A Systematic Review of Carotenoids in the Management of Age-Related Macular Degeneration
Abstract
:1. Introduction
1.1. Age-Related Macular Degeneration
1.2. Macular Pigment Optical Density in AMD—Background
1.3. Measuring MPOD
1.4. MPOD Biomarkers in Clinical AMD
2. Materials and Methods
2.1. Literature Search and Selection Strategy
2.2. Study Selection Criteria
2.3. Data Extraction, Reliability and Risk of Bias Assessment
3. Results
3.1. Search and Selection of Eligible Studies
3.2. Carotenoids and Risk of AMD (Observational Studies)
3.3. Carotenoids in the Management of AMD (Interventional Studies)
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- National Eye Institute. Age-Related Macular Degeneration. Available online: https://www.nei.nih.gov/learn-about-eye-health/eye-conditions-and-diseases/age-related-macular-degeneration (accessed on 12 May 2021).
- Bourne, R.R.A.; Jonas, J.B.; Bron, A.M.; Cicinelli, M.V.; Das, A.; Flaxman, S.R.; Friedman, D.S.; Keeffe, J.E.; Kempen, J.H.; Leasher, J.; et al. Prevalence and causes of vision loss in high-income countries and in Eastern and Central Europe in 2015: Magnitude, temporal trends and projections. Br. J. Ophthalmol. 2018, 102, 575–585. [Google Scholar] [CrossRef]
- Wong, W.L.; Su, X.; Li, X.; Cheung, C.M.; Klein, R.; Cheng, C.Y.; Wong, T.Y. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: A systematic review and meta-analysis. Lancet Glob. Health 2014, 2, e106–e116. [Google Scholar] [CrossRef] [Green Version]
- Jonas, J.B.; Cheung, C.M.G.; Panda-Jonas, S. Updates on the epidemiology of age-related macular degeneration. Asia Pac. J. Ophthalmol. 2017, 6, 493–497. [Google Scholar] [CrossRef]
- Lambert, N.G.; ElShelmani, H.; Singh, M.K.; Mansergh, F.C.; Wride, M.A.; Padilla, M.; Keegan, D.; Hogg, R.E.; Ambati, B.K. Risk factors and biomarkers of age-related macular degeneration. Prog. Retin. Eye Res. 2016, 54, 64–102. [Google Scholar] [CrossRef] [Green Version]
- Klein, R.; Klein, B.E.; Knudtson, M.D.; Wong, T.Y.; Cotch, M.F.; Liu, K.; Burke, G.; Saad, M.F.; Jacobs, D.R., Jr. Prevalence of age-related macular degeneration in 4 racial/ethnic groups in the multi-ethnic study of atherosclerosis. Ophthalmology 2006, 113, 373–380. [Google Scholar] [CrossRef]
- Priya, R.R.; Chew, E.Y.; Swaroop, A. Genetic studies of age-related macular degeneration: Lessons, challenges, and opportunities for disease management. Ophthalmology 2012, 119, 2526–2536. [Google Scholar] [CrossRef] [Green Version]
- Chakrabarti, M. Noncommunicable diseases and noncommunicable eye diseases: What is the way forward? Kerala J. Ophthalmol. 2020, 32, 224–231. [Google Scholar] [CrossRef]
- World Health Organization. World Report on Vision 2019. Available online: https://www.who.int/publications/i/item/world-report-on-vision (accessed on 5 April 2021).
- Resnikoff, S.; Kocur, I. Non-communicable eye diseases: Facing the future. Community Eye Health 2014, 27, 41–43. [Google Scholar] [PubMed]
- Kawasaki, R.; Yasuda, M.; Song, S.J.; Chen, S.J.; Jonas, J.B.; Wang, J.J.; Mitchell, P.; Wong, T.Y. The prevalence of age-related macular degeneration in Asians: A systematic review and meta-analysis. Ophthalmology 2010, 117, 921–927. [Google Scholar] [CrossRef]
- Klein, R.; Chou, C.F.; Klein, B.E.; Zhang, X.; Meuer, S.M.; Saaddine, J.B. Prevalence of age-related macular degeneration in the US population. Arch. Ophthalmol. 2011, 129, 75–80. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krishnan, T.; Ravindran, R.D.; Murthy, G.V.; Vashist, P.; Fitzpatrick, K.E.; Thulasiraj, R.D.; John, N.; Maraini, G.; Camparini, M.; Chakravarthy, U.; et al. Prevalence of early and late age-related macular degeneration in India: The INDEYE study. Investig. Ophthalmol. Vis. Sci. 2010, 51, 701–707. [Google Scholar] [CrossRef]
- Rein, D.B. Forecasting age-related macular degeneration through the year 2050. Arch. Ophthalmol. 2009, 127, 533. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitchell, P.; Liew, G.; Gopinath, B.; Wong, T.Y. Age-related macular degeneration. Lancet 2018, 392, 1147–1159. [Google Scholar] [CrossRef]
- Ho, L.; van Leeuwen, R.; Witteman, J.C.; van Duijn, C.M.; Uitterlinden, A.G.; Hofman, A.; de Jong, P.T.; Vingerling, J.R.; Klaver, C.C. Reducing the genetic risk of age-related macular degeneration with dietary antioxidants, zinc, and omega-3 fatty acids: The Rotterdam study. Arch. Ophthalmol. 2011, 129, 758–766. [Google Scholar] [CrossRef] [Green Version]
- Jabbarpoor Bonyadi, M.H.; Yaseri, M.; Nikkhah, H.; Bonyadi, M.; Soheilian, M. Association of risk genotypes of ARMS2/LOC387715 A69S and CFH Y402H with age-related macular degeneration with and without reticular pseudodrusen: A meta-analysis. Acta Ophthalmol. 2018, 96, e105–e110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meyers, K.J.; Johnson, E.J.; Bernstein, P.S.; Iyengar, S.K.; Engelman, C.D.; Karki, C.K.; Liu, Z.; Igo, R.P., Jr.; Truitt, B.; Klein, M.L.; et al. Genetic determinants of macular pigments in women of the Carotenoids in Age-Related Eye Disease Study. Investig. Ophthalmol. Vis. Sci. 2013, 54, 2333–2345. [Google Scholar] [CrossRef] [Green Version]
- Khanamiri, H.N.; Falavarjani, K.G.; Sanati, M.H.; Aryan, H.; Irani, A.; Hashemi, M.; Modarres, M.; Parvaresh, M.M.; Nikeghbali, A. Complement factor H Y402H and LOC387715 A69S polymorphisms in association with age-related macular degeneration in Iran. J. Ophthalmic Vis. Res. 2014, 9, 181–187. [Google Scholar] [PubMed]
- Vavvas, D.G.; Small, K.W.; Awh, C.C.; Zanke, B.W.; Tibshirani, R.J.; Kustra, R. CFH and ARMS2 genetic risk determines progression to neovascular age-related macular degeneration after antioxidant and zinc supplementation. Proc. Natl. Acad. Sci. USA 2018, 115, E696–E704. [Google Scholar] [CrossRef] [Green Version]
- Seddon, J.M.; Cote, J.; Page, W.F.; Aggen, S.H.; Neale, M.C. The US twin study of age-related macular degeneration: Relative roles of genetic and environmental influences. Arch. Ophthalmol. 2005, 123, 321–327. [Google Scholar] [CrossRef]
- Bonyadi, M.; Foruzandeh, Z.; Mohammadian, T.; Fotouhi, N.; Soheilian, M.; Jabbarpoor Bonyadi, M.H.; Javadzadeh, A.; Moein, H.; Yaseri, M. Evaluation of CC-cytokine ligand 2 and complementary factor H Y402H polymorphisms and their interactional association with age-related macular degeneration. Acta Ophthalmol. 2016, 94, e779–e785. [Google Scholar] [CrossRef]
- Rivera, A.; Fisher, S.A.; Fritsche, L.G.; Keilhauer, C.N.; Lichtner, P.; Meitinger, T.; Weber, B.H. Hypothetical LOC387715 is a second major susceptibility gene for age-related macular degeneration, contributing independently of complement factor H to disease risk. Hum. Mol. Genet. 2005, 14, 3227–3236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weeks, D.E.; Conley, Y.P.; Tsai, H.J.; Mah, T.S.; Schmidt, S.; Postel, E.A.; Agarwal, A.; Haines, J.L.; Pericak-Vance, M.A.; Rosenfeld, P.J.; et al. Age-related maculopathy: A genomewide scan with continued evidence of susceptibility loci within the 1q31, 10q26, and 17q25 regions. Am. J. Hum. Genet. 2004, 75, 174–189. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.J.; Buitendijk, G.H.; Rochtchina, E.; Lee, K.E.; Klein, B.E.; van Duijn, C.M.; Flood, V.M.; Meuer, S.M.; Attia, J.; Myers, C.; et al. Genetic susceptibility, dietary antioxidants, and long-term incidence of age-related macular degeneration in two populations. Ophthalmology 2014, 121, 667–675. [Google Scholar] [CrossRef] [PubMed]
- Caban-Martinez, A.J.; Davila, E.P.; Lam, B.L.; Dubovy, S.R.; McCollister, K.E.; Fleming, L.E.; Zheng, D.D.; Lee, D.J. Age-related macular degeneration and smoking cessation advice by eye care providers: A pilot study. Prev. Chronic Dis. 2011, 8, A147. [Google Scholar] [PubMed]
- Chakravarthy, U.; Augood, C.; Bentham, G.C.; de Jong, P.T.; Rahu, M.; Seland, J.; Soubrane, G.; Tomazzoli, L.; Topouzis, F.; Vingerling, J.R.; et al. Cigarette smoking and age-related macular degeneration in the EUREYE Study. Ophthalmology 2007, 114, 1157–1163. [Google Scholar] [CrossRef] [PubMed]
- Handa, S.; Woo, J.H.; Wagle, A.M.; Htoon, H.M.; Eong, K.A. Awareness of blindness and other smoking-related diseases and its impact on motivation for smoking cessation in eye patients. Eye 2011, 25, 1170–1176. [Google Scholar] [CrossRef] [Green Version]
- Choudhury, F.; Varma, R.; McKean-Cowdin, R.; Klein, R.; Azen, S.P. Risk factors for four-year incidence and progression of age-related macular degeneration: The Los Angeles Latino Eye Study. Am. J. Ophthalmol. 2011, 152, 385–395. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seddon, J.M. Progression of age-related macular degeneration. Arch. Ophthalmol. 2003, 121, 785. [Google Scholar] [CrossRef] [Green Version]
- Hammond, B.R., Jr.; Wooten, B.R.; Snodderly, D.M. Cigarette smoking and retinal carotenoids: Implications for age-related macular degeneration. Vis. Res. 1996, 36, 3003–3009. [Google Scholar] [CrossRef] [Green Version]
- Kaarniranta, K.; Kajdanek, J.; Morawiec, J.; Pawlowska, E.; Blasiak, J. PGC-1alpha protects RPE cells of the aging retina against oxidative stress-induced degeneration through the regulation of senescence and mitochondrial quality control. The significance for AMD pathogenesis. Int. J. Mol. Sci. 2018, 19, 2317. [Google Scholar] [CrossRef] [Green Version]
- Marazita, M.C.; Dugour, A.; Marquioni-Ramella, M.D.; Figueroa, J.M.; Suburo, A.M. Oxidative stress-induced premature senescence dysregulates VEGF and CFH expression in retinal pigment epithelial cells: Implications for age-related macular degeneration. Redox Biol. 2016, 7, 78–87. [Google Scholar] [CrossRef] [Green Version]
- Maugeri, A.; Barchitta, M.; Mazzone, M.G.; Giuliano, F.; Agodi, A. Complement System and age-related macular degeneration: Implications of gene-environment interaction for preventive and personalized medicine. Biomed. Res. Int. 2018, 2018, 7532507. [Google Scholar] [CrossRef] [Green Version]
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2008, 454, 428–435. [Google Scholar] [CrossRef]
- Nozaki, M.; Raisler, B.J.; Sakurai, E.; Sarma, J.V.; Barnum, S.R.; Lambris, J.D.; Chen, Y.; Zhang, K.; Ambati, B.K.; Baffi, J.Z.; et al. Drusen complement components C3a and C5a promote choroidal neovascularization. Proc. Natl. Acad. Sci. USA 2006, 103, 2328–2333. [Google Scholar] [CrossRef] [Green Version]
- Xu, H.; Chen, M. Targeting the complement system for the management of retinal inflammatory and degenerative diseases. Eur. J. Pharmacol. 2016, 787, 94–104. [Google Scholar] [CrossRef] [Green Version]
- Dong, A.; Xie, B.; Shen, J.; Yoshida, T.; Yokoi, K.; Hackett, S.F.; Campochiaro, P.A. Oxidative stress promotes ocular neovascularization. J. Cell. Physiol. 2009, 219, 544–552. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beatty, S.; Koh, H.; Phil, M.; Henson, D.; Boulton, M. The role of oxidative stress in the pathogenesis of age-related macular degeneration. Surv. Ophthalmol. 2000, 45, 115–134. [Google Scholar] [CrossRef] [Green Version]
- Chen, M.; Xu, H. Parainflammation, chronic inflammation, and age-related macular degeneration. J. Leukoc. Biol. 2015, 98, 713–725. [Google Scholar] [CrossRef] [Green Version]
- Xu, H.; Chen, M.; Forrester, J.V. Para-inflammation in the aging retina. Prog. Retin. Eye Res. 2009, 28, 348–368. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, P.S.; Li, B.; Vachali, P.P.; Gorusupudi, A.; Shyam, R.; Henriksen, B.S.; Nolan, J.M. Lutein, zeaxanthin, and meso-zeaxanthin: The basic and clinical science underlying carotenoid-based nutritional interventions against ocular disease. Prog. Retin. Eye Res. 2016, 50, 34–66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lem, D.W.; Gierhart, D.L.; Davey, P.G. Management of diabetic eye disease using carotenoids and nutrients. In Antioxidants—Benefits, Sources, and Mechanisms of Action; Waisundara, V.Y., Ed.; IntechOpen: London, UK, 2021. [Google Scholar]
- Beatty, S.; Boulton, M.; Henson, D.; Koh, H.H.; Murray, I.J. Macular pigment and age related macular degeneration. Br. J. Ophthalmol. 1999, 83, 867–877. [Google Scholar] [CrossRef]
- Cai, J.; Nelson, K.C.; Wu, M.; Sternberg, P., Jr.; Jones, D.P. Oxidative damage and protection of the RPE. Prog. Retin. Eye Res. 2000, 19, 205–221. [Google Scholar] [CrossRef]
- Georgieva, E.; Ivanova, D.; Zhelev, Z.; Bakalova, R.; Gulubova, M.; Aoki, I. Mitochondrial dysfunction and redox imbalance as a diagnostic marker of “free radical diseases”. Anticancer Res. 2017, 37, 5373–5381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lem, D.W.; Gierhart, D.L.; Davey, P.G. Carotenoids in the management of glaucoma: A systematic review of the evidence. Nutrients 2021, 13, 1949. [Google Scholar] [CrossRef] [PubMed]
- Lem, D.W.; Gierhart, D.L.; Davey, P.G. A systematic review of carotenoids in the management of diabetic retinopathy. Nutrients 2021, 13, 2441. [Google Scholar] [CrossRef]
- Shimomachi, M.; Hasan, M.Z.; Kawaichi, M.; Oka, C. HtrA1 is induced by oxidative stress and enhances cell senescence through p38 MAPK pathway. Exp. Eye Res. 2013, 112, 79–92. [Google Scholar] [CrossRef]
