Agomelatine: A Potential Multitarget Compound for Neurodevelopmental Disorders
Abstract
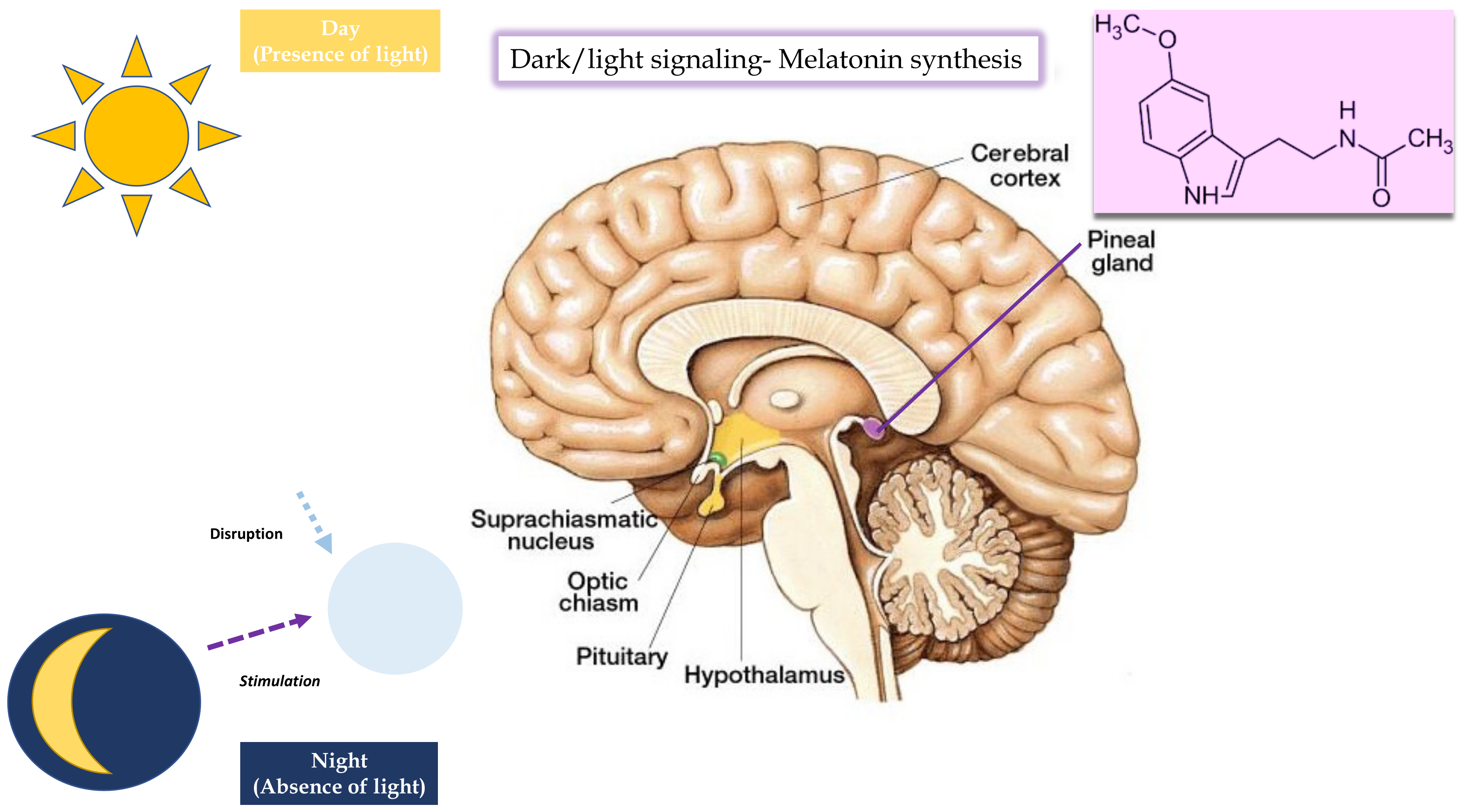
:1. Introduction
AGM Pharmacology
2. Clinical Use of Agomelatine in ADHD and ASD
2.1. ADHD
2.2. ASD
3. Potential Effects of AGM in Neurodevelopmental Disorders
3.1. Melatoninergic Action
3.2. Anti-Inflammatory and Oxidative Stress Action
3.3. Neurotrophic, Anti-Glutamatergic, and Anxiolytic Actions
3.4. 5-HT2C Antagonist Action
4. Potential Therapeutic Role of AGM against SARS-CoV-2
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- de Bodinat, C.; Guardiola-Lemaitre, B.; Mocaër, E.; Renard, P.; Muñoz, C.; Millan, M.J. AGM, the First Melatonergic Antidepressant: Discovery, Characterization and Development. Nat. Rev. Drug Discov. 2010, 9, 628–642. [Google Scholar] [CrossRef] [PubMed]
- Buoli, M.; Grassi, S.; Serati, M.; Altamura, A.C. AGM for the Treatment of Generalized Anxiety Disorder. Expert Opin. Pharmacother. 2017, 18, 1373–1379. [Google Scholar] [CrossRef] [PubMed]
- Pae, C.-U. AGM: A New Option for Treatment of Depression? Expert Opin. Pharmacother. 2014, 15, 443–447. [Google Scholar] [CrossRef] [PubMed]
- De Berardis, D.; Conti, C.M.; Marini, S.; Ferri, F.; Iasevoli, F.; Valchera, A.; Fornaro, M.; Cavuto, M.; Srinivasan, V.; Perna, G.; et al. Is There a Role for AGM in the Treatment of Anxiety Disorders? A Review of Published Data. Int. J. Immunopathol. Pharmacol. 2013, 26, 299–304. [Google Scholar] [CrossRef] [PubMed]
- De Berardis, D.; Fornaro, M.; Serroni, N.; Campanella, D.; Rapini, G.; Olivieri, L.; Srinivasan, V.; Iasevoli, F.; Tomasetti, C.; De Bartolomeis, A.; et al. AGM beyond Borders: Current Evidences of Its Efficacy in Disorders Other than Major Depression. Int. J. Mol. Sci. 2015, 16, 1111–1130. [Google Scholar] [CrossRef]
- Fornaro, M.; McCarthy, M.J.; De Berardis, D.; De Pasquale, C.; Tabaton, M.; Martino, M.; Colicchio, S.; Cattaneo, C.I.; D’Angelo, E.; Fornaro, P. Adjunctive AGM Therapy in the Treatment of Acute Bipolar II Depression: A Preliminary Open Label Study. Neuropsychiatr. Dis. Treat. 2013, 9, 243–251. [Google Scholar] [CrossRef]
- Gahr, M. AGM in the Treatment of Major Depressive Disorder: An Assessment of Benefits and Risks. Curr. Neuropharmacol. 2014, 12, 287–398. [Google Scholar] [CrossRef]
- Millan, M.J.; Gobert, A.; Lejeune, F.; Dekeyne, A.; Newman-Tancredi, A.; Pasteau, V.; Rivet, J.-M.; Cussac, D. The Novel Melatonin Agonist AGM (S20098) Is an Antagonist at 5-Hydroxytryptamine2C Receptors, Blockade of Which Enhances the Activity of Frontocortical Dopaminergic and Adrenergic Pathways. J. Pharmacol. Exp. Ther. 2003, 306, 954–964. [Google Scholar] [CrossRef]
