Abstract
Calcific aortic valve disease (CAVD) and coronary artery disease (CAD) are related cardiovascular diseases in which common mechanisms lead to tissue calcification. Oxidative stress plays a key role in these diseases and there is also evidence that the redox state of serum albumin exerts a significant influence on these conditions. To further explore this issue, we used multimarker scores (OxyScore and AntioxyScore) to assess the global oxidative status in patients with CAVD, with and without CAD, also evaluating their plasma thiol levels. In addition, valvular interstitial cells were treated with reduced, oxidized, and native albumin to study how this protein and its modifications affect cell calcification. The differences we found suggest that oxidative status is distinct in CAVD and CAD, with differences in redox markers and thiol levels. Importantly, the in vitro interstitial cell model revealed that modified albumin affects cell calcification, accelerating this process. Hence, we show here the importance of the redox system in the development of CAVD, emphasizing the relevance of multimarker scores, while also offering evidence of how the redox state of albumin influences vascular calcification. These data highlight the relevance of understanding the overall redox processes involved in these diseases, opening the door to new studies on antioxidants as potential therapies for these patients.
1. Introduction
Calcific aortic valve disease (CAVD), also known as aortic valve (AV) stenosis, and coronary artery disease (CAD), are both progressive conditions with certain similarities, including common clinical risk factors [1]. The initial stages of CAVD and CAD share pathogenic mechanisms, including the endothelial dysfunction that favors inflammatory cell infiltration and lipid deposition in the tissues [2,3,4]. However, as AVs and arteries differ structurally and functionally, tissue stiffness has distinct consequences, and the most severe clinical manifestations in these structures have different causes. Indeed, the clinical manifestations in CAVD are due to obstructed blood flow, while in CAD, plaque stability is critical to avoid the release of prothrombotic agents [5,6].
Oxidative stress, which refers to an imbalance between antioxidant defenses and the production of reactive oxygen species (ROS), plays an important role in the calcification of vascular tissues, including AVs and arteries [7,8,9,10]. It is known that there are important differences between ROS production and the activity of certain enzymes in CAVD and CAD [11,12]. Moreover, when CAD is a comorbidity in CAVD patients, it is associated with alterations to the redox proteome, implying differences in their oxidative statuses [13]. Unfortunately, oxidative stress is not easy to assess due to its complex and multifactorial nature. Measuring individual biomarkers only partially defines the oxidative state, such that they may give rise to erroneous conclusions [14,15]. Hence, no single parameter can be recommended as a gold standard to determine an individual’s redox status. By contrast, global scores of oxidative status can be used that not only include markers of oxidative damage but also biomarkers of antioxidant defenses, offering a more complete overview of oxidative status [16]. Antioxidant capacity can be assessed by chemical-based assays that analyze free radical scavenging activity, such as 2,2′-azinobis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS), 2,2-Diphenyl-1-picrylhydrazylradical (DPPH) or Ferric Reducing Antioxidant Power (FRAP) assays [17,18,19]. Other methods, such as the measurement of the activity of low-molecular-weight antioxidants or antioxidants enzymes, are also usually used, especially in biological samples. This approach, combining individual plasma biomarkers of oxidative damage and antioxidant capacity to assess the overall oxidative balance, has been used successfully in several studies on different cardiovascular diseases (CVD) [14,20,21,22,23].
Considerable attention has been afforded to one potential biomarker of oxidative stress, the redox state of human serum albumin (HSA), a protein that is the most abundant source of thiol groups in human plasma [24]. Several studies have shown that the redox state of HSA is associated with a variety of health conditions, including CVD and type 2 Diabetes Mellitus (T2DM) [25,26]. Classically, the antioxidant activity of HSA has been attributed to the presence of the free sulfhydryl group of cysteine at position 34 (Cys34) [27]. Nevertheless, S-thiolation has also been described at different disulfide bonds of HSA [28]. Moreover, we recently found two new sites of oxidation and highlighted the implication of oxidized HSA for the development of CAVD in patients with CAD, maybe due to the transport of low-molecular-weight thiols inside the AV tissue [13]. Interestingly, endothelial cells cultured with oxidized albumin exhibit an increase in the markers of endothelial damage, as well as enhanced expression of inflammatory cytokines that is also related to vascular calcification [29]. However, the effects of oxidized albumin on valvular interstitial cells (VICs), the major cell population in heart valves, has not yet been studied.
Despite the known association between CAVD and CAD, the effects of the presence of CAD in CAVD patients in terms of global oxidative status have not been described. Here we measured different markers of oxidative damage and antioxidant defense, including the thiol levels in the plasma of patients, to gain a deeper understanding of the implication of HSA in these conditions. In addition, we also studied the effects of oxidized, reduced and native HSA on VICs in vitro, demonstrating its importance in the calcification process (Figure 1). The use of in vitro models is currently a cost-effective way to develop new pharmacological treatments. Moreover, understanding the global redox state associated with CAVD and CAD is key to taking a step forward towards precision personalized medicine, facilitating the prescription of adequate therapies to these patients.
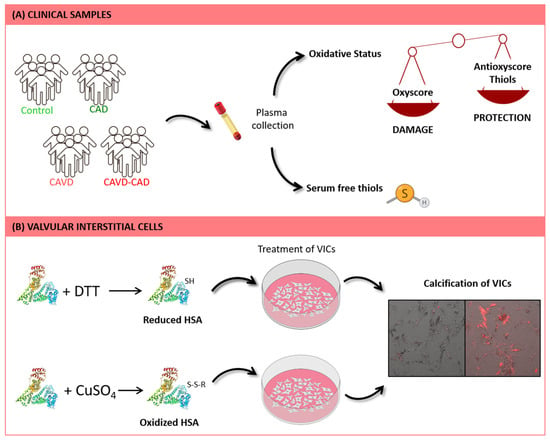
Figure 1.
Workflow of the study. (A) Different markers of oxidative damage and antioxidant defense, including thiol levels, were measured in plasma samples from patients with CAVD, with and without CAD. (B) Human VICs were treated with oxidized, reduced, and native HSA. Subsequently, calcification of these cells was assessed by using Alizarin Red staining. Microscope images size is 665.6 µm × 665.6 µm.
2. Materials and Methods
2.1. Patient Selection
In this cross-sectional study, peripheral blood samples from 4 groups of subjects were collected: (i) controls without CAVD or CAD (C, n = 19); (ii) subjects with CAD alone (CAD, n = 16); (iii) patients with CAVD alone (CAVD, n = 20); and (iv) patients with both CAVD and CAD (CAVD + CAD, n = 20). All the patients were recruited from the Hospital General Universitario de Toledo (Spain).
Patients with any severe morbidity (e.g., ischemic heart disease with ventricular dysfunction or end-stage chronic kidney disease—CKD), any type of DM, bicuspid AV, a family or personal history of aortopathy, rheumatic valve disease or moderate or severe mitral valve disease were excluded from the study. Significant differences in terms of cardiovascular risk factors (gender, obesity, hypertension, and dyslipidemia) were avoided. The blood samples were collected in tubes containing EDTA and centrifuged for 10 min at 1125× g, and the resulting supernatant was immediately frozen at −80 °C until analysis. Collection, processing and storage were constant among the samples to avoid differences due to experimental handling.
This study was carried out in accordance with the guidelines of the Helsinki Declaration and the study design was approved by the local ethics committee. Signed informed consent was obtained from all individuals prior to their inclusion in the study.
2.2. Biomarkers of Oxidative Damage
Oxidative damage to proteins, lipids and DNA was evaluated by measuring protein carbonyls, oxidized low-density lipoprotein (oxLDL) and 8-hydroxy-20-deoxyguanosine (8-OHdG) levels. Protein carbonyl groups were measured using 2,4-dinitrophenylhydrazine in a protocol adapted to a microplate reader [30], and they were expressed as nmol/mg of total protein. OxLDL and 8-OHdG were assayed using commercial enzyme-linked immunosorbent assay (ELISA) kits (Mercodia AB, Uppsala, Sweden, and Stress-MarqBiosciences Inc., Victoria, BC, Canada, respectively), following the manufacturer’s instructions. Pro-oxidant xanthine oxidase (XOD) activity was determined with the Amplex Red assay (Invitrogen, Carlsbad, CA, USA) and expressed as mU/mg of total protein.
