Abstract
Diabetes mellitus (DM) is a complex condition and serious health problem, with growing occurrence of DM-associated complications occurring globally. Persistent hyperglycemia is confirmed as promoting neurovascular dysfunction leading to irreversible endothelial cell dysfunction, increased neuronal cell apoptosis, oxidative stress and inflammation. These collaboratively and individually result in micro- and macroangiopathy as well as neuropathy demonstrated by progressive neuronal loss. Recently, major efforts have been pursued to select not only useful diagnostic and prognostic biomarkers, but also novel therapeutic approaches. Both microRNAs (miRNAs) and long non-coding RNAs (lncRNAs) belong to a class of non-coding RNAs identified in most of the body fluids i.e., peripheral blood, cerebrospinal fluid, brain tissue and neurons. Numerous miRNAs, lncRNAs and their target genes are able to modulate signaling pathways known to play a role in the pathophysiology of progressive neuronal dysfunction. Therefore, they pose as promising biomarkers and treatment for the vast majority of neurodegenerative disorders. This review provides an overall assessment of both miRNAs’ and lncRNAs’ utility in decelerating progressive nervous system impairment, including neurodegeneration in diabetic pathways.
1. Introduction
Diabetes mellitus (DM) is one of the most common chronic diseases worldwide [1]. In accordance with the latest edition of the International Diabetes Federation reports, currently almost half a billion people suffer from diabetes and by 2045 this count will reach 700 million [2]. DM is defined by persistent hyperglycemia and defective metabolism of carbohydrates caused by decreased secretion and increased resistance of insulin as a consequence of β-cells dysfunction [3]. Nearly half of patients present with poorly controlled diabetes which leads to a series of macro- and microvascular complications including cardiovascular disease (CVD), diabetic neuropathies such as retinopathy, and corneal neuropathy [4]. Although recent studies have shed light upon a correlation between DM and nervous system complications, i.e., neuropathy, neurovascular dysfunction, and neuroinflammation resulting from progressive neurodegeneration, particular underlying mechanisms are yet to be fully elucidated [5,6].
Type 1 diabetes mellitus (T1DM) is a chronic disease, in which the autoimmune system destroys the pancreatic beta cells responsible for insulin production [7]. Insulin possesses anabolic and anti-catabolic properties and maintains homeostasis of carbohydrate metabolism. Meanwhile, insulin deficiency leads to constant hyperglycemia [8]. The role of immune breakdown, including the expansion of CD4+ and CD8+ autoreactive T cells as well as B lymphocytes responsible for the production of autoantibodies, is underlined in the pathogenesis of T1DM [9]. Many scientists have researched the correlation of T1DM with retinal neurodegeneration, one of the earliest complications in T1DM [10,11].
Type 2 diabetes (T2DM) is a potentially reversible disease, characterized by high blood glucose, insulin resistance and relative lack of insulin. Insulin resistance is the earliest abnormality and major pathophysiological factor of T2DM. The role of insulin signaling defects, glucose transportation defects or lipo-toxicity are underlined in a study of insulin resistance pathophysiology [12]. Another key component of T2DM onset and progression is B-cells apoptosis. Although genetic abnormalities also play a role in the mechanism of T2DM, an unhealthy diet as well as a sedentary lifestyle lead to obesity and are crucial factors in the disease’s pathophysiology [3]. Various reports indicate that there is a link between T2DM and the development of neurodegenerative diseases as well as exacerbation of the neurodegenerative processes [13].
The alteration of microRNA (miRNA, miR) and long non-coding RNA (lncRNA) expression along with their relation to the pathophysiological mechanisms of chronic hyperglycemia was documented in diabetic neuropathy. LncRNA are a class of RNA non-protein transcripts longer than 200 nucleotides, serving as transcriptional regulators able to impact cellular processes, i.e., proliferation, differentiation or apoptosis [14,15,16,17,18]. They participate in numerous biological processes such as regulation of gene expression through mRNA splicing, transcription regulation, translation regulation and genomic imprinting [14,15,16,17,18,19]. LncRNAs are capable of modifying cellular responses through down- or up-regulation in microvascular degeneration and in high glucose-induced neuronal injury. This should be taken into consideration when considering these proteins as a novel therapeutic approach in diabetic-induced neurodegeneration, along with miRNAs (other promising molecules also broadly analyzed) [20,21]. MiRNAs are small (18–25 nucleotides), endogenous, single-stranded and non-coding RNAs, that are able to modulate approximately 60% of mammalian protein coding genes post-transcriptionally. MiRNAs are considered to play a pivotal role in many common disorders, e.g., DM, CVD as well as ischemic stroke [16,18]. Moreover, particular miRNAs were found to upregulate or downregulate particular cellular responses in diabetes-induced neurovascular injuries. Therefore, it is hypothesized that certain miRNAs and lncRNAs could be novel biomarkers and could direct a novel therapeutic approach in diabetic neuropathies [22]. This review aims to provide an overall overview of the current knowledge of miRNAs and lncRNAs in neurodegeneration and neuro-regenerative processes resulting from DM.
2. MiRNAs and Their Link to Neurodegenerative Changes in Metabolic Pathways Related to Diabetes
A DM-induced persistent state of hyperglycemia is suggested to be a major cause of a variety of pathological pathways, including oxidative stress, apoptosis, inflammation and neurodegeneration. Oxidative stress is associated with inflammation and neurodegeneration due to formation and augmented concentration of reactive oxygen species (ROS) [23]. Hyperglycemia increases the production of ROS which results in a dysfunction in neuronal cells. Additionally, oxidative stress enhances an imbalance between endogenous ROS and antioxidant defense systems, initiating chronic inflammation and tissue damage [24]. MiRNAs regulate many biological processes, and are correlated with different aspects of complex diseases. Differential expressions of miRNAs were observed in patients with neurodegenerative diseases as well as DM, as they play an important role in regulating diabetes-induced inflammatory and neurodegenerative responses [25].
2.1. MicroRNAs Involved in Neurodegeneration and Regeneration
2.1.1. MiRNAs in Diabetic Neuropathies
Diabetic peripheral neuropathy (DPN) is one of the most frequent persistent complications of all stages of DM, resulting in gradually spreading peripheral nerve damage. It is speculated that in a few years over 50 million diabetes patients worldwide will develop DPN [26]. DPN demonstrates symmetric, spreading proximally and mainly sensory progressive axonal loss as a result of chronic hyperglycemia and microangiopathy. Amongst the pathophysiological processes underlying DPN, increased oxidative stress, apoptosis ratio, mitochondrial dysfunction, chronic inflammation and accumulation of advanced glycation end products (AGEs) are deemed crucial. However, particular mechanisms have still not been fully elucidated [27,28]. Moreover, increasing evidence suggests that diabetic endothelial dysfunction is an early manifestation of DPN. Both DPN and diabetic endothelial dysfunction show similarities in induction of shared signaling pathways and seem to force each other [29,30]. Undoubtedly, diabetic endothelial dysfunction is a major cause of diminished neuronal perfusion resulting in reduced axonal reflexes, vasodilation and therefore, may promote neurodegeneration. Moreover, endothelial dysfunction is deemed crucial in the onset and progression of cerebral small vessel disease (CSVD), which may result in stroke and cognitive decline. Phosphodiesterase 3 (PDE3) is an enzyme detected in brain arteries which is known to play a key role in regulating endothelial function [31]. PDE3-targeting miR-221/miR-222 and miR-27a/miR-27b/miR-128 were identified in silico analysis. Overexpression of miR-27a-3p and miR-222-3p decreased the protein level of PDE3A in the in vitro model. MiR-221/miR-222 and miR-27a/miR-27b/miR-128 family impact pathways involved in immune modulation as well as cerebrovascular integrity and function. Targeting PDE3A by particular endothelial miRNAs may present as a suitable treatment for CSVD due to the capacity of these miRNAs to simultaneously affect various pathways related to CSVD and should consequently be studied more extensively in diabetic models [32].