- Sansone, R.A.; Sansone, L.A. AGM: A Novel Antidepressant. Innov. Clin. Neurosci. 2011, 8, 10–14. [Google Scholar]
- Cipriani, A.; Furukawa, T.A.; Salanti, G.; Chaimani, A.; Atkinson, L.Z.; Ogawa, Y.; Leucht, S.; Ruhe, H.G.; Turner, E.H.; Higgins, J.P.T.; et al. Comparative Efficacy and Acceptability of 21 Antidepressant Drugs for the Acute Treatment of Adults with Major Depressive Disorder: A Systematic Review and Network Meta-Analysis. Lancet 2018, 391, 1357–1366. [Google Scholar] [CrossRef]
- Potměšil, P. What Combinations of AGM with Other Antidepressants Could Be Successful during the Treatment of Major Depressive Disorder or Anxiety Disorders in Clinical Practice? Ther. Adv. Psychopharmacol. 2019, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Coles, M.E.; Goodman, M.H. A Systematic Review of Case Studies Testing a Melatonergic Agonist/ 5HT2c Antagonist for Individuals with Obsessive Compulsive Disorder. J. Anxiety Disord. 2020, 69, 102173. [Google Scholar] [CrossRef] [PubMed]
- Stein, D.J.; Ahokas, A.; Jarema, M.; Avedisova, A.S.; Vavrusova, L.; Chaban, O.; Gruget, C.; Olivier, V.; Picarel-Blanchot, F.; de Bodinat, C. Efficacy and Safety of AGM (10 or 25 Mg/Day) in Non-Depressed out-Patients with Generalized Anxiety Disorder: A 12-Week, Double- Blind, Placebo-Controlled Study. Eur. Neuropsychopharmacol. J. Eur. Coll. Neuropsychopharmacol. 2017, 27, 526–537. [Google Scholar] [CrossRef] [PubMed]
- Stein, D.J.; Khoo, J.-P.; Picarel-Blanchot, F.; Olivier, V.; Van Ameringen, M. Efficacy of AGM 25–50 Mg for the Treatment of Anxious Symptoms and Functional Impairment in Generalized Anxiety Disorder: A Meta-Analysis of Three Placebo-Controlled Studies. Adv. Ther. 2021, 38, 1567–1583. [Google Scholar] [CrossRef] [PubMed]
- Kong, W.; Deng, H.; Wan, J.; Zhou, Y.; Zhou, Y.; Song, B.; Wang, X. Comparative Remission Rates and Tolerability of Drugs for Generalised Anxiety Disorder: A Systematic Review and Network Meta-Analysis of Double-Blind Randomized Controlled Trials. Front. Pharmacol. 2020, 11, 580858. [Google Scholar] [CrossRef]
- Freiesleben, S.D.; Furczyk, K. A Systematic Review of AGM-Induced Liver Injury. J. Mol. Psychiatry 2015, 3, 4. [Google Scholar] [CrossRef]
- Perlemuter, G.; Cacoub, P.; Valla, D.; Guyader, D.; Saba, B.; Batailler, C.; Moore, K. Characterisation of AGM-Induced Increase in Liver Enzymes: Frequency and Risk Factors Determined from a Pooled Analysis of 7605 Treated Patients. CNS Drugs 2016, 30, 877–888. [Google Scholar] [CrossRef]
- Procedure No. EMEA/H/C/000916. Ema/97539/2009. Available online: https://www.ema.europa.eu/en/documents/assessment-report/thymanax-epar-public-assessment-report_en.pdf (accessed on 5 March 2023).