2.3. Biomarkers of Antioxidant Defense
Catalase and superoxide dismutase (SOD) activity was assessed as markers of enzymatic antioxidant activity. Plasma catalase activity was measured using the Amplex Red assay (Invitrogen) and expressed as U/mg of total protein. SOD activity was estimated using a colorimetric assay (Invitrogen, Carlsbad, CA, USA) and expressed as mU/mg of total protein. The overall activity of low-molecular-weight antioxidants or the total antioxidant capacity (TAC) was determined using an enhanced horseradish–peroxidase catalyzed-based luminol chemiluminescence assay adapted to a microplate reader [31]. Luminescence inhibition by the addition of plasma was used to calculate the area under the curve (AUC).
2.4. Serum Free Thiol Levels
Plasma thiol compounds were assayed using a SensoLyte® Thiol Quantitation Assay Kit (AnaSpec, Fremont, CA, USA) according to the manufacturer’s instructions. This assay is based on the reaction of the sulfhydryl group of thiols with Ellman’s Reagent, generating 2-nitro-5-thiobenzoic acid (TNB) that in turn produces a yellow color that can be detected at 420 nm absorbance. The intensity of the color produced is proportional to the thiol concentration and it is expressed as µM thiol.
2.5. OxyScore and AntioxyScore
Multimarker scores of oxidative damage were computed as described previously [14,32,33,34]. Briefly, markers of oxidative damage or antioxidant defenses were standardized for each subject, using healthy subjects as a reference. The sum of the standardized values for protein carbonyls, oxLDL, 8-OHdG, and XOD activity was used to calculate the score of oxidative damage (OxyScore), whereas the sum of the standardized values of catalase and SOD activity and that of the TAC were used to calculate the score of global antioxidant defense (AntioxyScore).
2.6. Valvular Interstitial Cell Culture
Human cardiac VICs (Innoprot, Derio, Spain, P10462) were used in this study. These cells were isolated from heart valves and cryopreserved at passage one, after which they are guaranteed to expand further by 10 population doublings under the conditions indicated in the data sheet. VICs were cultured in Fibroblast Medium-2 (FM-2: Innoprot, Derio, Spain), designed for the optimal growth of normal human cardiac fibroblasts in vitro, a medium that contains essential and non-essential amino acids, vitamins, organic and inorganic compounds, hormones, growth factors, trace minerals, and a low concentration of fetal bovine serum (FBS, 5%). In these experiments, the cells were cultured for 7 days in two different media: (i) a special medium for fibroblasts (FIBm) that favors a quiescent phenotype (Hyclone Dulbecco’s Modified Eagle Medium (DMEM)), supplemented with 2% heat-inactivated FBS, 150 U/mL penicillin/streptomycin, 2 mM L-glutamine, 10 ng/mL fibroblast growth factor (FGF-2) and 50 ng/mL insulin and (ii) osteogenic medium (OSTm), to induce osteogenic differentiation of the HAVICs (FIBm supplemented with 50 µg/mL ascorbic acid, 10 mM β-glycerophosphate and 100 nM dexamethasone) [35,36]. As we observed differences in calcification after 24 h of culture (Figure S1), different concentrations of HSA were added to both these media and treatment was maintained during that period of time: either native HSA, OxHSA or RedHSA at a final concentration of 0.5, 1 and 2 mg/mL.
2.7. Preparation of Modified HSA
Oxidized HSA (OxHSA) was prepared following the protocol described previously, with minor modifications [37]. Briefly, HSA (Sigma Aldrich, St. Louis, MO, USA, Ref. A9731-5G) was diluted to 0.01 mM and incubated with CuSO4 (15 µmol/L) for 24 h at 37 °C before adding 0.15 mmol/L EDTA to prevent further oxidation. OxHSA was concentrated using regenerated cellulose membrane columns with a cutoff of 10 kDa, and the protein concentration was determined using a NanoDrop (Thermo Scientific, Waltham, MA, USA). The level of protein oxidation was determined with the OxyBlot Protein Oxidation Detection Kit, according to manufacturer’s specifications (Sigma Aldrich, St. Louis, MO, USA, Ref. S7150). Briefly, the OxyBlot kit derivatizes carbonyl groups to a 2,4-dinitrophenylhydrazone (DNP) moiety, which can then be detected with anti-DNP antibodies.
Albumin reduction was achieved as described previously, with minor modifications [29]. Diluted HSA (0.01 mM) was incubated with a 0.5 mM DTT solution for 1.5 h at 37 °C. At the end of the incubation, DTT was removed using regenerated cellulose membrane columns with a 10 kDa cutoff and the protein concentration was determined using a NanoDrop. To analyze protein reduction, reduced HSA (RedHSA) was labeled with SulfoBiotics PEG-PCMal (Dojindo Molecular Technologies Inc., Rockville, MD, USA, Ref. SB20-01), a 5 kDa Protein-SHifter, in accordance with the manufacturer’s instructions. Subsequently, RedHSA was analyzed by immunodetection as described previously [13].
2.8. Alizarin Red Staining
The cells were washed with PBS, fixed with 4% paraformaldehyde for 15 min, and then incubated for 10 min with Alizarin Red S (Sigma Aldrich, St. Louis, MO, USA) [38]. After washing with deionized water, calcium deposition was visualized under an Olympus IX83 inverted microscope, capturing 49 images per well, which were analyzed with ScanR software (v3.4.1., Olympus, Tokyo, Japan). Each of these experiments were performed in triplicate.
2.9. Statistical Analysis
Statistical analyses were performed using GraphPad Prism software (v.8.0.2., GraphPad Software Inc., San Diego, CA, USA) and SPSS software for Windows (v..15.0., SPSS Inc., Chicago, IL, USA). First, normality was assessed with the Kolmogorov–Smirnov test, and consequently, normally distributed variables were analyzed by parametric tests and those distributed non-normally were analyzed with non-parametric tests. Differences between the groups for the clinical parameters with discrete variables were calculated using a chi-squared test, while continuous variables such as oxidative markers were calculated by one-way ANOVA with a Bonferroni’s post hoc analysis for the four-group comparisons and adjusted for age and dyslipidemia. The descriptive data were presented as the mean ± standard deviation (SD) or as percentages.
3. Results
In this work, we initially analyzed clinical samples to determine the global oxidative statuses of four groups of subjects: controls without CAVD or CAD, subjects with CAD alone, patients with CAVD alone, or patients with both CAVD and CAD. We assessed markers of oxidative damage and of antioxidant defenses, as well as evaluating any reduction in thiol levels. In addition, we used an in vitro model to study the direct effect of the HSA redox state on the calcification of VICs.
3.1. Characteristics of the Study Population
The clinical characteristics of the study groups were compared (Table 1) and there were no significant differences between the groups in terms of the main cardiovascular risk factors, with the exception of the lower mean age of the controls when compared with the other groups. As dyslipidemia was almost significant, results are adjusted for these two factors: age and presence of dyslipidemia. Patients with DM were excluded from the study in order to minimize the presence of confounding factors.

Table 1.
Clinical characteristics of the subjects studied: C, control without calcific aortic valve disease or coronary artery disease; CAD, control with coronary artery disease; CAVD, calcific aortic valve disease without coronary artery disease; CAD, calcific aortic valve disease with coronary artery disease. Abbreviations: M/F, male/female; AHT, Arterial Hypertension; p, p-value.