One of the crucial factors involved in neuropathological processes is the small acidic polypeptide thymosin β4 (Tβ4) which promotes neuro-regeneration and reduces inflammation in diabetes-induced injury. Alongside this, miRNAs are known to influence some pathways involved in the onset and progression of neuronal dysfunction. Therefore, Wang et al. [33] analyzed the neuroprotective effects of Tβ4 in DPN on miR-146a both in vivo and in vitro. It was found that the Tβ4 injection reversed the inhibitory effect of diabetes by miR-146a upregulation. Increased miR-146a expression ameliorated the motor and sensory function of nerves, and improved nerve fiber density and regional blood flow in animal models, whereas endogenous inhibition of miR-146a reversed the positive effect of Tβ4 on endothelial cells. High glucose levels caused downregulation of miR-146a and upregulation of its target genes IRAK1, TRAF6 and p-NFkB further causing increased pro-inflammatory mediators, such as MCP-1 and VCAM-1 levels. Taken together, the study showed that Tβ4 promoted the neuroprotective role via upregulation of miR-146a in diabetic subjects by the inhibition of inflammatory mediators [34]. Furthermore, the same research team aimed to analyze the effect of sildenafil on miRNA expression in distal axons of embryonic cortical neurons. Sildenafil is suggested to upregulate the expression of miR-146a in dorsal root ganglia (DRG) and ameliorates neuropathy [33,35]. In high glucose conditions, axonal miR-146a expression was significantly reduced. This resulted in a significant increase in target genes IRAK1 and TRAF6. MiR-146a treatment on DRG neurons diminished axonal mRNA and protein levels of IRAK1 and TRAF6 (proinflammatory mediators) forcing axonal lengthening. In line with these findings, sildenafil use reversed high glucose-induced axonal injury by downregulation of miR-146a. Taken together, miR-146a seems to serve as a novel therapeutic approach in high glucose-induced neuropathy [36].
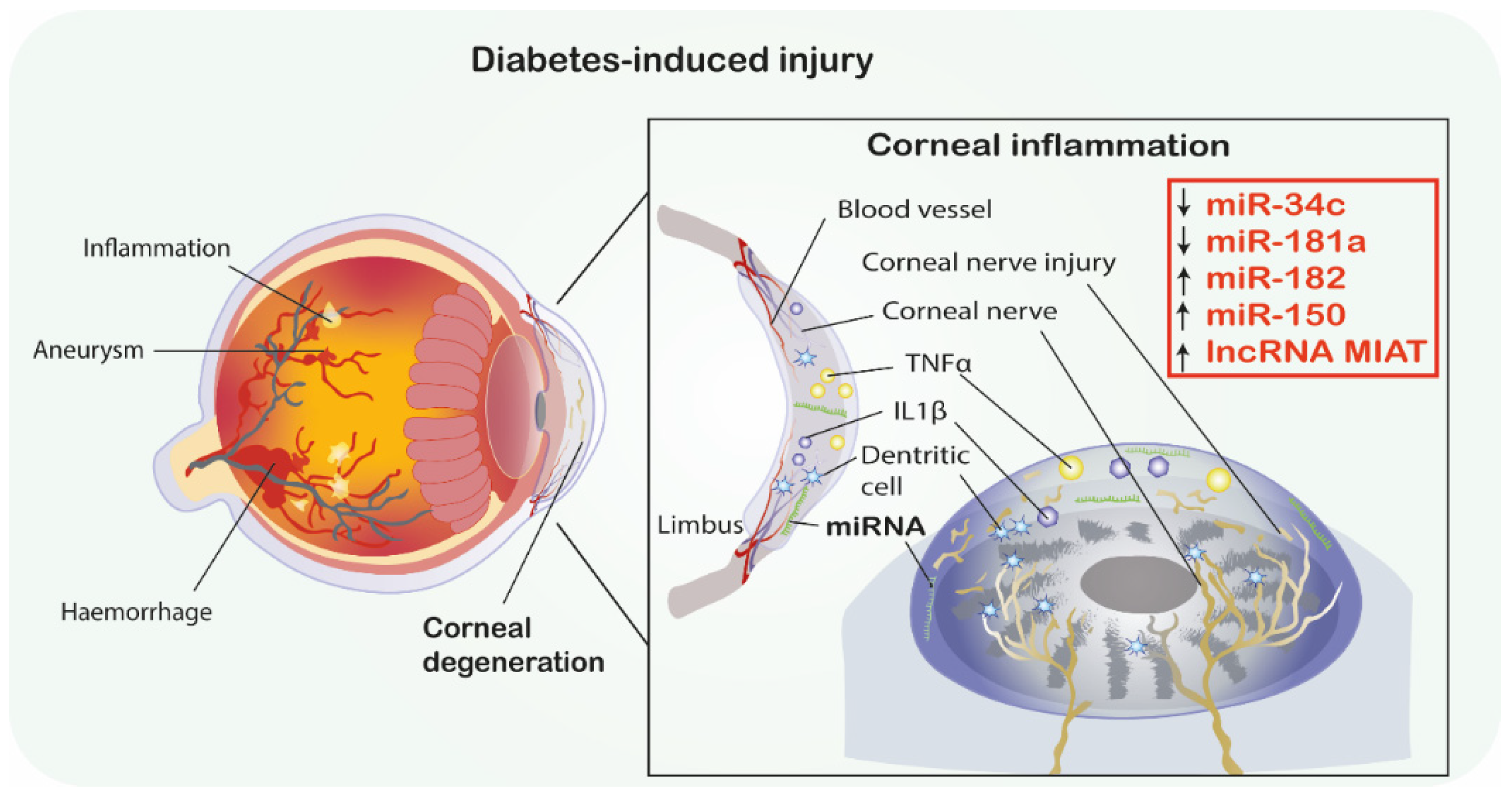
Diabetic corneal neuropathy (DCN) belongs to a variety of common diabetic-associated ophthalmic complications. DCN is demonstrated by simultaneously decreased neuronal fiber density and length, resulting in the onset of neurotrophic ulcer and progressive visual loss [37]. In this regard, Hu et al. conducted a study to analyze the role of miR-34c in DCN associated with T1DM. Researchers utilized both in vivo and in vitro studies. In trigeminal tissue in the diabetic mouse model, miR-34c expression was significantly increased compared to controls. The in vitro studies showed that inhibiting miR-34c resulted in an increased growth of neurites and increased total length of trigeminal sensory neurons. The in vivo study showed that subconjunctival injections of miR-34c antagomir (inhibitor of miRNA) increased corneal nerve density and promoted epithelial wound healing via increased nerve fiber regeneration. In terms of the underlying pathological mechanisms, the autophagy-related proteins, namely, Atg4B and LC3-II, which promote autophagy, were downregulated in diabetic mice trigeminal ganglia. This study predicted the possible interaction of miR-34a and Atg4B by using an in silico tool and confirmed it also with an in vitro experimental analysis. Thus, this study concluded that miR-34c silencing can promote neuroprotective effects in nerve injury via the upregulation of Atg4B and autophagy promotion. Ultimately, the silencing of this miRNA may present as a promising approach by enhancing corneal nerve regeneration and epithelial wound healing in diabetic corneal neuropathies [38] (Figure 1).
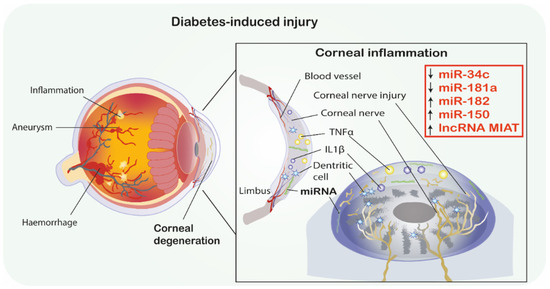
Figure 1.
Diabetic corneal neuropathy. MiRNAs as promising therapeutics in diabetes-induced corneal degeneration. ↑ indicates the mimic-use as treatment, ↓ indicates inhibitor-use as treatment. Abbreviations: IL-1β, Interleukin 1β; TNF-α, Tumor necrosis factor α; MIAT, Myocardial infarction associated transcript; miRNA-miR, microRNA; lncRNA, long non-coding RNA.
Moreover, the same research group investigated the neuroprotective role of miR-181a in mice trigeminal ganglia neurons serving as a model of the T1DM corneal nerve. Firstly, it was found that miR-181a is upregulated in diabetic trigeminal cells. The in vitro study showed that trigeminal cells (cultured in a high glucose environment and treated with miR-181a antagomir) showed a significant increase in axonal growth compared to the control groups. Moreover, subconjunctival injections of the miR-181a inhibitor, performed after epithelial scraping, accelerated the corneal epithelium damage repair in vivo. The density of the corneal plexus in the treated group was higher than in negative-controls. Furthermore, miR-181a antagomir therapy was correlated with increased expression of ATG5, LC3B-II and Bcl-2 proteins which are associated with autophagy promotion and apoptosis inhibition. In summary, the miR-181a antagomir treatment was correlated with the upregulation of anti-apoptotic and autophagy enhancing proteins as well as acceleration of neuronal axon growth and corneal tissue damage repair in diabetic mice. Thus, miR-181a inhibition seems to have a neuroprotective effect on diabetic corneal nerve [39] (Figure 1).