- Goodwin, G.M.; Emsley, R.; Rembry, S.; Rouillon, F.; AGM Study Group. AGM Prevents Relapse in Patients with Major Depressive Disorder without Evidence of a Discontinuation Syndrome: A 24-Week Randomized, Double-Blind, Placebo-Controlled Trial. J. Clin. Psychiatry 2009, 70, 1128–1137. [Google Scholar] [CrossRef]
- Arango, C.; Buitelaar, J.K.; Fegert, J.M.; Olivier, V.; Pénélaud, P.-F.; Marx, U.; Chimits, D.; Falissard, B.; Barylnik, J.; Birdeanu, L.; et al. Safety and Efficacy of AGM in Children and Adolescents with Major Depressive Disorder Receiving Psychosocial Counselling: A Double-Blind, Randomised, Controlled, Phase 3 Trial in Nine Countries. Lancet Psychiatry 2022, 9, 113–124. [Google Scholar] [CrossRef]
- Niederhofer, H. AGM Treatment with Adolescents with ADHD. J. Atten. Disord. 2012, 16, 530–532. [Google Scholar] [CrossRef]
- Salardini, E.; Zeinoddini, A.; Kohi, A.; Mohammadi, M.-R.; Mohammadinejad, P.; Khiabany, M.; Shahriari, M.; Akhondzadeh, S. AGM as a Treatment for Attention- Deficit/Hyperactivity Disorder in Children and Adolescents: A Double-Blind, Randomized Clinical Trial. J. Child Adolesc. Psychopharmacol. 2016, 26, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Haya Al Mutairi, A.N. AGM Augmenting Partial Stimulant Response in ADHD and Mitigating Stimulant-Induced Insomnia and Anxiety. J. Child Adolesc. Behav. 2015, 3, 1000208. [Google Scholar] [CrossRef]
- Naguy, A.; Alamiri, B. Successful AGM Monotherapy for an Adolescent with Attention Deficit Hyperactivity Disorder and Comorbid Migraine. CNS Spectr. 2020, 27, 134–135. [Google Scholar] [CrossRef] [PubMed]
- Purper-Ouakil, D.; Ramoz, N.; Lepagnol-Bestel, A.-M.; Gorwood, P.; Simonneau, M. Neurobiology of Attention Deficit/Hyperactivity Disorder. Pediatr. Res. 2011, 69, 69R–76R. [Google Scholar] [CrossRef] [PubMed]
- Curatolo, P.; Paloscia, C.; D’Agati, E.; Moavero, R.; Pasini, A. The Neurobiology of Attention Deficit/Hyperactivity Disorder. Eur. J. Paediatr. Neurol. EJPN Off. J. Eur. Paediatr. Neurol. Soc. 2009, 13, 299–304. [Google Scholar] [CrossRef]
- Niederhofer, H. Efficacy of Duloxetine and AGM Does Not Exceed That of Other Antidepressants in Patients with Autistic Disorder: Preliminary Results in 3 Patients. Prim. Care Companion CNS Disord. 2011, 13, PCC.10l01038. [Google Scholar] [CrossRef]
- Naguy, A.; Tajali, A.A. AGM Addressing Behavioural Facets in Autism. J. Psychiatry 2015, 18. [Google Scholar] [CrossRef]
- Ballester, P.; Martínez, M.J.; Inda, M.-M.; Javaloyes, A.; Richdale, A.L.; Muriel, J.; Belda, C.; Toral, N.; Morales, D.; Fernández, E.; et al. Evaluation of AGM for the Treatment of Sleep Problems in Adults with Autism Spectrum Disorder and Co-Morbid Intellectual Disability. J. Psychopharmacol. 2019, 33, 1395–1406. [Google Scholar] [CrossRef]
- Ballester, P.; Martínez, M.J.; Javaloyes, A.; Hernández, L.; Peiró, A.M. Agomelatine Effectiveness in Sleep Disturbances in Autism Spectrum Disorder. Clin. Ther. 2015, 37, e132–e133. [Google Scholar] [CrossRef]
- Kumar, H.; Sharma, B.M.; Sharma, B. Benefits of AGM in Behavioral, Neurochemical and Blood Brain Barrier Alterations in Prenatal Valproic Acid Induced Autism Spectrum Disorder. Neurochem. Int. 2015, 91, 34–45. [Google Scholar] [CrossRef]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar] [CrossRef]
- White, S.W.; Roberson-Nay, R. Anxiety, Social Deficits, and Loneliness in Youth with Autism Spectrum Disorders. J. Autism Dev. Disord. 2009, 39, 1006–1013. [Google Scholar] [CrossRef] [PubMed]
- Gagnon, K.; Godbout, R. Melatonin and Comorbidities in Children with Autism Spectrum Disorder. Curr. Dev. Disord. Rep. 2018, 5, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Hollocks, M.J.; Lerh, J.W.; Magiati, I.; Meiser-Stedman, R.; Brugha, T.S. Anxiety and Depression in Adults with Autism Spectrum Disorder: A Systematic Review and Meta-Analysis. Psychol. Med. 2019, 49, 559–572. [Google Scholar] [CrossRef] [PubMed]
- Gentile, I.; Zappulo, E.; Militerni, R.; Pascotto, A.; Borgia, G.; Bravaccio, C. Etiopathogenesis of Autism Spectrum Disorders: Fitting the Pieces of the Puzzle Together. Med. Hypotheses 2013, 81, 26–35. [Google Scholar] [CrossRef]
- Giana, G.; Romano, E.; Porfirio, M.C.; D’Ambrosio, R.; Giovinazzo, S.; Troianiello, M.; Barlocci, E.; Travaglini, D.; Granstrem, O.; Pascale, E.; et al. Detection of Auto-Antibodies to DAT in the Serum: Interactions with DAT Genotype and Psycho-Stimulant Therapy for ADHD. J. Neuroimmunol. 2015, 278, 212–222. [Google Scholar] [CrossRef]
