Fernandez, K.A.; Jeffers, P.W.; Lall, K.; Liberman, M.C.; Kujawa, S.G. Aging after Noise Exposure: Acceleration of Cochlear Synaptopathy in "Recovered" Ears. J. Neurosci. 2015, 35, 7509–7520. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Furman, A.C.; Kujawa, S.G.; Liberman, M.C. Noise-induced cochlear neuropathy is selective for fibers with low spontaneous rates. J. Neurophysiol. 2013, 110, 577–586. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.W.; Furman, A.C.; Kujawa, S.G.; Liberman, M.C. Primary neural degeneration in the Guinea pig cochlea after reversible noise-induced threshold shift. J Assoc Res Otolaryngol. 2011, 12, 605–616. [Google Scholar] [CrossRef] [Green Version]
- Lobarinas, E.; Spankovich, C.; Le Prell, C. Evidence of “hidden hearing loss” following noise exposures that produce robust TTS and ABR wave-I amplitude reductions. Hear. Res. 2017, 349, 155–163. [Google Scholar] [CrossRef]
- Johannesen, P.T.; Buzo, B.C.; Lopez-Poveda, E.A. Evidence for age-related cochlear synaptopathy in humans unconnected to speech-in-noise intelligibility deficits. Hear. Res. 2019, 374, 35–48. [Google Scholar] [CrossRef]
- Liberman, M.C.; Kujawa, S.G. Cochlear synaptopathy in acquired sensorineural hearing loss: Manifestations and mechanisms. Hear. Res. 2017, 349, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Bramhall, N.; Beach, E.F.; Epp, B.; Le Prell, C.G.; Lopez-Poveda, E.A.; Plack, C.J.; Schaette, R.; Verhulst, S.; Canlon, B. The search for noise-induced cochlear synaptopathy in humans: Mission impossible? Hear. Res. 2019, 377, 88–103. [Google Scholar] [CrossRef] [Green Version]
- Schaette, R.; McAlpine, D. Tinnitus with a normal audiogram: Physiological evidence for hidden hearing loss and computa-tional model. J. Neurosci. 2011, 31, 13452–13457. [Google Scholar] [CrossRef] [Green Version]
- Gu, J.W.; Herrmann, B.S.; Levine, R.A.; Melcher, J.R. Brainstem Auditory Evoked Potentials Suggest a Role for the Ventral Cochlear Nucleus in Tinnitus. J. Assoc. Res. Otolaryngol. 2012, 13, 819–833. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Axelsson, A.; Ringdahl, A. Tinnitus—A study of its prevalence and characteristics. Br. J. Audiol. 1989, 23, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Henry, J.A.; Dennis, K.C.; Schechter, M.A. General review of tinnitus: Prevalence, mechanisms, effects, and management. J Speech Lang Hear Res. 2005, 48, 1204–1235. [Google Scholar] [CrossRef]
- Baguley, D.; McFerran, D.; Hall, D. Tinnitus. Lancet 2013, 382, 1600–1607. [Google Scholar] [CrossRef] [Green Version]
- McCormack, A.; Edmondson-Jones, M.; Somerset, S.; Hall, D. A systematic review of the reporting of tinnitus prevalence and severity. Hear. Res. 2016, 337, 70–79. [Google Scholar] [CrossRef]
- Meikle, M.B.; Henry, J.A.; Griest, S.E.; Stewart, B.J.; Abrams, H.B.; McArdle, R.; Myers, P.J.; Newman, C.W.; Sandridge, S.; Turk, D.C.; et al. The tinnitus functional index: Development of a new clinical measure for chronic, intrusive tinnitus. Ear Hear. 2012, 33, 153–176. [Google Scholar] [CrossRef] [PubMed]
- Khalfa, S.; Dubal, S.; Veuillet, E.; Perez-Diaz, F.; Jouvent, R.; Collet, L. Psychometric normalization of a hyperacusis questionnaire. J Otorhinolaryngol Relat Spec. 2002, 64, 436–442. [Google Scholar] [CrossRef]
- Bauch, C.D.; Olsen, W.O. Comparison of ABR Amplitudes with TIPtrode™ and Mastoid Electrodes. Ear Hear. 1990, 11, 463–467. [Google Scholar] [CrossRef]
- Prendergast, G.; Tu, W.; Guest, H.; Millman, R.E.; Kluk, K.; Couth, S.; Munro, K.J.; Plack, C. Supra-threshold auditory brainstem response amplitudes in humans: Test-retest reliability, electrode montage and noise exposure. Hear. Res. 2018, 364, 38–47. [Google Scholar] [CrossRef]
- Bramhall, N.; Ong, B.; Ko, J.; Parker, M. Speech Perception Ability in Noise is Correlated with Auditory Brainstem Response Wave I Amplitude. J. Am. Acad. Audiol. 2015, 26, 509–517. [Google Scholar] [CrossRef]
- Jastreboff, P.J.; Jastreboff, M.M. Tinnitus Retraining Therapy (TRT) as a method for treatment of tinnitus and hyperacusis patients. J. Am. Acad. Audiol. 2000, 11, 162–177. [Google Scholar]
- Schecklmann, M.; Landgrebe, M.; Langguth, B.; Grp, T.D.S. Phenotypic Characteristics of Hyperacusis in Tinnitus. PLoS ONE 2014, 9, e86944. [Google Scholar] [CrossRef] [Green Version]
