Abstract
Purpose: Previous research has evaluated the effects of acute hypoxia exposure on cognitive function, notably executive function. No studies, to date, have evaluated the effects of acute hypoxia exposure on memory interference, which was the purpose of this experiment. Methods: A within-subjects, counterbalanced experimental design was employed, with condition (hypoxia vs. normoxia) and time (immediate vs. delayed) being the independent variables. Participants (N = 21; Mage = 21.0 years) completed two laboratory visits, involving 30 min of exposure to either hypoxia (FIO2 = 0.12) or normoxia (FIO2 = 0.21). Following this, they completed a memory interference task (AB/AC paradigm), assessing immediate and delayed proactive and retroactive interference. Results: For retroactive interference, we observed a significant main effect for condition, F(1, 20) = 5.48, p = 0.03, ƞ2 = 0.10, condition by time interaction, F(1, 20) = 4.96, p = 0.03, ƞ2 = 0.01, but no main effect for time, F(1, 20) = 1.75, p = 0.20, ƞ2 = 0.004. Conclusion: Our results demonstrate that acute hypoxia exposure was facilitative in reducing memory interference. We discuss these findings in the context of the potential therapeutic effects of acute hypoxia exposure on synaptic plasticity.
1. Introduction
Various populations, such as mountaineers or mountain rescuers, may be at risk of cognitive dysfunction when ascending to higher altitudes. During states of acute hypoxia, turnover of key cognitive-related neurotransmitters (e.g., dopamine and norepinephrine) is reduced as a result of the oxygen requirement during the synthesis, release, and metabolism of these neurotransmitters [1,2]. Such alterations may, in theory, alter cognitive function. Narrative reviews indicate that acute and chronic hypoxia exposure may negatively influence several cognitive parameters, such as attention and executive function [3]. A recent meta-analysis demonstrated that acute hypoxia exposure (<60 mmHg for 10 min to 5 days) impairs both central executive and non-executive (e.g., memory) cognitive tasks [4]. For example, a recent experiment [5] demonstrated that acute (90 min) gradual exposure going from 20.9% O2 to 13.5% O2 (simulated altitude of 4500 m) impaired reaction time, but such an effect was attenuated with an acute (60 min; 50% of VO2peak) bout of exercise. Importantly, though, other related work [6] shows that under hypoxia (~30 min of simulated altitude of 3800 to 4500 m), exercise-induced improvements in cognition (reaction time) are attenuated for individuals exhibiting greater decreases in peripheral oxygen saturation.
We recently evaluated the effects of acute (30 min) moderate hypoxia (partial pressure of inspired oxygen (PIO2): 117 mmHg; fraction of inspired oxygen (FIO2): 0.154; altitude equivalent of 2500 m) on reaction time and central executive cognitive performance (Go/No-Go task) [7]. Participants (N = 20, Mage = 23.9 years) were exposed to a normoxic or hypoxic condition for 30 min, completed a pre-exercise cognitive task (Go/No-Go), then engaged in an acute bout of high-intensity interval exercise (or rest), and then re-completed the cognitive task immediately after exercise. Results demonstrated no moderate-hypoxia-induced impairment effects on reaction time or response accuracy [7]. Following this, we evaluated the effects of acute (10 min) severe hypoxia (PIO2: 87 mmHg; FIO2: 0.12; altitude equivalent of 4000 m) on central executive cognitive performance (Go/No-Go task) [8]. Participants (N = 30, Mage = 22.6 years) were exposed to the normoxic or hypoxic conditions for 10 min, completed the pre-exercise cognitive task, then engaged in an acute bout of moderate-intensity continuous exercise (or rest), and then re-completed the cognitive task during exercise. Our results demonstrated no severe-hypoxia-induced impairment effects on response accuracy [8]. Relatedly, other recent work (N = 15, Mage = 21.9 years) demonstrates that 30 min of severe acute hypoxia (equivalent to 4500 m) does not impair behavioral performance on a central executive cognitive task (i.e., Go/No-Go task), but does impair neural activity during inhibitory processing (i.e., reduced the peak amplitudes of Go- and No-Go-P300) [9].
Collectively, these studies suggest that short-duration (10–30 min) acute hypoxia may not be sufficient to induce behavioral impairments in central executive cognitive tasks. Although it is possible that the degree of hypoxia (i.e., altitude equivalent of 2500 m to 4500 m) was not enough to impair cognitive behavior (however, other work shows cognitive impairment effects at this altitude [5,6]), it is also possible that the cognitive tasks utilized in these studies were not sufficient to observe a hypoxia-induced impairment of cognition. That is, for these studies, the commission and omission error rates were very low. The present experiment addresses this latter issue. We aim to evaluate whether short duration (30 min) hypoxia at an altitude equivalent of 4000 m is sufficient to impair cognition. In order to enhance the sensitivity of the cognitive measure, instead of a central executive cognitive task, we will employ a paired-associative memory task that is highly sensitive to inducing error rates [10]. Further, paired-associative learning is dependent on the hippocampus [11], which is a brain structure particularly vulnerable to hypoxia [12]. The hippocampus also plays an important role in pattern separation and attenuation of memory interference [13]. Via this paired-associative task, we utilize an AB/AC memory interference paradigm that assesses both proactive and retroactive memory interference. Proactive memory interference involves the disruption of a subsequent memory from an earlier memory. In contrast, retrospective memory interference involves a subsequent memory disrupting the retrieval of a previously encoded memory.
