High Concentration of Anti-SARS-CoV-2 Antibodies 2 Years after COVID-19 Vaccination Stems Not Only from Boosters but Also from Widespread, Often Unrecognized, Contact with the Virus
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants and Study Design
2.2. Laboratory Testing
2.3. Statistical Analysis
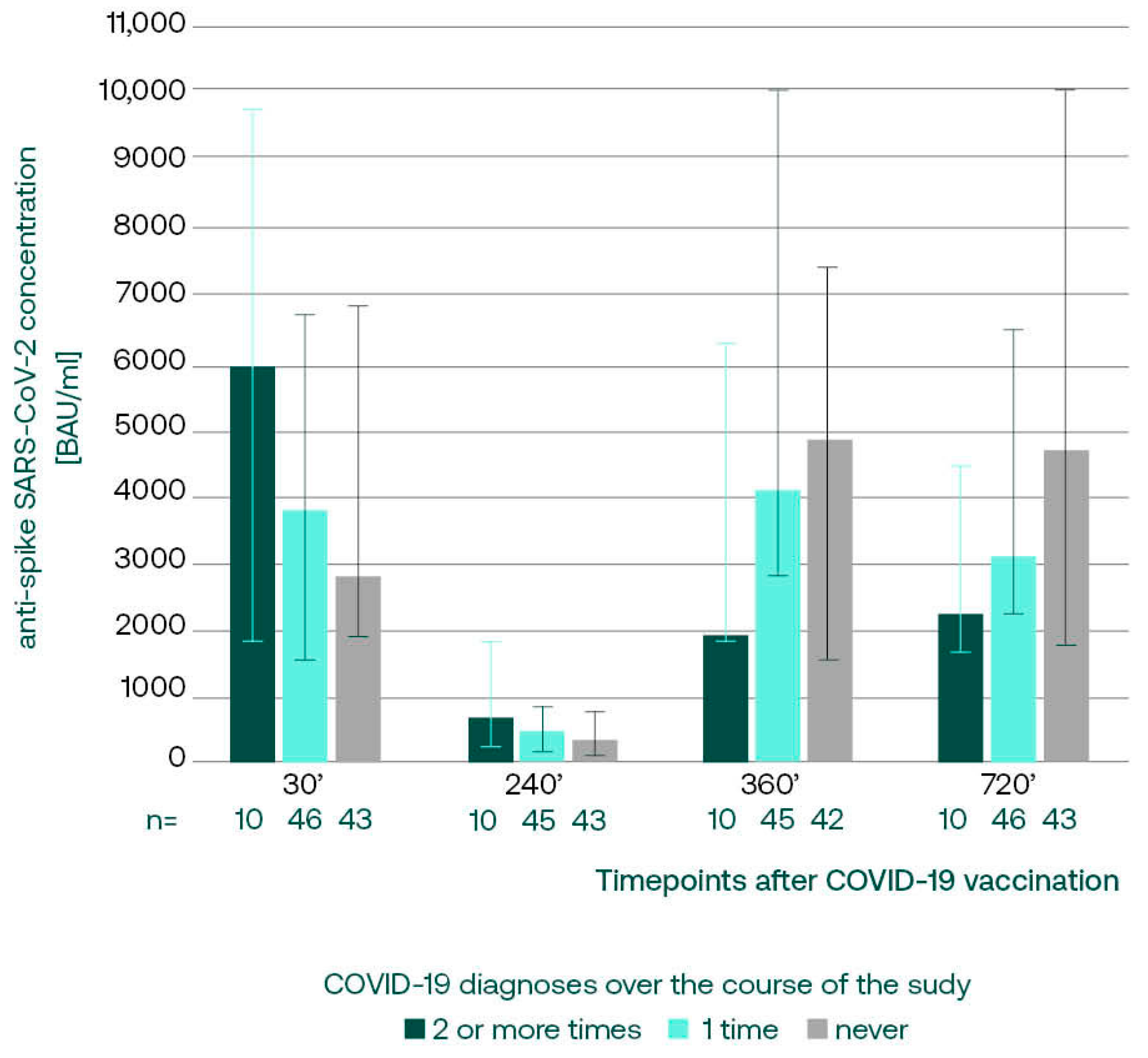
3. Results
3.1. Anti-Spike and Anti-Nucleocapsid SARS-CoV-2 IgG Concentrations Two Years after the Primary Vaccination
3.2. Relationship between Anti-Nucleocapsid SARS-CoV-2 IgG and the Reported COVID-19 Incidence
3.3. Factors Influencing Anti-Spike SARS-CoV-2 IgG Two Years after Vaccination