An important gene involved in neuroprotection, especially sensory neurons in diabetics, is SIRT1. Wang et al. [33] conducted a study to analyze the correlation between corneal tissue regeneration and miRNA associated with SIRT1 in diabetic mice with corneal neuropathy. The study reported that SIRT1 was downregulated in the trigeminal sensory neurons of diabetic mice. Additionally, SIRT1 was overexpressed via subconjunctival injection and in miRNA microarray and PCR validation analysis in trigeminal cells. Ultimately, the study showed that miR-182 was significantly upregulated by SIRT1 in these cells. Importantly, the overexpression of miR-182 promoted axonal growth in diabetic trigeminal cells and therefore reversed the negative impact of high glucose conditions. The in vivo investigations showed that miR-182 injections increased corneal nerve density, improved corneal sensation and reduced corneal epithelium defect. The in silico analysis showed that NOX4, associated with ROS production, is increased in diabetic trigeminal cells, and is additionally a direct target gene of miR-182. The miR-182 injection caused a downregulation of NOX4, thus promoting corneal nerve regeneration in diabetic mice. In conclusion, the research presented a correlation between SIRT1, miR-182 and NOX4 as well as their interactions regarding corneal healing and trigeminal nerve innervation in diabetes [40].
Wu et al. [41] analyzed retinal miRNA expression in diabetic retinopathies utilizing a STZ (streptozocin)-induced diabetic animal model. STZ injections caused retinal capillary dilatation, interstitial edema and various other pathological changes of the capillary basement membrane, endothelial cells and mitochondria. MiRNA microarray analysis showed that 37 retinal miRNAs were altered in diabetic rats. However, only 17 of these were confirmed in qRT-PCR analysis. It was found that miR-182, miR-96, miR-183, miR-211, miR-204 and miR-124 were increased with diabetic retinopathy development, whereas miR-199a-3p, miR-10b, miR-10a, miR-219-2-3p, miR-144 and miR-338 were decreased. Importantly, authors mentioned that important angiogenesis factors, including VEGF and PEDF, are direct targets of miR-199a-3p and miR-363, suggesting that these miRNAs could be crucial for capillary changes in diabetic retinopathy. Thus, specific miRNAs are associated with DR development and their modulation may be a novel therapeutic target for DR treatment [41], (Table 1).

Table 1.
Potential miRNAs involved in neurodegeneration.
2.1.2. MiRNAs Involved in Insulin Signaling Pathways in Neurodegeneration
Alzheimer’s disease (AD) is the most common neurodegenerative disorder demonstrated by β-amyloid and tau protein aggregation in the brain that results in gradual tissue atrophy, nerve loss and ultimately progressive cognitive impairment. The underlying mechanism of AD has been extensively researched [48]. One of the postulated hypotheses is the alteration of insulin signaling which has been linked to diabetes and neurodegenerative disease development [49]. The role of miR-302 in neuroprotection against neurotoxicity induced by amyloid-β (Aβ) has been studied both in vitro and in vivo. MiR-302 transfection protected against Aβ-induced apoptosis, mitochondrial dysfunction and insulin resistance via stimulating the PI3K/Akt signaling pathway. Additionally, miR-302 activated the Akt/GSK3β axis which attenuated tau hyperphosphorylation involved in AD pathogenesis. Besides, treatment of miR-302 inhibits ROS accumulation through Akt-induced upregulation of Nrf2/HO-1 pathway and therefore protects against Aβ-induced neurotoxicity. Nrf2 is particularly involved in protection against oxidative stress impairment via increasing antioxidant reaction agents such as HO-1 [50]. Further analysis showed that overexpression of miR-302 inhibited PTEN expression via Akt signaling pathway activation which induced Nanog expression. Of note, reduced Nanog expression was found in AD patients. Silencing of Nanog expression was associated with tau hyperphosphorylation and neurotoxicity due to Akt/GSK3 axis inhibition [51]. MiR-302 treatment re-activated Akt/GSK3 signaling and inhibited AD progression. In addition, miR-302 is encoded in the LARP7 gene and amongst patients with AD, the expression of LARP7 was markedly decreased [44,52]. Overall, miR-302 is able to prevent AD progression through the activation of the Akt signaling pathway [44].
Studies have shown that the inhibition of PDCD4 gene decreased apoptosis, and miR-21 was found as a direct target of PDCD4 [53,54]. This explains why miR-21 treatment in neuronal damage due to diabetes was studied in DM rats with cerebral infarction (CI). Notably, the DM+CI rats which were transfected with miR-21 mimic and those that were transfected with PDCD4 siRNA (si-PDCD4) demonstrate better scores in motoric tests than in control groups. Additionally, analysis of rat brain tissue with miR-21 mimics DM+CI demonstrated decreased expression of proapoptotic markers, namely PTEN, FasL and PDCD4. Neuron differentiation markers (NeuN protein and neural-specific βIII-tubulin) were highly upregulated [55,56]. This evidence suggested that miR-21 may promote nerve cell regeneration via diminishing the apoptosis ratio by PDCD4 downregulation [45].
Venkat et al. [46] analyzed the role of miR-126 and exosomes derived from mouse brain endothelial cells (EC-exo) as a treatment for stroke in diabetic mice. In vivo analysis showed DM-stroke mice were injected with EC-exo or inhibitors of miR-126 + EC-exo. In the DM-stroke model, expression of miR-126 both in serum and brain tissue were significantly decreased when compared to mice with stroke without diabetes. Treatment with EC-exo significantly increased miR-126 expression in serum and brain tissue. Importantly, DM-stroke mice treated with EC-exo had an increased axon and myelin density, vascular density and arterial diameter when compared to control groups. EC-exo treatment was correlated with improvement of cognitive abilities and neurological function. Interestingly, this positive effect was not observed in case of miR-126 knockdown in EC-exo treatment, thus miR-126 seems to play a regulatory role in EC-exo neuro-regenerative effect. Similar findings were reported in vitro, as downregulation of miR-126 resulted in reduced angiogenesis and axon outgrowth. Thus, studies have suggested that miR-126 may play a key role in EC-exo neurorestorative effects in DM-stroke mice [46].
TRPM7 is responsible for magnesium homeostasis, therefore is also involved in glucose and insulin metabolism, whereas miR-34a plays a role in beta-cell apoptosis [57,58]. Zhang et al. [43] conducted a study to investigate the effect of silencing TRPM7/miR-34a in mice with T1DM. Firstly, both TRPM7 and miR-34a were found to be upregulated in T1DM mice. Silencing of TRPM7/miR-34a resulted in improved spatial learning and memory abilities. Moreover, an increased number of neurons in the hippocampal region and improved neuronal structures were observed after TRPM7/miR-34a silencing compared to control and T1DM groups. Reduction in T1DM typical hippocampal changes like swollen mitochondria, vacuolar degeneration and apoptosis symptoms were observed after TRPM7/miR-34a silencing. Apoptosis rate was reduced as the expressions of pro-apoptotic Bax, cyt-c, and cleaved-caspase-3 were significantly decreased, while Bcl-2, an important anti-apoptotic protein, increased notably. Consequently, TRPM7/miR-34a silencing can improve spatial cognitive function and hippocampal neurogenesis in T1DM mice [43]. Further clinical investigations are needed to evaluate the role of TRPM7/miR-34a in T1DM treatment (Table 1).
2.2. Dicer
Dicer is an endoribonuclease acting in the maturation of miRNA [59]. By cooperation with various proteins, Dicer processes miRNA precursors into mature, fully functional miRNAs [60]. The role of Dicer in pituitary dysfunction, neurodegeneration and development of obesity was determined by Schneeberger et al. [42]. Specific hypothalamic neurons (i.e., Agouti-related protein—AgRP, neuropeptide Y—NPY, pro-opiomelanocortin—POMC and cocaine and amphetamine-related transcript—CART) expressing orexigenic and anorexigenic neuropeptides involved in metabolism regulation were studied. Dicer expression was found in both AgRP and POMC neurons. An association between nutrient availability and expression of Dicer was found, as fasting was positively correlated with Dicer. Furthermore, Dicer deficiency in POMC neurons was established in an animal model. Dicer-deficient mice exhibited obesity as well as energy balance and glucose metabolism alterations. In addition, a deletion of Dicer in mice has caused an altered pituitary-adrenal axis, defined as a distinct deficiency of adrenocorticotropic hormone, with normal levels of other pituitary hormones as well as secondary hypoadrenalism. Lack of Dicer also resulted in strict neurodegeneration of POMC neurons, showing a 70% decrease in this subset of neurons in young mice. Taken together, Dicer deletion in POMC neuronal tissue leads to neurodegeneration as well as early onset of obesity and its following metabolic complications. As Dicer is a crucial enzyme for miRNAs formation, its deficiency causes inhibition of miRNA maturation, which leads to disruption of metabolic homeostasis regulated by miRNAs. As discussed above, many miRNAs play an important role in neuronal damage in DM, therefore Dicer alteration is indirectly associated with neurodegeneration/neurogenesis due to diabetes [42].