Prior research has evaluated the effects of acute hypoxia on memory (not memory interference). Fowler et al. [14] evaluated the effects of acute hypoxia (arterial oxyhemoglobin saturation between 64%–66%) on short-term and long-term memory and found no evidence of a memory impairment effect. However, after 24 h of hypoxia exposure (simulated altitude of 4500 m), working memory was impaired [15]. Similar working memory impairments have been observed from shorter duration (30 min) acute hypoxia (5334 m or 7620 m) [16]. Although prior research has examined the effects of acute hypoxia on memory, no studies, to our knowledge, have evaluated the effects of acute hypoxia on memory interference specifically. This novel experiment is the first to evaluate the effects of acute hypoxia on immediate- and delayed-memory interference. Our a priori hypothesis was that acute hypoxia exposure would induce memory interference. The ultimate goal of this research was to establish this potential memory interference effect, and then in our subsequent research, evaluate the effects of other behaviors (e.g., acute exercise) on attenuating this potential hypoxia-induced memory interference effect.
2. Methods
2.1. Study Design
A within-subject, counterbalanced experimental design was employed. The experiment involved 2 visits, with each visit occurring approximately 24 h apart. Each participant completed both visits. Participants were blinded to the normoxia/hypoxia condition. Experimental procedures were approved by the ethics committee at the University of Mississippi and participants provided written informed consent prior to participation.
2.2. Participants
21 participants (10 males and 11 females) completed both visits. This was based on a power analysis indicating that 18 participants would be needed to achieve a power of 0.80, with inputs of 0.05 (α) and 0.08 (ƞ2). All participants were naïve, with no previous exposure to the normobaric procedure or exposure to laboratory-induced hypoxia. All participants lived at an approximate altitude of 154 m. Participants were excluded if they: (1) were outside the age range of 18–35 years, (2) exercised within 5 h of their visit, (3) consumed caffeine within 3 h of their visit, (4) were a daily smoker, (5) self-reported being pregnant, (6) had taken marijuana or other mind-altering substances in the previous 2 days, (7) had a diagnosis of attention deficit disorder/attention deficit hyperactivity disorder or took medications for these conditions, (8) consumed more than 1 alcoholic drink/day (female) or more than 2 alcoholic drinks/day (male), (9) had had a concussion in the past 30 days, (10) had a resting blood pressure ≥ 140/90 mmHg, (11) self-reported a previous episode of altitude sickness, (12) self-reported or diagnosed asthma, (13) were diagnosed with a neurological disorder, (14) measured body mass index > 35 kg/m2, or (15) had one or more of the following clotting risk factors: previously had a deep vein thrombosis (DVT), family history of DVT, previously had a thromboembolism, family history of thromboembolism, previous pulmonary embolism, family history of pulmonary embolism, Crohn’s disease diagnosis, currently taking anti-hypertensive medication, or previous orthopedic injury in the past 6 months.
2.3. Normoxia and Hypoxia
Similar to our previous work [7,8], normoxia and normobaric hypoxia was induced using the Hypoxic Training System (Everest Summit II Hypoxic Generator, New York, NY, USA). For these conditions, participants rested quietly in a seated position with a mask affixed over their mouth and nose for 30 min. The normoxic and hypoxic gas mixtures were generated through tubes that were connected to a breathing mask. The normoxic condition involved breathing a gas mixture involving FIO2 of 21%. The hypoxic condition involved breathing a gas mixture involving FIO2 of 12%.
Throughout this 30 min period, peripheral oxygen saturation (SpO2) was measured at 5 min intervals. The SpO2 was measured via a pulse oximeter (Zacurate, 500 BL) placed on the right index finger. During the 30 min period, and every five minutes, we also measured the participants’ perceptual responses (for safety reasons) using the 2018 Lake Louise Scoring System (LLSS) [17]. This questionnaire evaluates four symptoms (headache, gastrointestinal symptoms, fatigue/weakness, and dizziness/lightheadedness). Four items were assessed for each of these categories, with response options of 0 (no symptoms) to 3 (severe symptoms). The hypoxia exposure ceased if the total LLSS score was 3 or higher. Notably, no participants had a LLSS score of 3 or higher. Table 1 displays the schematic of the study protocol.

Table 1.
Study protocol.
2.4. Memory Function
On a computer, participants completed a memory interference task using the AB/AC paradigm [19]. See Table 2 for an illustration of this paradigm. Participants were exposed to List 1 (AB–DE), which consisted of eight word pairs (e.g., Baby–Hunter) and then exposed to the Cued Recall List 1 where they recalled the missing word from the word pair (e.g., Baby–_____). Participants completed a 20 s distractor task (i.e., mathematical problems) and then were exposed to List 2 (AC–FG), which consisted of eight word pairs with some overlap of A words (e.g., Baby–Salad). After another 20 s distractor, participants were exposed to the Cued Recall of List 2 and attempted to recall the missing word from the word pair. Lastly, a final 20 s distractor was implemented and then participants completed the Modified Modified Free Recall (MMFR) word list where they recalled a combined list of List 1 and List 2. The MMFR consisted of all of the previously learned word pairs, with some have only one missing word (DE, FG) and others have two missing word pair associations (AB, AC) (e.g., Shoe–Movie, Baby–Hunter and Salad). After this MMFR, which was used to establish immediate memory interference, participants played on-line games for 20 min and then recompleted the MMFR for a delayed assessment of memory interference.