- Age-Related Eye Disease Study 2 Research Group; Chew, E.Y.; Clemons, T.E.; SanGiovanni, J.P.; Danis, R.; Ferris, F.L., 3rd; Elman, M.; Antoszyk, A.; Ruby, A.; Orth, D.; et al. Lutein + zeaxanthin and omega-3 fatty acids for age-related macular degeneration: The Age-Related Eye Disease Study 2 (AREDS2) randomized clinical trial. JAMA 2013, 309, 2005–2015. [Google Scholar] [CrossRef]
- Age-Related Eye Disease Study Research Group; SanGiovanni, J.P.; Chew, E.Y.; Clemons, T.E.; Ferris, F.L., 3rd; Gensler, G.; Lindblad, A.S.; Milton, R.C.; Seddon, J.M.; Sperduto, R.D. The relationship of dietary carotenoid and vitamin A, E, and C intake with age-related macular degeneration in a case-control study: AREDS Report No. 22. Arch. Ophthalmol. 2007, 125, 1225–1232. [Google Scholar] [CrossRef]
- Chous, A.P.; Richer, S.P.; Gerson, J.D.; Kowluru, R.A. The diabetes visual function supplement study (DiVFuSS). Br. J. Ophthalmol. 2016, 100, 227–234. [Google Scholar] [CrossRef] [Green Version]
- Davey, P.G.; Henderson, T.; Lem, D.W.; Weis, R.; Amonoo-Monney, S.; Evans, D.W. Visual function and macular carotenoid changes in eyes with retinal drusen—An open label randomized controlled trial to compare a micronized lipid-based carotenoid liquid supplementation and AREDS-2 formula. Nutrients 2020, 12, 3271. [Google Scholar] [CrossRef]
- Johnson, E.J.; Avendano, E.E.; Mohn, E.S.; Raman, G. The association between macular pigment optical density and visual function outcomes: A systematic review and meta-analysis. Eye 2020, 35, 1620–1628. [Google Scholar] [CrossRef]
- Loughman, J.; Nolan, J.M.; Howard, A.N.; Connolly, E.; Meagher, K.; Beatty, S. The impact of macular pigment augmentation on visual performance using different carotenoid formulations. Investig. Ophthalmol. Vis. Sci. 2012, 53, 7871–7880. [Google Scholar] [CrossRef] [Green Version]
- Ma, L.; Liu, R.; Du, J.H.; Liu, T.; Wu, S.S.; Liu, X.H. Lutein, zeaxanthin and meso-zeaxanthin supplementation associated with macular pigment optical density. Nutrients 2016, 8, 426. [Google Scholar] [CrossRef]
- Nolan, J.M.; Loughman, J.; Akkali, M.C.; Stack, J.; Scanlon, G.; Davison, P.; Beatty, S. The impact of macular pigment augmentation on visual performance in normal subjects: COMPASS. Vis. Res. 2011, 51, 459–469. [Google Scholar] [CrossRef] [Green Version]
- Nolan, J.M.; Power, R.; Stringham, J.; Dennison, J.; Stack, J.; Kelly, D.; Moran, R.; Akuffo, K.O.; Corcoran, L.; Beatty, S. Enrichment of macular pigment enhances contrast sensitivity in subjects free of retinal disease: Central retinal enrichment supplementation trials—Report 1. Investig. Ophthalmol. Vis. Sci. 2016, 57, 3429–3439. [Google Scholar] [CrossRef] [PubMed]
- Guymer, R.; Wu, Z. Age-related macular degeneration (AMD): More than meets the eye. The role of multimodal imaging in today’s management of AMD-A review. Clin. Exp. Ophthalmol. 2020, 48, 983–995. [Google Scholar] [CrossRef]
- Miller, J.W.; Bagheri, S.; Vavvas, D.G. Advances in Age-related macular degeneration understanding and therapy. US Ophthalmic Rev. 2017, 10, 119–130. [Google Scholar] [CrossRef] [Green Version]
- Bird, A.C.; Bressler, N.M.; Bressler, S.B.; Chisholm, I.H.; Coscas, G.; Davis, M.D.; de Jong, P.T.; Klaver, C.C.; Klein, B.E.; Klein, R.; et al. An international classification and grading system for age-related maculopathy and age-related macular degeneration. The International ARM Epidemiological Study Group. Surv. Ophthalmol. 1995, 39, 367–374. [Google Scholar] [CrossRef] [Green Version]
- Klein, R.; Davis, M.D.; Magli, Y.L.; Segal, P.; Klein, B.E.; Hubbard, L. The Wisconsin age-related maculopathy grading system. Ophthalmology 1991, 98, 1128–1134. [Google Scholar] [CrossRef]
- Klein, R.; Meuer, S.M.; Myers, C.E.; Buitendijk, G.H.; Rochtchina, E.; Choudhury, F.; de Jong, P.T.; McKean-Cowdin, R.; Iyengar, S.K.; Gao, X.; et al. Harmonizing the classification of age-related macular degeneration in the three-continent AMD consortium. Ophthalmic Epidemiol. 2014, 21, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Age-Related Eye Disease Study Group; Davis, M.D.; Gangnon, R.E.; Lee, L.Y.; Hubbard, L.D.; Klein, B.E.; Klein, R.; Ferris, F.L.; Bressler, S.B.; Milton, R.C. The Age-Related Eye Disease Study severity scale for age-related macular degeneration: AREDS report No. 17. Arch. Ophthalmol. 2005, 123, 1484–1498. [Google Scholar] [CrossRef] [Green Version]
- Age-Related Eye Disease Study Research Group. Risk factors associated with age-related macular degeneration. A case-control study in the age-related eye disease study: Age-related eye disease study report number 3. Ophthalmology 2000, 107, 2224–2232. [Google Scholar] [CrossRef]
- Age-Related Eye Disease Study Research Group; Ferris, F.L.; Davis, M.D.; Clemons, T.E.; Lee, L.Y.; Chew, E.Y.; Lindblad, A.S.; Milton, R.C.; Bressler, S.B.; Klein, R. A simplified severity scale for age-related macular degeneration: AREDS report no. 18. Arch. Ophthalmol. 2005, 123, 1570–1574. [Google Scholar] [CrossRef] [PubMed]
- Liew, G.; Joachim, N.; Mitchell, P.; Burlutsky, G.; Wang, J.J. Validating the AREDS simplified severity scale of age-related macular degeneration with 5- and 10-year incident data in a population-based sample. Ophthalmology 2016, 123, 1874–1878. [Google Scholar] [CrossRef] [PubMed]
- Ferris, F.L., 3rd; Wilkinson, C.P.; Bird, A.; Chakravarthy, U.; Chew, E.; Csaky, K.; Sadda, S.R.; Beckman Initiative for Macular Research Classification Committee. Clinical classification of age-related macular degeneration. Ophthalmology 2013, 120, 844–851. [Google Scholar] [CrossRef]
- Sarks, S.; Cherepanoff, S.; Killingsworth, M.; Sarks, J. Relationship of Basal laminar deposit and membranous debris to the clinical presentation of early age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 2007, 48, 968–977. [Google Scholar] [CrossRef]
- Garrity, S.T.; Sarraf, D.; Freund, K.B.; Sadda, S.R. Multimodal imaging of nonneovascular age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 2018, 59, AMD48–AMD64. [Google Scholar] [CrossRef] [Green Version]
- Huisingh, C.; McGwin, G., Jr.; Neely, D.; Zarubina, A.; Clark, M.; Zhang, Y.; Curcio, C.A.; Owsley, C. The association between subretinal drusenoid deposits in older adults in normal macular health and incident age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 2016, 57, 739–745. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spaide, R.F.; Ooto, S.; Curcio, C.A. Subretinal drusenoid deposits AKA pseudodrusen. Surv. Ophthalmol. 2018, 63, 782–815. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Clark, M.E.; Crossman, D.K.; Kojima, K.; Messinger, J.D.; Mobley, J.A.; Curcio, C.A. Abundant lipid and protein components of drusen. PLoS ONE 2010, 5, e10329. [Google Scholar] [CrossRef]
- Wightman, A.J.; Guymer, R.H. Reticular pseudodrusen: Current understanding. Clin. Exp. Optom. 2019, 102, 455–462. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Owsley, C.; McGwin, G., Jr.; Clark, M.E.; Jackson, G.R.; Callahan, M.A.; Kline, L.B.; Witherspoon, C.D.; Curcio, C.A. Delayed rod-mediated dark adaptation is a functional biomarker for incident early age-related macular degeneration. Ophthalmology 2016, 123, 344–351. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dimitrov, P.N.; Guymer, R.H.; Zele, A.J.; Anderson, A.J.; Vingrys, A.J. Measuring rod and cone dynamics in age-related maculopathy. Investig. Ophthalmol. Vis. Sci. 2008, 49, 55–65. [Google Scholar] [CrossRef]
- Owsley, C.; Huisingh, C.; Clark, M.E.; Jackson, G.R.; McGwin, G., Jr. Comparison of visual function in older eyes in the earliest stages of age-related macular degeneration to those in normal macular health. Curr. Eye Res. 2016, 41, 266–272. [Google Scholar] [CrossRef] [Green Version]
- Owsley, C.; Jackson, G.R.; White, M.; Feist, R.; Edwards, D. Delays in rod-mediated dark adaptation in early age-related maculopathy. Ophthalmology 2001, 108, 1196–1202. [Google Scholar] [CrossRef]
- Owsley, C.; McGwin, G., Jr.; Jackson, G.R.; Kallies, K.; Clark, M. Cone- and rod-mediated dark adaptation impairment in age-related maculopathy. Ophthalmology 2007, 114, 1728–1735. [Google Scholar] [CrossRef]
- Steinmetz, R.L.; Haimovici, R.; Jubb, C.; Fitzke, F.W.; Bird, A.C. Symptomatic abnormalities of dark adaptation in patients with age-related Bruch’s membrane change. Br. J. Ophthalmol. 1993, 77, 549–554. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhutto, I.; Lutty, G. Understanding age-related macular degeneration (AMD): Relationships between the photoreceptor/retinal pigment epithelium/Bruch’s membrane/choriocapillaris complex. Mol. Aspects Med. 2012, 33, 295–317. [Google Scholar] [CrossRef] [Green Version]
- Freund, K.B.; Ho, I.V.; Barbazetto, I.A.; Koizumi, H.; Laud, K.; Ferrara, D.; Matsumoto, Y.; Sorenson, J.A.; Yannuzzi, L. Type 3 neovascularization: The expanded spectrum of retinal angiomatous proliferation. Retina 2008, 28, 201–211. [Google Scholar] [CrossRef]
- Kim, J.H.; Kim, J.W.; Kim, C.G.; Lee, D.W. Long-term treatment outcomes in type 3 neovascularization: Focus on the difference in outcomes between geographic atrophy and fibrotic scarring. J. Clin. Med. 2020, 9, 1145. [Google Scholar] [CrossRef]
- Naysan, J.; Jung, J.J.; Dansingani, K.K.; Balaratnasingam, C.; Freund, K.B. Type 2 (subretinal) neovascularization in age-related macular degeneration associated with pure reticular pseudodrusen phenotype. Retina 2016, 36, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Seddon, J.M.; McLeod, D.S.; Bhutto, I.A.; Villalonga, M.B.; Silver, R.E.; Wenick, A.S.; Edwards, M.M.; Lutty, G.A. Histopathological insights into choroidal vascular loss in clinically documented cases of age-related macular degeneration. JAMA Ophthalmol. 2016, 134, 1272–1280. [Google Scholar] [CrossRef]
- Yeo, N.J.Y.; Chan, E.J.J.; Cheung, C. Choroidal neovascularization: Mechanisms of endothelial dysfunction. Front. Pharmacol. 2019, 10, 1363. [Google Scholar] [CrossRef] [Green Version]
- Biesemeier, A.; Taubitz, T.; Julien, S.; Yoeruek, E.; Schraermeyer, U. Choriocapillaris breakdown precedes retinal degeneration in age-related macular degeneration. Neurobiol. Aging 2014, 35, 2562–2573. [Google Scholar] [CrossRef] [Green Version]
- Yannuzzi, L.A.; Negrao, S.; Iida, T.; Carvalho, C.; Rodriguez-Coleman, H.; Slakter, J.; Freund, K.B.; Sorenson, J.; Orlock, D.; Borodoker, N. Retinal angiomatous proliferation in age-related macular degeneration. Retina 2001, 21, 416–434. [Google Scholar] [CrossRef]
- Spaide, R.F.; Jaffe, G.J.; Sarraf, D.; Freund, K.B.; Sadda, S.R.; Staurenghi, G.; Waheed, N.K.; Chakravarthy, U.; Rosenfeld, P.J.; Holz, F.G.; et al. Consensus nomenclature for reporting neovascular age-related macular degeneration data: Consensus on neovascular age-related macular degeneration nomenclature study group. Ophthalmology 2020, 127, 616–636. [Google Scholar] [CrossRef]
- McLeod, D.S.; Grebe, R.; Bhutto, I.; Merges, C.; Baba, T.; Lutty, G.A. Relationship between RPE and choriocapillaris in age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 2009, 50, 4982–4991. [Google Scholar] [CrossRef] [Green Version]
- Sparrow, J.R.; Boulton, M. RPE lipofuscin and its role in retinal pathobiology. Exp. Eye Res. 2005, 80, 595–606. [Google Scholar] [CrossRef]
- Curcio, C.A.; Medeiros, N.E.; Millican, C.L. Photoreceptor loss in age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 1996, 37, 1236–1249. [Google Scholar]
- Pilotto, E.; Benetti, E.; Convento, E.; Guidolin, F.; Longhin, E.; Parrozzani, R.; Midena, E. Microperimetry, fundus autofluorescence, and retinal layer changes in progressing geographic atrophy. Can. J. Ophthalmol. 2013, 48, 386–393. [Google Scholar] [CrossRef] [PubMed]
- Rudolf, M.; Vogt, S.D.; Curcio, C.A.; Huisingh, C.; McGwin, G., Jr.; Wagner, A.; Grisanti, S.; Read, R.W. Histologic basis of variations in retinal pigment epithelium autofluorescence in eyes with geographic atrophy. Ophthalmology 2013, 120, 821–828. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yung, M.; Klufas, M.A.; Sarraf, D. Clinical applications of fundus autofluorescence in retinal disease. Int. J. Retina Vitr. 2016, 2, 12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bernstein, P.S.; Delori, F.C.; Richer, S.; van Kuijk, F.J.; Wenzel, A.J. The value of measurement of macular carotenoid pigment optical densities and distributions in age-related macular degeneration and other retinal disorders. Vis. Res. 2010, 50, 716–728. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gruszecki, W.I.; Sielewiesiuk, J. Orientation of xanthophylls in phosphatidylcholine multibilayers. Biochim. Biophys. Acta 1990, 1023, 405–412. [Google Scholar] [CrossRef]
- Li, B.; George, E.W.; Rognon, G.T.; Gorusupudi, A.; Ranganathan, A.; Chang, F.Y.; Shi, L.; Frederick, J.M.; Bernstein, P.S. Imaging lutein and zeaxanthin in the human retina with confocal resonance Raman microscopy. Proc. Natl. Acad. Sci. USA 2020, 117, 12352–12358. [Google Scholar] [CrossRef]
- Leung, I.Y. Macular pigment: New clinical methods of detection and the role of carotenoids in age-related macular degeneration. Optometry 2008, 79, 266–272. [Google Scholar] [CrossRef] [PubMed]