- Santos, P.; Herrmann, A.P.; Elisabetsky, E.; Piato, A. Anxiolytic Properties of Compounds That Counteract Oxidative Stress, Neuroinflammation, and Glutamatergic Dysfunction: A Review. Rev. Bras. Psiquiatr. Sao Paulo Braz. 1999 2019, 41, 168–178. [Google Scholar] [CrossRef]
- Savino, R.; Carotenuto, M.; Polito, A.N.; Di Noia, S.; Albenzio, M.; Scarinci, A.; Ambrosi, A.; Sessa, F.; Tartaglia, N.; Messina, G. Analyzing the Potential Biological Determinants of Autism Spectrum Disorder: From Neuroinflammation to the Kynurenine Pathway. Brain Sci. 2020, 10, 631. [Google Scholar] [CrossRef]
- Frye, R.E.; Delatorre, R.; Taylor, H.; Slattery, J.; Melnyk, S.; Chowdhury, N.; James, S.J. Redox Metabolism Abnormalities in Autistic Children Associated with Mitochondrial Disease. Transl. Psychiatry 2013, 3, e273. [Google Scholar] [CrossRef]
- Rossignol, D.A.; Frye, R.E. Melatonin in Autism Spectrum Disorders: A Systematic Review and Meta-Analysis: Review. Dev. Med. Child Neurol. 2011, 53, 783–792. [Google Scholar] [CrossRef]
- Ramírez-Rodríguez, G.; Klempin, F.; Babu, H.; Benítez-King, G.; Kempermann, G. Melatonin Modulates Cell Survival of New Neurons in the Hippocampus of Adult Mice. Neuropsychopharmacology 2009, 34, 2180–2191. [Google Scholar] [CrossRef]
- Lacivita, E.; Perrone, R.; Margari, L.; Leopoldo, M. Targets for Drug Therapy for Autism Spectrum Disorder: Challenges and Future Directions. J. Med. Chem. 2017, 60, 9114–9141. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Couture, J. A Review of the Pathophysiology, Etiology, and Treatment of Attention-Deficit Hyperactivity Disorder (ADHD). Ann. Pharmacother. 2014, 48, 209–225. [Google Scholar] [CrossRef] [PubMed]
- Lyon, G.J.; Coffey, B.J. Attention-Deficit Hyperactivity Disorder: A Handbook for Diagnosis and Treatment, 3rd Ed. J. Clin. Psychiatry 2008, 69, 1023. [Google Scholar] [CrossRef]
- Cortese, S.; Brown, T.E.; Corkum, P.; Gruber, R.; O’Brien, L.M.; Stein, M.; Weiss, M.; Owens, J. Assessment and Management of Sleep Problems in Youths with Attention- Deficit/Hyperactivity Disorder. J. Am. Acad. Child Adolesc. Psychiatry 2013, 52, 784–796. [Google Scholar] [CrossRef] [PubMed]
- on behalf of Lombardy ADHD Group; Reale, L.; Bartoli, B.; Cartabia, M.; Zanetti, M.; Costantino, M.A.; Canevini, M.P.; Termine, C.; Bonati, M. Comorbidity Prevalence and Treatment Outcome in Children and Adolescents with ADHD. Eur. Child Adolesc. Psychiatry 2017, 26, 1443–1457. [Google Scholar] [CrossRef]
- Andrade, C. Stahl′s Essential Psychopharmacology: Neuroscientific Basis and Practical Applications. Mens Sana Monogr. 2010, 8, 146. [Google Scholar] [CrossRef]
- Maenner, M.J.; Shaw, K.A.; Baio, J.; Washington, A.; Patrick, M.; DiRienzo, M.; Christensen, D.L.; Wiggins, L.D.; Pettygrove, S. Prevalence of Autism Spectrum Disorder Among Children Aged 8 Years—Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2016. MMWR Surveill. Summ. 2020, 69, 1–12. [Google Scholar] [CrossRef]
- DeFilippis, M.; Wagner, K.D. Management of Treatment-Resistant Depression in Children and Adolescents. Paediatr. Drugs 2014, 16, 353–361. [Google Scholar] [CrossRef]
- Goodyer, I.M. Editorial Perspective: Antidepressants and the Depressed Adolescent. Child Adolesc. Ment. Health 2018, 23, 137–140. [Google Scholar] [CrossRef]
- Merikangas, K.R.; He, J.-P.; Burstein, M.; Swanson, S.A.; Avenevoli, S.; Cui, L.; Benjet, C.; Georgiades, K.; Swendsen, J. Lifetime Prevalence of Mental Disorders in U.S. Adolescents: Results from the National Comorbidity Survey Replication--Adolescent Supplement (NCS-A). J. Am. Acad. Child Adolesc. Psychiatry 2010, 49, 980–989. [Google Scholar] [CrossRef]
- Amray, A.N.; Munir, K.; Jahan, N.; Motiwala, F.B.; Naveed, S. Psychopharmacology of Pediatric Anxiety Disorders: A Narrative Review. Cureus 2019, 11, e5487. [Google Scholar] [CrossRef] [PubMed]
- Kessler, R.C.; Petukhova, M.; Sampson, N.A.; Zaslavsky, A.M.; Wittchen, H.-U. Twelve-Month and Lifetime Prevalence and Lifetime Morbid Risk of Anxiety and Mood Disorders in the United States. Int. J. Methods Psychiatr. Res. 2012, 21, 169–184. [Google Scholar] [CrossRef] [PubMed]
- Blackburn, T.P. Depressive Disorders: Treatment Failures and Poor Prognosis over the Last 50 Years. Pharmacol. Res. Perspect. 2019, 7, e00472. [Google Scholar] [CrossRef] [PubMed]
- Baglioni, C.; Nanovska, S.; Regen, W.; Spiegelhalder, K.; Feige, B.; Nissen, C.; Reynolds, C.F.; Riemann, D. Sleep and Mental Disorders: A Meta-Analysis of Polysomnographic Research. Psychol. Bull. 2016, 142, 969–990. [Google Scholar] [CrossRef]