3.2. Markers of Oxidative Damage and of Antioxidant Defenses
Oxidative damage to DNA was measured using the 8-OHdG levels, and it was significantly higher in patients with CAVD and CAD (63.34 ± 16.09 ng/mL) than in the controls (41.84 ± 7.07 ng/mL, adjusted p-value = 0.00) or patients with CAD alone (34.56 ± 12.41 ng/mL, adjusted p-value = 0.00: Figure 2A). There were also differences in this parameter between controls and patients with CAVD alone (adjusted p-value = 0.012: Figure 2A). Stronger pro-oxidant XOD enzymatic activity was detected in both groups of patients with CAD, although the differences between these subjects with and without CAVD were not significant (Figure 2A). In contrast, the protein carbonylation and oxLDL levels did not vary significantly between the three groups (Figure 2A). Similarly, catalase and SOD activity did not differ between the three groups of patients studied (Figure 2B), and along similar lines, the luminescence in the TAC assay was also similar in each group (Figure 2B). All mean values and the remaining statistical details are shown in Supplementary Tables S1 and S2.
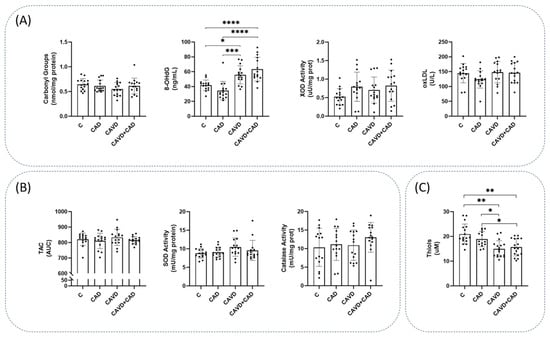
Figure 2.
Markers of oxidative status in plasma from the four groups of study. (A) Markers of oxidative damage, including protein carbonyls, 8-hydroxy-2′-deoxyguanosine (8-OHdG) xanthine oxidase (XOD) activity and oxidized LDL (oxLDL) (n = 15 subjects/each group). (B) Markers of antioxidants defense, including total antioxidant capacity (TAC), superoxide dismutase (SOD) activity and catalase (CAT) activity (n = 15 subjects/each group). (C) Free reduced thiols (n = 14 subjects/CAD group and n = 17 subjects/C, CAVD and CAVD + CAD group). Data are represented as the mean ± SD. AUC, Area under the curve; C, Controls; CAD, Coronary artery disease; CAVD, Calcific aortic valve disease. * p < 0.05, ** p < 0.01, *** p < 0.001, **** p < 0.0001.
3.3. Serum Thiol Levels
After measuring the thiols, we observed lower levels of reduced thiols in conjunction with the pathologies (controls = 20.94 ± 4.08 µM; CAD = 18.93 ± 3.05 µM; CAVD = 14.98 ± 3.31 µM; and CAVD + CAD = 15.63 ± 3.50 µM: Figure 2C). Significant differences existed between the controls and the CAVD patients, both without and with CAD (adjusted p-value < 0.01 in both cases), as well as between patients with CAD alone and patients with both pathologies (adjusted p-value < 0.05, see Supplementary Table S2 for the remaining statistical details).
3.4. Global Oxidative Status
We found that the multimarker score of oxidative damage (OxyScore) was significantly higher in patients with CAD and CAVD (2.54 ± 2.41) than in the control patients (−0.87 ± 1.85, adjusted p-value = 0.001) or in patients with CAD alone (−2.15 ± 2.09, adjusted p-value = 0.000). By contrast, there were no differences in the multimarker antioxidant defense scores (AntioxyScore) of the groups (Figure 3, see Supplementary Tables S1 and S2 for all the mean values and the remaining statistical details).
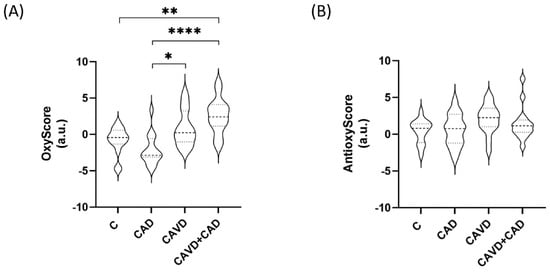
Figure 3.
Multimarker scores of (A) oxidative damage (OxyScore) and (B) antioxidant defense (AntiOxyscore) (n = 15 subjects/each group). C, Controls; CAD, Coronary artery disease; CAVD, Calcific aortic valve disease. * p < 0.05, ** p < 0.01, **** p < 0.0001.
3.5. Cell Calcification on Exposure to HSA
Human VICs were cultured in the presence of different concentrations of HSA, including basal, RedHSA and OxHSA. HSA modifications were confirmed by immunoblot (Figure 4A). After 24 h in culture with FIBm or OSTm, differences in calcification were evident, and specifically, the calcification of cells cultured in FIBm augmented when exposed to higher concentrations of OxHSA (Figure 4B,C: p-values < 0.005 in all cases). Surprisingly, cells respond in a different manner to HSA when cultured in FIBm or OSTm. In OSTm, the cells appeared to calcify more in the presence of high concentrations of RedHSA (Figure 4B,D), with significant differences when compared to the cells maintained in OSTm alone (p-values < 0.005 for the three concentrations, the mean values and the remaining statistical details can be found in Supplementary Tables S3 and S4).
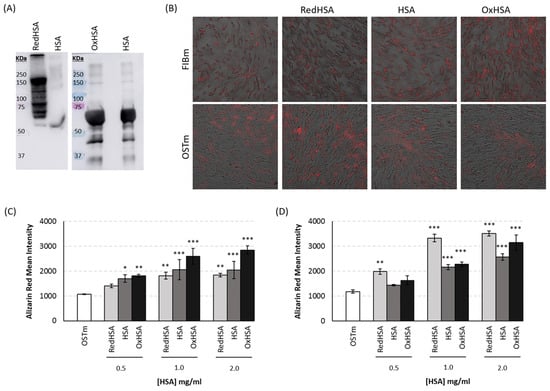
Figure 4.
Results from in vitro model. (A) Immunoblot of modified HSA. Reduced HSA (RedHSA) was labeled with SulfoBiotics-PEG-PCMal, which produces a mobility shift corresponding to approximately 5 kDa for each molecule of PEG-PCMal bound to a free thiol group of the target protein. Thus, after the blotting, several bands corresponding to RedHSA are observed (from 60 to 150 kDa), while the lane of native HSA only shows one band. Oxidized HSA (OxHSA) was evaluated using OxyBlot Protein Oxidation Detection Kit. This kit allows the immunodetection of carbonyl groups through DNP-derivatization and subsequent detection of this DNP moiety with a specific primary antibody. It can be observed that the bands corresponding to OxHSA are more intense than the bands corresponding to native HSA. (B) Representative images of the Alizarin Red staining at a concentration of 1 mg/mL of modified or native HSA after 24 h of treatment. Microscope images size is 665.6 µm × 665.6 µm. (C) Calcification levels of cells cultured for 24 h in medium for fibroblast (FIBm) when supplemented with different concentrations of modified or native HSA. (D) Calcification levels of cells cultured for 24 h in osteogenic medium (OSTm) when supplemented with different concentrations of modified or native HAS was measured using Alizarin red staining. All experiments were performed in triplicate. * p < 0.05, ** p < 0.01, *** p < 0.001.
4. Discussion
Oxidative stress represents an imbalance between the production of ROS and the ability to detoxify reactive products and/or repair the resulting damage. The toxic effects of oxidative stress caused by peroxide and free radical production produce damage in all cell components, affecting proteins, lipids and DNA. Despite the importance of oxidative stress in biology and medicine, it is a phenomenon that is still challenging to measure. ROS are highly reactive, and consequently, they have short half-lives in biological environments, making them difficult to measure directly. Indeed, even indirect estimates of their abundance and reactivity are complicated. As such, a common method to assess oxidative stress is to measure the macromolecules oxidized (lipids, proteins, and DNA) and the antioxidants (enzymatic and non-enzymatic antioxidants) [39,40].
Oxidative stress is implicated in pathophysiological vascular calcification, and it plays a significant role in the development and progression of CAVD and CAD, even being able to predict cardiovascular events [41,42,43,44,45,46]. Given this association, we evaluated here the global oxidative status associated with both these pathologies using the multimarker parameters, OxyScore and AntioxyScore, as well as the plasma thiol levels. In addition, the effect of the redox state of HSA on VICs in culture was also studied.