- De Kinkelder, R.; van der Veen, R.L.; Verbaak, F.D.; Faber, D.J.; van Leeuwen, T.G.; Berendschot, T.T. Macular pigment optical density measurements: Evaluation of a device using heterochromatic flicker photometry. Eye 2011, 25, 105–112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bone, R.A.; Landrum, J.T.; Hime, G.W.; Cains, A.; Zamor, J. Stereochemistry of the human macular carotenoids. Investig. Ophthalmol. Vis. Sci. 1993, 34, 2033–2040. [Google Scholar]
- Scripsema, N.K.; Hu, D.N.; Rosen, R.B. Lutein, zeaxanthin, and meso-zeaxanthin in the clinical management of eye disease. J. Ophthalmol. 2015, 2015, 865179. [Google Scholar] [CrossRef] [Green Version]
- Howells, O.; Eperjesi, F.; Bartlett, H. Measuring macular pigment optical density in vivo: A review of techniques. Graefes Arch. Clin. Exp. Ophthalmol. 2011, 249, 315–347. [Google Scholar] [CrossRef] [Green Version]
- Li, L.H.; Lee, J.C.; Leung, H.H.; Lam, W.C.; Fu, Z.; Lo, A.C.Y. Lutein supplementation for eye diseases. Nutrients 2020, 12, 1721. [Google Scholar] [CrossRef] [PubMed]
- Gorusupudi, A.; Shyam, R.; Li, B.; Vachali, P.; Subhani, Y.K.; Nelson, K.; Bernstein, P.S. Developmentally regulated production of meso-zeaxanthin in chicken retinal pigment epithelium/choroid and retina. Investig. Ophthalmol. Vis. Sci. 2016, 57, 1853–1861. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shyam, R.; Gorusupudi, A.; Nelson, K.; Horvath, M.P.; Bernstein, P.S. RPE65 has an additional function as the lutein to meso-zeaxanthin isomerase in the vertebrate eye. Proc. Natl. Acad. Sci. USA 2017, 114, 10882–10887. [Google Scholar] [CrossRef] [Green Version]
- Bone, R.A.; Landrum, J.T.; Mayne, S.T.; Gomez, C.M.; Tibor, S.E.; Twaroska, E.E. Macular pigment in donor eyes with and without AMD: A case-control study. Investig. Ophthalmol. Vis. Sci. 2001, 42, 235–240. [Google Scholar]
- Howells, O.; Eperjesi, F.; Bartlett, H. Improving the repeatability of heterochromatic flicker photometry for measurement of macular pigment optical density. Graefes Arch. Clin. Exp. Ophthalmol. 2013, 251, 871–880. [Google Scholar] [CrossRef]
- Zhou, H.; Zhao, X.; Johnson, E.J.; Lim, A.; Sun, E.; Yu, J.; Zhang, Y.; Liu, X.; Snellingen, T.; Shang, F.; et al. Serum carotenoids and risk of age-related macular degeneration in a chinese population sample. Investig. Ophthalmol. Vis. Sci. 2011, 52, 4338–4344. [Google Scholar] [CrossRef]
- Nolan, J.M.; Stack, J.; O’Donovan, O.; Loane, E.; Beatty, S. Risk factors for age-related maculopathy are associated with a relative lack of macular pigment. Exp. Eye Res. 2007, 84, 61–74. [Google Scholar] [CrossRef]
- Landrum, J.T.; Bone, R.A.; Joa, H.; Kilburn, M.D.; Moore, L.L.; Sprague, K.E. A one year study of the macular pigment: The effect of 140 days of a lutein supplement. Exp. Eye Res. 1997, 65, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Bone, R.A.; Landrum, J.T.; Cao, Y.; Howard, A.N.; Alvarez-Calderon, F. Macular pigment response to a supplement containing meso-zeaxanthin, lutein and zeaxanthin. Nutr. Metab. 2007, 4, 12. [Google Scholar] [CrossRef] [Green Version]
- Akuffo, K.O.; Beatty, S.; Stack, J.; Dennison, J.; O’Regan, S.; Meagher, K.A.; Peto, T.; Nolan, J. Central retinal enrichment supplementation trials (CREST): Design and methodology of the CREST randomized controlled trials. Ophthalmic Epidemiol. 2014, 21, 111–123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berendschot, T.T.; Goldbohm, R.A.; Klopping, W.A.; van de Kraats, J.; van Norel, J.; van Norren, D. Influence of lutein supplementation on macular pigment, assessed with two objective techniques. Investig. Ophthalmol. Vis. Sci. 2000, 41, 3322–3326. [Google Scholar]
- Huang, Y.M.; Dou, H.L.; Huang, F.F.; Xu, X.R.; Zou, Z.Y.; Lin, X.M. Effect of supplemental lutein and zeaxanthin on serum, macular pigmentation, and visual performance in patients with early age-related macular degeneration. Biomed. Res. Int. 2015, 2015, 564738. [Google Scholar] [CrossRef]
- Koh, H.H.; Murray, I.J.; Nolan, D.; Carden, D.; Feather, J.; Beatty, S. Plasma and macular responses to lutein supplement in subjects with and without age-related maculopathy: A pilot study. Exp. Eye Res. 2004, 79, 21–27. [Google Scholar] [CrossRef]
- Ma, L.; Yan, S.F.; Huang, Y.M.; Lu, X.R.; Qian, F.; Pang, H.L.; Xu, X.R.; Zou, Z.Y.; Dong, P.C.; Xiao, X.; et al. Effect of lutein and zeaxanthin on macular pigment and visual function in patients with early age-related macular degeneration. Ophthalmology 2012, 119, 2290–2297. [Google Scholar] [CrossRef]
- Richer, S.; Devenport, J.; Lang, J.C. LAST II: Differential temporal responses of macular pigment optical density in patients with atrophic age-related macular degeneration to dietary supplementation with xanthophylls. Optometry 2007, 78, 213–219. [Google Scholar] [CrossRef]
- Trieschmann, M.; Beatty, S.; Nolan, J.M.; Hense, H.W.; Heimes, B.; Austermann, U.; Fobker, M.; Pauleikhoff, D. Changes in macular pigment optical density and serum concentrations of its constituent carotenoids following supplemental lutein and zeaxanthin: The LUNA study. Exp. Eye Res. 2007, 84, 718–728. [Google Scholar] [CrossRef] [PubMed]
- Weigert, G.; Kaya, S.; Pemp, B.; Sacu, S.; Lasta, M.; Werkmeister, R.M.; Dragostinoff, N.; Simader, C.; Garhofer, G.; Schmidt-Erfurth, U.; et al. Effects of lutein supplementation on macular pigment optical density and visual acuity in patients with age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 2011, 52, 8174–8178. [Google Scholar] [CrossRef] [PubMed]
- Khachik, F.; Bernstein, P.S.; Garland, D.L. Identification of lutein and zeaxanthin oxidation products in human and monkey retinas. Investig. Ophthalmol. Vis. Sci. 1997, 38, 1802–1811. [Google Scholar]
- Bone, R.A.; Davey, P.G.; Roman, B.O.; Evans, D.W. Efficacy of commercially available nutritional supplements: Analysis of Serum uptake, macular pigment optical density and visual functional response. Nutrients 2020, 12, 1321. [Google Scholar] [CrossRef]
- Kijlstra, A.; Tian, Y.; Kelly, E.R.; Berendschot, T.T. Lutein: More than just a filter for blue light. Prog. Retin. Eye Res. 2012, 31, 303–315. [Google Scholar] [CrossRef]
- Junghans, A.; Sies, H.; Stahl, W. Macular pigments lutein and zeaxanthin as blue light filters studied in liposomes. Arch. Biochem. Biophys. 2001, 391, 160–164. [Google Scholar] [CrossRef] [PubMed]
- Krinsky, N.I.; Johnson, E.J. Carotenoid actions and their relation to health and disease. Mol. Aspects Med. 2005, 26, 459–516. [Google Scholar] [CrossRef]
- Landrum, J.T.; Bone, R.A. Mechanistic Evidence for Eye Disease and Carotenoids; Krinsky, N.I., Mayne, S.T., Sies, H., Eds.; CRC Press: New York, NY, USA, 2004. [Google Scholar]
- Li, S.Y.; Fu, Z.J.; Lo, A.C. Hypoxia-induced oxidative stress in ischemic retinopathy. Oxid. Med. Cell. Longev. 2012, 2012, 426769. [Google Scholar] [CrossRef] [Green Version]
- Suarez-Berumen, K.; Davey, P.G. Macular pigments optical density: A Review of techniques of measurements and factors influencing their levels. JSM Ophthalmol. 2014, 3, 4. [Google Scholar]
- Bartlett, H.; Eperjesi, F. Apparent motion photometry: Evaluation and reliability of a novel method for the measurement of macular pigment. Br. J. Ophthalmol. 2011, 95, 662–665. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davey, P.G.; Alvarez, S.D.; Lee, J.Y. Macular pigment optical density: Repeatability, intereye correlation, and effect of ocular dominance. Clin. Ophthalmol. 2016, 10, 1671–1678. [Google Scholar] [CrossRef] [Green Version]
- Bartlett, H.; Howells, O.; Eperjesi, F. The role of macular pigment assessment in clinical practice: A review. Clin. Exp. Optom. 2010, 93, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Delori, F.C. Autofluorescence method to measure macular pigment optical densities fluorometry and autofluorescence imaging. Arch. Biochem. Biophys. 2004, 430, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, H.; Eperjesi, F. Use of fundus imaging in quantification of age-related macular change. Surv. Ophthalmol. 2007, 52, 655–671. [Google Scholar] [CrossRef]
- Delori, F.C.; Goger, D.G.; Hammond, B.R.; Snodderly, D.M.; Burns, S.A. Macular pigment density measured by autofluorescence spectrometry: Comparison with reflectometry and heterochromatic flicker photometry. J. Opt. Soc. Am. A Opt. Image Sci. Vis. 2001, 18, 1212–1230. [Google Scholar] [CrossRef] [Green Version]
- Dennison, J.L.; Stack, J.; Beatty, S.; Nolan, J.M. Concordance of macular pigment measurements obtained using customized heterochromatic flicker photometry, dual-wavelength autofluorescence, and single-wavelength reflectance. Exp. Eye Res. 2013, 116, 190–198. [Google Scholar] [CrossRef] [PubMed]
- Lapierre-Landry, M.; Carroll, J.; Skala, M.C. Imaging retinal melanin: A review of current technologies. J. Biol. Eng. 2018, 12, 29. [Google Scholar] [CrossRef] [PubMed]
- Bone, R.A.; Mukherjee, A. Innovative Troxler-free measurement of macular pigment and lens density with correction of the former for the aging lens. J. Biomed. Opt. 2013, 18, 107003. [Google Scholar] [CrossRef]
- Lee, B.B.; Martin, P.R.; Valberg, A. The physiological basis of heterochromatic flicker photometry demonstrated in the ganglion cells of the macaque retina. J. Physiol. 1988, 404, 323–347. [Google Scholar] [CrossRef]
- Obana, A.; Gohto, Y.; Moriyama, T.; Seto, T.; Sasano, H.; Okazaki, S. Reliability of a commercially available heterochromatic flicker photometer, the MPS2, for measuring the macular pigment optical density of a Japanese population. Jpn. J. Ophthalmol. 2018, 62, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Berendschot, T.T.; van Norren, D. Objective determination of the macular pigment optical density using fundus reflectance spectroscopy. Arch. Biochem. Biophys. 2004, 430, 149–155. [Google Scholar] [CrossRef]
- Davey, P.G.; Ngo, A.; Cross, J.; Gierhart, D.L. Macular pigment reflectometry: Development and evaluation of a novel clinical device for rapid objective assessment of the macular carotenoids. In Proceedings of the Ophthalmic Technologies XXIX, San Francisco, CA, USA, 2–3 February 2019; Manns, F., Soderberg, P.G., Ho, A., Eds.; SPIE—International Society for Optics and Photonics: Bellingham, WA, USA, 2019; Volume 10858. [Google Scholar]
- Huang, H.; Guan, C.; Ng, D.S.; Liu, X.; Chen, H. Macular Pigment optical density measured by a single wavelength reflection photometry with and without mydriasis. Curr. Eye Res. 2019, 44, 324–328. [Google Scholar] [CrossRef] [PubMed]
- Kilbride, P.E.; Alexander, K.R.; Fishman, M.; Fishman, G.A. Human macular pigment assessed by imaging fundus reflectometry. Vis. Res. 1989, 29, 663–674. [Google Scholar] [CrossRef]
- Sanabria, J.C.; Bass, J.; Spors, F.; Gierhart, D.L.; Davey, P.G. Measurement of carotenoids in perifovea using the macular pigment reflectometer. J. Vis. Exp. 2020, 9. [Google Scholar] [CrossRef] [Green Version]
- You, Q.S.; Bartsch, D.U.; Espina, M.; Alam, M.; Camacho, N.; Mendoza, N.; Freeman, W.R. Reproducibility of macular pigment optical density measurement by two-wavelength autofluorescence in a clinical setting. Retina 2016, 36, 1381–1387. [Google Scholar] [CrossRef] [Green Version]
- Bernstein, P.S.; Zhao, D.Y.; Sharifzadeh, M.; Ermakov, I.V.; Gellermann, W. Resonance Raman measurement of macular carotenoids in the living human eye. Arch. Biochem. Biophys. 2004, 430, 163–169. [Google Scholar] [CrossRef]
- Gellermann, W.; Ermakov, I.V.; Ermakova, M.R.; McClane, R.W.; Zhao, D.Y.; Bernstein, P.S. In vivo resonant Raman measurement of macular carotenoid pigments in the young and the aging human retina. J. Opt. Soc. Am. A Opt. Image Sci. Vis. 2002, 19, 1172–1186. [Google Scholar] [CrossRef]
- Obana, A.; Gohto, Y.; Tanito, M.; Okazaki, S.; Gellermann, W.; Bernstein, P.S.; Ohira, A. Effect of age and other factors on macular pigment optical density measured with resonance Raman spectroscopy. Graefes Arch. Clin. Exp. Ophthalmol. 2014, 252, 1221–1228. [Google Scholar] [CrossRef] [PubMed]
- Obana, A.; Tanito, M.; Gohto, Y.; Gellermann, W.; Okazaki, S.; Ohira, A. Macular pigment changes in pseudophakic eyes quantified with resonance Raman spectroscopy. Ophthalmology 2011, 118, 1852–1858. [Google Scholar] [CrossRef]
- Christaras, D.; Ginis, H.; Pennos, A.; Mompean, J.; Artal, P. Objective method for measuring the macular pigment optical density in the eye. Biomed. Opt. Express 2019, 10, 3572–3583. [Google Scholar] [CrossRef] [PubMed]
- Fasler, K.; Fu, D.J.; Moraes, G.; Wagner, S.; Gokhale, E.; Kortuem, K.; Chopra, R.; Faes, L.; Preston, G.; Pontikos, N.; et al. Moorfields AMD database report 2: Fellow eye involvement with neovascular age-related macular degeneration. Br. J. Ophthalmol. 2020, 104, 684–690. [Google Scholar] [CrossRef] [PubMed]
- Rozing, M.P.; Durhuus, J.A.; Nielsen, M.K.; Subhi, Y.; Kirkwood, T.B.; Westendorp, R.G.; Sorensen, T.L. Age-related macular degeneration: A two-level model hypothesis. Prog. Retin. Eye Res. 2020, 76, 100825. [Google Scholar] [CrossRef]
- Sene, A.; Chin-Yee, D.; Apte, R.S. Seeing through VEGF: Innate and adaptive immunity in pathological angiogenesis in the eye. Trends Mol. Med. 2015, 21, 43–51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Califf, R.M. Biomarker definitions and their applications. Exp. Biol. Med. 2018, 243, 213–221. [Google Scholar] [CrossRef]