- Landmann, N.; Kuhn, M.; Piosczyk, H.; Feige, B.; Baglioni, C.; Spiegelhalder, K.; Frase, L.; Riemann, D.; Sterr, A.; Nissen, C. The Reorganisation of Memory during Sleep. Sleep Med. Rev. 2014, 18, 531–541. [Google Scholar] [CrossRef]
- Landmann, N.; Kuhn, M.; Maier, J.-G.; Spiegelhalder, K.; Baglioni, C.; Frase, L.; Riemann, D.; Sterr, A.; Nissen, C. REM Sleep and Memory Reorganization: Potential Relevance for Psychiatry and Psychotherapy. Neurobiol. Learn. Mem. 2015, 122, 28–40. [Google Scholar] [CrossRef]
- Gordon, A.M.; Chen, S. The Role of Sleep in Interpersonal Conflict: Do Sleepless Nights Mean Worse Fights? Soc. Psychol. Personal. Sci. 2014, 5, 168–175. [Google Scholar] [CrossRef]
- Guadagni, V.; Burles, F.; Ferrara, M.; Iaria, G. The Effects of Sleep Deprivation on Emotional Empathy. J. Sleep Res. 2014, 23, 657–663. [Google Scholar] [CrossRef]
- Harvey, A.G.; Murray, G.; Chandler, R.A.; Soehner, A. Sleep Disturbance as Transdiagnostic: Consideration of Neurobiological Mechanisms. Clin. Psychol. Rev. 2011, 31, 225–235. [Google Scholar] [CrossRef]
- Harvey, A.G. A Transdiagnostic Approach to Treating Sleep Disturbance in Psychiatric Disorders. Cogn. Behav. Ther. 2009, 38 (Suppl. 1), 35–42. [Google Scholar] [CrossRef]
- Marotta, R.; Risoleo, M.C.; Messina, G.; Parisi, L.; Carotenuto, M.; Vetri, L.; Roccella, M. The Neurochemistry of Autism. Brain Sci. 2020, 10, 163. [Google Scholar] [CrossRef] [PubMed]
- Alfano, C.A.; Beidel, D.C.; Turner, S.M.; Lewin, D.S. Preliminary Evidence for Sleep Complaints among Children Referred for Anxiety. Sleep Med. 2006, 7, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Ritvo, E.R.; Ritvo, R.; Yuwiler, A.; Brothers, A.; Freeman, B.J.; Plotkin, S. Elevated Daytime Melatonin Concentrations in Autism: A Pilot Study. Eur. Child Adolesc. Psychiatry 1993, 2, 75–78. [Google Scholar] [CrossRef] [PubMed]
- Esposito, S.; Laino, D.; D’Alonzo, R.; Mencarelli, A.; Di Genova, L.; Fattorusso, A.; Argentiero, A.; Mencaroni, E. Pediatric Sleep Disturbances and Treatment with Melatonin. J. Transl. Med. 2019, 17, 77. [Google Scholar] [CrossRef] [PubMed]
- Lalanne, S.; Fougerou-Leurent, C.; Anderson, G.M.; Schroder, C.M.; Nir, T.; Chokron, S.; Delorme, R.; Claustrat, B.; Bellissant, E.; Kermarrec, S.; et al. Melatonin: From Pharmacokinetics to Clinical Use in Autism Spectrum Disorder. Int. J. Mol. Sci. 2021, 22, 1490. [Google Scholar] [CrossRef] [PubMed]
- Grivas, T.B.; Savvidou, O.D. Melatonin the “Light of Night” in Human Biology and Adolescent Idiopathic Scoliosis. Scoliosis 2007, 2, 6. [Google Scholar] [CrossRef]
- Sadeh, A. Sleep and Melatonin in Infants: A Preliminary Study. Sleep 1997, 20, 185–191. [Google Scholar]
- Rossignol, D.A.; Frye, R.E. Mitochondrial Dysfunction in Autism Spectrum Disorders: A Systematic Review and Meta-Analysis. Mol. Psychiatry 2012, 17, 290–314. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G.; Maes, M. Interactions of Tryptophan and Its Catabolites with Melatonin and the Alpha 7 Nicotinic Receptor in Central Nervous System and Psychiatric Disorders: Role of the Aryl Hydrocarbon Receptor and Direct Mitochondria Regulation. Int. J. Tryptophan Res. IJTR 2017, 10, 1178646917691738. [Google Scholar] [CrossRef] [PubMed]
- Schwarcz, R.; Stone, T.W. The Kynurenine Pathway and the Brain: Challenges, Controversies and Promises. Neuropharmacology 2017, 112, 237–247. [Google Scholar] [CrossRef]
- Fujigaki, H.; Yamamoto, Y.; Saito, K. L-Tryptophan-Kynurenine Pathway Enzymes Are Therapeutic Target for Neuropsychiatric Diseases: Focus on Cell Type Differences. Neuropharmacology 2017, 112, 264–274. [Google Scholar] [CrossRef] [PubMed]
- Wigner, P.; Synowiec, E.; Jóźwiak, P.; Czarny, P.; Bijak, M.; Barszczewska, G.; Białek, K.; Szemraj, J.; Gruca, P.; Papp, M.; et al. The Changes of Expression and Methylation of Genes Involved in Oxidative Stress in Course of Chronic Mild Stress and Antidepressant Therapy with AGM. Genes 2020, 11, 644. [Google Scholar] [CrossRef] [PubMed]
- Wigner, P.; Czarny, P.; Synowiec, E.; Bijak, M.; Talarowska, M.; Galecki, P.; Szemraj, J.; Sliwinski, T. Variation of Genes Encoding KAT1, AADAT and IDO1 as a Potential Risk of Depression Development. Eur. Psychiatry J. Assoc. Eur. Psychiatr. 2018, 52, 95–103. [Google Scholar] [CrossRef]
- Anderson, G.; Maes, M. Redox Regulation and the Autistic Spectrum: Role of Tryptophan Catabolites, Immuno-Inflammation, Autoimmunity and the Amygdala. Curr. Neuropharmacol. 2014, 12, 148–167. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.F. Quest for Biomarkers of Treatment-Resistant Depression: Shifting the Paradigm toward Risk. Front. Psychiatry 2013, 4, 57. [Google Scholar] [CrossRef]