- Ferraro, J.A.; Ferguson, R. Tympanic ECochG and conventional ABR: A combined approach for the identification of wave I and the I-V interwave interval. Ear Hear. 1989, 10, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Möhrle, D.; Hofmeier, B.; Amend, M.; Wolpert, S.; Ni, K.; Bing, D.; Klose, U.; Pichler, B.; Knipper, M.; Rüttiger, L. Enhanced Central Neural Gain Compensates Acoustic Trauma-induced Cochlear Impairment, but Unlikely Correlates with Tinnitus and Hyperacusis. Neurosci. 2019, 407, 146–169. [Google Scholar] [CrossRef]
- Paul, B.T.; Bruce, I.C.; Roberts, L.E. Evidence that hidden hearing loss underlies amplitude modulation encoding deficits in individuals with and without tinnitus. Hear. Res. 2017, 344, 170–182. [Google Scholar] [CrossRef] [PubMed]
- Moon, I.J.; Won, J.H.; Kang, H.W.; Kim, D.H.; An, Y.-H.; Shim, H.J. Influence of Tinnitus on Auditory Spectral and Temporal Resolution and Speech Perception in Tinnitus Patients. J. Neurosci. 2015, 35, 14260–14269. [Google Scholar] [CrossRef] [Green Version]
- Mehraei, G.; Hickox, A.E.; Bharadwaj, H.M.; Goldberg, H.; Verhulst, S.; Liberman, M.C.; Shinn-Cunningham, B.G. Auditory Brainstem Response Latency in Noise as a Marker of Cochlear Synaptopathy. J. Neurosci. 2016, 36, 3755–3764. [Google Scholar] [CrossRef] [PubMed]
- Lockwood, A.H.; Salvi, R.J.; Burkard, R.F. Tinnitus. N. Engl. J. Med. 2002, 347, 904–910. [Google Scholar] [CrossRef]

| Wave I Amplitude at 70 dB nHL (μV) | Wave I Amplitude Slope (μV/dB) | ||||||
|---|---|---|---|---|---|---|---|
| Stimulus | Tinnitus | Mean | 95% CI | p Value | Mean | 95% CI | p Value |
| Click | No | 0.2192 | 0.1483−0.2901 | 0.66 | 0.0040 | 0.0022−0.0058 | 0.49 |
| Yes | 0.2397 | 0.1841−0.2953 | 0.0048 | 0.0035−0.0061 | |||
| 8000 Hz Tone | No | 0.1111 | 0.0728−0.1494 | 0.86 | 0.0014 | 0.0004−0.0024 | 0.80 |
| Yes | 0.1069 | 0.0794−0.1344 | 0.0016 | 0.0010−0.0022 | |||
| 4000 Hz Tone | No | 0.1413 | 0.1031−0.1795 | 0.18 | 0.0026 | 0.0016−0.0036 | 0.18 |
| Yes | 0.1830 | 0.1374−0.2286 | 0.0037 | 0.0025−0.0049 | |||
| 1000 Hz Tone | No | 0.0614 | 0.0475−0.0753 | 0.45 | 0.0007 | 0.0002−0.0012 | 0.25 |
| Yes | 0.0693 | 0.0548−0.0838 | 0.0010 | 0.0006−0.0014 | |||
| Wave I Amplitude at 70 dB nHL (μV) | Wave I Amplitude Slope (μV/dB) | ||||||
|---|---|---|---|---|---|---|---|
| Stimulus | Tinnitus | Mean | 95% CI | p Value | Mean | 95% CI | p Value |
| Click | No | 0.3452 | 0.2496−0.4408 | 0.81 | 0.0063 | 0.0033−0.0093 | 0.73 |
| Yes | 0.3293 | 0.2442−0.4144 | 0.0069 | 0.0050−0.0088 | |||
| 8000 Hz Tone | No | 0.1633 | 0.1106−0.2160 | 0.78 | 0.0022 | 0.0005−0.0039 | 0.97 |
| Yes | 0.1540 | 0.1205−0.1875 | 0.0023 | 0.0015−0.0031 | |||
| 4000 Hz Tone | No | 0.2114 | 0.1665−0.2563 | 0.07 | 0.0041 | 0.0028−0.0054 | 0.06 |
| Yes | 0.2668 | 0.2090−0.3246 | 0.0061 | 0.0049−0.0073 | |||
| 1000 Hz Tone | No | 0.0776 | 0.0619−0.0933 | 0.06 | 0.0090 | 0.0067−0.0113 | 0.49 |
| Yes | 0.0990 | 0.0855−0.1125 | 0.0100 | 0.0086−0.0114 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Turner, K.; Moshtaghi, O.; Saez, N.; Richardson, M.; Djalilian, H.; Zeng, F.-G.; Lin, H. Auditory Brainstem Response Wave I Amplitude Has Limited Clinical Utility in Diagnosing Tinnitus in Humans. Brain Sci. 2022, 12, 142. https://doi.org/10.3390/brainsci12020142
Turner K, Moshtaghi O, Saez N, Richardson M, Djalilian H, Zeng F-G, Lin H. Auditory Brainstem Response Wave I Amplitude Has Limited Clinical Utility in Diagnosing Tinnitus in Humans. Brain Sciences. 2022; 12(2):142. https://doi.org/10.3390/brainsci12020142
Chicago/Turabian StyleTurner, Katie, Omid Moshtaghi, Neil Saez, Matthew Richardson, Hamid Djalilian, Fan-Gang Zeng, and Harrison Lin. 2022. "Auditory Brainstem Response Wave I Amplitude Has Limited Clinical Utility in Diagnosing Tinnitus in Humans" Brain Sciences 12, no. 2: 142. https://doi.org/10.3390/brainsci12020142
APA StyleTurner, K., Moshtaghi, O., Saez, N., Richardson, M., Djalilian, H., Zeng, F.-G., & Lin, H. (2022). Auditory Brainstem Response Wave I Amplitude Has Limited Clinical Utility in Diagnosing Tinnitus in Humans. Brain Sciences, 12(2), 142. https://doi.org/10.3390/brainsci12020142