- FDA-NIH Biomarker Working Group. BEST (Biomarkers, EndpointS, and Other Tools) Resource; Food and Drug Administration: Silver Spring, MD, USA, 2016. [Google Scholar]
- Wickstrom, K.; Moseley, J. Biomarkers and surrogate endpoints in drug development: A European regulatory view. Investig. Ophthalmol. Vis. Sci. 2017, 58, BIO27–BIO33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robb, M.A.; McInnes, P.M.; Califf, R.M. Biomarkers and surrogate endpoints: Developing common terminology and definitions. JAMA 2016, 315, 1107–1108. [Google Scholar] [CrossRef]
- Obana, A.; Hiramitsu, T.; Gohto, Y.; Ohira, A.; Mizuno, S.; Hirano, T.; Bernstein, P.S.; Fujii, H.; Iseki, K.; Tanito, M.; et al. Macular carotenoid levels of normal subjects and age-related maculopathy patients in a Japanese population. Ophthalmology 2008, 115, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Beatty, S.; Murray, I.J.; Henson, D.B.; Carden, D.; Koh, H.; Boulton, M.E. Macular pigment and risk for age-related macular degeneration in subjects from a Northern European population. Investig. Ophthalmol. Vis. Sci. 2001, 42, 439–446. [Google Scholar]
- Bernstein, P.S.; Zhao, D.Y.; Wintch, S.W.; Ermakov, I.V.; McClane, R.W.; Gellermann, W. Resonance Raman measurement of macular carotenoids in normal subjects and in age-related macular degeneration patients. Ophthalmology 2002, 109, 1780–1787. [Google Scholar] [CrossRef] [Green Version]
- Dietzel, M.; Zeimer, M.; Heimes, B.; Claes, B.; Pauleikhoff, D.; Hense, H.W. Determinants of macular pigment optical density and its relation to age-related maculopathy: Results from the Muenster Aging and Retina Study (MARS). Investig. Ophthalmol. Vis. Sci. 2011, 52, 3452–3457. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- LaRowe, T.L.; Mares, J.A.; Snodderly, D.M.; Klein, M.L.; Wooten, B.R.; Chappell, R.; Group, C.M.P.S. Macular pigment density and age-related maculopathy in the carotenoids in age-related eye disease study. An ancillary study of the women’s health initiative. Ophthalmology 2008, 115, 876–883. [Google Scholar] [CrossRef]
- Mares, J.A.; LaRowe, T.L.; Snodderly, D.M.; Moeller, S.M.; Gruber, M.J.; Klein, M.L.; Wooten, B.R.; Johnson, E.J.; Chappell, R.J.; Group, C.M.P.S.; et al. Predictors of optical density of lutein and zeaxanthin in retinas of older women in the Carotenoids in Age-Related Eye Disease Study, an ancillary study of the Women’s Health Initiative. Am. J. Clin. Nutr. 2006, 84, 1107–1122. [Google Scholar] [CrossRef] [Green Version]
- Raman, R.; Biswas, S.; Gupta, A.; Kulothungan, V.; Sharma, T. Association of macular pigment optical density with risk factors for wet age-related macular degeneration in the Indian population. Eye 2012, 26, 950–957. [Google Scholar] [CrossRef] [Green Version]
- American Optometric Association. Macular pigment and healthy vision. Optometry 2009, 80, 592–598. [Google Scholar] [CrossRef]
- Hammond, B.R., Jr.; Curran-Celentano, J.; Judd, S.; Fuld, K.; Krinsky, N.I.; Wooten, B.R.; Snodderly, D.M. Sex differences in macular pigment optical density: Relation to plasma carotenoid concentrations and dietary patterns. Vis. Res. 1996, 36, 2001–2012. [Google Scholar] [CrossRef] [Green Version]
- Hammond, B.R., Jr.; Fuld, K.; Snodderly, D.M. Iris color and macular pigment optical density. Exp. Eye Res. 1996, 62, 293–297. [Google Scholar] [CrossRef]
- Ozawa, Y.; Shigeno, Y.; Nagai, N.; Suzuki, M.; Kurihara, T.; Minami, S.; Hirano, E.; Shinoda, H.; Kobayashi, S.; Tsubota, K. Absolute and estimated values of macular pigment optical density in young and aged Asian participants with or without age-related macular degeneration. BMC Ophthalmol. 2017, 17, 161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagai, N.; Asato, T.; Minami, S.; Suzuki, M.; Shinoda, H.; Kurihara, T.; Sonobe, H.; Watanabe, K.; Uchida, A.; Ban, N.; et al. Correlation between macular pigment optical density and neural thickness and volume of the retina. Nutrients 2020, 12, 888. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brandl, C.; Brucklmayer, C.; Gunther, F.; Zimmermann, M.E.; Kuchenhoff, H.; Helbig, H.; Weber, B.H.F.; Heid, I.M.; Stark, K.J. Retinal layer thicknesses in early age-related macular degeneration: Results from the German AugUR study. Investig. Ophthalmol. Vis. Sci. 2019, 60, 1581–1594. [Google Scholar] [CrossRef] [Green Version]
- Lamin, A.; Oakley, J.D.; Dubis, A.M.; Russakoff, D.B.; Sivaprasad, S. Changes in volume of various retinal layers over time in early and intermediate age-related macular degeneration. Eye 2019, 33, 428–434. [Google Scholar] [CrossRef] [Green Version]
- Ferrara, D.; Silver, R.E.; Louzada, R.N.; Novais, E.A.; Collins, G.K.; Seddon, J.M. Optical Coherence tomography features preceding the onset of advanced age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 2017, 58, 3519–3529. [Google Scholar] [CrossRef]
- Curcio, C.A.; Messinger, J.D.; Sloan, K.R.; Mitra, A.; McGwin, G.; Spaide, R.F. Human chorioretinal layer thicknesses measured in macula-wide, high-resolution histologic sections. Investig. Ophthalmol. Vis. Sci. 2011, 52, 3943–3954. [Google Scholar] [CrossRef]
- Jackson, G.R.; Owsley, C.; Curcio, C.A. Photoreceptor degeneration and dysfunction in aging and age-related maculopathy. Ageing Res. Rev. 2002, 1, 381–396. [Google Scholar] [CrossRef]
- Medeiros, N.E.; Curcio, C.A. Preservation of ganglion cell layer neurons in age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 2001, 42, 795–803. [Google Scholar]
- Lee, E.K.; Yu, H.G. Ganglion cell-inner plexiform layer and peripapillary retinal nerve fiber layer thicknesses in age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 2015, 56, 3976–3983. [Google Scholar] [CrossRef]
- Muftuoglu, I.K.; Ramkumar, H.L.; Bartsch, D.U.; Meshi, A.; Gaber, R.; Freeman, W.R. Quantitative analysis of the inner retinal layer thicknesses in age-related macular degeneration using corrected optical coherence tomography segmentation. Retina 2018, 38, 1478–1484. [Google Scholar] [CrossRef]
- Savastano, M.C.; Minnella, A.M.; Tamburrino, A.; Giovinco, G.; Ventre, S.; Falsini, B. Differential vulnerability of retinal layers to early age-related macular degeneration: Evidence by SD-OCT segmentation analysis. Investig. Ophthalmol. Vis. Sci. 2014, 55, 560–566. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yenice, E.; Sengun, A.; Demirok, G.S.; Turacli, E. Ganglion cell complex thickness in nonexudative age-related macular degeneration. Eye 2015, 29, 1076–1080. [Google Scholar] [CrossRef] [Green Version]
- Zucchiatti, I.; Parodi, M.B.; Pierro, L.; Cicinelli, M.V.; Gagliardi, M.; Castellino, N.; Bandello, F. Macular ganglion cell complex and retinal nerve fiber layer comparison in different stages of age-related macular degeneration. Am. J. Ophthalmol. 2015, 160, 602–607. [Google Scholar] [CrossRef] [PubMed]
- Age-Related Eye Disease Study Research Group. A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E, beta carotene, and zinc for age-related macular degeneration and vision loss: AREDS report no. 8. Arch. Ophthalmol. 2001, 119, 1417–1436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amissah-Arthur, K.N.; Panneerselvam, S.; Narendran, N.; Yang, Y.C. Optical coherence tomography changes before the development of choroidal neovascularization in second eyes of patients with bilateral wet macular degeneration. Eye 2012, 26, 394–399. [Google Scholar] [CrossRef] [Green Version]
- Nagai, N.; Minami, S.; Suzuki, M.; Shinoda, H.; Kurihara, T.; Sonobe, H.; Watanabe, K.; Uchida, A.; Ban, N.; Tsubota, K.; et al. Macular pigment optical density and photoreceptor outer segment length as predisease biomarkers for age-related macular degeneration. J. Clin. Med. 2020, 9, 1347. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. J. Clin. Epidemiol. 2009, 62, 1006–1012. [Google Scholar] [CrossRef]
- Higgins, J.P.; Altman, D.G.; Gotzsche, P.C.; Juni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.; et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef] [Green Version]
- Arslan, S.; Kadayifcilar, S.; Samur, G. The potential role of dietary antioxidant capacity in preventing age-related macular degeneration. J. Am. Coll. Nutr. 2019, 38, 424–432. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.; Hankinson, S.E.; Rosner, B.; Willett, W.C.; Colditz, G.A. Prospective study of lutein/zeaxanthin intake and risk of age-related macular degeneration. Am. J. Clin. Nutr. 2008, 87, 1837–1843. [Google Scholar] [CrossRef]
- Cho, E.; Seddon, J.M.; Rosner, B.; Willett, W.C.; Hankinson, S.E. Prospective study of intake of fruits, vegetables, vitamins, and carotenoids and risk of age-related maculopathy. Arch. Ophthalmol. 2004, 122, 883–892. [Google Scholar] [CrossRef] [Green Version]
- Dasch, B.; Fuhs, A.; Schmidt, J.; Behrens, T.; Meister, A.; Wellmann, J.; Fobker, M.; Pauleikhoff, D.; Hense, H.W. Serum levels of macular carotenoids in relation to age-related maculopathy: The Muenster Aging and Retina Study (MARS). Graefes Arch. Clin. Exp. Ophthalmol. 2005, 243, 1028–1035. [Google Scholar] [CrossRef] [PubMed]
- Delcourt, C.; Carriere, I.; Delage, M.; Barberger-Gateau, P.; Schalch, W.; The POLA Study Group. Plasma lutein and zeaxanthin and other carotenoids as modifiable risk factors for age-related maculopathy and cataract: The POLA Study. Investig. Ophthalmol. Vis. Sci. 2006, 47, 2329–2335. [Google Scholar] [CrossRef] [PubMed]
- Eye Disease Case-Control Study Group. Antioxidant status and neovascular age-related macular degeneration. Eye Disease Case-Control Study Group. Arch. Ophthalmol. 1993, 111, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Gale, C.R.; Hall, N.F.; Phillips, D.I.; Martyn, C.N. Lutein and zeaxanthin status and risk of age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 2003, 44, 2461–2465. [Google Scholar] [CrossRef] [Green Version]
- Mares-Perlman, J.A.; Brady, W.E.; Klein, R.; Klein, B.E.; Bowen, P.; Stacewicz-Sapuntzakis, M.; Palta, M. Serum antioxidants and age-related macular degeneration in a population-based case-control study. Arch. Ophthalmol. 1995, 113, 1518–1523. [Google Scholar] [CrossRef] [PubMed]
- Mares-Perlman, J.A.; Fisher, A.I.; Klein, R.; Palta, M.; Block, G.; Millen, A.E.; Wright, J.D. Lutein and zeaxanthin in the diet and serum and their relation to age-related maculopathy in the third national health and nutrition examination survey. Am. J. Epidemiol. 2001, 153, 424–432. [Google Scholar] [CrossRef] [Green Version]
- Michikawa, T.; Ishida, S.; Nishiwaki, Y.; Kikuchi, Y.; Tsuboi, T.; Hosoda, K.; Ishigami, A.; Iwasawa, S.; Nakano, M.; Takebayashi, T. Serum antioxidants and age-related macular degeneration among older Japanese. Asia Pac. J. Clin. Nutr. 2009, 18, 1–7. [Google Scholar]
- Moeller, S.M.; Parekh, N.; Tinker, L.; Ritenbaugh, C.; Blodi, B.; Wallace, R.B.; Mares, J.A.; CAREDS Research Study Group. Associations between intermediate age-related macular degeneration and lutein and zeaxanthin in the Carotenoids in Age-related Eye Disease Study (CAREDS): Ancillary study of the Women’s Health Initiative. Arch. Ophthalmol. 2006, 124, 1151–1162. [Google Scholar] [CrossRef]
- Seddon, J.M.; Ajani, U.A.; Sperduto, R.D.; Hiller, R.; Blair, N.; Burton, T.C.; Farber, M.D.; Gragoudas, E.S.; Haller, J.; Miller, D.T.; et al. Dietary carotenoids, vitamins A, C, and E, and advanced age-related macular degeneration. Eye Disease Case-Control Study Group. JAMA 1994, 272, 1413–1420. [Google Scholar] [CrossRef]
- Snellen, E.L.; Verbeek, A.L.; Van Den Hoogen, G.W.; Cruysberg, J.R.; Hoyng, C.B. Neovascular age-related macular degeneration and its relationship to antioxidant intake. Acta Ophthalmol. Scand. 2002, 80, 368–371. [Google Scholar] [CrossRef]
- Tan, J.S.; Wang, J.J.; Flood, V.; Rochtchina, E.; Smith, W.; Mitchell, P. Dietary antioxidants and the long-term incidence of age-related macular degeneration: The Blue Mountains Eye Study. Ophthalmology 2008, 115, 334–341. [Google Scholar] [CrossRef]
- Van Leeuwen, R.; Boekhoorn, S.; Vingerling, J.R.; Witteman, J.C.; Klaver, C.C.; Hofman, A.; de Jong, P.T. Dietary intake of antioxidants and risk of age-related macular degeneration. JAMA 2005, 294, 3101–3107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- VandenLangenberg, G.M.; Mares-Perlman, J.A.; Klein, R.; Klein, B.E.; Brady, W.E.; Palta, M. Associations between antioxidant and zinc intake and the 5-year incidence of early age-related maculopathy in the Beaver Dam Eye Study. Am. J. Epidemiol. 1998, 148, 204–214. [Google Scholar] [CrossRef] [Green Version]
- Wu, J.; Cho, E.; Willett, W.C.; Sastry, S.M.; Schaumberg, D.A. Intakes of lutein, zeaxanthin, and other carotenoids and age-related macular degeneration during 2 decades of prospective follow-up. JAMA Ophthalmol. 2015, 133, 1415–1424. [Google Scholar] [CrossRef] [PubMed]
- Age-Related Eye Disease Study 2 Research Group; Chew, E.Y.; Clemons, T.E.; Sangiovanni, J.P.; Danis, R.P.; Ferris, F.L., 3rd; Elman, M.J.; Antoszyk, A.N.; Ruby, A.J.; Orth, D.; et al. Secondary analyses of the effects of lutein/zeaxanthin on age-related macular degeneration progression: AREDS2 report no. 3. JAMA Ophthalmol. 2014, 132, 142–149. [Google Scholar] [CrossRef]