3.3.1. Anti-S SARS-CoV-2 IgG in Subgroups with Different COVID-19 Incidence
3.3.2. Anti-S SARS-CoV-2 IgG, Booster Acceptance, and COVID-19 Incidence in Age Subgroups
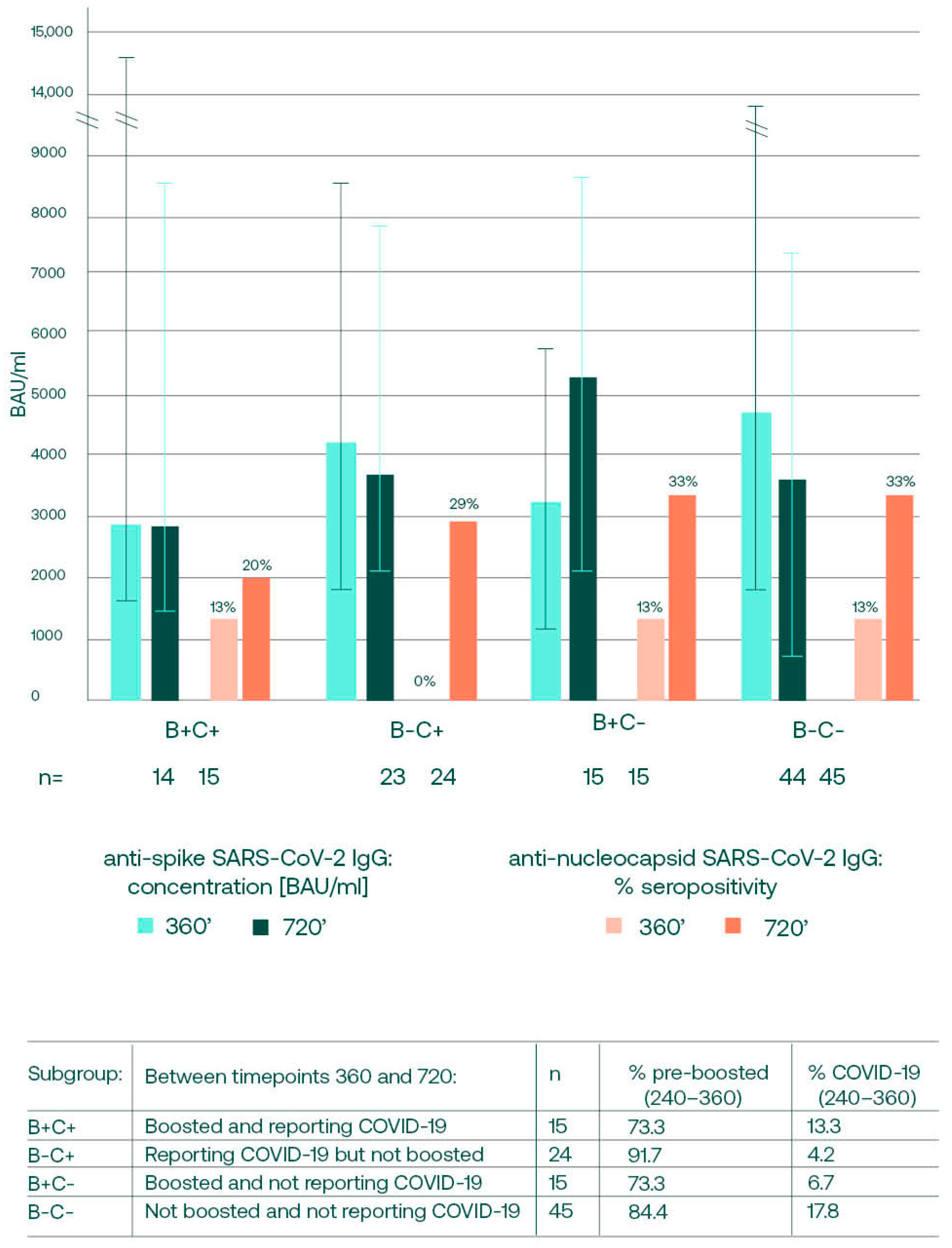
3.4. Anti-S SARS-CoV-2 Changes between Days 360 and 720, in Relation to Booster and Convalescence Status
3.5. Anti-S SARS-CoV-2 IgG Concentrations on Day 360 and the Future Infections
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- WHO—COVID-19 Epidemiological Update—16 February 2024, Edition 164. Available online: https://www.who.int/publications/m/item/covid-19-epidemiological-update-16-february-2024 (accessed on 27 February 2024).