3. LncRNAs and Their Links to Neurogenesis and Nerve Regeneration in Diabetes
Recent evaluations strengthen the major role of lncRNAs in a variety of DM-induced disorders demonstrated by progressive nerve loss i.e., AD, Parkinson’s disease (PD), Huntington disease (HD) or IS. LncRNAs utility is broadly discussed with regard not only to diagnosis or prognosis but also as a therapeutic approach. Studies also suggest their crucial role in reversing these effects via promoting signaling pathways involved in nerve regeneration. Therefore, analysis of particular mechanisms involved in both neuronal injury and repairment seems crucial.
3.1. LncRNAs Involved in Neurodegeneration and Regeneration
3.1.1. LncRNAs in Diabetic Retinopathy
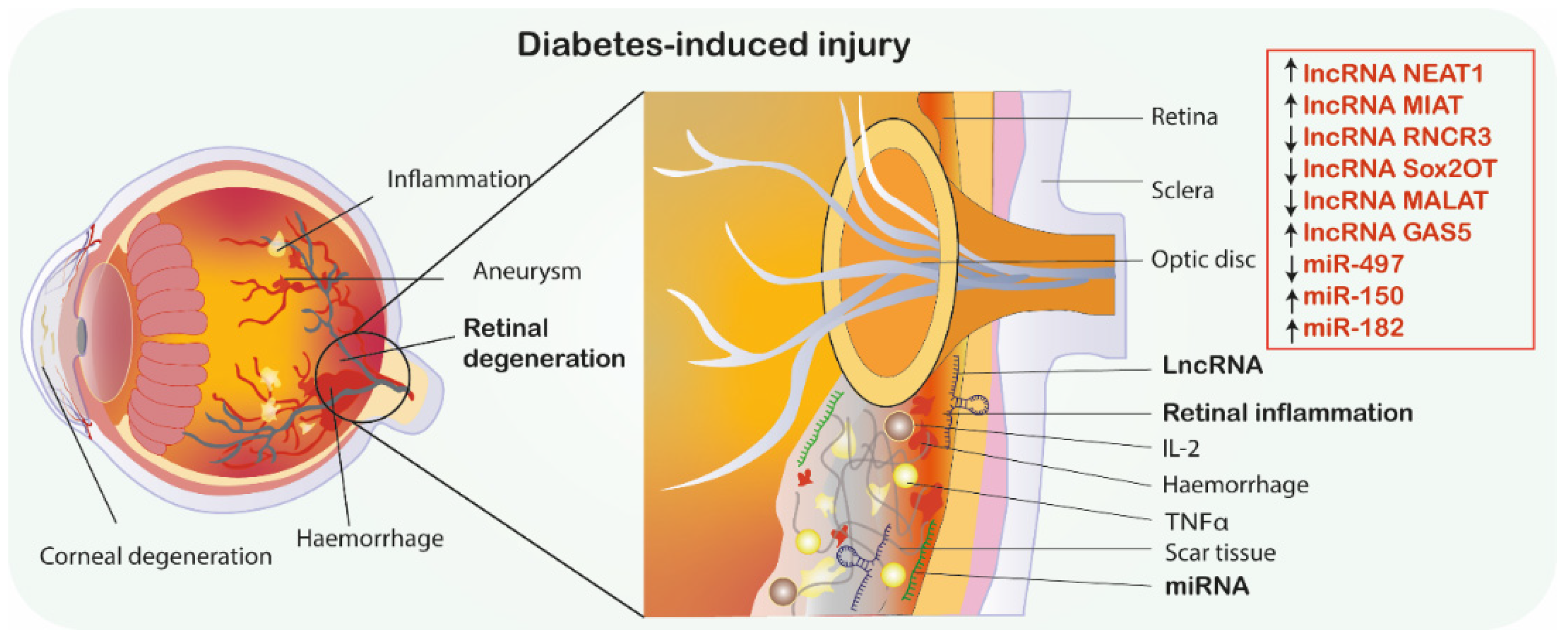
Neurovascular dysfunction is a primary and major cause of diabetes complications, and nervous and vascular systems are regulated by mutual mediators. Studies showed that alteration of lncRNAs is involved in microvascular degeneration and high glucose-induced neuron injury [20,61,62]. Understanding underlying mechanisms responsible for neurovascular interactions could contribute to discovering novel therapeutic strategies. Therefore, Jiang et al. [19] analyzed the role of lncRNA myocardial infarction associated transcript (MIAT), which is found to be highly expressed in neurons and glial cells under hypoxia and oxidative stress conditions in diabetic retinopathy. In vitro evaluation revealed that MIAT inhibition significantly diminished gliosis via downregulation of both glial fibrillary acidic protein (GFAP) and vimentin (known as markers of Müller glial cells). Downregulation of those mediators by MIAT inhibition increased oxidative stress, apoptosis ratio, mitochondrial depolarization and reduced Müller cells viability. These results suggested that MIAT knockdown promoted neurovascular damage. MIAT knockdown significantly enhanced microvascular progressive damage. Further evaluation demonstrated that expression of neurovascular regulators, namely BDNF, NGF, NT3, Ang-1 and VEGF was downregulated by MIAT knockdown. Moreover, the study found that miR-150-5p can directly target MIAT and VEGF, suggesting its involvement in maintenance of neurovascular functionality. Hence, authors showed that lncRNA MIAT also plays an important role in microvascular dysfunction induced by DM and MIAT/miR-150/VEGF axis may represent a further pharmacological target for treating neurovascular-related disorders [19] (Figure 2, Table 2).
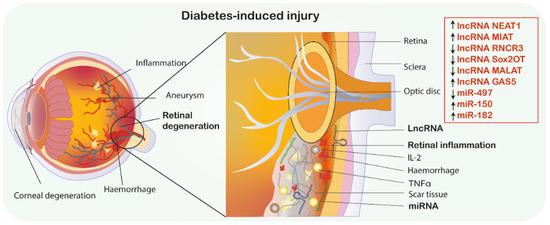
Figure 2.
Diabetic retinopathy. MiRNAs/lncRNAs as promising therapeutics in diabetes-induced retinal degeneration. ↑ indicates the mimic-use as treatment, ↓ indicates inhibitor-use as treatment. Abbreviations: GAS5, Growth arrest-specific transcript 5; IL-2, Interleukin 2; MIAT, Myocardial infarction associated transcript; NEAT1, Nuclear paraspeckle assembly transcript 1; RNCR3, Retinal non-coding RNA3, Sox2OT, Sox2 overlapping transcript; TNF-α, Tumor necrosis factor α; MALAT1, metastasis-associated lung adenocarcinoma transcript 1; miRNA-miR, microRNA; lncRNA, long non-coding RNA.

Table 2.
Potential lncRNAs involved in neurodegeneration.
Growth factors modulate the physiological growth, formation and restoration of all tissues, including neuro-regeneration, and are also affected by pathological conditions [63,64]. VEGF is primarily responsible for functioning of endothelial cell proliferation and migration, as well as collagen production. Importantly, VEGF has been correlated with induced permeability of the blood–retina barrier and increased neovascularization in DM [65]. While VEGF is suggested as a promising biomarker of early stages of diabetic retinopathy, further correlation analysis should be conducted between lncRNAs and VEGF. Apart from VEGF, another important growth factor, which is involved in both neurodegenerative disease and diabetes, is BDNF [66,67,68,69]. It plays an important role in nervous system maturation while supporting the survival of neurons and neuro-regeneration after injuries. Many studies have shown the correlation of BDNF with miRNAs in diabetes-induced neurodegeneration. Studies are not only limited to in vitro and in vivo analysis, but several human studies were also conducted regarding BDNF-miRNAs interaction in diabetes, AD, PD, HD, multiple sclerosis and ischemic stroke [70]. On the other hand, very limited studies have shown the relation of lncRNAs with BDNF in neurodegenerative disease and diabetes, which are mainly in vitro and in vivo analysis [47,71,72,73,74]. For example, a previous study showed that high glucose condition caused Müller glial cells apoptosis by downregulating lncRNA NEAT1. NEAT1 treatment suppressed the apoptosis of retina Müller glial cells after diabetic retinopathy through modulating miR-497/BDNF cascade (by downregulating miR-497 and upregulating BDNF) [47]. Therefore, further studies are needed aiming to analyze the interaction between BDNF and lncRNAs in diabetes-induced neurodegeneration (Figure 2).