- Zhang, X.; Gainetdinov, R.R.; Beaulieu, J.-M.; Sotnikova, T.D.; Burch, L.H.; Williams, R.B.; Schwartz, D.A.; Krishnan, K.R.R.; Caron, M.G. Loss-of-Function Mutation in Tryptophan Hydroxylase-2 Identified in Unipolar Major Depression. Neuron 2005, 45, 11–16. [Google Scholar] [CrossRef]
- Chumboatong, W.; Khamchai, S.; Tocharus, C.; Govitrapong, P.; Tocharus, J. AGM Exerts an Anti-Inflammatory Effect by Inhibiting Microglial Activation Through TLR4/NLRP3 Pathway in PMCAO Rats. Neurotox. Res. 2022, 40, 259–266. [Google Scholar] [CrossRef]
- Cankara, F.N.; Günaydın, C.; Çelik, Z.B.; Şahin, Y.; Pekgöz, Ş.; Erzurumlu, Y.; Gülle, K. AGM Confers Neuroprotection against Cisplatin-Induced Hippocampal Neurotoxicity. Metab. Brain Dis. 2021, 36, 339–349. [Google Scholar] [CrossRef]
- Manda, K.; Reiter, R.J. Melatonin Maintains Adult Hippocampal Neurogenesis and Cognitive Functions after Irradiation. Prog. Neurobiol. 2010, 90, 60–68. [Google Scholar] [CrossRef]
- Markham, A.; Bains, R.; Franklin, P.; Spedding, M. Changes in Mitochondrial Function Are Pivotal in Neurodegenerative and Psychiatric Disorders: How Important Is BDNF? Br. J. Pharmacol. 2014, 171, 2206–2229. [Google Scholar] [CrossRef]
- Sasaki, R.; Kojima, S.; Onishi, H. Do Brain-Derived Neurotrophic Factor Genetic Polymorphisms Modulate the Efficacy of Motor Cortex Plasticity Induced by Non-Invasive Brain Stimulation? A Systematic Review. Front. Hum. Neurosci. 2021, 15, 742373. [Google Scholar] [CrossRef] [PubMed]
- Abellaneda-Pérez, K.; Martin-Trias, P.; Cassé-Perrot, C.; Vaqué-Alcázar, L.; Lanteaume, L.; Solana, E.; Babiloni, C.; Lizio, R.; Junqué, C.; Bargalló, N.; et al. BDNF Val66Met Gene Polymorphism Modulates Brain Activity Following RTMS-Induced Memory Impairment. Sci. Rep. 2022, 12, 176. [Google Scholar] [CrossRef] [PubMed]
- Cheeran, B.; Talelli, P.; Mori, F.; Koch, G.; Suppa, A.; Edwards, M.; Houlden, H.; Bhatia, K.; Greenwood, R.; Rothwell, J.C. A Common Polymorphism in the Brain-Derived Neurotrophic Factor Gene (BDNF) Modulates Human Cortical Plasticity and the Response to RTMS. J. Physiol. 2008, 586, 5717–5725. [Google Scholar] [CrossRef] [PubMed]
- Su, B.; Ji, Y.-S.; Sun, X.; Liu, X.-H.; Chen, Z.-Y. Brain-Derived Neurotrophic Factor (BDNF)-Induced Mitochondrial Motility Arrest and Presynaptic Docking Contribute to BDNF- Enhanced Synaptic Transmission. J. Biol. Chem. 2014, 289, 1213–1226. [Google Scholar] [CrossRef]
- Penrod, R.D.; Kumar, J.; Smith, L.N.; McCalley, D.; Nentwig, T.B.; Hughes, B.W.; Barry, G.M.; Glover, K.; Taniguchi, M.; Cowan, C.W. Activity-regulated cytoskeleton-associated protein (Arc/Arg3.1) regulates anxiety- and novelty-related behaviors. Genes Brain Behavior. 2019, 18, e12561. [Google Scholar] [CrossRef]
- Lim, C.K.; Essa, M.M.; de Paula Martins, R.; Lovejoy, D.B.; Bilgin, A.A.; Waly, M.I.; Al-Farsi, Y.M.; Al-Sharbati, M.; Al-Shaffae, M.A.; Guillemin, G.J. Altered Kynurenine Pathway Metabolism in Autism: Implication for Immune-Induced Glutamatergic Activity: Altered Kynurenine Pathway Metabolism in ASD. Autism Res. 2016, 9, 621–631. [Google Scholar] [CrossRef]
- Musazzi, L.; Racagni, G.; Popoli, M. Stress, Glucocorticoids and Glutamate Release: Effects of Antidepressant Drugs. Neurochem. Int. 2011, 59, 138–149. [Google Scholar] [CrossRef]
- Casanova, M.F.; Shaban, M.; Ghazal, M.; El-Baz, A.S.; Casanova, E.L.; Sokhadze, E.M. Ringing Decay of Gamma Oscillations and Transcranial Magnetic Stimulation Therapy in Autism Spectrum Disorder. Appl. Psychophysiol. Biofeedback 2021, 46, 161–173. [Google Scholar] [CrossRef]
- Sonmez, A.I.; Almorsy, A.; Ramsey, L.B.; Strawn, J.R.; Croarkin, P.E. Novel Pharmacological Treatments for Generalized Anxiety Disorder: Pediatric Considerations. Depress. Anxiety 2020, 37, 747–759. [Google Scholar] [CrossRef]
- Harvey, B.H.; Regenass, W.; Dreyer, W.; Möller, M. Social Isolation Rearing-Induced Anxiety and Response to AGM in Male and Female Rats: Role of Corticosterone, Oxytocin, and Vasopressin. J. Psychopharmacol. Oxf. Engl. 2019, 33, 640–646. [Google Scholar] [CrossRef]
- McClung, C.A. How Might Circadian Rhythms Control Mood? Let Me Count the Ways. Biol. Psychiatry 2013, 74, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Floden, D.; Vallesi, A.; Stuss, D.T. Task Context and Frontal Lobe Activation in the Stroop Task. J. Cogn. Neurosci. 2011, 23, 867–879. [Google Scholar] [CrossRef] [PubMed]