Table 2.
Example protocol for evaluating memory interference. MMFR, Modified Modified Free Recall.
Using the participant’s MMFR results, the number of correctly recalled words from the subset of words (AB, DE are within List 1 and AC, FG are within List 2) were calculated. Using the cued recall results from List 1 and 2, proactive interference was measured by subtracting the percentage of correctly recalled FG pairs from the percentage of AC pairs (e.g., AC–FG) and retroactive interference was calculated by subtracting the percentage of correctly recalled DE pairs from the percentage of AB pairs (e.g., AB–DE). Higher interference scores indicate proactive and retroactive facilitation and thus correspond with decreased memory interference. That is, a lower interference score indicates a greater (worse, unfavorable) memory interference effect.
2.5. Statistical Analyses
JASP (vs. 0.10; The Netherlands) statistical software was utilized to analyze the data. A two-factor (condition and time) repeated measures ANOVA was employed. Specifically, a 2 (condition; normoxia vs. hypoxia) × 2 (time; immediate memory vs. delayed memory) repeated measures ANOVA was employed separately for proactive interference and retroactive interference. Statistical significance was set at an alpha lower than 0.05. Eta-squared (ƞ2) was calculated as an effect size estimate.
3. Results
Table 3 displays the characteristics of the sample. The sample was young (21.0 years), of near equal gender distribution (52.4% female) and heterogeneous regarding race-ethnicity (50.0% white).

Table 3.
Participant characteristics.
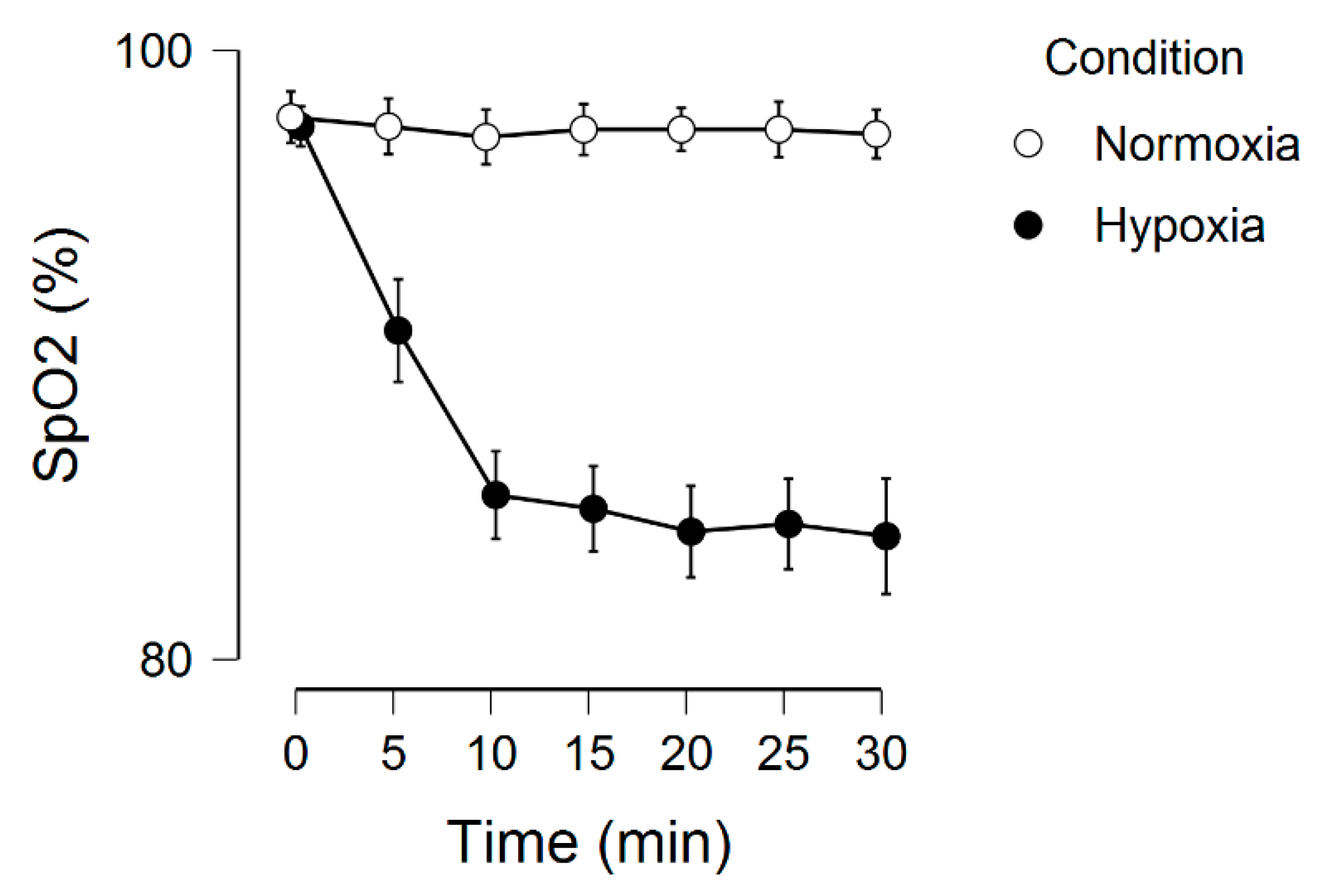
Figure 1 displays the SpO2 across the two experimental conditions. There was a significant main effect for condition, F(1, 19) = 239.0, p < 0.001, ƞ2 = 0.56, main effect for time, F(6, 114) = 54.8, p < 0.001, ƞ2 = 0.13, and condition by time interaction, F(6, 114) = 54.0, p < 0.001, ƞ2 = 0.11.