4.1. Oxidative Status
Higher levels of 8-OHdG were found in CAVD patients relative to the controls, which indicates more severe oxidative damage to DNA. Higher levels of 8-OHdG were associated with an unfavorable 30-day prognosis following AV replacement and better discrimination of standard clinical models predicting the 30-day and 1-year risk of standardized end-points [47,48]. Indeed, we found higher levels of 8-OHdG in patients with both CAVD and CAD, consistent with the fact that CAD is a negative predictive indicator in patients with CAVD [49,50,51]. It is important to highlight here that the differences between patients with and without CAD are not significant, although a clear tendency does exist. Previous studies showed the importance of 8-OHdG in the atherosclerotic process, and high levels of 8-OHdG have been found in fragments of aorta from patients with severe atherosclerotic lesions. In addition, 8-OHdG levels have been correlated with the number of vessels affected [52,53] and, in leukocyte mtDNA in diabetic patients, with coronary stenosis severity [54]. An extended meta-analysis of 14 studies showed that the association between 8-OHdG levels and CVD is largely independent of diabetes, hyperlipidemia, body mass index and smoking habits. By contrast, the association between 8-OHdG levels and CVD was stronger in younger subjects, and a higher prevalence of hypertension was associated with smaller differences in 8-OHdG levels between CVD patients and controls [55]. This phenomenon might be explained by the reduced DNA turnover associated with aging [56] and by the higher levels of 8-OHdG found in hypertensive subjects relative to normotensive controls [57]. The use of ELISA kits for the measurement of 8-OHdG should also be highlighted. Although the gold standard for 8-OHdG is high performance liquid chromatography coupled with an electrochemical detector, immunological methods, such as ELISA, are less costly and time consuming than chromatography. Furthermore, ELISA kits are tools widely used in clinical practice as they are used in surveillance and disease monitoring as well as diagnostic tools. The use of these methodologies at these stages of the research is a step forward in looking for clinical application and facilitates its implementation. It is important to note that the cohort of patients with severe CAVD studied here has an average age above 75 years old. Moreover, hypertension is more prevalent in patients with CAVD, such that we cannot rule out this comorbidity from the study [58]. To overcome this issue, we avoided significant differences in hypertension among the study groups. For those reasons, the differences we found in 8-OHdG levels may be less pronounced than those described elsewhere.
Although we do not find more significant differences in markers of oxidative damage, by focusing on XOD activity, a non-significant increase in ROS was evident in CAD patients relative to non-CAD patients. This enzyme is implicated in ROS production [59], and it is believed to promote inflammatory responses and atherosclerotic plaque formation [60], consistent with the enhanced activity found in these patients.
Regarding antioxidant levels, it has been previously described that expression and activity of antioxidant enzymes are reduced in calcified regions of stenotic AV when compared with non-calcified regions. Additionally, although oxidative stress appears to be increased in stenotic AV, the mechanisms that account for this oxidative stress differ greatly from those observed in atherosclerotic arteries [11]. Supporting our data from previous studies, our results showed no differences in individual markers of antioxidant defense [14].
Importantly, we found fewer reduced thiol groups in patients with CAVD, groups that offer protection against oxidative stress through ROS scavenging. In fact, low levels of serum free thiols are associated with a higher risk of cardiovascular events [61]. Recently, an analysis of four cohorts from different European countries observed associations of both high derivatives of reactive oxygen metabolites levels and low total thiol levels with fatal myocardial infarction and stroke, two atherosclerotic diseases [62]. The results suggest an important contribution of an imbalanced redox system to the etiology of mainly fatal MI and stroke events. Indeed, the results indicated that thiol levels may be an important factor in the development and progression of CVD, including AV disease in the context of atherosclerosis, as we hypothesized previously [63].
In considering the global scores, we observed a gradual increase in the OxyScore of patients with CAVD, both with and without CAD, relative to the controls. By contrast, the AntioxyScore does not vary significantly. There is evidence that oxidative stress may activate antioxidant defense mechanisms, for example through the activity of transcription factors like Nrf2 [64]. However, other studies showed that an excessive amount of ROS will produce an impairment in the antioxidant system, dampening the activity of antioxidant enzymes [65]. We previously found that despite an increase in oxidative end-products, patients with CAVD and T2DM do not respond to oxidative insults [14], as evident here. In fact, patients with CAVD and CAD have higher OxyScore and lower AntiOxyscore values than patients with CAVD alone, although these differences were not significant. In the light of this, we hypothesized that these patients have weaker cellular resilience to oxidative stressors, and as such, they are unable to counteract the harmful effects of ROS through such mechanisms.
4.2. How the Human Serum Albumin Redox State Affects Valvular Interstitial Cells
As we have discussed above, patients with CAVD have lower levels of reduced thiol groups than controls, i.e., a lower capacity for protection against ROS. This result highlights the importance of HSA in both CAVD and CAD, as this protein plays a key role in the antioxidative capacity of blood plasma and its ability to manage ROS [66] given that it is responsible for trapping more than 70% of the free radicals in plasma [67]. However, more studies will be needed to fully understand the role of thiols and HSA in CAVD and CAD.
The redox state of HSA has been associated with greater cardiovascular risk in patients with other comorbidities, such as CKD or COVID-19 infection [68,69,70]. Moreover, patients with CAVD and CAD have more circulating OxHSA in their plasma [13]. Here, to assess if this circulating HSA may be harmful to VICs, we studied the effects of different concentrations of modified HSA in vitro. As a result, calcification was observed to be affected by exposure to RedHSA and OxHSA, although interestingly, differences were found when HSA was added to FIBm or OSTm. Cell calcification is stronger when FIBm is supplemented with OxHSA, consistent with the previous hypothesis that oxidized circulating HSA might act as a carrier of low molecular weight thiols, which could in turn be released into the subendothelial space and aggravate the oxidative stress suffered by local tissues [13]. In other cell populations, such as endothelial cells or neutrophils, OxHSA produces an increase in apoptosis, inflammation, senescence and intracellular ROS production, thereby enhancing oxidative stress and cell damage [37,71] (Figure 5). Importantly, we also found enhanced calcification with non-modified HSA, which makes sense, as it usually undergoes some degree of oxidation. Alternatively, calcification is more pronounced in OSTm supplemented with RedHSA, the antioxidant activity of which is known to be stronger than that of OxHSA [72]. The binding properties of HSA depend on the three-dimensional structure of the binding sites, a structure that is altered by redox modifications [73]. Due to these structural changes, the redox state influences the drug binding properties of HSA. Thus, we hypothesize that RedHSA has a stronger affinity for the osteogenic components of OSTm (ascorbic acid, β-glycerophosphate, and dexamethasone), increasing the uptake of these substances by VICs (Figure 4). This would explain why these cells calcified in a more intense manner. If we consider the important role of has as a drug carrier, this result could be key in the clinical field for therapeutic purposes.
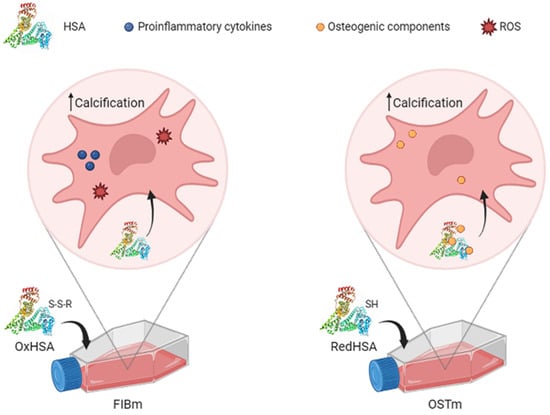
Figure 5.
Hypothesis about the effect of oxidized HSA (OxHSA) in medium for fibroblasts (FIBm) and reduced HSA (RedHSA) in osteogenic medium (OSTm). In both cases, an intense calcification is observed in valvular interstitial cells, but OxHSA is believed to induce proinflammatory cytokines and reactive oxygen species, while RedHSA may act as a carrier of the osteogenic components that have the OSTm.