- Koresh, O.; Kozlovsky, N.; Kaplan, Z.; Zohar, J.; Matar, M.A.; Cohen, H. The Long-Term Abnormalities in Circadian Expression of Period 1 and Period 2 Genes in Response to Stress Is Normalized by AGM Administered Immediately after Exposure. Eur. Neuropsychopharmacol. J. Eur. Coll. Neuropsychopharmacol. 2012, 22, 205–221. [Google Scholar] [CrossRef] [PubMed]
- Coull, J.T.; Frith, C.D.; Büchel, C.; Nobre, A.C. Orienting Attention in Time: Behavioural and Neuroanatomical Distinction between Exogenous and Endogenous Shifts. Neuropsychologia 2000, 38, 808–819. [Google Scholar] [CrossRef]
- Davidson, R.J.; Pizzagalli, D.; Nitschke, J.B.; Putnam, K. Depression: Perspectives from Affective Neuroscience. Annu. Rev. Psychol. 2002, 53, 545–574. [Google Scholar] [CrossRef]
- Casey, B.J.; Castellanos, F.X.; Giedd, J.N.; Marsh, W.L.; Hamburger, S.D.; Schubert, A.B.; Vauss, Y.C.; Vaituzis, A.C.; Dickstein, D.P.; Sarfatti, S.E.; et al. Implication of Right Frontostriatal Circuitry in Response Inhibition and Attention-Deficit/Hyperactivity Disorder. J. Am. Acad. Child Adolesc. Psychiatry 1997, 36, 374–383. [Google Scholar] [CrossRef]
- Castellanos, F.X.; Giedd, J.N.; Marsh, W.L.; Hamburger, S.D.; Vaituzis, A.C.; Dickstein, D.P.; Sarfatti, S.E.; Vauss, Y.C.; Snell, J.W.; Lange, N.; et al. Quantitative Brain Magnetic Resonance Imaging in Attention- Deficit Hyperactivity Disorder. Arch. Gen. Psychiatry 1996, 53, 607–616. [Google Scholar] [CrossRef]
- Filipek, P.A.; Semrud-Clikeman, M.; Steingard, R.J.; Renshaw, P.F.; Kennedy, D.N.; Biederman, J. Volumetric MRI Analysis Comparing Subjects Having Attention-Deficit Hyperactivity Disorder with Normal Controls. Neurology 1997, 48, 589–601. [Google Scholar] [CrossRef]
- Mataró, M.; Garcia-Sánchez, C.; Junqué, C.; Estévez-González, A.; Pujol, J. Magnetic Resonance Imaging Measurement of the Caudate Nucleus in Adolescents with Attention-Deficit Hyperactivity Disorder and Its Relationship with Neuropsychological and Behavioral Measures. Arch. Neurol. 1997, 54, 963–968. [Google Scholar] [CrossRef]
- Silberstein, R.B.; Farrow, M.; Levy, F.; Pipingas, A.; Hay, D.A.; Jarman, F.C. Functional Brain Electrical Activity Mapping in Boys with Attention-Deficit/Hyperactivity Disorder. Arch. Gen. Psychiatry 1998, 55, 1105–1112. [Google Scholar] [CrossRef]
- Cheng, W.; Rolls, E.T.; Gu, H.; Zhang, J.; Feng, J. Autism: Reduced Connectivity between Cortical Areas Involved in Face Expression, Theory of Mind, and the Sense of Self. Brain J. Neurol. 2015, 138 Pt 5, 1382–1393. [Google Scholar] [CrossRef]
- Kosaka, H.; Omori, M.; Munesue, T.; Ishitobi, M.; Matsumura, Y.; Takahashi, T.; Narita, K.; Murata, T.; Saito, D.N.; Uchiyama, H.; et al. Smaller Insula and Inferior Frontal Volumes in Young Adults with Pervasive Developmental Disorders. NeuroImage 2010, 50, 1357–1363. [Google Scholar] [CrossRef] [PubMed]
- Casanova, M.F.; Sokhadze, E.; Opris, I.; Wang, Y.; Li, X. Autism Spectrum Disorders: Linking Neuropathological Findings to Treatment with Transcranial Magnetic Stimulation. Acta Paediatr. Oslo Nor. 1992 2015, 104, 346–355. [Google Scholar] [CrossRef] [PubMed]
- Bauman, M.L.; Kemper, T.L. Neuroanatomic Observations of the Brain in Autism: A Review and Future Directions. Int. J. Dev. Neurosci. Off. J. Int. Soc. Dev. Neurosci. 2005, 23, 183–187. [Google Scholar] [CrossRef]
- Niederhofer, H. Treating ADHD with AGM. J. Atten. Disord. 2012, 16, 346–348. [Google Scholar] [CrossRef]
- Sara, S.J. The Locus Coeruleus and Noradrenergic Modulation of Cognition. Nat. Rev. Neurosci. 2009, 10, 211–223. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Hou, Y.; Shen, J.; Huang, Y.; Martin, W.; Cheng, F. Network-Based Drug Repurposing for Novel Coronavirus 2019-NCoV/SARS-CoV-2. Cell Discov. 2020, 6, 14. [Google Scholar] [CrossRef] [PubMed]
- Schneider, S.A.; Hennig, A.; Martino, D. Relationship between COVID 19 and Movement Disorders: A Narrative Review. Eur. J. Neurol. 2021, 29, 1243–1253. [Google Scholar] [CrossRef]
- Anderson, G.; Reiter, R.J. Melatonin: Roles in Influenza, COVID-19, and Other Viral Infections. Rev. Med. Virol. 2020, 30, e2109. [Google Scholar] [CrossRef]
- Yadalam, P.K.; Balaji, T.M.; Varadarajan, S.; Alzahrani, K.J.; Al-Ghamdi, M.S.; Baeshen, H.A.; Alfarhan, M.F.A.; Khurshid, Z.; Bhandi, S.; Jagannathan, R.; et al. Assessing the Therapeutic Potential of AGM, Ramelteon, and Melatonin against SARS-CoV-2. Saudi J. Biol. Sci. 2022, 29, 3140–3150. [Google Scholar] [CrossRef]