Another similar study aimed to analyze whether lncRNA RNCR3 mediate DM-induced retinal neurodegeneration by assessment of retinal glial reactivity which is an early manifestation of retinopathy. Both in vivo and in vitro evaluation revealed that RNCR3 knockdown lead to significant reduction of glial cell reactivity, demonstrated by diminished expression of GFAP, vimentin, proinflammatory cytokines such as IL-2, VEGF, MCP-1 or TNFα and decreased ratio of apoptotic retinal cells. Additionally, in vitro analysis revealed that RNCR3 knockdown can reverse progressive visual function loss. Thus, RNCR3 may be a promising target in preventing DM-related retinal neurodegeneration [75].
The role of lncRNA Sox2OT in diabetic retinopathy was also investigated. Previous study involved in vitro model of retinal ganglion cells (RGCs) and in vivo model of diabetic mice. Firstly, it was found that high glucose levels and oxidative stress downregulated Sox2OT expression in vitro. Similar findings were also reported in an animal model. Additionally, mice that were injected with a Sox2OT-inhibitor improved visual function, showing the protective role of Sox2OT inhibitor on RGC. Moreover, Sox2OT knockdown decreased ROS production and upregulated the activity of oxidative stress-related enzymes, which decreased oxidative stress-induced neuronal dysfunction both in in vitro and in vivo analysis. Furthermore, Sox2OT knockdown caused activation of NRF2/ARE signaling pathway. NRF2 is an important transactivator and activates ARE-dependent gene expression of antioxidative and cytoprotective proteins including HO-1 [80]. Taken together, Sox2OT knockdown protects against high glucose-induced RGC injury through the NRF2/HO1 signaling pathway, and seems to be a promising therapeutic approach for the prevention and treatment of diabetes-induced retinal neurodegeneration [77].
Another promising lncRNA MALAT1 on diabetic retinal neurodegeneration was studied by Zhang et al. [78], who found that retinal MALAT1 was upregulated in STZ-induced diabetic animal models. MALAT1 was silenced by MALAT1-siRNA injection in order to define its role in the retinal neurodegenerative process. Scotopic and photopic electroretinograms were performed to reflect the morphological and functional changes in rod and cone cells. The a-wave and b-wave amplitudes as well as outer nuclear layer thickness were lower both in diabetic control and diabetic MALAT1-siRNA models compared to normal control group. However, the amplitudes and outer nuclear layer were still significantly higher and thicker in the MALAT1-siRNA mice compared to diabetic control group, suggesting that silencing of MALAT1 shows protective effects on retinal photoreceptors and decreases retinal neurodegeneration due to diabetes [78] (Figure 2, Table 2).
LncRNA GAS5 has been documented to serve an important role in numerous signaling pathways, including insulin, mTOR and AKT [81,82,83]. Jiang et al. [79] investigated the influence of lncRNA GAS5 on endoplasmic reticulum stress and apoptosis in diabetic retinopathy. In this study, HG condition in retinal pigment epithelial cells resulted in increased endoplasmic reticulum stress, enhanced apoptosis and inflammation. The treatment of lncRNA GAS5 under HG conditions significantly reduced ER stress, apoptosis as well as inflammatory cytokines, such as TNF-α, IL-1β and IL-6. Consequently, the study indicates that lncRNA GAS5 plays a crucial role in diabetic retinopathy pathogenesis and further studies are needed to clarify its therapeutic potential [79].
3.1.2. LncRNA Involved in Neuronal Apoptosis, Autophagy and Oxidative Stress
DM associated oxidative stress and apoptosis are strongly suggested as crucial factors in progressive neurodegeneration and play a role in promoting earlier onset of various diseases including dementia, AD. Accumulating evidence indicates a significant role of lncRNAs in inhibition of pathways involved in progressive neurogenesis, therefore their potential therapeutic usage is postulated. The study by Yu et al. [76] aimed to investigate both in vitro and in vivo lncRNA H19 roles in DM-induced oxidative stress and apoptosis in hippocampal neurons. LncRNA H19 is known as a crucial glucose metabolism factor and is associated with insulin-like growth factor 2 (IGF2), a regulator of oxidative stress and apoptosis via targeting Bax, caspase-3 and Bcl-2. Downregulation of lncRNA H19 suppressed IGF2 methylation, which in turn increased the expression of IGF2 and therefore inhibited apoptosis as well as oxidative stress in hippocampal neurons. Therefore, it was hypothesized that downregulation of lncRNA H19 could be a potential therapeutic target in diminishing neurodegeneration [76].
4. Concluding Remarks and Limitations
Increasing incidence of DM and its complications constitute a major medical care burden worldwide. Asymptomatic onset and constant progression of DM-associated neurovascular complications in line with limited diagnostic and therapeutic strategies are deemed crucial in searching for novel biomarkers and therapeutics. Since miRNAs and lncRNAs seem to play pivotal roles in modulation of DM-induced neurodegeneration, their molecular relation is broadly discussed. Despite the fact that underlying mechanisms of diabetic neurodegeneration are still not fully recognized, prior studies highlighted the pivotal role of apoptosis, oxidative stress, inflammation and mitochondrial dysfunction. High glucose-induced cell apoptosis and nerve degeneration along with a reduced rate of nerve regeneration play an important role in the progression of peripheral neuropathy among diabetic patients [84]. The correlation between non-coding RNAs (including miRNAs and lncRNAs) and nerve regeneration has been demonstrated in numerous studies. There are several non-coding RNAs found to be downregulated or upregulated in DM. Several studies demonstrated the promising role of these molecules as potential therapeutic approaches in miRNA- and lncRNA-based novel treatments. As described above, this novel treatment can be achieved by using antagonists or mimics of miRNAs/lncRNAs, as some of those molecules’ silencing shows the protective effect, whereas some of these show the protective effect by overexpressing. Yet the detailed mechanism of action of described miRNAs and lncRNAs on neurodegeneration due to diabetes-related oxidative stress and inflammation has not been fully explained and more studies need to be conducted.
Author Contributions
J.J.-P., C.E. contributed to the data collection and elaboration, writing and approval of manuscript; and is guarantor of the article. L.S., M.W., A.G., T.A., P.C., D.J., W.-L.L., D.M.-G., M.P., and C.E. contributed discussion and writing and approval of manuscript. D.M.-G. contributed valuable revision of manuscript. C.E. contributed valuable contributions to graphical designs. The corresponding author attests that all listed authors meet authorship criteria and that no others meeting the criteria have been omitted. All authors have read and agreed to the published version of the manuscript.
Funding
M.P. supported financially as part of the research grant ‘OPUS’ from the National Science Center, Poland (grant number 2018/31/B/NZ7/01137) and internal funding of the Department of Experimental and Clinical Pharmacology, Medical University of Warsaw, Centre for Preclinical Research and Technology CEPT, Warsaw, Poland.
Acknowledgments
This paper was written as a part of cooperation of the international scientific group I-COMET (International Cardiovascular and Cardiometabolic Research Team, www.icomet.science).
Conflicts of Interest
The authors state there are no conflict of interest.