- Coronavirus (COVID-19) Cases—Our World in Data. Available online: https://ourworldindata.org/covid-cases#confirmed-deaths-and-cases-our-data-source (accessed on 27 February 2024).
- Mathieu, E.; Ritchie, H.; Rodés-Guirao, L.; Appel, C.; Giattino, C.; Hasell, J.; Macdonald, B.; Dattani, S.; Beltekian, D.; Ortiz-Ospina, E.; et al. Coronavirus Pandemic (COVID-19). Published online at OurWorldInData.org. 2020. Available online: https://ourworldindata.org/coronavirus (accessed on 27 February 2024).
- Tenforde, M.W.; Self, W.H.; Adams, K.; Gaglani, M.; Ginde, A.A.; McNeal, T.; Ghamande, S.; Douin, D.J.; Talbot, H.K.; Casey, J.D.; et al. Influenza and Other Viruses in the Acutely Ill (IVY) Network. Association Between mRNA Vaccination and COVID-19 Hospitalization and Disease Severity. JAMA 2021, 326, 2043–2054. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Suzuki, R.; Yamasoba, D.; Kimura, I.; Wang, L.; Kishimoto, M.; Ito, J.; Morioka, Y.; Nao, N.; Nasser, H.; Uriu, K.; et al. Attenuated fusogenicity and pathogenicity of SARS-CoV-2 Omicron variant. Nature 2022, 603, 700–705. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Brüssow, H. COVID-19: Omicron—The latest, the least virulent, but probably not the last variant of concern of SARS-CoV-2. Microb. Biotechnol. 2022, 15, 1927–1939. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mohamed, K.; Rzymski, P.; Islam, M.S.; Makuku, R.; Mushtaq, A.; Khan, A.; Ivanovska, M.; Makka, S.A.; Hashem, F.; Marquez, L.; et al. COVID-19 vaccinations: The unknowns, challenges, and hopes. J. Med. Virol. 2022, 94, 1336–1349. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- 2023 Timeline of EU Action in the Fight against Coronavirus. Available online: https://ec.europa.eu/info/live-work-travel-eu/coronavirus-response/timeline-eu-action_en (accessed on 27 February 2024).