Here, we obtain evidence of the importance of the redox system in the development of CAVD. We found that in patients with both CAVD and CAD, CAVD has a stronger effect on the oxidative status than CAD. There has been considerable interest in antioxidants in recent years for their protective role in CVD, even in conjunction with CAVD and CAD, and through different mechanisms such as NADPH Oxidase 2 inhibition [42,74,75]. Better understanding of which redox processes are affected in these conditions will be essential to selecting the most adequate therapy for each patient. The application of multimarker scores may play a significant role in selecting such therapies, as they provide information about the different mechanisms that are implicated in maintaining the redox balance. In addition, in vitro models are important to understand the effects of different substances on specific tissues. We demonstrate that the redox state of HSA influences calcification, and we have evidence of differential transport rates according to that state. It is important to highlight the importance of this kind of study to assess the influence of different mediators of oxidative stress. These molecules have a physiological role, and in many cases, the therapeutic goals of non-targeted broad-spectrum antioxidants fail [76]. For that reason, such studies must adopt a precision medicine perspective. Indeed, understanding the specific redox changes that occur in patients with CAVD and different comorbidities, such as CAD, is essential, as patients with different comorbidities could have an additive accumulation of redox biomarkers [77]. These studies will pave the way for the design of new therapeutic strategies that may slow down the course of the disease, such as through the development of targeted drugs.
The main limitation of this study is that the sample size included is relatively small, as well as the heterogeneity in age. Although we excluded patients with DM, it was impossible to avoid other risk factors, such as hypertension, due to its elevated prevalence in patients with developed CVD. Thus, further studies on larger cohorts will be necessary to obtain more information about the effects of these comorbidities and to achieve better patient stratification based on their clinical characteristics. Importantly, different cardiovascular drugs, such as statins, have antioxidant properties that may affect outcomes. Unfortunately, information about patient medication is lacking, which is also a limitation of this study. Furthermore, additional in vitro studies should be performed to characterize the different structural modifications albumin undergoes.
5. Conclusions
Here, we provide information about the redox alterations found in patients with CAVD and CAD, which indicates that these impair the antioxidant responses in these patients such that they fail to fully neutralize the harmful effects of ROS. In addition, we demonstrate that different HSA redox states affect valvular cell calcification in different manners, potentially accelerating this process. These results highlight the importance of understanding the whole redox processes taking place in these diseases, as these mechanisms differ in each pathology. As such, future studies might focus on antioxidants as personalized therapies for these patients from a precision medicine perspective.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/antiox13010108/s1, Figure S1. Calcification levels of cells cultured for 7 days in medium for fibroblast (FIBm) and osteogenic medium (OSTm). Results show significant differences in calcification after 24 h of culture. ** p < 0.01, *** p < 0.001; Table S1. Antioxidant defense markers of oxidative damage (the mean ± SD is shown); Table S2. Comparison of antioxidant defense markers and markers of oxidative damage between the different groups studied (age and dyslipidemia-adjusted p-values are shown); Table S3. Calcification of VICs (the mean intensity of Alizarin Red staining is shown): FIBm, medium for fibroblast medium; HSA, human serum albumin; OSTm, osteogenic medium; OxHSA, oxidized human serum albumin; RedHSA, reduced human serum albumin; Table S4. Comparison of the treatments of VICs in vitro (p-values are shown): FIBm, medium for fibroblast medium; HSA, human serum albumin; OSTm, osteogenic medium; OxHSA, oxidized human serum albumin; RedHSA, reduced human serum albumin. The p-values < 0.05 are shown in bold and italics.
Author Contributions
Conceptualization: L.M.-A., M.G.B. and G.R.-H.; methodology: L.M.-A., M.G.B. and G.R.-H.; validation: T.S.-O., N.C.-A., E.R.-S., E.M.-G., I.P.-S., G.H.-F., C.J.-A. and E.M.-N.; formal analysis: L.M.-A., N.L.-A., G.R.-H. and E.M.-N.; investigation: T.S.-O., N.C.-A., E.R.-S., E.M.-G., I.P.-S., G.H.-F., C.J.-A. and E.M.-N.; resources: L.F.L.-A., T.T., L.R.P. and P.L.S.; writing—original draft: L.M.-A. and M.G.B.; writing—review and editing: L.M.-A., M.G.B., G.R.-H., N.L.-A., L.R.P., T.T., L.F.L.-A. and P.L.S.; visualization: L.M.-A. and M.G.B.; supervision: L.M.-A., M.G.B., G.R.-H. and N.L.-A.; project administration: M.G.B.; funding acquisition: M.G.B. All authors have read and agreed to the published version of the manuscript.
Funding
This study has been funded by Instituto de Salud Carlos III through the projects PI18/00995, PI20/00763, PI21/00384, PI23/00182, AC 22/00022 (European Joint Programme Rare disease –EJPRD2249-), CPII20/00022, FI19/00157 and co-funded by the European Union, Sociedad Española de Cardiología, 2020, the Spanish Society of Cardiology (SEC)/Spanish Foundation of Heart (SEC/FEC-INV-BAS 23), European Union, Biomedicine Network Comunidad de Madrid (P2022/BMD-7223 CIFRA_COR-CM), Spanish Network (RED2022-134511-T), Junta de Comunidades de Castilla-La Mancha (JCCM, co-funded by the European Regional Development Fund, SBPLY/19/180501/000226, SBPLY/21/180501/000078). These results are aligned with the Spanish initiative on the Human Proteome Project (SpHPP).
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of “Complejo Hospitalario Universitario de Toledo” (Toledo, Spain) (protocol code 878).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
Data are contained within the article and supplementary material.
Acknowledgments
The authors thank the Service of Microscopy and Image Analysis at the HNP.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Katz, R.; Wong, N.D.; Kronmal, R.; Takasu, J.; Shavelle, D.M.; Probstfield, J.L.; Bertoni, A.G.; Budoff, M.J.; O’Brien, K.D. Features of the metabolic syndrome and diabetes mellitus as predictors of aortic valve calcification in the Multi-Ethnic Study of Atherosclerosis. Circulation 2006, 113, 2113–2119. [Google Scholar] [CrossRef]