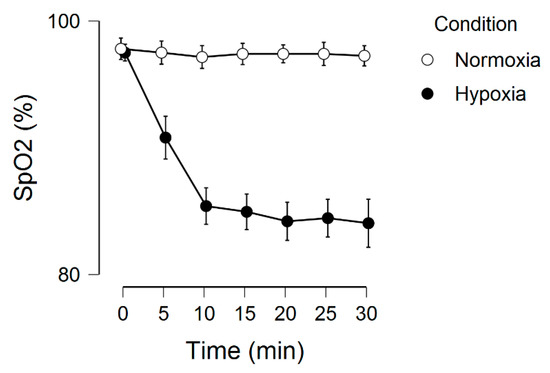
Figure 1.
Peripheral oxygen saturation as a function of time and condition. Error bars represent 95% confidence intervals.
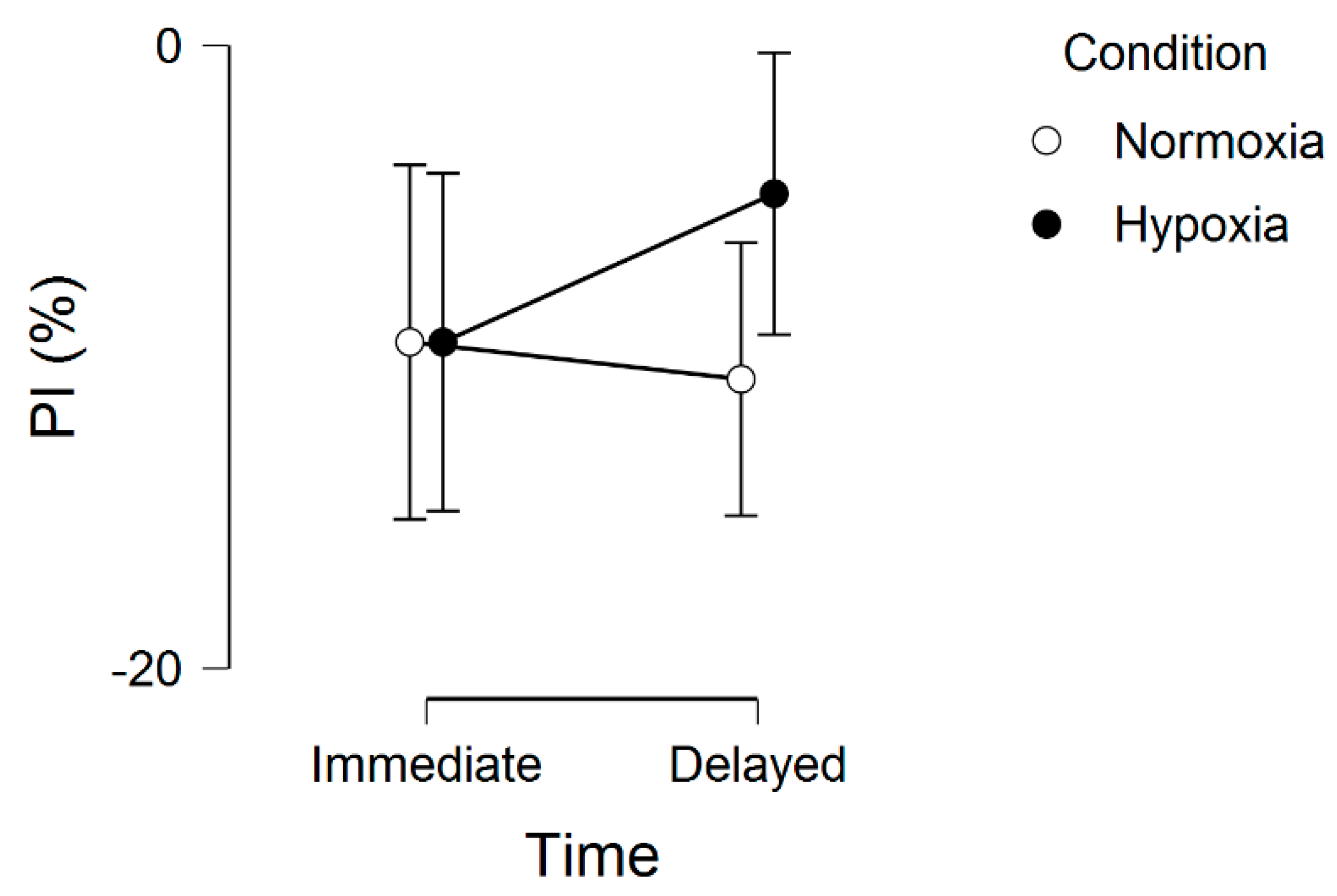
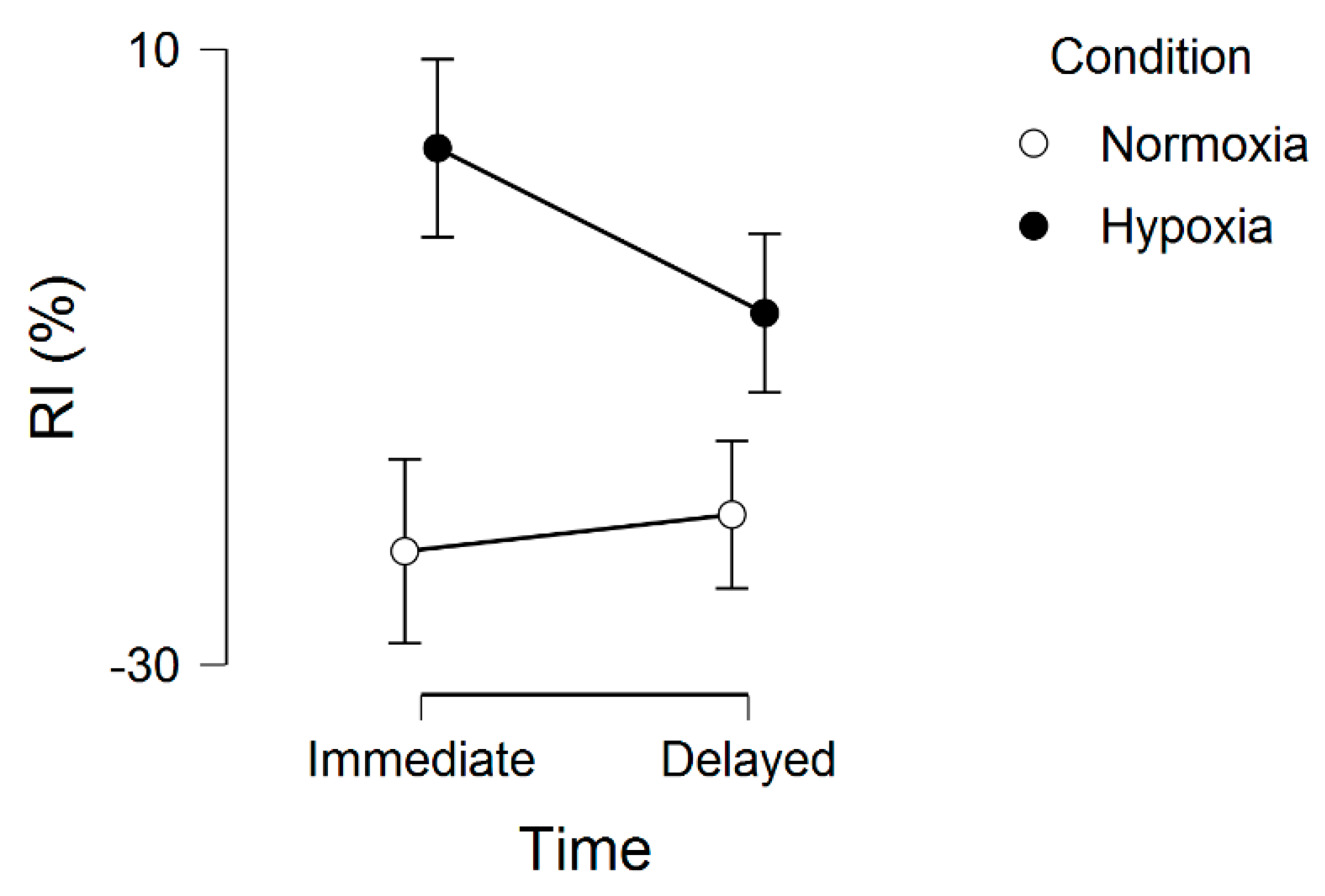
Regarding the memory interference outcomes, for proactive interference, there were no significant effects, including main effect for condition, F(1, 20) = 0.16, p = 0.69, ƞ2 = 0.002, main effect for time, F(1, 20) = 0.34, p = 0.56, ƞ2 = 0.001, and condition by time interaction, F(1, 20) = 0.74, p = 0.40, ƞ2 = 0.002. These results are graphically shown in Figure 2. For retroactive interference, we observed a significant main effect for condition, F(1, 20) = 5.48, p = 0.03, ƞ2 = 0.10, condition by time interaction, F(1, 20) = 4.96, p = 0.03, ƞ2 = 0.01, but no main effect for time, F(1, 20) = 1.75, p = 0.20, ƞ2 = 0.004. These results are graphically shown in Figure 3. Given that a lower interference score indicates a greater (worse, unfavorable) memory interference effect, these results demonstrate that the hypoxia condition had a higher score, suggesting a reduced memory interference effect.