- Chivu-Economescu, M.; Vremera, T.; Ruta, S.M.; Grancea, C.; Leustean, M.; Chiriac, D.; David, A.; Matei, L.; Diaconu, C.C.; Gatea, A.; et al. Assessment of the Humoral Immune Response Following COVID-19 Vaccination in Healthcare Workers: A One Year Longitudinal Study. Biomedicines 2022, 10, 1526. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhong, D.; Xiao, S.; Debes, A.K.; Egbert, E.R.; Caturegli, P.; Colantuoni, E.; Milstone, A.M. Durability of Antibody Levels After Vaccination With mRNA SARS-CoV-2 Vaccine in Individuals With or Without Prior Infection. JAMA 2021, 326, 2524–2526. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Long, Q.X.; Liu, B.Z.; Deng, H.J.; Wu, G.C.; Deng, K.; Chen, Y.K.; Liao, P.; Qiu, J.F.; Lin, Y.; Cai, X.F.; et al. Antibody responses to SARS-CoV-2 in patients with COVID-19. Nat. Med. 2020, 26, 845–848. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Yuan, Q.; Wang, H.; Liu, W.; Liao, X.; Su, Y.; Wang, X.; Yuan, J.; Li, T.; Li, J.; et al. Antibody Responses to SARS-CoV-2 in Patients With Novel Coronavirus Disease 2019. Clin. Infect. Dis. 2020, 71, 2027–2034. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lippi, G.; Henry, B.M.; Plebani, M. Anti-SARS-CoV-2 Antibodies Testing in Recipients of COVID-19 Vaccination: Why, When, and How? Diagnostics 2021, 11, 941. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Swadźba, J.; Anyszek, T.; Panek, A.; Martin, E. Anti-Spike SARS-CoV-2 IgG Assessment with a Commercial Assay during a 4-Month Course after COVID-19 Vaccination. Vaccines 2021, 9, 1367. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ferrari, D.; Clementi, N.; Criscuolo, E.; Ambrosi, A.; Corea, F.; Di Resta, C.; Tomaiuolo, R.; Mancini, N.; Locatelli, M.; Plebani, M.; et al. Antibody Titer Kinetics and SARS-CoV-2 Infections Six Months after Administration with the BNT162b2 Vaccine. Vaccines 2021, 9, 1357. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rastawicki, W.; Rokosz-Chudziak, N. Characteristics and assessment of the usefulness of serological tests in the diagnostic of infections caused by coronavirus SARS-CoV-2 on the basis of available manufacturer’s data and literature review. Prz. Epidemiol. 2020, 74, 49–68. [Google Scholar] [CrossRef] [PubMed]
- Swadźba, J.; Kozłowska, D.; Anyszek, T.; Dorycka, M.; Martin, E.; Piotrowska-Mietelska, A. Atypical pneumonia diagnosed as coronavirus disease 2019 by a serologic test (patient-1 in Poland). Pol. Arch. Intern. Med. 2020, 130, 444–445. [Google Scholar] [CrossRef] [PubMed]
- Swadźba, J.; Bednarczyk, M.; Anyszek, T.; Martin, E. A comparison of 7 commercial anti-SARS-CoV-2 antibody immunoassays. Arch. Med. Sci. 2020, 19, 1281–1288. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Swadźba, J.; Bednarczyk, M.; Anyszek, T.; Kozlowska, D.; Panek, A.; Martin, E. The real life performance of 7 automated anti-SARS-CoV-2 IgG and IgM/IgA immunoassays. Pract. Lab. Med. 2021, 25, e00212. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- First WHO International Standard for Anti-SARS-CoV-2 Immunoglobulin (Human). NIBSC Code: 20/136. Instructions for Use (Version 2.0, Dated 17 December 2020). Available online: https://www.nibsc.org/documents/ifu/20-136.pdf (accessed on 4 March 2022).