Abbreviations
| AGEs | Advanced glycation end products |
| AgRP | Agouti-related protein |
| AD | Alzheimer’s disease |
| Aβ | Amyloid-β |
| Ang-1 | Angiopoietin 1 |
| ARC | Arcuate nucleus of the hypothalamus |
| Atg4B | Autophagy related gene 4B |
| Bcl-2 | B-cell lymphoma 2 |
| BDNF | Brain-derived neurotrophic factor |
| CNS | Central nervous system |
| CSVD | Cerebral small vessel disease |
| CART | Cocaine and amphetamine-related transcript |
| DM | Diabetes Mellitus |
| DCN | Diabetic corneal neuropathy |
| DPN | Diabetic peripheral neuropathy |
| Dicer | Endoribonuclease |
| DIO | Diet-induced obesity |
| DRG | Dorsal root ganglia |
| ECs | Endothelial cells |
| GAS5 | Growth arrest-specific transcript 5 |
| GFAP | Glial fibrillary acidic protein |
| GFP | Green fluorescent protein |
| HO-1 | Heme oxygenase-1 |
| HG | High glucose |
| hCMEC/D3 | Human cerebral endothelial cell model |
| HD | Huntington’s Disease |
| IRS-1 | Increased insulin receptor substrate 1 |
| iPSC | Induced pluripotent stem cells |
| IRS-1 | Insulin receptor substrate 1 |
| IRAK1 | Interleukin 1 Receptor Associated Kinase 1 |
| IL-1β | Interleukin-1β |
| IL-2 | Interleukin 2 |
| IL-6 | Interleukin 6 |
| IP | Intraperitoneal |
| LARP7 | La ribonucleoprotein domain family member 7 |
| lncRNA | Long noncoding RNA, |
| MSC | Mesenchymal stromal cells, |
| MALAT1 | Metastasis-associated lung adenocarcinoma transcript 1 |
| miRs | MicroRNAs, miRNAs |
| LC3-II | Microtubule-associated protein 1 light chain 3 |
| miRISC | MiRNA induced silencing complex |
| MCP-1 | Monocyte chemotactic protein-1 |
| NOX4 | NADPH oxidase 4 |
| NTC | Negative Treated Control |
| NPY | Neuropeptide Y |
| NEAT1 | Nuclear paraspeckle assembly transcript 1 |
| Nrf2 | Nuclear factor erythroid 2-related factor 2 |
| NF-κB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| PD | Parkinson’s Disease, |
| PTIP 1 | Pax transactivation domain-interacting protein 1 |
| p-NFkB | Phospho NFkB |
| PDE3A | Phosphodiesterase 3a |
| POMC | Pro-opiomelanocortin |
| PDCD4 | Programmed cell death protein 4 |
| Ago-2 | Protein argonaute-2 |
| qPCR | Quantitative real-time PCR |
| ROS | Reactive oxygen species |
| RAGE | Receptor for advanced glycation end |
| RGCs | Retinal ganglion cells |
| Sirt1 | Silent mating type information regulation 2 homolog 1 |
| Sox2OT | Sox2 overlapping transcript |
| STZ | Streptozocin |
| Tβ4 | Thymosin beta 4 |
| TRAF6 | TNF Receptor Associated Factor 6 |
| TLR4 | Toll-like receptor 4 |
| TRPM7 | Transient receptor potential melastatin 7 |
| TRAF6 | Tumor necrosis factor (TNFR)-associated factor 6 |
| TNF-α | Tumor necrosis factor α |
| T1DM | Type 1 Diabetes Mellitus |
| UTR | 30 untranslated region |
| VCAM-1 | Vascular cell adhesion molecule-1 |
| VEGF | Vascular endothelial growth factor |
| YFP | Yellow fluorescent protein |
References
- American Diabetes Association Diagnosis and classification of diabetes mellitus. Diabetes Care 2014, 37 (Suppl. 1), S81–S90. [CrossRef] [PubMed]
- Saeedi, P.; Petersohn, I.; Salpea, P.; Malanda, B.; Karuranga, S.; Unwin, N.; Colagiuri, S.; Guariguata, L.; Motala, A.A.; Ogurtsova, K.; et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res. Clin. Pract. 2019, 157, 107843. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Ley, S.H.; Hu, F.B. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat. Rev. Endocrinol. 2018, 14, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Forbes, J.M.; Cooper, M.E. Mechanisms of diabetic complications. Physiol. Rev. 2013, 93, 137–188. [Google Scholar] [CrossRef]
- Ramos-Rodriguez, J.J.; Ortiz, O.; Jimenez-Palomares, M.; Kay, K.R.; Berrocoso, E.; Murillo-Carretero, M.I.; Perdomo, G.; Spires-Jones, T.; Cozar-Castellano, I.; Lechuga-Sancho, A.M.; et al. Differential central pathology and cognitive impairment in pre-diabetic and diabetic mice. Psychoneuroendocrinology 2013, 38, 2462–2475. [Google Scholar] [CrossRef]
- Moran, C.; Beare, R.; Wang, W.; Callisaya, M.; Srikanth, V. For the Alzheimer’s Disease Neuroimaging Initiative (ADNI) Type 2 diabetes mellitus, brain atrophy, and cognitive decline. Neurology 2019, 92, 823–830. [Google Scholar] [CrossRef]
- Oikawa, Y.; Shimada, A. [Type 1 diabetes]. Nihon Rinsho 2015, 73, 1997–2002. [Google Scholar]
- Thevis, M.; Thomas, A.; Schänzer, W. Insulin. Handb. Exp. Pharm. 2010, 209–226. [Google Scholar] [CrossRef]
- Bluestone, J.A.; Herold, K.; Eisenbarth, G. Genetics, pathogenesis and clinical interventions in type 1 diabetes. Nature 2010, 464, 1293–1300. [Google Scholar] [CrossRef]
- Pemp, B.; Palkovits, S.; Howorka, K.; Pumprla, J.; Sacu, S.; Garhöfer, G.; Bayerle-Eder, M.; Schmetterer, L.; Schmidt-Erfurth, U. Correlation of retinal neurodegeneration with measures of peripheral autonomic neuropathy in type 1 diabetes. Acta Ophthalmol. 2018, 96, e804–e810. [Google Scholar] [CrossRef]
- Gundogan, F.C.; Akay, F.; Uzun, S.; Yolcu, U.; Çağıltay, E.; Toyran, S. Early Neurodegeneration of the Inner Retinal Layers in Type 1 Diabetes Mellitus. Ophthalmologica 2016, 235, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Taylor, R. Type 2 diabetes: Etiology and reversibility. Diabetes Care 2013, 36, 1047–1055. [Google Scholar] [CrossRef]
- Bharadwaj, P.; Wijesekara, N.; Liyanapathirana, M.; Newsholme, P.; Ittner, L.; Fraser, P.; Verdile, G. The Link between Type 2 Diabetes and Neurodegeneration: Roles for Amyloid-β, Amylin, and Tau Proteins. J. Alzheimer’s Dis. 2017, 59, 421–432. [Google Scholar] [CrossRef]
- Yoneda, R.; Ueda, N.; Uranishi, K.; Hirasaki, M.; Kurokawa, R. Long noncoding RNA reduces cyclin D1 gene expression and arrests cell cycle through RNA mA modification. J. Biol. Chem. 2020, 295, 5626–5639. [Google Scholar] [CrossRef] [PubMed]
- Wolska, M.; Jarosz-Popek, J.; Junger, E.; Wicik, Z.; Porshoor, T.; Sharif, L.; Czajka, P.; Postula, M.; Mirowska-Guzel, D.; Czlonkowska, A.; et al. Long Non-coding RNAs as Promising Therapeutic Approach in Ischemic Stroke: A Comprehensive Review. Mol. Neurobiol. 2020, 1–9. [Google Scholar] [CrossRef]
- Eyileten, C.; Wicik, Z.; De Rosa, S.; Mirowska-Guzel, D.; Soplinska, A.; Indolfi, C.; Jastrzebska-Kurkowska, I.; Czlonkowska, A.; Postula, M. MicroRNAs as Diagnostic and Prognostic Biomarkers in Ischemic Stroke-A Comprehensive Review and Bioinformatic Analysis. Cells 2018, 7, 249. [Google Scholar] [CrossRef] [PubMed]
- Pordzik, J.; Pisarz, K.; De Rosa, S.; Jones, A.D.; Eyileten, C.; Indolfi, C.; Malek, L.; Postula, M. The Potential Role of Platelet-Related microRNAs in the Development of Cardiovascular Events in High-Risk Populations, Including Diabetic Patients: A Review. Front. Endocrinol. 2018, 9, 74. [Google Scholar] [CrossRef]