- Feitosa, E.L.; Júnior, F.T.D.S.S.; Nery Neto, J.A.D.O.; Matos, L.F.L.; Moura, M.H.D.S.; Rosales, T.O.; De Freitas, G.B.L. COVID-19: Rational Discovery of the Therapeutic Potential of Melatonin as a SARS-CoV-2 Main Protease Inhibitor. Int. J. Med. Sci. 2020, 17, 2133–2146. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Moore, M.J.; Vasilieva, N.; Sui, J.; Wong, S.K.; Berne, M.A.; Somasundaran, M.; Sullivan, J.L.; Luzuriaga, K.; Greenough, T.C.; et al. Angiotensin-Converting Enzyme 2 Is a Functional Receptor for the SARS Coronavirus. Nature 2003, 426, 450–454. [Google Scholar] [CrossRef] [PubMed]
- Lan, J.; Ge, J.; Yu, J.; Shan, S.; Zhou, H.; Fan, S.; Zhang, Q.; Shi, X.; Wang, Q.; Zhang, L.; et al. Structure of the SARS-CoV-2 Spike Receptor-Binding Domain Bound to the ACE2 Receptor. Nature 2020, 581, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Cecon, E.; Izabelle, C.; Poder, S.L.; Real, F.; Zhu, A.; Tu, L.; Ghigna, M.R.; Klonjkowski, B.; Bomsel, M.; Jockers, R.; et al. Therapeutic Potential of Melatonin and Melatonergic Drugs on K18 HACE2 Mice Infected with SARS-CoV-2. J. Pineal Res. 2022, 72, e12772. [Google Scholar] [CrossRef] [PubMed]
- Brusco, L.I.; Cruz, P.; Cangas, A.V.; Rojas, C.G.; Vigo, D.E.; Cardinali, D.P. Efficacy of Melatonin in Non-Intensive Care Unit Patients with COVID-19 Pneumonia and Sleep Dysregulation. Melatonin Res. 2021, 4, 173–188. [Google Scholar] [CrossRef]
- Savino, R.; Polito, A.N.; Arcidiacono, G.; Poliseno, M.; Lo Caputo, S. Neuropsychiatric Disorders in Pediatric Long COVID-19: A Case Series. Brain Sci. 2022, 12, 514. [Google Scholar] [CrossRef]
- de Sousa Moreira, J.L.; Barbosa, S.M.B.; Vieira, J.G.; Chaves, N.C.B.; Felix, E.B.G.; Feitosa, P.W.G.; da Cruz, I.S.; da Silva, C.G.L.; Neto, M.L.R. The Psychiatric and Neuropsychiatric Repercussions Associated with Severe Infections of COVID-19 and Other Coronaviruses. Prog. Neuropsychopharmacol. Biol. Psychiatry 2021, 106, 110159. [Google Scholar] [CrossRef]

| Authors | Year | Article Type | Age (Year) | Number of Patients | Gender | Diagnosis/Comorbidity | Dose of Agomelatine |
|---|---|---|---|---|---|---|---|
| Niederhofer H., et al. [21] | 2012 | Placebo-controlled study | 17–19 years old | 10 | M:F = 8:2 | Severe ADHD | 25 mg/day |
| Naguy A. and Al-tajali A. [22] | 2015 | Case report | 13 years old | 1 | F | Severe ADHD | 25 mg/day |
| Salardini E., et al. [23] | 2016 | Double-blind randomized controlled trial | 6–15 years old | 54 | Not available | Severe ADHD | 15 mg/day in patients with weight ≥30 kg and 25 mg/day in patients with weight ≥45 kg |
| Naguy A. and Alamiri B. [24] | 2020 | Case report | 15 years old | 1 | F | Severe ADHD/migraine | 25 mg/day |
| Authors | Year | Article Type | Age (Year) | Number of Patients | Gender | Diagnosis/Comorbidity | Dose of Agomelatine |
|---|---|---|---|---|---|---|---|
| Niederhofer H., et al. [27] | 2011 | Case report: 10 week clinical trial | Adults | 2:1 | M | Severe ASD/ID | 25 mg/day |
| Naguy A. and Ali Al Tajali [28] | 2015 | Case report | 10 years old | 1 | M | Severe ASD/behavioral disorder and insomnia | 25 mg/day |
| Ballester P., et al. [29] | 2015 | Randomized, cross-double-blind, multicenter study | 30–32 years old | 25 | M:F = 20:5 | Severe ASD | 25 mg/day |
| Ballester P., et al. [30] | 2019 | Cross-sectional, randomized, triple-blind, placebo-controlled study | 35 ± 12 years old | 23 | M:F = 19:4 | ASD/ID and sleep disturbance | 25 mg/day |
| Kumar H., et al. [31] | 2015 | Study on animal models | / | / | / | Autism VPA-induced | / |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Savino, R.; Polito, A.N.; Marsala, G.; Ventriglio, A.; Di Salvatore, M.; De Stefano, M.I.; Valenzano, A.; Marinaccio, L.; Bellomo, A.; Cibelli, G.; et al. Agomelatine: A Potential Multitarget Compound for Neurodevelopmental Disorders. Brain Sci. 2023, 13, 734. https://doi.org/10.3390/brainsci13050734
Savino R, Polito AN, Marsala G, Ventriglio A, Di Salvatore M, De Stefano MI, Valenzano A, Marinaccio L, Bellomo A, Cibelli G, et al. Agomelatine: A Potential Multitarget Compound for Neurodevelopmental Disorders. Brain Sciences. 2023; 13(5):734. https://doi.org/10.3390/brainsci13050734
Chicago/Turabian StyleSavino, Rosa, Anna Nunzia Polito, Gabriella Marsala, Antonio Ventriglio, Melanie Di Salvatore, Maria Ida De Stefano, Anna Valenzano, Luigi Marinaccio, Antonello Bellomo, Giuseppe Cibelli, and et al. 2023. "Agomelatine: A Potential Multitarget Compound for Neurodevelopmental Disorders" Brain Sciences 13, no. 5: 734. https://doi.org/10.3390/brainsci13050734