- Ross, R. Atherosclerosis—An inflammatory disease. N. Engl. J. Med. 1999, 340, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Otto, C.M.; Kuusisto, J.; Reichenbach, D.D.; Gown, A.M.; O’Brien, K.D. Characterization of the early lesion of ‘degenerative’ valvular aortic stenosis. Histological and immunohistochemical studies. Circulation 1994, 90, 844–853. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, K.D.; Reichenbach, D.D.; Marcovina, S.M.; Kuusisto, J.; Alpers, C.E.; Otto, C.M. Apolipoproteins B, (a), and E accumulate in the morphologically early lesion of ‘degenerative’ valvular aortic stenosis. Arter. Thromb. Vasc. Biol. 1996, 16, 523–532. [Google Scholar] [CrossRef] [PubMed]
- van der Wal, A.C.; Becker, A.E.; van der Loos, C.M.; Das, P.K. Site of intimal rupture or erosion of thrombosed coronary atherosclerotic plaques is characterized by an inflammatory process irrespective of the dominant plaque morphology. Circulation 1994, 89, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Naghavi, M.; Libby, P.; Falk, E.; Casscells, S.W.; Litovsky, S.; Rumberger, J.; Badimon, J.J.; Stefanadis, C.; Moreno, P.; Pasterkamp, G.; et al. From vulnerable plaque to vulnerable patient: A call for new definitions and risk assessment strategies: Part I. Circulation 2003, 108, 1664–1672. [Google Scholar] [CrossRef]
- Kimura, Y.; Tsukui, D.; Kono, H. Uric acid in inflammation and the pathogenesis of atherosclerosis. Int. J. Mol. Sci. 2021, 22, 12394. [Google Scholar] [CrossRef] [PubMed]
- Vichova, T.; Motovska, Z. Oxidative stress: Predictive marker for coronary artery disease. Exp. Clin. Cardiol. 2013, 18, e88–e91. [Google Scholar]
- Heistad, D.D.; Wakisaka, Y.; Miller, J.; Chu, Y.; Pena-Silva, R. Novel aspects of oxidative stress in cardiovascular diseases. Circ. J. 2009, 73, 201–207. [Google Scholar] [CrossRef]
- Miller, J.D.; Weiss, R.M.; Heistad, D.D. Calcific aortic valve stenosis: Methods, models, and mechanisms. Circ. Res. 2011, 108, 1392–1412. [Google Scholar] [CrossRef]
- Miller, J.D.; Chu, Y.; Brooks, R.M.; Richenbacher, W.E.; Peña-Silva, R.; Heistad, D.D. Dysregulation of antioxidant mechanisms contributes to increased oxidative stress in calcific aortic valvular stenosis in humans. J. Am. Coll. Cardiol. 2008, 52, 843–850. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.E.; Sung, J.M.; Andreini, D.; Al-Mallah, M.H.; Budoff, M.J.; Cademartiri, F.; Chinnaiyan, K.; Choi, J.H.; Chun, E.J.; Conte, E.; et al. Association between aortic valve calcification progression and coronary atherosclerotic plaque volume progression in the PARADIGM registry. Radiology 2021, 300, 79–86. [Google Scholar] [CrossRef]
- Sastre-Oliva, T.; Corbacho-Alonso, N.; Albo-Escalona, D.; Lopez, J.A.; Lopez-Almodovar, L.F.; Vázquez, J.; Padial, L.R.; Mourino-Alvarez, L.; Barderas, M.G. The influence of coronary artery disease in the development of aortic stenosis and the importance of the albumin redox state. Antioxidants 2022, 11, 317. [Google Scholar] [CrossRef] [PubMed]
- Corbacho-Alonso, N.; Rodriguez-Sanchez, E.; Sastre-Oliva, T.; Mercado-Garcia, E.; Perales-Sanchez, I.; Juarez-Alia, C.; Lopez-Almodovar, L.F.; Padial, L.R.; Tejerina, T.; Mourino-Alvarez, L.; et al. Global oxidative status is linked to calcific aortic stenosis: The differences due to Diabetes Mellitus and the effects of metformin. Antioxidants 2023, 12, 1024. [Google Scholar] [CrossRef] [PubMed]
- Veglia, F.; Cighetti, G.; De Franceschi, M.; Zingaro, L.; Boccotti, L.; Tremoli, E.; Cavalca, V. Age- and gender-related oxidative status determined in healthy subjects by means of OXY-SCORE, a potential new comprehensive index. Biomarkers 2006, 11, 562–573. [Google Scholar] [CrossRef]
- Griendling, K.K.; Touyz, R.M.; Zweier, J.L.; Dikalov, S.; Chilian, W.; Chen, Y.R.; Harrison, D.G.; Bhatnagar, A. Measurement of reactive oxygen species, reactive nitrogen species, and redox-dependent signaling in the cardiovascular system: A scientific statement from the American Heart Association. Circ. Res. 2016, 119, e39–e75. [Google Scholar] [CrossRef] [PubMed]
- Soetan, O.A.; Ajao, F.O.; Ajayi, A.F. Blood glucose lowering and anti-oxidant potential of erythritol: An in vitro and in vivo study. J. Diabetes Metab. Disord. 2023, 22, 1217–1229. [Google Scholar] [CrossRef] [PubMed]
- Jasim, L.M.M.; Homayouni Tabrizi, M.; Darabi, E.; Jaseem, M.M.M. The antioxidant, anti-angiogenic, and anticancer impact of chitosan-coated herniarin-graphene oxide nanoparticles (CHG-NPs). Heliyon 2023, 9, e20042. [Google Scholar] [CrossRef]
- Plácido, A.; do Pais do Amaral, C.; Teixeira, C.; Nogueira, A.; Brango-Vanegas, J.; Alves Barbosa, E.; Moreira, D.C.; Silva-Carvalho, A.; da Silva, M.D.G.; do Nascimento Dias, J.; et al. Neuroprotective effects on microglia and insights into the structure-activity relationship of an antioxidant peptide isolated from Pelophylax perezi. J. Cell. Mol. Med. 2022, 26, 2793–2807. [Google Scholar] [CrossRef]
- Condezo-Hoyos, L.; Rubio, M.; Arribas, S.M.; España-Caparrós, G.; Rodríguez-Rodríguez, P.; Mujica-Pacheco, E.; González, M.C. A plasma oxidative stress global index in early stages of chronic venous insufficiency. J. Vasc. Surg. 2013, 57, 205–213. [Google Scholar] [CrossRef]
- Veglia, F.; Werba, J.P.; Tremoli, E.; Squellerio, I.; Sisillo, E.; Parolari, A.; Minardi, F.; Cavalca, V. Assessment of oxidative stress in coronary artery bypass surgery: Comparison between the global index OXY-SCORE and individual biomarkers. Biomarkers 2009, 14, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Vassalle, C.; Pratali, L.; Boni, C.; Mercuri, A.; Ndreu, R. An oxidative stress score as a combined measure of the pro-oxidant and anti-oxidant counterparts in patients with coronary artery disease. Clin. Biochem. 2008, 41, 1162–1167. [Google Scholar] [CrossRef]
- Ruiz-Hurtado, G.; Condezo-Hoyos, L.; Pulido-Olmo, H.; Aranguez, I.; Del Carmen Gonzalez, M.; Arribas, S.; Cerezo, C.; Segura, J.; Praga, M.; Fernández-Alfonso, M.S.; et al. Development of albuminuria and enhancement of oxidative stress during chronic renin-angiotensin system suppression. J. Hypertens. 2014, 32, 2082–2091; discussion 2091. [Google Scholar] [CrossRef]
- Turell, L.; Radi, R.; Alvarez, B. The thiol pool in human plasma: The central contribution of albumin to redox processes. Free Radic. Biol. Med. 2013, 65, 244–253. [Google Scholar] [CrossRef] [PubMed]
- Ishimaru, Y.; Adachi, T.; Ashikawa, H.; Hori, M.; Shimozato, T.; Ohtake, H.; Shimizu, S.; Ueyama, J.; Yamada, S. Association between the redox state of human serum albumin and exercise capacity in patients with cardiac disease. Am. J. Cardiol. 2023, 189, 56–60. [Google Scholar] [CrossRef]
- Paramasivan, S.; Adav, S.S.; Ngan, S.C.; Dalan, R.; Leow, M.K.; Ho, H.H.; Sze, S.K. Serum albumin cysteine trioxidation is a potential oxidative stress biomarker of type 2 diabetes mellitus. Sci. Rep. 2020, 10, 6475. [Google Scholar] [CrossRef]
- Sugio, S.; Kashima, A.; Mochizuki, S.; Noda, M.; Kobayashi, K. Crystal structure of human serum albumin at 2.5 A resolution. Protein Eng. 1999, 12, 439–446. [Google Scholar] [CrossRef]