- Giavarina, D.; Carta, M. Improvements and limits of anti SARS-CoV-2 antibodies assays by WHO (NIBSC 20/136) standardization. Diagnosis 2021, 9, 274–279. [Google Scholar] [CrossRef] [PubMed]
- Tré-Hardy, M.; Cupaiolo, R.; Wilmet, A.; Antoine-Moussiaux, T.; Della Vecchia, A.; Horeanga, A.; Papleux, E.; Vekemans, M.; Beukinga, I.; Blairon, L. Six-month interim analysis of ongoing immunogenicity surveillance of the mRNA-1273 vaccine in healthcare workers: A third dose is expected. J. Infect. 2021, 83, 559–564. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Carta, M.; Marinello, I.; Cappelletti, A.; Rodolfi, A.; Cerrito, E.; Bernasconi, C.; Gottardo, M.; Dal Lago, F.; Rizzetto, D.; Barzon, E.; et al. Comparison of Anti-SARS-CoV-2 S1 Receptor-Binding Domain Antibody Immunoassays in Health Care Workers Before and After the BNT162b2 mRNA Vaccine. Am. J. Clin. Pathol. 2022, 157, 212–218. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hibino, M.; Watanabe, S.; Kamada, R.; Tobe, S.; Maeda, K.; Horiuchi, S.; Kondo, T. Antibody Responses to the BNT162b2 mRNA Vaccine in Healthcare Workers in a General Hospital in Japan: A Comparison of Two Assays for Anti-spike Protein Immunoglobulin G. Intern. Med. 2021, 61, 811–819. [Google Scholar] [CrossRef] [PubMed]
- Lukaszuk, K.; Kiewisz, J.; Rozanska, K.; Dabrowska, M.; Podolak, A.; Jakiel, G.; Woclawek-Potocka, I.; Lukaszuk, A.; Rabalski, L. Usefulness of IVD Kits for the Assessment of SARS-CoV-2 Antibodies to Evaluate the Humoral Response to Vaccination. Vaccines 2021, 9, 840. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Salvagno, G.L.; Henry, B.M.; di Piazza, G.; Pighi, L.; De Nitto, S.; Bragantini, D.; Gianfilippi, G.L.; Lippi, G. Anti-SARS-CoV-2 Receptor-Binding Domain Total Antibodies Response in Seropositive and Seronegative Healthcare Workers Undergoing COVID-19 mRNA BNT162b2 Vaccination. Diagnostics 2021, 11, 832. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ogrič, M.; Žigon, P.; Podovšovnik, E.; Lakota, K.; Sodin-Semrl, S.; Rotar, Ž.; Čučnik, S. Differences in SARS-CoV-2-Specific Antibody Responses After the First, Second, and Third Doses of BNT162b2 in Naïve and Previously Infected Individuals: A 1-Year Observational Study in Healthcare Professionals. Front. Immunol. 2022, 13, 876533. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zurac, S.; Vladan, C.; Dinca, O.; Constantin, C.; Neagu, M. Immunogenicity evaluation after BNT162b2 booster vaccination in healthcare workers. Sci. Rep. 2022, 12, 12716. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lumley, S.F.; O’Donnell, D.; Stoesser, N.E.; Matthews, P.C.; Howarth, A.; Hatch, S.B.; Marsden, B.D.; Cox, S.; James, T.; Warren, F.; et al. Antibody Status and Incidence of SARS-CoV-2 Infection in Health Care Workers. N. Engl. J. Med. 2021, 384, 533–540. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Okba, N.M.A.; Müller, M.A.; Li, W.; Wang, C.; GeurtsvanKessel, C.H.; Corman, V.M.; Lamers, M.M.; Sikkema, R.S.; de Bruin, E.; Chandler, F.D.; et al. Severe Acute Respiratory Syndrome Coronavirus 2-Specific Antibody Responses in Coronavirus Disease 2019 Patients. Emerg. Infect. Dis. 2020, 26, 1478–1488. [Google Scholar] [CrossRef] [PubMed]