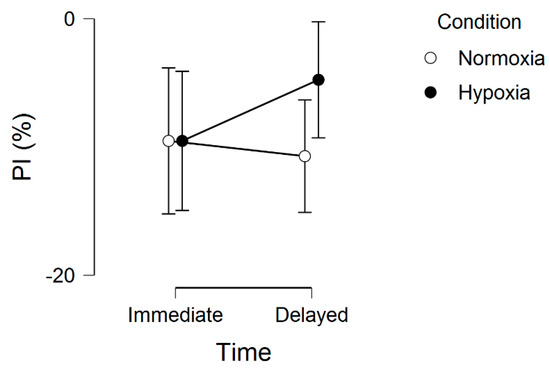
Figure 2.
Proactive interference (PI) across normoxia and hypoxia for both the immediate and delayed memory assessments. Error bars represent standard errors.
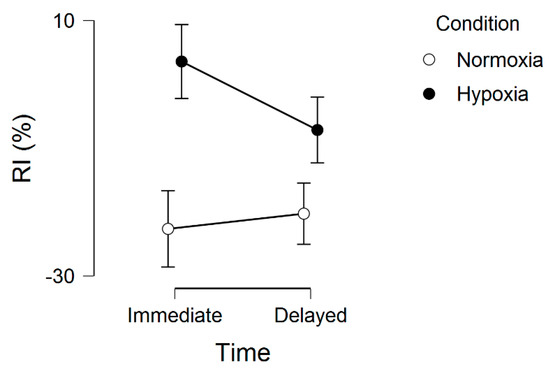
Figure 3.
Retroactive interference (RI) across normoxia and hypoxia for both the immediate and delayed memory assessments. Error bars represent standard errors.
4. Discussion
The purpose of this experiment was to evaluate the effects of normoxia/hypoxia on proactive and retroactive memory interference. The motivation for this experiment came from our earlier work evaluating the effects of hypoxia on other cognitive outcomes that did not reveal a detrimental effect. We anticipated that these null findings were a result of the cognitive measure used, and, as such, we employed a measure that we expected would be more sensitive to hypoxia. Further, the ultimate goal of our future work is to evaluate the extent to which acute exercise may attenuate hypoxia-induced memory function, including memory interference. Based on this, the present experiment was designed to first examine the extent to which hypoxia may induce memory interference. To our surprise, however, the result of the present experiment demonstrated that hypoxia induced retroactive memory facilitation, i.e., reduced retroactive memory interference.
As stated, we anticipated that hypoxia would induce a memory impairment effect by specifically inducing memory interference. Unexpectedly, our results showed that hypoxia reduced a retroactive memory interference effect, with a trend towards also reducing a proactive interference effect. Upon careful consideration of this observation, we identified emerging research showing that, in certain situations, acute hypoxia may enhance brain function. This possibility has been thoroughly discussed elsewhere [20,21]. Other research has also shown that, for various conditions (e.g., metabolic syndrome), both normobaric and hypobaric hypoxia may have therapeutic effects [22,23].
The dose of hypoxia is what determines whether a therapeutic benefit or net pathological effect ensues. That is, low doses of acute hypoxia do not elicit detectable pathologies, such as hippocampal apoptosis or reactive gliosis [20]. Beneficial effects of a single exposure of acute hypoxia has occurred from intermittent protocols involving 3 to 15 episodes, 5 min in duration, with 5 min intervals (i.e., 15 min to 75 min of hypoxia exposure) [20]. This 15–75 min exposure aligns with our present experiment (i.e., 30 min of exposure), but, notably, we acknowledge that the physiological and neurochemical responses to intermittent hypoxia protocols may not be the same as continuous protocols [24,25]. However, two of the recently recognized unexpected benefits of low-dose acute hypoxia include improved respiratory and non-respiratory somatic motor function and increased growth factor expression in the central nervous system.
Increased growth factor production, including upregulation of brain-derived neurotrophic factor (BDNF), plays a critical role in subserving memory function [26]. Tropomyosin-regulated kinase B (TrkB), a BDNF receptor, is regulated by hypoxia-inducible factor-1, a transcription factor regulated by hypoxia-sensitive genes [27]. Thus, TrkB regulation is upregulated in the whole brain following acute hypoxia exposure. These acute effects may facilitate memory function, as BDNF has been shown to upregulate the expression and function of key memory-related receptors, namely NMDA [28,29,30]. Further, a key mechanistic correlate of episodic memory function is long-term potentiation, that is, sustained neurotransmitter release to maintain excitatory post-synaptic potentiation. Downstream of the BDNF/TrkB, pathway, PI3K-AKT activation helps maintain LTP via NMDA activity [31]. Thus, hypoxia-induced BDNF production may help facilitate memory via increasing synaptic strength (i.e., plasticity). In addition to BDNF-related improvements in episodic memory function, BDNF also appears to play a critical role in neuronal pattern separation of similar content [32], which has implications in reducing memory interference.