- Nakashima, F.; Shibata, T.; Kamiya, K.; Yoshitake, J.; Kikuchi, R.; Matsushita, T.; Ishii, I.; Giménez-Bastida, J.A.; Schneider, C.; Uchida, K. Structural and functional insights into S-thiolation of human serum albumins. Sci. Rep. 2018, 8, 932. [Google Scholar] [CrossRef]
- Magzal, F.; Sela, S.; Szuchman-Sapir, A.; Tamir, S.; Michelis, R.; Kristal, B. In-vivo oxidized albumin- a pro-inflammatory agent in hypoalbuminemia. PLoS ONE 2017, 12, e0177799. [Google Scholar] [CrossRef]
- Hawkins, C.L.; Morgan, P.E.; Davies, M.J. Quantification of protein modification by oxidants. Free Radic. Biol. Med. 2009, 46, 965–988. [Google Scholar] [CrossRef] [PubMed]
- Saleh, L.; Plieth, C. Total low-molecular-weight antioxidants as a summary parameter, quantified in biological samples by a chemiluminescence inhibition assay. Nat. Protoc. 2010, 5, 1627–1634. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Garcia, J.A.; Rodriguez-Sanchez, E.; Aceves-Ripoll, J.; Abarca-Zabalia, J.; Susmozas-Sanchez, A.; Gonzalez Lafuente, L.; Bada-Bosch, T.; Hernandez, E.; Merida-Herrero, E.; Praga, M.; et al. Oxidative status before and after renal replacement therapy: Differences between conventional high flux hemodialysis and on-line hemodiafiltration. Nutrients 2019, 11, 2809. [Google Scholar] [CrossRef]
- Rodriguez-Sanchez, E.; Navarro-Garcia, J.A.; Aceves-Ripoll, J.; Gonzalez-Lafuente, L.; Corbacho-Alonso, N.; Baldan-Martin, M.; Madruga, F.; Alvarez-Llamas, G.; Barderas, M.G.; Ruilope, L.M.; et al. Analysis of global oxidative status using multimarker scores reveals a specific association between renal dysfunction and diuretic therapy in older adults. J. Gerontol. A Biol. Sci. Med. Sci. 2021, 76, 1198–1205. [Google Scholar] [CrossRef]
- Rodriguez-Sanchez, E.; Navarro-Garcia, J.A.; Aceves-Ripoll, J.; Gonzalez-Lafuente, L.; Corbacho-Alonso, N.; Martinez, P.; Calvo-Bonacho, E.; Alvarez-Llamas, G.; Barderas, M.G.; Ruilope, L.M.; et al. Lifetime cardiovascular risk is associated with a multimarker score of systemic oxidative status in young adults independently of traditional risk factors. Transl. Res. J. Lab. Clin. Med. 2019, 212, 54–66. [Google Scholar] [CrossRef] [PubMed]
- Latif, N.; Quillon, A.; Sarathchandra, P.; McCormack, A.; Lozanoski, A.; Yacoub, M.H.; Chester, A.H. Modulation of human valve interstitial cell phenotype and function using a Fibroblast Growth Factor 2 formulation. PLoS ONE 2015, 10, e0127844. [Google Scholar] [CrossRef]
- van Engeland, N.C.A.; Bertazzo, S.; Sarathchandra, P.; McCormack, A.; Bouten, C.V.C.; Yacoub, M.H.; Chester, A.H.; Latif, N. Aortic calcified particles modulate valvular endothelial and interstitial cells. Cardiovasc. Pathol. 2017, 28, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Luna, C.; Alique, M.; Navalmoral, E.; Noci, M.V.; Bohorquez-Magro, L.; Carracedo, J.; Ramírez, R. Aging-associated oxidized albumin promotes cellular senescence and endothelial damage. Clin. Interv. Aging 2016, 11, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Gregory, C.A.; Gunn, W.G.; Peister, A.; Prockop, D.J. An Alizarin red-based assay of mineralization by adherent cells in culture: Comparison with cetylpyridinium chloride extraction. Anal. Biochem. 2004, 329, 77–84. [Google Scholar] [CrossRef]
- Sies, H.; Berndt, C.; Jones, D.P. Oxidative Stress. Annu. Rev. Biochem. 2017, 86, 715–748. [Google Scholar] [CrossRef]
- Marrocco, I.; Altieri, F.; Peluso, I. Measurement and clinical significance of biomarkers of oxidative stress in humans. Oxidative Med. Cell. Longev. 2017, 2017, 6501046. [Google Scholar] [CrossRef]
- Madamanchi, N.R.; Vendrov, A.; Runge, M.S. Oxidative stress and vascular disease. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 29–38. [Google Scholar] [CrossRef]
- Adhikari, R.; Shiwakoti, S.; Ko, J.Y.; Dhakal, B.; Park, S.H.; Choi, I.J.; Kim, H.J.; Oak, M.H. Oxidative stress in calcific aortic valve stenosis: Protective role of natural antioxidants. Antioxidants 2022, 11, 1169. [Google Scholar] [CrossRef] [PubMed]
- Romuk, E.; Wojciechowska, C.; Jacheć, W.; Zemła-Woszek, A.; Momot, A.; Buczkowska, M.; Rozentryt, P. Malondialdehyde and Uric Acid as Predictors of Adverse Outcome in Patients with Chronic Heart Failure. Oxidative Med. Cell. Longev. 2019, 2019, 9246138. [Google Scholar] [CrossRef] [PubMed]
- Roest, M.; Voorbij, H.A.; Van der Schouw, Y.T.; Peeters, P.H.; Teerlink, T.; Scheffer, P.G. High levels of urinary F2-isoprostanes predict cardiovascular mortality in postmenopausal women. J. Clin. Lipidol. 2008, 2, 298–303. [Google Scholar] [CrossRef]
- Shishehbor, M.H.; Aviles, R.J.; Brennan, M.L.; Fu, X.; Goormastic, M.; Pearce, G.L.; Gokce, N.; Keaney, J.F., Jr.; Penn, M.S.; Sprecher, D.L.; et al. Association of nitrotyrosine levels with cardiovascular disease and modulation by statin therapy. JAMA 2003, 289, 1675–1680. [Google Scholar] [CrossRef]
- Walter, M.F.; Jacob, R.F.; Jeffers, B.; Ghadanfar, M.M.; Preston, G.M.; Buch, J.; Mason, R.P. Serum levels of thiobarbituric acid reactive substances predict cardiovascular events in patients with stable coronary artery disease: A longitudinal analysis of the PREVENT study. J. Am. Coll. Cardiol. 2004, 44, 1996–2002. [Google Scholar] [CrossRef]
- Leon, M.B.; Piazza, N.; Nikolsky, E.; Blackstone, E.H.; Cutlip, D.E.; Kappetein, A.P.; Krucoff, M.W.; Mack, M.; Mehran, R.; Miller, C.; et al. Standardized endpoint definitions for transcatheter aortic valve implantation clinical trials: A consensus report from the Valve Academic Research Consortium. Eur. Heart J. 2011, 32, 205–217. [Google Scholar] [CrossRef]
- Parenica, J.; Nemec, P.; Tomandl, J.; Ondrasek, J.; Pavkova-Goldbergova, M.; Tretina, M.; Jarkovsky, J.; Littnerova, S.; Poloczek, M.; Pokorny, P.; et al. Prognostic utility of biomarkers in predicting of one-year outcomes in patients with aortic stenosis treated with transcatheter or surgical aortic valve implantation. PLoS ONE 2012, 7, e48851. [Google Scholar] [CrossRef][Green Version]
- Meijboom, W.B.; Mollet, N.R.; Van Mieghem, C.A.; Kluin, J.; Weustink, A.C.; Pugliese, F.; Vourvouri, E.; Cademartiri, F.; Bogers, A.J.; Krestin, G.P.; et al. Pre-operative computed tomography coronary angiography to detect significant coronary artery disease in patients referred for cardiac valve surgery. J. Am. Coll. Cardiol. 2006, 48, 1658–1665. [Google Scholar] [CrossRef]
- Di Minno, M.N.D.; Poggio, P.; Conte, E.; Myasoedova, V.; Songia, P.; Mushtaq, S.; Cavallotti, L.; Moschetta, D.; Di Minno, A.; Spadarella, G.; et al. Cardiovascular morbidity and mortality in patients with aortic valve calcification: A systematic review and meta-analysis. J. Cardiovasc. Comput. Tomogr. 2019, 13, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Sankaramangalam, K.; Banerjee, K.; Kandregula, K.; Mohananey, D.; Parashar, A.; Jones, B.M.; Jobanputra, Y.; Mick, S.; Krishnaswamy, A.; Svensson, L.G.; et al. Impact of coronary artery disease on 30-day and 1-year mortality in patients undergoing transcatheter aortic valve replacement: A meta-analysis. J. Am. Heart Assoc. 2017, 6, e006092. [Google Scholar] [CrossRef] [PubMed]