- Pordzik, J.; Jakubik, D.; Jarosz-Popek, J.; Wicik, Z.; Eyileten, C.; De Rosa, S.; Indolfi, C.; Siller-Matula, J.M.; Czajka, P.; Postula, M. Significance of circulating microRNAs in diabetes mellitus type 2 and platelet reactivity: Bioinformatic analysis and review. Cardiovasc. Diabetol. 2019, 18, 113. [Google Scholar] [CrossRef]
- Jiang, Q.; Shan, K.; Qun-Wang, X.; Zhou, R.-M.; Yang, H.; Liu, C.; Li, Y.-J.; Yao, J.; Li, X.-M.; Shen, Y.; et al. Long non-coding RNA-MIAT promotes neurovascular remodeling in the eye and brain. Oncotarget 2016, 7, 49688–49698. [Google Scholar] [CrossRef]
- Raut, S.K.; Khullar, M. The Big Entity of New RNA World: Long Non-Coding RNAs in Microvascular Complications of Diabetes. Front. Endocrinol. 2018, 9, 300. [Google Scholar] [CrossRef]
- Biswas, S.; Sarabusky, M.; Chakrabarti, S. Diabetic Retinopathy, lncRNAs, and Inflammation: A Dynamic, Interconnected Network. J. Clin. Med. Res. 2019, 8, 1033. [Google Scholar] [CrossRef] [PubMed]
- Ciccacci, C.; Latini, A.; Colantuono, A.; Politi, C.; D’Amato, C.; Greco, C.; Rinaldi, M.E.; Lauro, D.; Novelli, G.; Spallone, V.; et al. Expression study of candidate miRNAs and evaluation of their potential use as biomarkers of diabetic neuropathy. Epigenomics 2020, 12, 575–585. [Google Scholar] [CrossRef] [PubMed]
- Robles-Rivera, R.R.; Castellanos-González, J.A.; Olvera-Montaño, C.; Flores-Martin, R.A.; López-Contreras, A.K.; Arevalo-Simental, D.E.; Cardona-Muñoz, E.G.; Roman-Pintos, L.M.; Rodríguez-Carrizalez, A.D. Adjuvant Therapies in Diabetic Retinopathy as an Early Approach to Delay Its Progression: The Importance of Oxidative Stress and Inflammation. Oxid. Med. Cell. Longev. 2020, 2020, 3096470. [Google Scholar] [CrossRef] [PubMed]
- Yildirim, S.S.; Akman, D.; Catalucci, D.; Turan, B. Relationship between downregulation of miRNAs and increase of oxidative stress in the development of diabetic cardiac dysfunction: Junctin as a target protein of miR-1. Cell Biochem. Biophys. 2013, 67, 1397–1408. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, J.C.R.; Acuña, S.M.; Aoki, J.I.; Floeter-Winter, L.M.; Muxel, S.M. Long Non-Coding RNAs in the Regulation of Gene Expression: Physiology and Disease. Noncoding Rna 2019, 5, 17. [Google Scholar] [CrossRef]
- Iqbal, Z.; Azmi, S.; Yadav, R.; Ferdousi, M.; Kumar, M.; Cuthbertson, D.J.; Lim, J.; Malik, R.A.; Alam, U. Diabetic Peripheral Neuropathy: Epidemiology, Diagnosis, and Pharmacotherapy. Clin. Ther. 2018, 40, 828–849. [Google Scholar] [CrossRef]
- Singh, R.; Kishore, L.; Kaur, N. Diabetic peripheral neuropathy: Current perspective and future directions. Pharm. Res. 2014, 80, 21–35. [Google Scholar] [CrossRef]
- Juster-Switlyk, K.; Smith, A.G. Updates in diabetic peripheral neuropathy. F1000Research 2016, 5, F1000 Faculty Rev-738. [Google Scholar] [CrossRef]
- Rask-Madsen, C.; King, G.L. Vascular complications of diabetes: Mechanisms of injury and protective factors. Cell Metab. 2013, 17, 20–33. [Google Scholar] [CrossRef]
- Roustit, M.; Loader, J.; Deusenbery, C.; Baltzis, D.; Veves, A. Endothelial Dysfunction as a Link Between Cardiovascular Risk Factors and Peripheral Neuropathy in Diabetes. J. Clin. Endocrinol. Metab. 2016, 101, 3401–3408. [Google Scholar] [CrossRef]
- Poggesi, A.; Pasi, M.; Pescini, F.; Pantoni, L.; Inzitari, D. Circulating biologic markers of endothelial dysfunction in cerebral small vessel disease: A review. J. Cereb. Blood Flow Metab. 2016, 36, 72–94. [Google Scholar] [CrossRef] [PubMed]
- Yasmeen, S.; Kaur, S.; Mirza, A.H.; Brodin, B.; Pociot, F.; Kruuse, C. miRNA-27a-3p and miRNA-222-3p as Novel Modulators of Phosphodiesterase 3a (PDE3A) in Cerebral Microvascular Endothelial Cells. Mol. Neurobiol. 2019, 56, 5304–5314. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Chopp, M.; Szalad, A.; Zhang, Y.; Wang, X.; Zhang, R.L.; Liu, X.S.; Jia, L.; Zhang, Z.G. The role of miR-146a in dorsal root ganglia neurons of experimental diabetic peripheral neuropathy. Neuroscience 2014, 259, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Chopp, M.; Lu, X.; Szalad, A.; Jia, L.; Liu, X.S.; Wu, K.-H.; Lu, M.; Zhang, Z.G. miR-146a mediates thymosin β4 induced neurovascular remodeling of diabetic peripheral neuropathy in type-II diabetic mice. Brain Res. 2019, 1707, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chopp, M.; Liu, X.S.; Kassis, H.; Wang, X.; Li, C.; An, G.; Zhang, Z.G. MicroRNAs in the axon locally mediate the effects of chondroitin sulfate proteoglycans and cGMP on axonal growth. Dev. Neurobiol. 2015, 75, 1402–1419. [Google Scholar] [CrossRef] [PubMed]
- Jia, L.; Wang, L.; Chopp, M.; Zhang, Y.; Szalad, A.; Zhang, Z.G. MicroRNA 146a locally mediates distal axonal growth of dorsal root ganglia neurons under high glucose and sildenafil conditions. Neuroscience 2016, 329, 43–53. [Google Scholar] [CrossRef]
- Ziegler, D.; Papanas, N.; Zhivov, A.; Allgeier, S.; Winter, K.; Ziegler, I.; Brüggemann, J.; Strom, A.; Peschel, S.; Köhler, B.; et al. Early detection of nerve fiber loss by corneal confocal microscopy and skin biopsy in recently diagnosed type 2 diabetes. Diabetes 2014, 63, 2454–2463. [Google Scholar] [CrossRef]
- Hu, J.; Hu, X.; Kan, T. MiR-34c Participates in Diabetic Corneal Neuropathy Via Regulation of Autophagy. Investig. Ophthalmol. Vis. Sci. 2019, 60, 16–25. [Google Scholar] [CrossRef]
- Hu, J.; Huang, Y.; Lin, Y.; Lin, J. Protective effect inhibiting the expression of miR-181a on the diabetic corneal nerve in a mouse model. Exp. Eye Res. 2020, 192, 107925. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao, X.; Wu, X.; Dai, Y.; Chen, P.; Xie, L. microRNA-182 Mediates Sirt1-Induced Diabetic Corneal Nerve Regeneration. Diabetes 2016, 65, 2020–2031. [Google Scholar] [CrossRef]
- Wu, J.-H.; Gao, Y.; Ren, A.-J.; Zhao, S.-H.; Zhong, M.; Peng, Y.-J.; Shen, W.; Jing, M.; Liu, L. Altered microRNA expression profiles in retinas with diabetic retinopathy. Ophthalmic Res. 2012, 47, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Schneeberger, M.; Altirriba, J.; García, A.; Esteban, Y.; Castaño, C.; García-Lavandeira, M.; Alvarez, C.V.; Gomis, R.; Claret, M. Deletion of miRNA processing enzyme Dicer in POMC-expressing cells leads to pituitary dysfunction, neurodegeneration and development of obesity. Mol. Metab. 2012, 2, 74–85. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.-J.; Li, J.; Zhang, S.-Y. Effects of TRPM7/miR-34a Gene Silencing on Spatial Cognitive Function and Hippocampal Neurogenesis in Mice with Type 1 Diabetes Mellitus. Mol. Neurobiol. 2018, 55, 1568–1579. [Google Scholar] [CrossRef] [PubMed]
- Li, H.-H.; Lin, S.-L.; Huang, C.-N.; Lu, F.-J.; Chiu, P.-Y.; Huang, W.-N.; Lai, T.-J.; Lin, C.-L. miR-302 Attenuates Amyloid-β-Induced Neurotoxicity through Activation of Akt Signaling. J. Alzheimers. Dis. 2016, 50, 1083–1098. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.-B.; Ji, T.-F.; Zhou, H.-W.; Yu, J.-L. Effects of microRNA-21 on Nerve Cell Regeneration and Neural Function Recovery in Diabetes Mellitus Combined with Cerebral Infarction Rats by Targeting PDCD4. Mol. Neurobiol. 2018, 55, 2494–2505. [Google Scholar] [CrossRef] [PubMed]