- Movsisyan, M.; Chopikyan, A.; Kasparova, I.; Hakobjanyan, G.; Carrat, F.; Sukiasyan, M.; Rushanyan, M.; Chalabyan, M.; Shariff, S.; Kantawala, B.; et al. Kinetics of anti-nucleocapsid IgG response in COVID-19 immunocompetent convalescent patients. Sci. Rep. 2022, 12, 12403. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Pinto, D.; Park, Y.J.; Beltramello, M.; Walls, A.C.; Tortorici, M.A.; Bianchi, S.; Jaconi, S.; Culap, K.; Zatta, F.; De Marco, A.; et al. Cross-neutralization of SARS-CoV-2 by a human monoclonal SARS-CoV antibody. Nature 2020, 583, 290–295. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.P.; Yansouni, C.P.; Basta, N.E.; Desjardins, M.; Kanjilal, S.; Paquette, K.; Caya, C.; Semret, M.; Quach, C.; Libman, M.; et al. Serodiagnostics for Severe Acute Respiratory Syndrome-Related Coronavirus 2: A Narrative Review. Ann. Intern. Med. 2020, 173, 450–460. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Manisty, C.; Otter, A.D.; Treibel, T.A.; McKnight, Á.; Altmann, D.M.; Brooks, T.; Noursadeghi, M.; Boyton, R.J.; Semper, A.; Moon, J.C. Antibody response to first BNT162b2 dose in previously SARS-CoV-2-infected individuals. Lancet 2021, 397, 1057–1058. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lyke, K.E.; Atmar, R.L.; Islas, C.D.; Posavad, C.M.; Szydlo, D.; Paul Chourdhury, R.; Deming, M.E.; Eaton, A.; Jackson, L.A.; Branche, A.R.; et al. Rapid decline in vaccine-boosted neutralizing antibodies against SARS-CoV-2 Omicron variant. Cell Rep. Med. 2022, 3, 100679. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Peng, D.; Yang, L.; Jin, C.; Feng, J.; Cao, M.; Liu, Y. Effect of second booster vaccination on clinical outcomes of Omicron-variant breakthrough infection: A propensity score matching cohort study. Heliyon 2023, 10, e23344. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Marklund, E.; Leach, S.; Nyström, K.; Lundgren, A.; Liljeqvist, J.Å.; Nilsson, S.; Yilmaz, A.; Andersson, L.M.; Bemark, M.; Gisslén, M. Longitudinal Follow Up of Immune Responses to SARS-CoV-2 in Health Care Workers in Sweden With Several Different Commercial IgG-Assays, Measurement of Neutralizing Antibodies and CD4+ T-Cell Responses. Front. Immunol. 2021, 12, 750448. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lee, N.; Jeong, S.; Lee, S.K.; Cho, E.J.; Hyun, J.; Park, M.J.; Song, W.; Kim, H.S. Quantitative Analysis of Anti-N and Anti-S Antibody Titers of SARS-CoV-2 Infection after the Third Dose of COVID-19 Vaccination. Vaccines 2022, 10, 1143. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Coronavirus (COVID-19) Cases—Our World in Data. Available online: https://ourworldindata.org/covid-cases (accessed on 1 December 2023).
- Cromer, D.; Steain, M.; Reynaldi, A.; Schlub, T.E.; Wheatley, A.K.; Juno, J.A.; Kent, S.J.; Triccas, J.A.; Khoury, D.S.; Davenport, M.P. Neutralising antibody titres as predictors of protection against SARS-CoV-2 variants and the impact of boosting: A meta-analysis. Lancet Microbe 2022, 3, e52–e61. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yu, Y.; Esposito, D.; Kang, Z.; Lu, J.; Remaley, A.T.; De Giorgi, V.; Chen, L.N.; West, K.; Cao, L. mRNA vaccine-induced antibodies more effective than natural immunity in neutralizing SARS-CoV-2 and its high affinity variants. Sci. Rep. 2022, 12, 2628. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Focosi, D.; Baj, A.; Maggi, F. Is a single COVID-19 vaccine dose enough in convalescents? Hum. Vaccines Immunother. 2021, 17, 2959–2961. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Goldblatt, D.; Alter, G.; Crotty, S.; Plotkin, S.A. Correlates of protection against SARS-CoV-2 infection and COVID-19 disease. Immunol. Rev. 2022, 310, 6–26. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Swadźba, J.; Anyszek, T.; Panek, A.; Chojęta, A.; Wyrzykowska, K.; Martin, E. Head-to-Head Comparison of 5 Anti-SARS-CoV-2 Assays Performance in One Hundred COVID-19 Vaccinees, over an 8-Month Course. Diagnostics 2022, 12, 1426. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Swadźba, J.; Anyszek, T.; Panek, A.; Chojęta, A.; Piotrowska-Mietelska, A.; Martin, E. The Influence of Booster Shot and SARS-CoV-2 Infection on the Anti-Spike Antibody Concentration One Year after the First COVID-19 Vaccine Dose Administration. Vaccines 2023, 11, 278. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Müller, L.; Andrée, M.; Moskorz, W.; Drexler, I.; Walotka, L.; Grothmann, R.; Ptok, J.; Hillebrandt, J.; Ritchie, A.; Rabl, D.; et al. Age-dependent immune response to the Biontech/Pfizer BNT162b2 COVID-19 vaccination. Clin. Infect. Dis. 2021, 73, 2065–2072. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sasso, B.L.; Giglio, R.V.; Vidali, M.; Scazzone, C.; Bivona, G.; Gambino, C.M.; Ciaccio, A.M.; Agnello, L.; Ciaccio, M. Evaluation of Anti-SARS-Cov-2 S-RBD IgG Antibodies after COVID-19 mRNA BNT162b2 Vaccine. Diagnostics 2021, 11, 1135. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Terpos, E.; Trougakos, I.P.; Apostolakou, F.; Charitaki, I.; Sklirou, A.D.; Mavrianou, N.; Papanagnou, E.D.; Liacos, C.I.; Gumeni, S.; Rentziou, G.; et al. Age-dependent and gender-dependent antibody responses against SARS-CoV-2 in health workers and octogenarians after vaccination with the BNT162b2 mRNA vaccine. Am. J. Hematol. 2021, 96, E257–E259. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Stærke, N.B.; Reekie, J.; Nielsen, H.; Benfield, T.; Wiese, L.; Knudsen, L.S.; Iversen, M.B.; Iversen, K.; Fogh, K.; Bodilsen, J.; et al. Levels of SARS-CoV-2 antibodies among fully vaccinated individuals with Delta or Omicron variant breakthrough infections. Nat. Commun. 2022, 13, 4466. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wei, J.; Matthews, P.C.; Stoesser, N.; Newton, J.N.; Diamond, I.; Studley, R.; Taylor, N.; Bell, J.I.; Farrar, J.; Kolenchery, J.; et al. Protection against SARS-CoV-2 Omicron BA.4/5 variant following booster vaccination or breakthrough infection in the UK. Nat. Commun. 2023, 14, 2799. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Feng, S.; Phillips, D.J.; White, T.; Sayal, H.; Aley, P.K.; Bibi, S.; Dold, C.; Fuskova, M.; Gilbert, S.C.; Hirsch, I.; et al. Correlates of protection against symptomatic and asymptomatic SARS-CoV-2 infection. Nat. Med. 2021, 27, 2032–2040. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bergwerk, M.; Gonen, T.; Lustig, Y.; Amit, S.; Lipsitch, M.; Cohen, C. COVID-19 breakthrough infections in vaccinated health care workers. N. Engl. J. Med. 2021, 385, 1629–1630. [Google Scholar] [CrossRef] [PubMed]
- Khoury, D.S.; Cromer, D.; Reynaldi, A.; Schlub, T.E.; Wheatley, A.K.; Juno, J.A.; Subbarao, K.; Kent, S.J.; Triccas, J.A.; Davenport, M.P. Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infection. Nat. Med. 2021, 27, 1205–1211. [Google Scholar] [CrossRef] [PubMed]