In conclusion, this experiment is the first to demonstrate that acute hypoxia exposure can reduce retroactive memory interference. If replicated, this has important implications on multiple levels. For example, it would be worthwhile to investigate whether acute hypoxia can attenuate memory interference among populations (e.g., older adults) that are vulnerable to memory interference. It would also be interesting to investigate whether acute hypoxia, coupled with other strategies thought to enhance memory (e.g., acute exercise), have a synergistic effect on enhancing memory and reducing memory interference. Such future work should aim to overcome the limitations of this experiment, which includes, for example, the relatively small, homogeneous sample, as well as the lack of a mechanistic measure (e.g., BDNF). Further, future work should more precisely and comprehensively evaluate the physiological responses to hypoxia exposure. For example, when feasible, more accurate measures of arterial oxygen saturation (via arterial blood assessment) should be evaluated. Further, there is evidence [33,34] to suggest that hypobaric and normobaric hypoxia environments may induce unique physiological responses (e.g., slightly different minute ventilation and nitric oxide levels), and, as such, future work may wish to evaluate whether the type of hypoxia exposure has a unique effect on memory interference. Relatedly, such work should carefully evaluate the rate of breathing during hypoxia exposure, as the rate and depth of breathing can reduce the partial pressure of arterial carbon dioxide (PaCO2). Such resultant hypocapnia effects may cause respiratory alkalosis and cerebral vasoconstriction, ultimately exacerbating cerebral hypoxia. The present experiment also did not measure water vapor pressure (PH2O), which may be an important parameter to evaluate, as it may influence the inspired partial pressure of oxygen. Such recommendations will be critical in future work that aims to evaluate the effects of specific doses of hypoxia on memory interference.
Author Contributions
Author P.D.L. conceptualized the study, analyzed the data and prepared the initial draft of the manuscript. Author L.C. supervised data collection. Author A.M. collected the data. All authors provided feedback on the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Gibson, G.E.; Peterson, C. Decreases in the release of acetylcholine in vitro with low oxygen. Biochem. Pharmacol. 1982, 31, 111–115. [Google Scholar] [CrossRef]
- Gibson, G.E.; Pulsinelli, W.; Blass, J.P.; Duffy, T.E. Brain dysfunction in mild to moderate hypoxia. Am. J. Med. 1981, 70, 1247–1254. [Google Scholar] [CrossRef]
- Virues-Ortega, J.; Buela-Casal, G.; Garrido, E.; Alcazar, B. Neuropsychological functioning associated with high-altitude exposure. Neuropsychol. Rev. 2004, 14, 197–224. [Google Scholar] [CrossRef]
- McMorris, T.; Hale, B.J.; Barwood, M.; Costello, J.; Corbett, J. Effect of acute hypoxia on cognition: A systematic review and meta-regression analysis. Neurosci. Biobehav. Rev. 2017, 74, 225–232. [Google Scholar] [CrossRef]
- Aquino-Lemos, V.; Santos, R.V.; Antunes, H.K.; Lira, F.S.; Luz Bittar, I.G.; Caris, A.V. Acute physical exercise under hypoxia improves sleep, mood and reaction time. Physiol. Behav. 2016, 154, 90–99. [Google Scholar] [CrossRef]
- Komiyama, T.; Katayama, K.; Sudo, M.; Ishida, K.; Higaki, Y.; Ando, S. Cognitive function during exercise under severe hypoxia. Sci. Rep. 2017, 7, 10000. [Google Scholar] [CrossRef]
- Sun, S.; Loprinzi, P.D.; Guan, H.; Zou, L.; Kong, Z.; Hu, Y. The Effects of High-Intensity Interval Exercise and Hypoxia on Cognition in Sedentary Young Adults. Medicina 2019, 55, 43. [Google Scholar] [CrossRef]
- Lei, O.K.; Kong, Z.; Loprinzi, P.D.; Shi, Q.; Sun, S.; Zou, L. Severe Hypoxia Does Not Offset the Benefits of Exercise on Cognitive Function in Sedentary Young Women. Int. J. Environ. Res. Public Health 2019, 16, 1003. [Google Scholar] [CrossRef]
- Nakata, H.; Miyamoto, T.; Ogoh, S.; Kakigi, R.; Shibasaki, M. Effects of acute hypoxia on human cognitive processing: A study using ERPs and SEPs. J. Appl. Physiol. 2017, 123, 1246–1255. [Google Scholar] [CrossRef]
- Burton, R.L.; Lek, I.; Caplan, J.B. Associative independence revisited: Competition between conflicting associations can be resolved or even reversed in one trial. Q. J. Exp. Psychol. (Hove) 2017, 70, 832–857. [Google Scholar] [CrossRef]
- Yoon, J.; Seo, Y.; Kim, J.; Lee, I. Hippocampus is required for paired associate memory with neither delay nor trial uniqueness. Learn. Mem. 2012, 19, 1–8. [Google Scholar] [CrossRef][Green Version]
- Hossmann, K.A. The hypoxic brain. Insights from ischemia research. Adv. Exp. Med. Biol. 1999, 474, 155–169. [Google Scholar]