- Akuffo, K.O.; Beatty, S.; Peto, T.; Stack, J.; Stringham, J.; Kelly, D.; Leung, I.; Corcoran, L.; Nolan, J.M. The impact of supplemental antioxidants on visual function in nonadvanced age-related macular degeneration: A head-to-head randomized clinical trial. Investig. Ophthalmol. Vis. Sci. 2017, 58, 5347–5360. [Google Scholar] [CrossRef] [PubMed]
- Akuffo, K.O.; Nolan, J.M.; Howard, A.N.; Moran, R.; Stack, J.; Klein, R.; Klein, B.E.; Meuer, S.M.; Sabour-Pickett, S.; Thurnham, D.I.; et al. Sustained supplementation and monitored response with differing carotenoid formulations in early age-related macular degeneration. Eye 2015, 29, 902–912. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arnold, C.; Jentsch, S.; Dawczynski, J.; Bohm, V. Age-related macular degeneration: Effects of a short-term intervention with an oleaginous kale extract—A pilot study. Nutrition 2013, 29, 1412–1417. [Google Scholar] [CrossRef]
- Arnold, C.; Winter, L.; Frohlich, K.; Jentsch, S.; Dawczynski, J.; Jahreis, G.; Bohm, V. Macular xanthophylls and omega-3 long-chain polyunsaturated fatty acids in age-related macular degeneration: A randomized trial. JAMA Ophthalmol. 2013, 131, 564–572. [Google Scholar] [CrossRef]
- Azar, G.; Quaranta-El Maftouhi, M.; Masella, J.J.; Mauget-Faysse, M. Macular pigment density variation after supplementation of lutein and zeaxanthin using the Visucam((R)) 200 pigment module: Impact of age-related macular degeneration and lens status. J. Fr. Ophtalmol. 2017, 40, 303–313. [Google Scholar] [CrossRef]
- Bartlett, H.E.; Eperjesi, F. Effect of lutein and antioxidant dietary supplementation on contrast sensitivity in age-related macular disease: A randomized controlled trial. Eur. J. Clin. Nutr. 2007, 61, 1121–1127. [Google Scholar] [CrossRef] [PubMed]
- Beatty, S.; Chakravarthy, U.; Nolan, J.M.; Muldrew, K.A.; Woodside, J.V.; Denny, F.; Stevenson, M.R. Secondary outcomes in a clinical trial of carotenoids with coantioxidants versus placebo in early age-related macular degeneration. Ophthalmology 2013, 120, 600–606. [Google Scholar] [CrossRef]
- Berrow, E.J.; Bartlett, H.E.; Eperjesi, F.; Gibson, J.M. The effects of a lutein-based supplement on objective and subjective measures of retinal and visual function in eyes with age-related maculopathy—A randomised controlled trial. Br. J. Nutr. 2013, 109, 2008–2014. [Google Scholar] [CrossRef] [Green Version]
- Cangemi, F.E. TOZAL study: An open case control study of an oral antioxidant and omega-3 supplement for dry AMD. BMC Ophthalmol. 2007, 7, 3. [Google Scholar] [CrossRef] [Green Version]
- Connolly, E.E.; Beatty, S.; Thurnham, D.I.; Loughman, J.; Howard, A.N.; Stack, J.; Nolan, J.M. Augmentation of macular pigment following supplementation with all three macular carotenoids: An exploratory study. Curr. Eye Res. 2010, 35, 335–351. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Corvi, F.; Souied, E.H.; Falfoul, Y.; Georges, A.; Jung, C.; Querques, L.; Querques, G. Pilot evaluation of short-term changes in macular pigment and retinal sensitivity in different phenotypes of early age-related macular degeneration after carotenoid supplementation. Br. J. Ophthalmol. 2017, 101, 770–773. [Google Scholar] [CrossRef]
- Dawczynski, J.; Jentsch, S.; Schweitzer, D.; Hammer, M.; Lang, G.E.; Strobel, J. Long term effects of lutein, zeaxanthin and omega-3-LCPUFAs supplementation on optical density of macular pigment in AMD patients: The LUTEGA study. Graefes Arch. Clin. Exp. Ophthalmol. 2013, 251, 2711–2723. [Google Scholar] [CrossRef]
- Garcia-Layana, A.; Recalde, S.; Alaman, A.S.; Robredo, P.F. Effects of lutein and docosahexaenoic Acid supplementation on macular pigment optical density in a randomized controlled trial. Nutrients 2013, 5, 543–551. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garcia-Layana, A.; Recalde, S.; Hernandez, M.; Abraldes, M.J.; Nascimento, J.; Hernandez-Galilea, E.; Olmedilla-Alonso, B.; Escobar-Barranco, J.J.; Zapata, M.A.; Silva, R.; et al. A randomized study of nutritional supplementation in patients with unilateral wet age-related macular degeneration. Nutrients 2021, 13, 1253. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.M.; Dou, H.L.; Huang, F.F.; Xu, X.R.; Zou, Z.Y.; Lu, X.R.; Lin, X.M. Changes following supplementation with lutein and zeaxanthin in retinal function in eyes with early age-related macular degeneration: A randomised, double-blind, placebo-controlled trial. Br. J. Ophthalmol. 2015, 99, 371–375. [Google Scholar] [CrossRef]
- Huang, Y.M.; Yan, S.F.; Ma, L.; Zou, Z.Y.; Xu, X.R.; Dou, H.L.; Lin, X.M. Serum and macular responses to multiple xanthophyll supplements in patients with early age-related macular degeneration. Nutrition 2013, 29, 387–392. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Dou, H.L.; Huang, Y.M.; Lu, X.R.; Xu, X.R.; Qian, F.; Zou, Z.Y.; Pang, H.L.; Dong, P.C.; Xiao, X.; et al. Improvement of retinal function in early age-related macular degeneration after lutein and zeaxanthin supplementation: A randomized, double-masked, placebo-controlled trial. Am. J. Ophthalmol. 2012, 154, 625–634. [Google Scholar] [CrossRef] [PubMed]
- Meagher, K.A.; Thurnham, D.I.; Beatty, S.; Howard, A.N.; Connolly, E.; Cummins, W.; Nolan, J.M. Serum response to supplemental macular carotenoids in subjects with and without age-related macular degeneration. Br. J. Nutr. 2013, 110, 289–300. [Google Scholar] [CrossRef] [Green Version]
- Murray, I.J.; Makridaki, M.; van der Veen, R.L.; Carden, D.; Parry, N.R.; Berendschot, T.T. Lutein supplementation over a one-year period in early AMD might have a mild beneficial effect on visual acuity: The CLEAR study. Investig. Ophthalmol. Vis. Sci. 2013, 54, 1781–1788. [Google Scholar] [CrossRef] [Green Version]
- Parisi, V.; Tedeschi, M.; Gallinaro, G.; Varano, M.; Saviano, S.; Piermarocchi, S.; Group, C.S. Carotenoids and antioxidants in age-related maculopathy italian study: Multifocal electroretinogram modifications after 1 year. Ophthalmology 2008, 115, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Piermarocchi, S.; Saviano, S.; Parisi, V.; Tedeschi, M.; Panozzo, G.; Scarpa, G.; Boschi, G.; Lo Giudice, G.; Carmis Study, G. Carotenoids in Age-related Maculopathy Italian Study (CARMIS): Two-year results of a randomized study. Eur. J. Ophthalmol. 2012, 22, 216–225. [Google Scholar] [CrossRef]
- Richer, S.; Stiles, W.; Statkute, L.; Pulido, J.; Frankowski, J.; Rudy, D.; Pei, K.; Tsipursky, M.; Nyland, J. Double-masked, placebo-controlled, randomized trial of lutein and antioxidant supplementation in the intervention of atrophic age-related macular degeneration: The Veterans LAST study (Lutein Antioxidant Supplementation Trial). Optometry 2004, 75, 216–230. [Google Scholar] [CrossRef]
- Richer, S.P.; Stiles, W.; Graham-Hoffman, K.; Levin, M.; Ruskin, D.; Wrobel, J.; Park, D.W.; Thomas, C. Randomized, double-blind, placebo-controlled study of zeaxanthin and visual function in patients with atrophic age-related macular degeneration: The Zeaxanthin and Visual Function Study (ZVF) FDA IND #78, 973. Optometry 2011, 82, 667–680. [Google Scholar] [CrossRef]
- Rosenthal, J.M.; Kim, J.; de Monasterio, F.; Thompson, D.J.; Bone, R.A.; Landrum, J.T.; de Moura, F.F.; Khachik, F.; Chen, H.; Schleicher, R.L.; et al. Dose-ranging study of lutein supplementation in persons aged 60 years or older. Investig. Ophthalmol. Vis. Sci. 2006, 47, 5227–5233. [Google Scholar] [CrossRef] [PubMed]
- Sabour-Pickett, S.; Beatty, S.; Connolly, E.; Loughman, J.; Stack, J.; Howard, A.; Klein, R.; Klein, B.E.; Meuer, S.M.; Myers, C.E.; et al. Supplementation with three different macular carotenoid formulations in patients with early age-related macular degeneration. Retina 2014, 34, 1757–1766. [Google Scholar] [CrossRef] [Green Version]
- Sawa, M.; Shunto, T.; Nishiyama, I.; Yokoyama, A.; Shigeta, R.; Miura, S.; Kawasaki, R. Effects of lutein supplementation in Japanese patients with unilateral age-related macular degeneration: The Sakai lutein Study. Sci. Rep. 2020, 10, 5958. [Google Scholar] [CrossRef] [Green Version]
- Thurnham, D.I.; Nolan, J.M.; Howard, A.N.; Beatty, S. Macular response to supplementation with differing xanthophyll formulations in subjects with and without age-related macular degeneration. Graefes Arch. Clin. Exp. Ophthalmol. 2015, 253, 1231–1243. [Google Scholar] [CrossRef] [PubMed]
- Wolf-Schnurrbusch, U.E.; Zinkernagel, M.S.; Munk, M.R.; Ebneter, A.; Wolf, S. Oral lutein supplementation enhances macular pigment density and contrast sensitivity but not in combination with polyunsaturated fatty acids. Investig. Ophthalmol. Vis. Sci. 2015, 56, 8069–8074. [Google Scholar] [CrossRef] [Green Version]
- Meyers, K.J.; Liu, Z.; Millen, A.E.; Iyengar, S.K.; Blodi, B.A.; Johnson, E.; Snodderly, D.M.; Klein, M.L.; Gehrs, K.M.; Tinker, L.; et al. Joint associations of diet, lifestyle, and genes with age-related macular degeneration. Ophthalmology 2015, 122, 2286–2294. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Age-Related Eye Disease Study Research Group; Clemons, T.E.; Milton, R.C.; Klein, R.; Seddon, J.M.; Ferris, F.L., 3rd. Risk factors for the incidence of Advanced Age-Related Macular Degeneration in the Age-Related Eye Disease Study (AREDS) AREDS report no. 19. Ophthalmology 2005, 112, 533–539. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abdel-Aal, E.-S.M.; Akhtar, H.; Zaheer, K.; Ali, R. Dietary sources of lutein and zeaxanthin carotenoids and their role in eye health. Nutrients 2013, 5, 1169–1185. [Google Scholar] [CrossRef] [Green Version]
- Eisenhauer, B.; Natoli, S.; Liew, G.; Flood, V.M. Lutein and zeaxanthin-food sources, bioavailability and dietary variety in age-related macular degeneration protection. Nutrients 2017, 9, 120. [Google Scholar] [CrossRef]
- Mageney, V.; Baldermann, S.; Albach, D.C. Intraspecific variation in carotenoids of Brassica oleracea var. sabellica. J. Agric. Food Chem. 2016, 64, 3251–3257. [Google Scholar] [CrossRef]
- Perry, A.; Rasmussen, H.; Johnson, E.J. Xanthophyll (lutein, zeaxanthin) content in fruits, vegetables and corn and egg products. J. Food Compos. Anal. 2009, 22, 9–15. [Google Scholar] [CrossRef]
- Ma, L.; Dou, H.L.; Wu, Y.Q.; Huang, Y.M.; Huang, Y.B.; Xu, X.R.; Zou, Z.Y.; Lin, X.M. Lutein and zeaxanthin intake and the risk of age-related macular degeneration: A systematic review and meta-analysis. Br. J. Nutr. 2012, 107, 350–359. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.P.; Lin, Y.W.; Huang, Y.C.; Tsai, F.J. Mitochondrial dysfunction as a novel target for neuroprotective nutraceuticals in ocular diseases. Nutrients 2020, 12, 1950. [Google Scholar] [CrossRef] [PubMed]
- Barot, M.; Gokulgandhi, M.R.; Mitra, A.K. Mitochondrial dysfunction in retinal diseases. Curr. Eye. Res. 2011, 36, 1069–1077. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Armstrong, J.S. Mitochondrial medicine: Pharmacological targeting of mitochondria in disease. Br. J. Pharmacol. 2007, 151, 1154–1165. [Google Scholar] [CrossRef] [PubMed]
- Feher, J.; Papale, A.; Mannino, G.; Gualdi, L.; Gabrieli, C.B. Mitotropic compounds for the treatment of age-related macular degeneration. The metabolic approach and a pilot study. Ophthalmologica 2003, 217, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Bohn, T. Bioavailability of non-provitamin A carotenoids. Curr. Nutr. Food Sci. 2008, 4, 240–258. [Google Scholar] [CrossRef]
- Bray, G.A.; Nielsen, S.J.; Popkin, B.M. Consumption of high-fructose corn syrup in beverages may play a role in the epidemic of obesity. Am. J. Clin. Nutr. 2004, 79, 537–543. [Google Scholar] [CrossRef]
- Castenmiller, J.J.; West, C.E. Bioavailability and bioconversion of carotenoids. Annu. Rev. Nutr. 1998, 18, 19–38. [Google Scholar] [CrossRef]
- Gross, L.S.; Li, L.; Ford, E.S.; Liu, S. Increased consumption of refined carbohydrates and the epidemic of type 2 diabetes in the United States: An ecologic assessment. Am. J. Clin. Nutr. 2004, 79, 774–779. [Google Scholar] [CrossRef]
- Ludwig, D.S.; Majzoub, J.A.; Al-Zahrani, A.; Dallal, G.E.; Blanco, I.; Roberts, S.B. High glycemic index foods, overeating, and obesity. Pediatrics 1999, 103, E26. [Google Scholar] [CrossRef] [Green Version]
- Nagao, A. Absorption and metabolism of dietary carotenoids. BioFactors 2011, 37, 83–87. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, O.F.; Ryan, L.; O’Brien, N.M. Xanthophyll carotenoids are more bioaccessible from fruits than dark green vegetables. Nutr. Res. 2007, 27, 258–264. [Google Scholar] [CrossRef]
- Van het Hof, K.H.; West, C.E.; Weststrate, J.A.; Hautvast, J.G.A.J. Dietary factors that affect the bioavailability of carotenoids. J. Nutr. 2000, 130, 503–506. [Google Scholar] [CrossRef]
- Barreau, E.; Brossas, J.Y.; Courtois, Y.; Tréton, J.A. Accumulation of mitochondrial DNA deletions in human retina during aging. Investig. Ophthalmol. Vis. Sci. 1996, 37, 384–391. [Google Scholar]