- Nagayoshi, Y.; Kawano, H.; Hokamaki, J.; Uemura, T.; Soejima, H.; Kaikita, K.; Sugiyama, S.; Yamabe, H.; Shioji, I.; Sasaki, S.; et al. Differences in oxidative stress markers based on the aetiology of heart failure: Comparison of oxidative stress in patients with and without coronary artery disease. Free Radic. Res. 2009, 43, 1159–1166. [Google Scholar] [CrossRef]
- Xiang, F.; Shuanglun, X.; Jingfeng, W.; Ruqiong, N.; Yuan, Z.; Yongqing, L.; Jun, Z. Association of serum 8-hydroxy-2’-deoxyguanosine levels with the presence and severity of coronary artery disease. Coron. Artery Dis. 2011, 22, 223–227. [Google Scholar] [CrossRef]
- Wang, X.-b.; Cui, N.-h.; Liu, X.n.; Liu, X. Mitochondrial 8-hydroxy-2′-deoxyguanosine and coronary artery disease in patients with type 2 diabetes mellitus. Cardiovasc. Diabetol. 2020, 19, 22. [Google Scholar] [CrossRef] [PubMed]
- Di Minno, A.; Turnu, L.; Porro, B.; Squellerio, I.; Cavalca, V.; Tremoli, E.; Di Minno, M.N. 8-Hydroxy-2-Deoxyguanosine levels and cardiovascular disease: A systematic review and meta-analysis of the literature. Antioxid. Redox Signal 2016, 24, 548–555. [Google Scholar] [CrossRef]
- Gorbunova, V.; Seluanov, A.; Mao, Z.; Hine, C. Changes in DNA repair during aging. Nucleic Acids Res. 2007, 35, 7466–7474. [Google Scholar] [CrossRef]
- Redon, J.; Oliva, M.R.; Tormos, C.; Giner, V.; Chaves, J.; Iradi, A.; Sáez, G.T. Antioxidant activities and oxidative stress byproducts in human hypertension. Hypertension 2003, 41, 1096–1101. [Google Scholar] [CrossRef]
- Basile, C.; Fucile, I.; Lembo, M.; Manzi, M.V.; Ilardi, F.; Franzone, A.; Mancusi, C. Arterial hypertension in aortic valve stenosis: A critical update. J. Clin. Med. 2021, 10, 5553. [Google Scholar] [CrossRef]
- Polito, L.; Bortolotti, M.; Battelli, M.G.; Bolognesi, A. Xanthine oxidoreductase: A leading actor in cardiovascular disease drama. Redox Biol. 2021, 48, 102195. [Google Scholar] [CrossRef] [PubMed]
- Battelli, M.G.; Polito, L.; Bolognesi, A. Xanthine oxidoreductase in atherosclerosis pathogenesis: Not only oxidative stress. Atherosclerosis 2014, 237, 562–567. [Google Scholar] [CrossRef]
- Abdulle, A.E.; Bourgonje, A.R.; Kieneker, L.M.; Koning, A.M.; la Bastide-van Gemert, S.; Bulthuis, M.L.C.; Dijkstra, G.; Faber, K.N.; Dullaart, R.P.F.; Bakker, S.J.L.; et al. Serum free thiols predict cardiovascular events and all-cause mortality in the general population: A prospective cohort study. BMC Med. 2020, 18, 130. [Google Scholar] [CrossRef]
- Xuan, Y.; Bobak, M.; Anusruti, A.; Jansen, E.; Pająk, A.; Tamosiunas, A.; Saum, K.U.; Holleczek, B.; Gao, X.; Brenner, H.; et al. Association of serum markers of oxidative stress with myocardial infarction and stroke: Pooled results from four large European cohort studies. Eur. J. Epidemiol. 2019, 34, 471–481. [Google Scholar] [CrossRef] [PubMed]
- Corbacho-Alonso, N.; Baldán-Martín, M.; López, J.A.; Rodríguez-Sánchez, E.; Martínez, P.J.; Mourino-Alvarez, L.; Sastre-Oliva, T.; Cabrera, M.; Calvo, E.; Padial, L.R.; et al. Cardiovascular risk stratification based on oxidative stress for early detection of pathology. Antioxid. Redox Signal 2021, 35, 602–617. [Google Scholar] [CrossRef]
- Ungvari, Z.; Tarantini, S.; Nyúl-Tóth, Á.; Kiss, T.; Yabluchanskiy, A.; Csipo, T.; Balasubramanian, P.; Lipecz, A.; Benyo, Z.; Csiszar, A. Nrf2 dysfunction and impaired cellular resilience to oxidative stressors in the aged vasculature: From increased cellular senescence to the pathogenesis of age-related vascular diseases. GeroScience 2019, 41, 727–738. [Google Scholar] [CrossRef] [PubMed]
- Kattoor, A.J.; Pothineni, N.V.K.; Palagiri, D.; Mehta, J.L. Oxidative stress in atherosclerosis. Curr. Atheroscler. Rep. 2017, 19, 42. [Google Scholar] [CrossRef]
- Taverna, M.; Marie, A.L.; Mira, J.P.; Guidet, B. Specific antioxidant properties of human serum albumin. Ann. Intensive Care 2013, 3, 4. [Google Scholar] [CrossRef] [PubMed]
- Bourdon, E.; Blache, D. The importance of proteins in defense against oxidation. Antioxid. Redox Signal 2001, 3, 293–311. [Google Scholar] [CrossRef]
- Lim, P.S.; Jeng, Y.; Wu, M.Y.; Pai, M.A.; Wu, T.K.; Liu, C.S.; Chen, C.H.; Kuo, Y.C.; Chien, S.W.; Chen, H.P. Serum oxidized albumin and cardiovascular mortality in normoalbuminemic hemodialysis patients: A cohort study. PLoS ONE 2013, 8, e70822. [Google Scholar] [CrossRef]
- Terawaki, H.; Takada, Y.; Era, S.; Funakoshi, Y.; Nakayama, K.; Nakayama, M.; Ogura, M.; Ito, S.; Hosoya, T. The redox state of albumin and serious cardiovascular incidence in hemodialysis patients. Ther. Apher. Dial. 2010, 14, 465–471. [Google Scholar] [CrossRef]
- Rahmani-Kukia, N.; Abbasi, A.; Pakravan, N.; Hassan, Z.M. Measurement of oxidized albumin: An opportunity for diagnoses or treatment of COVID-19. Bioorg. Chem. 2020, 105, 104429. [Google Scholar] [CrossRef]
- Das, S.; Maras, J.S.; Hussain, M.S.; Sharma, S.; David, P.; Sukriti, S.; Shasthry, S.M.; Maiwall, R.; Trehanpati, N.; Singh, T.P.; et al. Hyperoxidized albumin modulates neutrophils to induce oxidative stress and inflammation in severe alcoholic hepatitis. Hepatology 2017, 65, 631–646. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, A.; Kubota, K.; Yamada, N.; Tagami, U.; Takehana, K.; Sonaka, I.; Suzuki, E.; Hirayama, K. Identification and characterization of oxidized human serum albumin: A slight structural change impairs its ligand-binding and antioxidant functions. FEBS J. 2006, 273, 3346–3357. [Google Scholar] [CrossRef] [PubMed]
- Oettl, K.; Stauber, R.E. Physiological and pathological changes in the redox state of human serum albumin critically influence its binding properties. Br. J. Pharmacol. 2007, 151, 580–590. [Google Scholar] [CrossRef]
- Wang, W.; Kang, P.M. Oxidative stress and antioxidant treatments in cardiovascular diseases. Antioxidants 2020, 9, 1292. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Wang, L.; Pan, Y.; Wang, X.; Ding, Y.; Zhou, C.; Shah, A.M.; Zhao, G.; Zhang, M. Celastrol alleviates aortic valve calcification via inhibition of NADPH oxidase 2 in valvular interstitial cells. JACC Basic Transl. Sci. 2020, 5, 35–49. [Google Scholar] [CrossRef]
- Tretter, V.; Hochreiter, B.; Zach, M.L.; Krenn, K.; Klein, K.U. Understanding Cellular Redox Homeostasis: A Challenge for Precision Medicine. Int. J. Mol. Sci. 2021, 23, 106. [Google Scholar] [CrossRef]
- Daiber, A.; Hahad, O.; Andreadou, I.; Steven, S.; Daub, S.; Münzel, T. Redox-related biomarkers in human cardiovascular disease—Classical footprints and beyond. Redox Biol. 2021, 42, 101875. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).