- Koolschijn, R.S.; Emir, U.E.; Pantelides, A.C.; Nili, H.; Behrens, T.E.J.; Barron, H.C. The Hippocampus and Neocortical Inhibitory Engrams Protect against Memory Interference. Neuron 2019, 101, 528–541. [Google Scholar] [CrossRef]
- Fowler, B.; Prlic, H.; Brabant, M. Acute hypoxia fails to influence two aspects of short-term memory: Implications for the source of cognitive deficits. Aviat. Space Environ. Med. 1994, 65, 641–645. [Google Scholar]
- Aquino Lemos, V.; Antunes, H.K.; Santos, R.V.; Lira, F.S.; Tufik, S.; Mello, M.T. High altitude exposure impairs sleep patterns, mood, and cognitive functions. Psychophysiology 2012, 49, 1298–1306. [Google Scholar] [CrossRef]
- Asmaro, D.; Mayall, J.; Ferguson, S. Cognition at altitude: Impairment in executive and memory processes under hypoxic conditions. Aviat. Space Environ. Med. 2013, 84, 1159–1165. [Google Scholar] [CrossRef]
- Roach, R.C.; Hackett, P.H.; Oelz, O.; Bartsch, P.; Luks, A.M.; MacInnis, M.J. The 2018 Lake Louise Acute Mountain Sickness Score. High Alt. Med. Biol. 2018, 19, 4–6. [Google Scholar] [CrossRef]
- Blough, J.; Loprinzi, P.D. Experimental manipulation of psychological control scenarios: Implications for exercise and memory research. Psych 2019, 1, 279–289. [Google Scholar] [CrossRef]
- Crawford, L.; Loprinzi, P.D. Effects of intensity-specific acute exercise on paired-associative memory and memory interference. Psych 2019, 1, 290–305. [Google Scholar] [CrossRef]
- Dale, E.A.; Ben Mabrouk, F.; Mitchell, G.S. Unexpected benefits of intermittent hypoxia: Enhanced respiratory and nonrespiratory motor function. Physiology 2014, 29, 9–48. [Google Scholar] [CrossRef]
- Taylor, L.; Watkins, S.L.; Marshall, H.; Dascombe, B.J.; Foster, J. The Impact of Different Environmental Conditions on Cognitive Function: A Focused Review. Front. Physiol. 2015, 6, 372. [Google Scholar] [CrossRef]
- Cai, Z.; Manalo, D.J.; Wei, G.; Rodriguez, E.R.; Fox-Talbot, K.; Lu, H. Hearts from rodents exposed to intermittent hypoxia or erythropoietin are protected against ischemia-reperfusion injury. Circulation 2003, 108, 79–85. [Google Scholar] [CrossRef]
- Costa, D.C.; Alva, N.; Trigueros, L.; Gamez, A.; Carbonell, T.; Rama, R. Intermittent hypobaric hypoxia induces neuroprotection in kainate-induced oxidative stress in rats. J. Mol. Neurosci. 2013, 50, 402–410. [Google Scholar] [CrossRef]
- Cummings, K.J.; Wilson, R.J. Time-dependent modulation of carotid body afferent activity during and after intermittent hypoxia. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2005, 288, R1571–R1580. [Google Scholar] [CrossRef]
- Neubauer, J.A. Invited review: Physiological and pathophysiological responses to intermittent hypoxia. J. Appl. Physiol. 2001, 90, 1593–1599. [Google Scholar] [CrossRef]
- Loprinzi, P.D.; Frith, E. A brief primer on the mediational role of BDNF in the exercise-memory link. Clin. Physiol. Funct. Imaging 2019, 39, 9–14. [Google Scholar] [CrossRef]
- Martens, L.K.; Kirschner, K.M.; Warnecke, C.; Scholz, H. Hypoxia-inducible factor-1 (HIF-1) is a transcriptional activator of the TrkB neurotrophin receptor gene. J. Biol. Chem. 2007, 282, 14379–14388. [Google Scholar] [CrossRef]
- Clarke, R.J.; Johnson, J.W. Voltage-dependent gating of NR1/2B NMDA receptors. J. Physiol. 2008, 586, 5727–5741. [Google Scholar] [CrossRef]
- Caldeira, M.V.; Melo, C.V.; Pereira, D.B.; Carvalho, R.F.; Carvalho, A.L.; Duarte, C.B. BDNF regulates the expression and traffic of NMDA receptors in cultured hippocampal neurons. Mol. Cell. Neurosci. 2007, 35, 208–219. [Google Scholar] [CrossRef]
- Kim, J.H.; Roberts, D.S.; Hu, Y.; Lau, G.C.; Brooks-Kayal, A.R.; Farb, D.H. Brain-derived neurotrophic factor uses CREB and Egr3 to regulate NMDA receptor levels in cortical neurons. J. Neurochem. 2012, 120, 210–219. [Google Scholar] [CrossRef]
- Nakai, T.; Nagai, T.; Tanaka, M.; Itoh, N.; Asai, N.; Enomoto, A. Girdin phosphorylation is crucial for synaptic plasticity and memory: A potential role in the interaction of BDNF/TrkB/Akt signaling with NMDA receptor. J. Neurosci. 2014, 34, 14995–15008. [Google Scholar] [CrossRef]
- Bekinschtein, P.; Kent, B.A.; Oomen, C.A.; Clemenson, G.D.; Gage, F.H.; Saksida, L.M. BDNF in the dentate gyrus is required for consolidation of “pattern-separated” memories. Cell Rep. 2013, 5, 759–768. [Google Scholar] [CrossRef]
- Fulco, C.S.; Beidleman, B.A.; Muza, S.R. Effectiveness of preacclimatization strategies for high-altitude exposure. Exerc. Sport. Sci. Rev. 2013, 41, 55–63. [Google Scholar] [CrossRef]
- Coppel, J.; Hennis, P.; Gilbert-Kawai, E.; Grocott, M.P. The physiological effects of hypobaric hypoxia versus normobaric hypoxia: A systematic review of crossover trials. Extrem. Physiol. Med. 2015, 4, 2. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).