- Decanini, A.; Nordgaard, C.L.; Feng, X.; Ferrington, D.A.; Olsen, T.W. Changes in select redox proteins of the retinal pigment epithelium in age-related macular degeneration. Am. J. Ophthalmol. 2007, 143, 607–615. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dunaief, J.L. The role of apoptosis in age-related macular degeneration. Arch. Ophthalmol. 2002, 120, 1435. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feher, J.; Kovacs, I.; Artico, M.; Cavallotti, C.; Papale, A.; Gabrieli, C.B. Mitochondrial alterations of retinal pigment epithelium in age-related macular degeneration. Neurobiol. Aging 2006, 27, 983–993. [Google Scholar] [CrossRef] [PubMed]
- Kaarniranta, K.; Pawlowska, E.; Szczepanska, J.; Jablkowska, A.; Blasiak, J. Role of mitochondrial DNA damage in ROS-mediated pathogenesis of age-related macular degeneration (AMD). Int. J. Mol. Sci. 2019, 20, 2374. [Google Scholar] [CrossRef] [Green Version]
- Kennedy, C.J.; Rakoczy, P.E.; Constable, I.J. Lipofuscin of the retinal pigment epithelium: A review. Eye 1995, 9, 763–771. [Google Scholar] [CrossRef] [Green Version]
- Liang, F.-Q.; Godley, B.F. Oxidative stress-induced mitochondrial DNA damage in human retinal pigment epithelial cells: A possible mechanism for RPE aging and age-related macular degeneration. Exp. Eye Res. 2003, 76, 397–403. [Google Scholar] [CrossRef]
- Nordgaard, C.L.; Berg, K.M.; Kapphahn, R.J.; Reilly, C.; Feng, X.; Olsen, T.W.; Ferrington, D.A. Proteomics of the retinal pigment epithelium reveals altered protein expression at progressive stages of age-related macular degeneration. Investig. Opthalmol. Vis. Sci. 2006, 47, 815. [Google Scholar] [CrossRef]
- Terluk, M.R.; Kapphahn, R.J.; Soukup, L.M.; Gong, H.; Gallardo, C.; Montezuma, S.R.; Ferrington, D.A. Investigating mitochondria as a target for treating age-related macular degeneration. J. Neurosci. 2015, 35, 7304–7311. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Terman, A.; Brunk, U.T. Oxidative stress, accumulation of biological ‘garbage’, and aging. Antioxid. Redox Signal. 2006, 8, 197–204. [Google Scholar] [CrossRef]
- Mukhopadhyay, A.; Weiner, H. Delivery of drugs and macromolecules to mitochondria. Adv. Drug Deliv. Rev. 2007, 59, 729–738. [Google Scholar] [CrossRef] [Green Version]
- Neroev, V.V.; Archipova, M.M.; Bakeeva, L.E.; Fursova, A.; Grigorian, E.N.; Grishanova, A.Y.; Iomdina, E.N.; Ivashchenko, Z.N.; Katargina, L.A.; Khoroshilova-Maslova, I.P.; et al. Mitochondria-targeted plastoquinone derivatives as tools to interrupt execution of the aging program. 4. Age-related eye disease. SkQ1 returns vision to blind animals. Biochemistry 2008, 73, 1317–1328. [Google Scholar] [CrossRef] [PubMed]
- Al-Aubaidy, H.A.; Jelinek, H.F. Oxidative DNA damage and obesity in type 2 diabetes mellitus. Eur. J. Endocrinol. 2011, 164, 899–904. [Google Scholar] [CrossRef]
- Castro, A.M.; Macedo-de la Concha, L.E.; Pantoja-Meléndez, C.A. Low-grade inflammation and its relation to obesity and chronic degenerative diseases. Rev. Méd. Hosp. Gen. México 2017, 80, 101–105. [Google Scholar] [CrossRef]
- Chung, H.Y.; Ferreira, A.L.; Epstein, S.; Paiva, S.A.; Castaneda-Sceppa, C.; Johnson, E.J. Site-specific concentrations of carotenoids in adipose tissue: Relations with dietary and serum carotenoid concentrations in healthy adults. Am. J. Clin. Nutr. 2009, 90, 533–539. [Google Scholar] [CrossRef] [Green Version]
- Connor, W.E.; Duell, P.B.; Kean, R.; Wang, Y. The prime role of HDL to transport lutein into the retina: Evidence from HDL-deficient WHAM chicks having a mutant ABCA1 transporter. Investig. Ophthalmol. Vis. Sci. 2007, 48, 4226–4231. [Google Scholar] [CrossRef] [Green Version]
- Fernandez-Sanchez, A.; Madrigal-Santillan, E.; Bautista, M.; Esquivel-Soto, J.; Morales-Gonzalez, A.; Esquivel-Chirino, C.; Durante-Montiel, I.; Sanchez-Rivera, G.; Valadez-Vega, C.; Morales-Gonzalez, J.A. Inflammation, oxidative stress, and obesity. Int. J. Mol. Sci. 2011, 12, 3117–3132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frayn, K.N.; Coppack, S.W.; Fielding, B.A.; Humphreys, S.M. Coordinated regulation of hormone-sensitive lipase and lipoprotein lipase in human adipose tissue in vivo: Implications for the control of fat storage and fat mobilization. Adv. Enzyme Regul. 1995, 35, 163–178. [Google Scholar] [CrossRef]
- Kwon, H.; Pessin, J.E. Adipokines mediate inflammation and insulin resistance. Front. Endocrinol. 2013, 4, 71. [Google Scholar] [CrossRef] [Green Version]
- Lim, C.; Kim, D.-W.; Sim, T.; Hoang, N.H.; Lee, J.W.; Lee, E.S.; Youn, Y.S.; Oh, K.T. Preparation and characterization of a lutein loading nanoemulsion system for ophthalmic eye drops. J. Drug Deliv. Sci. Technol. 2016, 36, 168–174. [Google Scholar] [CrossRef]
- Loane, E.; Nolan, J.M.; Beatty, S. The respective relationships between lipoprotein profile, macular pigment optical density, and serum concentrations of lutein and zeaxanthin. Investig. Ophthalmol. Vis. Sci. 2010, 51, 5897–5905. [Google Scholar] [CrossRef] [Green Version]
- Rosen, B.S.; Cook, K.S.; Yaglom, J.; Groves, D.L.; Volanakis, J.E.; Damm, D.; White, T.; Spiegelman, B.M. Adipsin and complement factor D activity: An immune-related defect in obesity. Science 1989, 244, 1483–1487. [Google Scholar] [CrossRef] [PubMed]
- Savini, I.; Catani, M.V.; Evangelista, D.; Gasperi, V.; Avigliano, L. Obesity-associated oxidative stress: Strategies finalized to improve redox state. Int. J. Mol. Sci. 2013, 14, 10497–10538. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tilg, H.; Moschen, A.R. Adipocytokines: Mediators linking adipose tissue, inflammation and immunity. Nat. Rev. Immunol. 2006, 6, 772–783. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Connor, S.L.; Johnson, E.J.; Klein, M.L.; Hughes, S.; Connor, W.E. Effect of dietary lutein and zeaxanthin on plasma carotenoids and their transport in lipoproteins in age-related macular degeneration. Am. J. Clin. Nutr. 2007, 85, 762–769. [Google Scholar] [CrossRef]
- Bovier, E.R.; Lewis, R.D.; Hammond, B.R., Jr. The relationship between lutein and zeaxanthin status and body fat. Nutrients 2013, 5, 750–757. [Google Scholar] [CrossRef]
- Broekmans, W.M.; Berendschot, T.T.; Klopping-Ketelaars, I.A.; de Vries, A.J.; Goldbohm, R.A.; Tijburg, L.B.; Kardinaal, A.F.; van Poppel, G. Macular pigment density in relation to serum and adipose tissue concentrations of lutein and serum concentrations of zeaxanthin. Am. J. Clin. Nutr. 2002, 76, 595–603. [Google Scholar] [CrossRef] [Green Version]
- Johnson, E.J.; Hammond, B.R.; Yeum, K.J.; Qin, J.; Wang, X.D.; Castaneda, C.; Snodderly, D.M.; Russell, R.M. Relation among serum and tissue concentrations of lutein and zeaxanthin and macular pigment density. Am. J. Clin. Nutr. 2000, 71, 1555–1562. [Google Scholar] [CrossRef] [Green Version]
- Hammond, B.R., Jr.; Ciulla, T.A.; Snodderly, D.M. Macular pigment density is reduced in obese subjects. Investig. Ophthalmol. Vis. Sci. 2002, 43, 47–50. [Google Scholar]
- Nieves, D.J.; Cnop, M.; Retzlaff, B.; Walden, C.E.; Brunzell, J.D.; Knopp, R.H.; Kahn, S.E. The atherogenic lipoprotein profile associated with obesity and insulin resistance is largely attributable to intra-abdominal fat. Diabetes 2003, 52, 172–179. [Google Scholar] [CrossRef] [Green Version]
- Nolan, J.; O’Donovan, O.; Kavanagh, H.; Stack, J.; Harrison, M.; Muldoon, A.; Mellerio, J.; Beatty, S. Macular pigment and percentage of body fat. Investig. Ophthalmol. Vis. Sci. 2004, 45, 3940–3950. [Google Scholar] [CrossRef] [PubMed]
- Nolan, J.M.; Feeney, J.; Kenny, R.A.; Cronin, H.; O’Regan, C.; Savva, G.M.; Loughman, J.; Finucane, C.; Connolly, E.; Meagher, K.; et al. Education is positively associated with macular pigment: The Irish Longitudinal Study on Ageing (TILDA). Investig. Ophthalmol. Vis. Sci. 2012, 53, 7855–7861. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Flammer, J.; Konieczka, K.; Flammer, A.J. The primary vascular dysregulation syndrome: Implications for eye diseases. EPMA J. 2013, 4, 14. [Google Scholar] [CrossRef] [Green Version]
- Jeganathan, V.S.; Kawasaki, R.; Wang, J.J.; Aung, T.; Mitchell, P.; Saw, S.M.; Wong, T.Y. Retinal vascular caliber and age-related macular degeneration: The Singapore Malay Eye Study. Am. J. Ophthalmol. 2008, 146, 954–959. [Google Scholar] [CrossRef]
- Kumari, N.; Cher, J.; Chua, E.; Hamzah, H.; Wong, T.Y.; Cheung, C.Y. Association of serum lutein and zeaxanthin with quantitative measures of retinal vascular parameters. PLoS ONE 2018, 13, e0203868. [Google Scholar] [CrossRef] [PubMed]
- Parker, R.S. Carotenoids in human blood and tissues. J. Nutr. 1989, 119, 101–104. [Google Scholar] [CrossRef] [Green Version]
- Alpha-Tocopherol Beta Carotene Cancer Prevention Study Group. The effect of vitamin E and beta carotene on the incidence of lung cancer and other cancers in male smokers. N. Engl. J. Med. 1994, 330, 1029–1035. [Google Scholar] [CrossRef] [PubMed]
- Middha, P.; Weinstein, S.J.; Männistö, S.; Albanes, D.; Mondul, A.M. β-carotene supplementation and lung cancer incidence in the alpha-tocopherol, beta-carotene cancer prevention study: The role of tar and nicotine. Nicotine Tob. Res. 2019, 21, 1045–1050. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Age-Related Eye Disease Study 2 Research Group; Chew, E.Y.; Clemons, T.; SanGiovanni, J.P.; Danis, R.; Domalpally, A.; McBee, W.; Sperduto, R.; Ferris, F.L. The Age-Related Eye Disease Study 2 (AREDS2): Study design and baseline characteristics (AREDS2 report number 1). Ophthalmology 2012, 119, 2282–2289. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hammond, B.R., Jr.; Fletcher, L.M.; Elliott, J.G. Glare disability, photostress recovery, and chromatic contrast: Relation to macular pigment and serum lutein and zeaxanthin. Investig. Ophthalmol. Vis. Sci. 2013, 54, 476–481. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hammond, B.R.; Fletcher, L.M.; Roos, F.; Wittwer, J.; Schalch, W. A double-blind, placebo-controlled study on the effects of lutein and zeaxanthin on photostress recovery, glare disability, and chromatic contrast. Investig. Ophthalmol. Vis. Sci. 2014, 55, 8583–8589. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loughman, J.; Akkali, M.C.; Beatty, S.; Scanlon, G.; Davison, P.A.; O’Dwyer, V.; Cantwell, T.; Major, P.; Stack, J.; Nolan, J.M. The relationship between macular pigment and visual performance. Vis. Res. 2010, 50, 1249–1256. [Google Scholar] [CrossRef] [Green Version]
- Stringham, J.M.; Garcia, P.V.; Smith, P.A.; Hiers, P.L.; McLin, L.N.; Kuyk, T.K.; Foutch, B.K. Macular pigment and visual performance in low-light conditions. Investig. Ophthalmol. Vis. Sci. 2015, 56, 2459–2468. [Google Scholar] [CrossRef] [Green Version]
- Stringham, J.M.; Garcia, P.V.; Smith, P.A.; McLin, L.N.; Foutch, B.K. Macular pigment and visual performance in glare: Benefits for photostress recovery, disability glare, and visual discomfort. Investig. Ophthalmol. Vis. Sci. 2011, 52, 7406–7415. [Google Scholar] [CrossRef] [Green Version]
- Stringham, J.M.; Hammond, B.R., Jr. The glare hypothesis of macular pigment function. Optom. Vis. Sci. 2007, 84, 859–864. [Google Scholar] [CrossRef] [Green Version]
- Stringham, J.M.; Hammond, B.R. Macular pigment and visual performance under glare conditions. Optom. Vis. Sci. 2008, 85, 82–88. [Google Scholar] [CrossRef]
- Stringham, J.M.; O’Brien, K.J.; Stringham, N.T. Macular carotenoid supplementation improves disability glare performance and dynamics of photostress recovery. Eye Vis. 2016, 3, 30. [Google Scholar] [CrossRef] [Green Version]
- Stringham, J.M.; O’Brien, K.J.; Stringham, N.T. Contrast sensitivity and lateral inhibition are enhanced with macular carotenoid supplementation. Investig. Ophthalmol. Vis. Sci. 2017, 58, 2291–2295. [Google Scholar] [CrossRef]
- Stringham, J.M.; Stringham, N.T.; O’Brien, K.J. Macular carotenoid supplementation improves visual performance, sleep quality, and adverse physical symptoms in those with high screen time exposure. Foods 2017, 6, 47. [Google Scholar] [CrossRef] [Green Version]
- Khachik, F.; de Moura, F.F.; Chew, E.Y.; Douglass, L.W.; Ferris, F.L., 3rd; Kim, J.; Thompson, D.J. The effect of lutein and zeaxanthin supplementation on metabolites of these carotenoids in the serum of persons aged 60 or older. Investig. Ophthalmol. Vis. Sci. 2006, 47, 5234–5242. [Google Scholar] [CrossRef] [PubMed]
- Bone, R.A.; Landrum, J.T. Dose-dependent response of serum lutein and macular pigment optical density to supplementation with lutein esters. Arch. Biochem. Biophys. 2010, 504, 50–55. [Google Scholar] [CrossRef] [Green Version]
- Bovier, E.R.; Hammond, B.R. A randomized placebo-controlled study on the effects of lutein and zeaxanthin on visual processing speed in young healthy subjects. Arch. Biochem. Biophys. 2015, 572, 54–57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnson, E.J.; Chung, H.Y.; Caldarella, S.M.; Snodderly, D.M. The influence of supplemental lutein and docosahexaenoic acid on serum, lipoproteins, and macular pigmentation. Am. J. Clin. Nutr. 2008, 87, 1521–1529. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kvansakul, J.; Rodriguez-Carmona, M.; Edgar, D.F.; Barker, F.M.; Kopcke, W.; Schalch, W.; Barbur, J.L. Supplementation with the carotenoids lutein or zeaxanthin improves human visual performance. Ophthalmic Physiol. Opt. 2006, 26, 362–371. [Google Scholar] [CrossRef]