- Goldblatt, D.; Fiore-Gartland, A.; Johnson, M.; Hunt, A.; Bengt, C.; Zavadska, D.; Snipe, H.D.; Brown, J.S.; Workman, L.; Zar, H.J.; et al. Towards a population-based threshold of protection for COVID-19 vaccines. Vaccine 2022, 40, 306–315. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Dimeglio, C.; Herin, F.; Martin-Blondel, G.; Miedougé, M.; Izopet, J. Antibody titers and protection against a SARS-CoV-2 infection. J. Infect. 2022, 84, 248–288. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Takahashi, E.; Sawabuchi, T.; Homma, T.; Fukuda, Y.; Sagara, H.; Kinjo, T.; Fujita, K.; Suga, S.; Kimoto, T.; Sakai, S.; et al. Clinical Utility of SARS-CoV-2 Antibody Titer Multiplied by Binding Avidity of Receptor-Binding Domain (RBD) in Monitoring Protective Immunity and Clinical Severity. Viruses 2023, 15, 1662. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Perry, J.; Osman, S.; Wright, J.; Richard-Greenblatt, M.; Buchan, S.A.; Sadarangani, M.; Bolotin, S. Does a humoral correlate of protection exist for SARS-CoV-2? A systematic review. PLoS ONE 2022, 17, e0266852. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sobhani, K.; Cheng, S.; Binder, R.A.; Mantis, N.J.; Crawford, J.M.; Okoye, N.; Braun, J.G.; Joung, S.; Wang, M.; Lozanski, G.; et al. Clinical Utility of SARS-CoV-2 Serological Testing and Defining a Correlate of Protection. Vaccines 2023, 11, 1644. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]


| Subgroups | N | Median Anti-S SARS-CoV-2 IgG [BAU/mL] 2 Years after the Vaccination | Significance | |
|---|---|---|---|---|
| Sex | Female | 85 | 3980 | NS (p = 0.4454) U Mann–Whitney test |
| Male | 14 | 3010 | ||
| Age | <60 | 84 | 3090 | p = 0.001 U Mann–Whitney test |
| >60 | 15 | 6460 | ||
| Number of vaccine doses received | 2 | 9 | 3520 | NS (p = 0.4175) Kruskal–Wallis test |
| 3 | 66 | 3140 | ||
| 4 | 24 | 4950 | ||
| Timing of the last booster | No booster | 9 | 3520 | NS (p = 0.5578) Kruskal–Wallis test |
| Before 360 | 60 | 3660 | ||
| After 360 | 30 | 3880 | ||
| COVID-19 history (self-reported) | No | 39 | 4640 | NS (p = 0.0642) Kruskal–Wallis test |
| 1 | 50 | 3090 | ||
| ≥2 | 10 | 2190 | ||
| COVID-19 history between days 360 and 720 | Yes (self-reported) | 39 | 3140 | NS (p = 0.8467) U Mann–Whitney test |
| No | 60 | 3850 | ||
| Anti-nucleocapsid seropositivity on day 720 | Anti-N positive | 30 | 3010 | NS (p = 0.8088) U Mann–Whitney test |
| Anti-N negative | 67 | 4180 | ||
| Other respiratory infections over the past 3 months | Yes | 31 | 3140 | NS (p = 1.0000) U Mann–Whitney test |
| No | 68 | 3850 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Swadźba, J.; Panek, A.; Wąsowicz, P.; Anyszek, T.; Martin, E. High Concentration of Anti-SARS-CoV-2 Antibodies 2 Years after COVID-19 Vaccination Stems Not Only from Boosters but Also from Widespread, Often Unrecognized, Contact with the Virus. Vaccines 2024, 12, 471. https://doi.org/10.3390/vaccines12050471
Swadźba J, Panek A, Wąsowicz P, Anyszek T, Martin E. High Concentration of Anti-SARS-CoV-2 Antibodies 2 Years after COVID-19 Vaccination Stems Not Only from Boosters but Also from Widespread, Often Unrecognized, Contact with the Virus. Vaccines. 2024; 12(5):471. https://doi.org/10.3390/vaccines12050471
Chicago/Turabian StyleSwadźba, Jakub, Andrzej Panek, Paweł Wąsowicz, Tomasz Anyszek, and Emilia Martin. 2024. "High Concentration of Anti-SARS-CoV-2 Antibodies 2 Years after COVID-19 Vaccination Stems Not Only from Boosters but Also from Widespread, Often Unrecognized, Contact with the Virus" Vaccines 12, no. 5: 471. https://doi.org/10.3390/vaccines12050471
APA StyleSwadźba, J., Panek, A., Wąsowicz, P., Anyszek, T., & Martin, E. (2024). High Concentration of Anti-SARS-CoV-2 Antibodies 2 Years after COVID-19 Vaccination Stems Not Only from Boosters but Also from Widespread, Often Unrecognized, Contact with the Virus. Vaccines, 12(5), 471. https://doi.org/10.3390/vaccines12050471