- Landrum, J.; Bone, R.; Mendez, V.; Valenciaga, A.; Babino, D. Comparison of dietary supplementation with lutein diacetate and lutein: A pilot study of the effects on serum and macular pigment. Acta Biochim. Pol. 2012, 59, 167–169. [Google Scholar] [CrossRef] [Green Version]
- Yao, Y.; Qiu, Q.H.; Wu, X.W.; Cai, Z.Y.; Xu, S.; Liang, X.Q. Lutein supplementation improves visual performance in Chinese drivers: 1-year randomized, double-blind, placebo-controlled study. Nutrition 2013, 29, 958–964. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Wang, T.; Zhang, B.; Qin, L.; Wu, C.; Li, Q.; Ma, L. Lutein and zeaxanthin supplementation and association with visual function in age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 2014, 56, 252–258. [Google Scholar] [CrossRef] [Green Version]
- Buhren, J.; Terzi, E.; Bach, M.; Wesemann, W.; Kohnen, T. Measuring contrast sensitivity under different lighting conditions: Comparison of three tests. Optom. Vis. Sci. 2006, 83, 290–298. [Google Scholar] [CrossRef]
- Chen, X.; Li, Q.; Liu, X.; Yang, L.; Xia, W.; Tao, L. Visual acuity evaluated by pattern-reversal visual-evoked potential is affected by check size/visual angle. Neurosci. Bull. 2012, 28, 737–745. [Google Scholar] [CrossRef] [Green Version]
- Stangos, N.; Voutas, S.; Topouzis, F.; Karampatakis, V. Contrast sensitivity evaluation in eyes predisposed to age-related macular degeneration and presenting normal visual acuity. Ophthalmologica 1995, 209, 194–198. [Google Scholar] [CrossRef]
- Berrow, E.J.; Bartlett, H.E.; Eperjesi, F.; Gibson, J.M. The electroretinogram: A useful tool for evaluating age-related macular disease? Doc. Ophthalmol. 2010, 121, 51–62. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, C.; Wu, L.; Wu, D.; Huang, S.; Wen, F.; Luo, G.; Long, S. The local cone and rod system function in early age-related macular degeneration. Doc. Ophthalmol. 2004, 109, 1–8. [Google Scholar] [CrossRef]
- Feigl, B.; Lovie-Kitchin, J.; Brown, B. Objective functional assessment of age-related maculopathy: A special application for the multifocal electroretinogram. Clin. Exp. Optom. 2005, 88, 304–312. [Google Scholar] [CrossRef] [Green Version]
- Gerth, C.; Delahunt, P.B.; Alam, S.; Morse, L.S.; Werner, J.S. Cone-mediated multifocal electroretinogram in age-related macular degeneration: Progression over a long-term follow-up. Arch. Ophthalmol. 2006, 124, 345–352. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gin, T.J.; Luu, C.D.; Guymer, R.H. Central retinal function as measured by the multifocal electroretinogram and flicker perimetry in early age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 2011, 52, 9267–9274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hood, D.C.; Frishman, L.J.; Saszik, S.; Viswanathan, S. Retinal origins of the primate multifocal ERG: Implications for the human response. Investig. Ophthalmol. Vis. Sci. 2002, 43, 1673–1685. [Google Scholar]
- Hood, D.C.; Odel, J.G.; Chen, C.S.; Winn, B.J. The multifocal electroretinogram. J. Neuroophthalmol. 2003, 23, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Wu, D.; Jiang, F.; Ma, J.; Wu, L.; Liang, J.; Luo, G. The multifocal electroretinogram in age-related maculopathies. Doc. Ophthalmol. 2000, 101, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Tso, M.O.; Lam, T.T. Reduced amplitude and delayed latency in foveal response of multifocal electroretinogram in early age related macular degeneration. Br. J. Ophthalmol. 2001, 85, 287–290. [Google Scholar] [CrossRef] [Green Version]
- Parisi, V.; Perillo, L.; Tedeschi, M.; Scassa, C.; Gallinaro, G.; Capaldo, N.; Varano, M. Macular function in eyes with early age-related macular degeneration with or without contralateral late age-related macular degeneration. Retina 2007, 27, 879–890. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sutter, E.E.; Tran, D. The field topography of ERG components in man—I. The photopic luminance response. Vis. Res. 1992, 32, 433–446. [Google Scholar] [CrossRef]
- Droge, W. Free radicals in the physiological control of cell function. Physiol. Rev. 2002, 82, 47–95. [Google Scholar] [CrossRef]
- Pinazo-Durán, M.D.; Zanón-Moreno, V.; Gallego-Pinazo, R.; García-Medina, J.J. Chapter 6—Oxidative stress and mitochondrial failure in the pathogenesis of glaucoma neurodegeneration. In Progress in Brain Research; Bagetta, G., Nucci, C., Eds.; Elsevier: Amsterdam, The Netherlands, 2015; Volume 220, pp. 127–153. [Google Scholar]
- Toma, C.; De Cilla, S.; Palumbo, A.; Garhwal, D.P.; Grossini, E. Oxidative and nitrosative stress in age-related macular degeneration: A Review of their role in different stages of disease. Antioxidants 2021, 10, 653. [Google Scholar] [CrossRef]
- Johnson, E.J.; Vishwanathan, R.; Johnson, M.A.; Hausman, D.B.; Davey, A.; Scott, T.M.; Green, R.C.; Miller, L.S.; Gearing, M.; Woodard, J.; et al. Relationship between serum and brain carotenoids, alpha-tocopherol, and retinol concentrations and cognitive performance in the oldest old from the Georgia Centenarian Study. J. Aging Res. 2013, 2013, 951786. [Google Scholar] [CrossRef] [Green Version]
- Johnson, E.J. Role of lutein and zeaxanthin in visual and cognitive function throughout the lifespan. Nutr. Rev. 2014, 72, 605–612. [Google Scholar] [CrossRef]
- Olk, R.J.; Peralta, E.; Gierhart, D.L.; Brown, G.C.; Brown, M.M. Triple combination therapy and zeaxanthin for the treatment of neovascular age-related macular degeneration: An interventional comparative study and cost-effectiveness analysis. Int. J. Retina Vitr. 2015, 1, 22. [Google Scholar] [CrossRef] [Green Version]
- Rosen, R.; Vagaggini, T.; Chen, Y.; Hu, D.N. Zeaxanthin inhibits hypoxia-induced VEGF secretion by RPE cells through decreased protein levels of hypoxia-inducible factors-1alpha. Biomed. Res. Int. 2015, 2015, 687386. [Google Scholar] [CrossRef] [Green Version]
- Lima, V.C.; Rosen, R.B.; Maia, M.; Prata, T.S.; Dorairaj, S.; Farah, M.E.; Sallum, J. Macular pigment optical density measured by dual-wavelength autofluorescence imaging in diabetic and nondiabetic patients: A comparative study. Investig. Ophthalmol. Vis. Sci. 2010, 51, 5840–5845. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Conti, B.; Bucolo, C.; Giannavola, C.; Puglisi, G.; Giunchedi, P.; Conte, U. Biodegradable microspheres for the intravitreal administration of acyclovir: In vitro/in vivo evaluation. Eur. J. Pharm. Sci. 1997, 5, 287–293. [Google Scholar] [CrossRef]
- Jaiswal, M.; Dudhe, R.; Sharma, P.K. Nanoemulsion: An advanced mode of drug delivery system. 3 Biotech 2015, 5, 123–127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, M.; Feng, Z.; Deng, Y.; Zhong, C.; Liu, Y.; Liu, J.; Zhao, X.; Fu, Y. Liquid antisolvent precipitation: An effective method for ocular targeting of lutein esters. Int. J. Nanomed. 2019, 14, 2667–2681. [Google Scholar] [CrossRef] [Green Version]


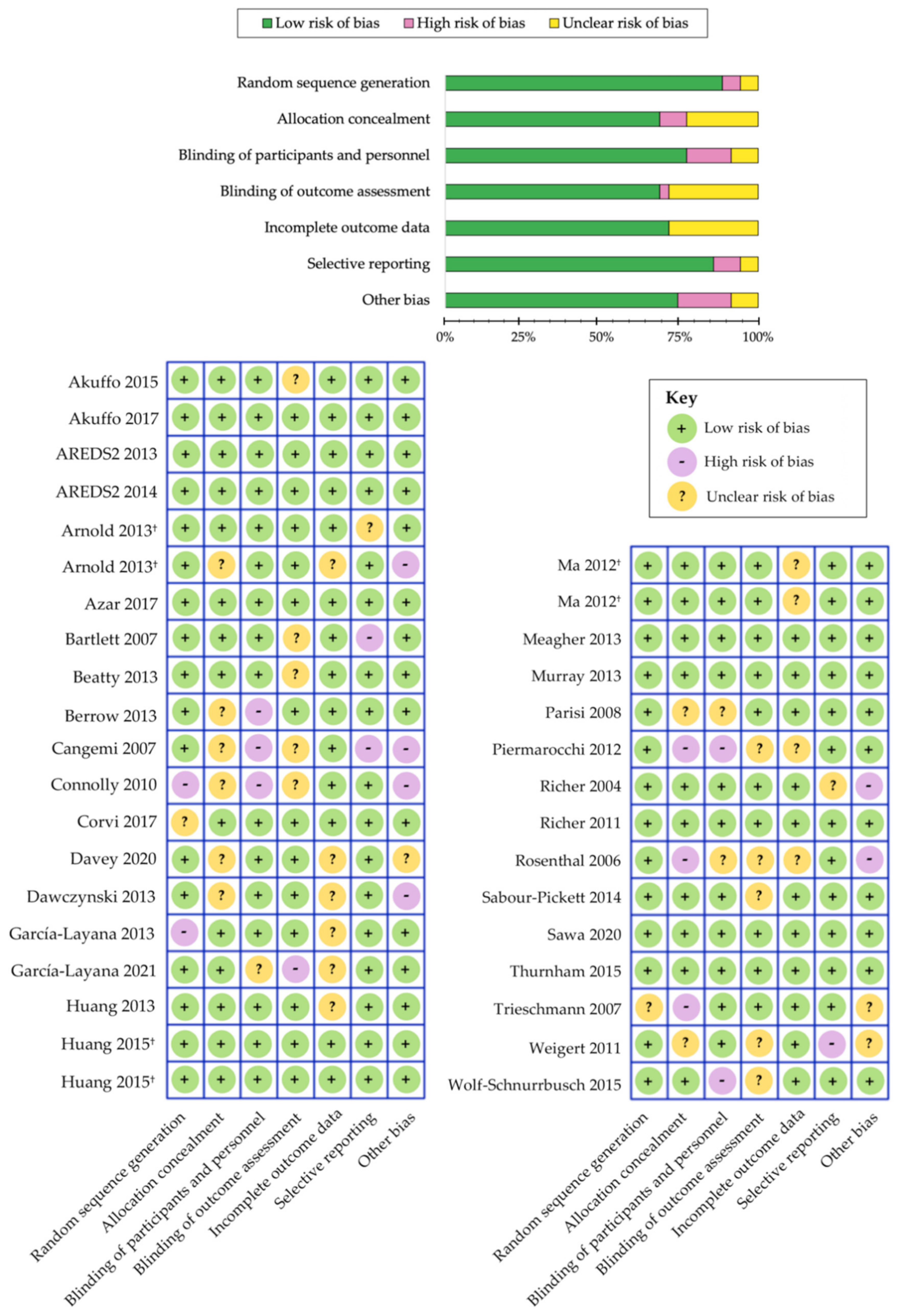
| Category | Age-Related Eye Disease Study (AREDS) Classification |
|---|---|
| 1 | No drusen, or non-extensive small drusen only in both eyes |
| 2 | Extensive small drusen, non-extensive intermediate drusen, or the presence of pigment abnormalities in at least one eye |
| 3 | Extensive intermediate drusen, large drusen, or non-central geographic atrophy in at least one eye |
| 4 | Advanced AMD as defined by at least one of the following: geographic atrophy, retinal pigment epithelial detachment in one eye, choroidal neovascularization, or scars of confluent photocoagulation; or visual acuity less than 20/32 associated with lesions from non-advanced age-related macular degeneration, including large drusen in the fovea, in only one eye |
| Beckman Clinical Classification [68] | |||
|---|---|---|---|
| AMD Classification 1 | Drusen | Pigmentary Abnormalities 2 | Additional Features |
| No apparent aging | None | None | n/a |
| Normal aging changes | Small (≤63 μm) | None | n/a |
| Early AMD | Medium (>63 μm and ≤125 μm) | None | n/a |
| Intermediate AMD | Large (>125 μm) | Abnormalities present 2 | n/a |
| Late AMD | Large (>125 μm) | Abnormalities present 2 | Neovascular AMD and/or any geographic atrophy |
| Authors (Year) | Study Name | Participants | Follow-Up | Assessment of L/Z | Results |
|---|---|---|---|---|---|
| Seddon (1994) [197] | EDCCS | 356 AMD patients, 520 controls in USA; aged 55–80 years | - | Dietary L/Z | Highest quintile of L/Z intake, such as spinach and collard greens, strongly associated with reduced risk of late AMD |
| VandenLangenberg (1998) [201] | Beaver Dam Eye Study | 1709 individuals in USA; aged 43–84 years | 5 years | Dietary L/Z | No significant association reported between incident large drusen and dietary intake |
| Mares-Perlman (2001) [194] | NHANES III | 8596 individuals in USA; aged ≥40 years | - | Dietary L/Z | Significantly lower risk of pigmentary abnormalities and late AMD in highest L/Z quintiles |
| Snellen (2002) [198] | - | 72 AMD patients, 66 controls in Netherlands; aged ≥60 years | - | Dietary L/Z | Low dietary intake significantly associated with higher risk of neovascular AMD |
| Cho (2004) [188] | NHS and HPFS | 77,562 female and 40,866 male health professionals in USA; aged ≥50 years | 18 years; 12 years | Dietary L/Z | No significant association between relative risk of age-related maculopathy and vegetable consumption or carotenoid intake |
| Van Leeuwen (2005) [200] | The Rotterdam Study | 4170 individuals in Netherlands; aged 55–95 years | 8 years | Dietary L/Z | No significant association reported between dietary L/Z intake and incident AMD |
| Moeller (2006) [196] | CAREDS | 1787 women in USA; aged 50–79 years | 7 years | Dietary L/Z | Protective association among adult women (<75 years) with stable dietary intake and no history of chronic disease |
| AREDS Research Group (2007) [51] | AREDS | 4159 AREDS participants in USA; aged 60–80 years | - | Dietary L/Z | Top quintile of dietary L/Z inversely associated with large drusen, neovascular AMD, and geographic atrophy |
| Tan (2008) [199] | Blue Mountains Eye Study | 2454 individuals in Australia; aged 49–93 years | 10.5 years | Dietary L/Z | Greater intake of L/Z saw reduced risk developing soft/reticular drusen and neovascular AMD progression |
| Cho (2008) [187] | NHS and HPFS | 71,494 female and 41,564 male health professionals in USA; aged 50–79 years | 18 years; 16 years | Dietary L/Z | A non-linear, inverse association seen among top quintiles of L/Z intake and neovascular AMD in both cohorts |
| Ho (2011) [16] | The Rotterdam Study | 2167 individuals in Netherlands; aged ≥55 years | 8 years | Dietary L/Z | Top tertile of L/Z intake significantly reduced incident early AMD in those with greater genetic risk |
| Wu (2015) [202] | NHS and HPFS | 63,443 female and 38,603 male health professionals in USA; aged 50–90 years | 26 years; 24 years | Dietary L/Z | Greater consumption of cooked spinach (0.5 cup, >1 serving/wk) inversely associated with intermediate AMD. Late AMD risk significantly lowered by up to 40% with higher L/Z intake |
| Arslan (2019) [186] | - | 100 AMD patients, 100 controls in Turkey; aged ≥50 years | - | Dietary L/Z | Non-significant association observed between serum L/Z |
| EDCCS Group (1993) [191] | EDCCS | 421 AMD patients, 615 controls in USA; aged 55–80 years | - | Serum L/Z | Protective association with greater serum L/Z levels and risk of neovascular AMD |
| Mares-Perlman (1995) [193] | Beaver Dam Eye Study | 167 AMD patients, 167 controls in USA; aged 43–84 years | - | Serum L/Z | No overall association between serum L/Z and risk of late AMD |
| Gale (2003) [192] | - | 380 individuals in Sheffield, United Kingdom; aged ≥60 years | - | Serum L/Z | Serum Z strongly associated with risk of incident early and late AMD |
| Dasch (2005) [189] | MARS | 586 AMD patients, 182 controls in Germany; aged 59–82 years | - | Serum L/Z | No significant association reported between serum L/Z levels |
| Delcourt (2006) [190] | POLA | 640 individuals in Sète, France; aged ≥60 years | - | Serum L/Z | Highest combined serum L/Z has significantly reduced risk |
| Michikawa (2009) [195] | - | 722 individuals in Karabuchi Town of Takasaki City, Japan; aged ≥65 years | - | Serum L/Z | No significant association found between serum L/Z |
| Zhou (2011) [109] | - | 174 AMD patients, 89 controls in China; aged 50–88 years | - | Serum L/Z | Significant inverse association between serum Z and neovascular AMD |
| Authors (Year) | Study | Participants | Duration | Interventions | Serum | Main Findings |
|---|---|---|---|---|---|---|
| Rosenthal (2006) [227] | - | 30 patients with intermediate or late AMD; aged 60–91 years in USA | 6 months | 2.5 mg L; 5 mg L; 10 mg L | L and Z | Mean serum concentrations rose in each dosage group by 2-fold, 2.9-fold and 4-fold, respectively (p < 0.001 for all) |
| Trieschmann (2007) [119] | LUNA | 100 patients with AMD; aged (71.5 ± 7.1) years in Germany | 6 months | 12 mg L and 1 mg Z * (multivitamin); placebo | L and Z | Substantial increase in L (4-fold rise; p < 0.001) and Z (p = 0.007) concentrations |
| Connolly (2010) [213] | MOST | 5 patients with early AMD; aged (72.0 ± 11.0) years in Ireland | 2 months | 7.3 mg MZ, 3.7 mg L and 0.8 mg Z | L, Z and MZ | Significant time effect between rise in all three carotenoid serum levels (p < 0.003 for all) |
| AREDS2 Research Group (2013) [50] | AREDS2 | 4203 patients with intermediate or late AMD; aged (73.1 ± 7.7) years in USA | 5 years | 10 mg L and 2 mg Z * (multivitamin); 10 mg L, 2 mg Z and omega-3 fatty acids * (multivitamin); “placebo” | L and Z | Total serum L + Z levels increased by 190% to 210% from baseline (p < 0.001) |
| Arnold (2013) [206] | - | 20 patients with atrophic AMD; aged (66.0 ± 8.0) years in Germany | 4 weeks | 10 mg L and 3 mg Z, in oleaginous kale extract | L and Z | Statistically significant rise in serum L and serum Z after 4 weeks (p < 0.001 for both) |
| Arnold (2013) [207] | LUTEGA | 172 patients with atrophic AMD; aged (69.0 ± 10.0) years in Germany | 12 months | 10 mg L and 1 mg Z * (multivitamin); 20 mg L and 2 mg Z * (multivitamin); placebo | L and Z | Beneficial alterations seen in both treatment groups (p < 0.05) after one-month and values remained elevated until trial completion |
| Huang (2013) [219] | - | 108 patients with early AMD; aged 50–81 years in China | 48 weeks | 10 mg L; 20 mg L; 10 mg L and 10 mg Z; placebo | L and Z | Greater increase in serum L and Z with high-dose L (6.23-fold) and L + Z formula (3.11-fold), respectively (p < 0.001 for both) |
| Meagher (2013) [221] | - | 27 patients with early AMD; aged (66.0 ± 9.0) years in Ireland | 8 weeks | 20 mg L, 2 mg Z and 0.3 mg MZ; 10 mg L, 2 mg Z and 10 mg MZ; 3 mg L, 2 mg Z and 17 mg MZ | L, Z and MZ | Serum L and Z increased only with higher-dose L (Groups 1 and 2; p < 0.001) while serum MZ increased in all three groups (p < 0.01 for all) |
| Murray (2013) [222] | CLEAR | 72 patients with early AMD; aged (70.5 ± 8.7) years in United Kingdom | 12 months | 10 mg L; placebo | L | Marked increase in serum L (p < 0.001) compared to placebo control |
| Akuffo (2015) [205] | MOST | 52 patients with early AMD; aged (66.0 ± 8.0) years in Ireland | 3 years | 20 mg L and 2 mg Z; 10 mg L, 2 mg Z and 10 mg MZ; 3 mg L, 2 mg Z and 17 mg MZ | L, Z and MZ | Statistically significant time x treatment effect revealed for changes serum L and MZ (p < 0.05 for both) concentrations |
| Huang (2015) [115] | - | 112 patients with early AMD; aged (69.1 ± 7.4) years in China | 24 months | 10 mg L; 20 mg L; 10 mg L and 10 mg Z; placebo | L and Z | Highly significant time x treatment interaction observed for both serum L and Z (p < 0.001 for both) |
| Wolf-Schnurrbusch (2015) [231] | - | 79 patients with early/intermediate AMD; aged 55–88 years in Switzerland | 6 months | 10 mg L and 1 mg Z * (multivitamin); 10 mg L, 1 mg Z and omega-3 fatty acids * (multivitamin); placebo | L and Z | Increases in serum L and Z (p < 0.05) only reported in Group 1 (carotenoid treatment without omega-3 fatty acids in formula) |
| Akuffo (2017) [204] | CREST | 121 patients with early/intermediate AMD; aged (64.7 ± 9.0) years in Ireland | 24 months | 10 mg L and 2 mg Z * (multivitamin); 10 mg L and 10 mg Z * (multivitamin); placebo | L, Z and MZ | Remarkable increase in all three serum concentrations (p < 0.0005); time x group interaction effect only for serum Z and MZ (p < 0.005 for both) |
| Sawa (2020) [229] | Sakai Lutein Study | 39 patients with neovascular AMD; aged (70.7 ± 5.3) years in Japan | 6 months | 20 mg L and 3 mg Z (beeswax capsule); 20 mg L and 3 mg Z (glycerol capsule) | L | Serum L increased in both treatment groups at 3- and 6 months (p < 0.01 for both) |
| García-Layana (2021) [217] | - | 109 patients with neovascular AMD; aged (77.1 ± 7.6) years in Spain | 12 months | 10 mg L and 2.6 mg Z * (multivitamin); original AREDS formula (no L/Z) | L and Z | Substantial increase in serum L and Z (p < 0.001 for both) with a large effect size after 12 months (Cohen’s d of ≥0.80 for both) |
| Authors (Year) | Study | Participants | Duration | Interventions | MPOD | Main Findings |
|---|---|---|---|---|---|---|
| Richer (2004) [225] | LAST | 90 patients with atrophic AMD; aged (74.7 ± 7.4) years in USA | 12 months | 10 mg L; 10 mg L * (multivitamin); placebo | HFP | Significant benefit in MPOD (p < 0.001), BCVA (p < 0.01) and CS at low/middle spatial frequencies (p < 0.05 for all) |
| Bartlett (2007) [209] | - | 25 patients with atrophic AMD; aged (69.2 ± 7.8) years in USA | 9 months | 6 mg L; placebo | - | Non-significant trend towards improvement in CS reported |
| Cangemi (2007) [212] | TOZAL | 37 patients with atrophic AMD; aged (76.3 ± 7.8) years in USA | 6 months | 8 mg L and 0.4 mg Z * (multivitamin) | - | Modest improvements observed in BCVA (p = 0.045) |
| Trieschmann (2007) [119] | LUNA | 100 patients with AMD; aged (71.5 ± 7.1) years in Germany | 6 months | 12 mg L and 1 mg Z * (multivitamin);placebo | Fundus AFI | Mean increase of +15.9% in MPOD measured at 0.5° eccentricity (p < 0.001) compared to control |
| Parisi (2008) [223] | CARMIS | 27 patients with atrophic AMD; aged (65.5 ± 5.1) years in Italy | 12 months | 10 mg L + 1 mg Z * (multivitamin); placebo | - | Enhanced improvement in central retinal function measures on mfERG (ring 1 and ring 2; p < 0.01 for both) |
| Connolly (2010) [213] | MOST | 5 patients with early AMD; aged (72.0 ± 11.0) years in Ireland | 2 months | 7.3 mg MZ, 3.7 mg L and 0.8 mg Z | cHFP | Significant increase in MPOD measured at 0.25° and 1° eccentricity with respect to time (p < 0.05 for all) |
| Richer (2011) [226] | ZVF | 60 patients with early/intermediate AMD; aged (74.9 ± 10.0) years in USA | 12 months | 8 mg Z; 8 mg and 9 mg L; 9 mg L | HFP | Central (1°) MPOD increased in all three groups (p < 0.03 for all); significant improvement in measures of foveal vision greater in Z-only group, while benefits in parafoveal vision were greater in L-only group |
| Weigert (2011) [120] | LISA | 126 patients with early/intermediate AMD; aged (71.6 ± 8.6) years in Austria | 6 months | 20 mg L for 3 months, then 10 mg L for 3 months; placebo | Reflectometry | Average increase of +27.9% in MPOD (p < 0.001); trend toward improvement in BCVA did not reach statistical significance |
| Ma (2012) [117] | - | 108 patients with early AMD; aged 50–81 years in China | 48 weeks | 10 mg L; 20 mg L; 10 mg L and 10 mg Z; placebo | Fundus AFI | Significant dose-response effect with increased MPOD (p < 0.01) positively related to benefits in CS (p < 0.05) and central retina function on mfERG (p < 0.01) |
| Piermarocchi (2012) [224] | CARMIS | 145 patients with atrophic AMD; aged (72.5 ± 7.0) years in Italy | 24 months | 10 mg L + 1 mg Z * (multivitamin); placebo | - | Reported significant benefits in BCVA and CS at 6-, 12-, and 24 months (p < 0.01 for all) compared to placebo |
| AREDS2 Research Group (2013) [50] | AREDS2 | 4203 patients with intermediate or late AMD; aged (73.1 ± 7.7) years in USA | 5 years | 10 mg L and 2 mg Z * (multivitamin); 10 mg L, 2 mg Z and omega-3 fatty acids * (multivitamin); “placebo” | - | Reduced hazard ratios of 0.82 (95% CI:0.69–0.96; p = 0.02) for late AMD and 0.76 (95% CI: 0.64–0.94; p = 0.01) for neovascular AMD compared to β-carotene in formulation † |
| Arnold (2013) [206] | - | 20 patients with atrophic AMD; aged (66.0 ± 8.0) years in Germany | 4 weeks | 10 mg L and 3 mg Z, in oleaginous kale extract | Reflectometry | Enhanced augmentation of macular pigment parameters including volume, area and maxOD (p < 0.001 for all) |
| Beatty (2013) [210] | CARMA | 433 patients with early AMD; aged (73.9 ± 8.1) years in Ireland | 12 months | 12 mg L and 0.6 mg Z * (multivitamin); placebo | Raman spectroscopy | Statistically significant increase in MPOD with a positive linear trend during trial period (p < 0.01 for all) |
| Berrow (2013) [211] | - | 14 patients with early AMD; aged (67.6 ± 8.4) years in UK | 40 weeks | 12 mg L and 0.6 mg Z * (multivitamin); placebo | - | Remarkable benefits in mfERG N1P1 response amplitude densities in ring 3 (p = 0.027); no differential changes observed in BCVA and CS |
| Dawczynski (2013) [215] | LUTEGA | 145 patients with atrophic AMD; aged (70.0 ± 10.0) years in Germany | 12 months | 10 mg L and 1 mg Z * (multivitamin); 20 mg L and 2 mg Z * (multivitamin); placebo | Reflectometry | Significant improvements observed for MPOD parameters (volume, area, maxOD and mean OD) and BCVA (p < 0.001 for all) in both treatment groups |
| García-Layana (2013) [216] | - | 44 patients with early AMD; aged (68.5 ± 8.5) years in Spain | 12 months | 12 mg L and 0.6 mg Z * (multivitamin); placebo | HFP | Considerable rise in MPOD (+0.162 ODU; p < 0.01); however, no significant changes seen in BCVA and CS |
| Murray (2013) [222] | CLEAR | 72 patients with early AMD; aged (70.5 ± 8.7) years in United Kingdom | 12 months | 10 mg L; placebo | cHFP | Highly statistically significant increase in MPOD (+39.5%; p < 0.001) when compared to placebo |
| Sabour-Pickett (2014) [228] | MOST | 52 patients with early AMD; aged (66.0 ± 8.0) years in Ireland | 12 months | 20 mg L and 2 mg Z; 10 mg L, 2 mg Z and 10 mg MZ; 3 mg L, 2 mg Z and 17 mg MZ | cHFP | Robust improvements in MPOD spatial profile observed in those supplemented all three carotenoids in formulation (Group 2, p < 0.005; Group 3, p < 0.05) |
| Akuffo (2015) [205] | MOST | 52 patients with early AMD; aged (66.0 ± 8.0) years in Ireland | 3 years | 20 mg L and 2 mg Z; 10 mg L, 2 mg Z and 10 mg MZ; 3 mg L, 2 mg Z and 17 mg MZ | cHFP | Clinically meaningful CS benefits were seen in all three groups (p < 0.05 for all): Group 1 (15.15 cpd), Group 2 (1.2-, 6- and 9.6 cpd) and Group 3 (6-, 9.6- and 15.15 cpd) |
| Huang (2015) [115] | - | 112 patients with early AMD; aged (69.1 ± 7.4) years in China | 24 months | 10 mg L; 20 mg L; 10 mg L and 10 mg Z; placebo | Fundus AFI | Highly significant time x treatment interaction (p < 0.001) between changes in MPOD and central retinal function improvements (mfERG and MRS; p < 0.05 for both) |
| Thurnham (2015) [230] | - | 32 patients with early AMD; aged (66.0 ± 9.0) years in Ireland | 8 weeks | 20 mg L, 2 mg Z and 0.3 mg MZ; 10 mg L, 2 mg Z and 10 mg MZ; 3 mg L, 2 mg Z and 17 mg MZ | cHFP | Significant increase in all three groups (p < 0.05); Group 2 formulation (10 mg L, 2 mg Z and 10 mg MZ) may offer greater improvement to macular pigment spatial profile |
| Wolf-Schnurrbusch (2015) [231] | - | 79 patients with early/intermediate AMD; aged between 55–88 years in Switzerland | 6 months | 10 mg L and 1 mg Z * (multivitamin); 10 mg L, 1 mg Z and omega-3 fatty acids * (multivitamin); placebo | Fundus AFI | Demonstrable benefits in MPOD (p < 0.005) and CS scores (p < 0.01) observed in Group 1 only (carotenoid treatment without omega-3 fatty acids in formulation) |
| Akuffo (2017) [204] | CREST | 121 patients with early/intermediate AMD; aged (64.7 ± 9.0) years in Ireland | 24 months | 10 mg L, 2 mg Z and 10 mg MZ * (AREDS2 multivitamin); 10 mg L and 10 mg Z * (AREDS2 multivitamin) | cHFP | Augmentation of MPOD (p < 0.001) with clinically meaningful benefits in visual function (CS and GD under mesopic and photopic conditions, photostress recovery, and mean/max reading speed; p < 0.05 for all) |
| Azar (2017) [208] | - | 79 patients with neovascular AMD; aged (75.3 ± 7.6) years in France | 8 months | 5 mg L and 1 mg Z * (multivitamin); placebo | Reflectometry | Non-significant trend toward greater MPOD levels reported in patients with neovascular AMD |
| Corvi (2017) [214] | - | 39 patients with early AMD; aged (78.0 ± 6.5) years in France | 3 months | 10 mg L and 2 mg Z * (multivitamin) | HFP | Significant rise in MPOD only in eyes with reticular pseudodrusen (n = 19; p = 0.002) after 3 months |
| Davey (2020) [53] | - | 56 patients with subclinical AMD; aged (68.4 ± 5.3) years in USA | 6 months | 15 mg L, 10 mg MZ and 2 mg Z * (Lumega-Z); 10 mg L and 2 mg Z * (AREDS-2 multivitamin); placebo | HFP | Statistically significant CS improvements for Lumega-Z group only (p < 0.001) with a positive linear trend with treatment time (p < 0.001) |
| Sawa (2020) [229] | Sakai Lutein Study | 39 patients with neovascular AMD; aged (70.7 ± 5.3) years in Japan | 6 months | 20 mg L and 3 mg Z (beeswax capsule); 20 mg L and 3 mg Z (glycerol capsule) | Fundus AFI | No significant changes were observed in CS, mesopic glare or MPOD in both treatment groups |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lem, D.W.; Davey, P.G.; Gierhart, D.L.; Rosen, R.B. A Systematic Review of Carotenoids in the Management of Age-Related Macular Degeneration. Antioxidants 2021, 10, 1255. https://doi.org/10.3390/antiox10081255
Lem DW, Davey PG, Gierhart DL, Rosen RB. A Systematic Review of Carotenoids in the Management of Age-Related Macular Degeneration. Antioxidants. 2021; 10(8):1255. https://doi.org/10.3390/antiox10081255
Chicago/Turabian StyleLem, Drake W., Pinakin Gunvant Davey, Dennis L. Gierhart, and Richard B. Rosen. 2021. "A Systematic Review of Carotenoids in the Management of Age-Related Macular Degeneration" Antioxidants 10, no. 8: 1255. https://doi.org/10.3390/antiox10081255
APA StyleLem, D. W., Davey, P. G., Gierhart, D. L., & Rosen, R. B. (2021). A Systematic Review of Carotenoids in the Management of Age-Related Macular Degeneration. Antioxidants, 10(8), 1255. https://doi.org/10.3390/antiox10081255