- Venkat, P.; Cui, C.; Chopp, M.; Zacharek, A.; Wang, F.; Landschoot-Ward, J.; Shen, Y.; Chen, J. MiR-126 Mediates Brain Endothelial Cell Exosome Treatment-Induced Neurorestorative Effects After Stroke in Type 2 Diabetes Mellitus Mice. Stroke 2019, 50, 2865–2874. [Google Scholar] [CrossRef]
- Li, X.-J. Long non-coding RNA nuclear paraspeckle assembly transcript 1 inhibits the apoptosis of retina Müller cells after diabetic retinopathy through regulating miR-497/brain-derived neurotrophic factor axis. Diab. Vasc. Dis. Res. 2018, 15, 204–213. [Google Scholar] [CrossRef]
- Mattsson, N.; Andreasson, U.; Zetterberg, H.; Blennow, K. Alzheimer’s Disease Neuroimaging Initiative Association of Plasma Neurofilament Light With Neurodegeneration in Patients with Alzheimer Disease. JAMA Neurol. 2017, 74, 557–566. [Google Scholar] [CrossRef]
- Spielman, L.J.; Little, J.P.; Klegeris, A. Inflammation and insulin/IGF-1 resistance as the possible link between obesity and neurodegeneration. J. Neuroimmunol. 2014, 273, 8–21. [Google Scholar] [CrossRef]
- Ma, Q. Role of nrf2 in oxidative stress and toxicity. Annu. Rev. Pharm. Toxicol. 2013, 53, 401–426. [Google Scholar] [CrossRef]
- Sarlak, G.; Vincent, B. The Roles of the Stem Cell-Controlling Sox2 Transcription Factor: From Neuroectoderm Development to Alzheimer’s Disease? Mol. Neurobiol. 2016, 53, 1679–1698. [Google Scholar] [CrossRef] [PubMed]
- Bayfield, M.A.; Yang, R.; Maraia, R.J. Conserved and divergent features of the structure and function of La and La-related proteins (LARPs). Biochim. Biophys. Acta 2010, 1799, 365–378. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Li, X.; Li, Q. Curcumin accelerates the repair of sciatic nerve injury in rats through reducing Schwann cells apoptosis and promoting myelinization. Biomed. Pharm. 2017, 92, 1103–1110. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Zhu, P.; Yang, J.; Liu, X.; Dong, S.; Wang, X.; Chun, B.; Zhuang, J.; Zhang, C. Ischaemic preconditioning-regulated miR-21 protects heart against ischaemia/reperfusion injury via anti-apoptosis through its target PDCD4. Cardiovasc. Res. 2010, 87, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Gusel’nikova, V.V.; Korzhevskiy, D.E. NeuN As a Neuronal Nuclear Antigen and Neuron Differentiation Marker. Acta Nat. 2015, 7, 42–47. [Google Scholar] [CrossRef]
- Person, F.; Wilczak, W.; Hube-Magg, C.; Burdelski, C.; Möller-Koop, C.; Simon, R.; Noriega, M.; Sauter, G.; Steurer, S.; Burdak-Rothkamm, S.; et al. Prevalence of βIII-tubulin (TUBB3) expression in human normal tissues and cancers. Tumour Biol. 2017, 39, 1010428317712166. [Google Scholar] [CrossRef]
- Schlingmann, K.P.; Waldegger, S.; Konrad, M.; Chubanov, V.; Gudermann, T. TRPM6 and TRPM7--Gatekeepers of human magnesium metabolism. Biochim. Biophys. Acta 2007, 1772, 813–821. [Google Scholar] [CrossRef]
- Lin, X.; Guan, H.; Huang, Z.; Liu, J.; Li, H.; Wei, G.; Cao, X.; Li, Y. Downregulation of Bcl-2 expression by miR-34a mediates palmitate-induced Min6 cells apoptosis. J. Diabetes Res. 2014, 2014, 258695. [Google Scholar] [CrossRef]
- Song, M.-S.; Rossi, J.J. Molecular mechanisms of Dicer: Endonuclease and enzymatic activity. Biochem. J. 2017, 474, 1603–1618. [Google Scholar] [CrossRef]
- Koscianska, E.; Starega-Roslan, J.; Krzyzosiak, W.J. The role of Dicer protein partners in the processing of microRNA precursors. PLoS ONE 2011, 6, e28548. [Google Scholar] [CrossRef]
- Shan, K.; Jiang, Q.; Wang, X.-Q.; Wang, Y.-N.-Z.; Yang, H.; Yao, M.-D.; Liu, C.; Li, X.-M.; Yao, J.; Liu, B.; et al. Role of long non-coding RNA-RNCR3 in atherosclerosis-related vascular dysfunction. Cell Death Dis. 2016, 7, e2248. [Google Scholar] [CrossRef] [PubMed]
- Leung, A.; Natarajan, R. Long Noncoding RNAs in Diabetes and Diabetic Complications. Antioxid. Redox Signal. 2018, 29, 1064–1073. [Google Scholar] [CrossRef] [PubMed]
- Gasecka, A.; Siwik, D.; Gajewska, M.; Jaguszewski, M.J.; Mazurek, T.; Filipiak, K.J.; Postuła, M.; Eyileten, C. Early Biomarkers of Neurodegenerative and Neurovascular Disorders in Diabetes. J. Clin. Med. Res. 2020, 9, 2807. [Google Scholar] [CrossRef] [PubMed]
- Adamska, A.; Pilacinski, S.; Zozulinska-Ziolkiewicz, D.; Gandecka, A.; Grzelka, A.; Konwerska, A.; Malinska, A.; Nowicki, M.; Araszkiewicz, A. An increased skin microvessel density is associated with neurovascular complications in type 1 diabetes mellitus. Diab. Vasc. Dis. Res. 2019, 16, 513–522. [Google Scholar] [CrossRef]
- Zhang, Q.; Fang, W.; Ma, L.; Wang, Z.-D.; Yang, Y.-M.; Lu, Y.-Q. VEGF levels in plasma in relation to metabolic control, inflammation, and microvascular complications in type-2 diabetes: A cohort study. Medicine 2018, 97, e0415. [Google Scholar] [CrossRef]
- Eyileten, C.; Mirowska-Guzel, D.; Milanowski, L.; Zaremba, M.; Rosiak, M.; Cudna, A.; Kaplon-Cieslicka, A.; Opolski, G.; Filipiak, K.J.; Malek, L.; et al. Serum Brain-Derived Neurotrophic Factor is Related to Platelet Reactivity and Metformin Treatment in Adult Patients with Type 2 Diabetes Mellitus. Can. J. Diabetes 2019, 43, 19–26. [Google Scholar] [CrossRef]
- Eyileten, C.; Kaplon-Cieslicka, A.; Mirowska-Guzel, D.; Malek, L.; Postula, M. Antidiabetic Effect of Brain-Derived Neurotrophic Factor and Its Association with Inflammation in Type 2 Diabetes Mellitus. J. Diabetes Res. 2017, 2017, 2823671. [Google Scholar] [CrossRef]
- Eyileten, C.; Zaremba, M.; Janicki, P.K.; Rosiak, M.; Cudna, A.; Kapłon-Cieślicka, A.; Opolski, G.; Filipiak, K.J.; Kosior, D.A.; Mirowska-Guzel, D.; et al. Serum Brain-Derived Neurotrophic Factor is Related to Platelet Reactivity but not to Genetic Polymorphisms within BDNF Encoding Gene in Patients with Type 2 Diabetes. Med. Sci. Monit. 2016, 22, 69–76. [Google Scholar] [CrossRef]
- Mirowska-Guzel, D. The role of neurotrophic factors in the pathology and treatment of multiple sclerosis. Immunopharmacol. Immunotoxicol. 2009, 31, 32–38. [Google Scholar] [CrossRef]
- Eyileten, C.; Sharif, L.; Wicik, Z.; Jakubik, D.; Jarosz-Popek, J.; Soplinska, A.; Postula, M.; Czlonkowska, A.; Kaplon-Cieslicka, A.; Mirowska-Guzel, D. The Relation of the Brain-Derived Neurotrophic Factor with MicroRNAs in Neurodegenerative Diseases and Ischemic Stroke. Mol. Neurobiol. 2020, 58, 329–347. [Google Scholar] [CrossRef]
- Li, Y.; Xu, F.; Xiao, H.; Han, F. Long noncoding RNA BDNF-AS inversely regulated BDNF and modulated high-glucose induced apoptosis in human retinal pigment epithelial cells. J. Cell. Biochem. 2018, 119, 817–823. [Google Scholar] [CrossRef]
- Luo, L.; Ji, L.-D.; Cai, J.-J.; Feng, M.; Zhou, M.; Hu, S.-P.; Xu, J.; Zhou, W.-H. Microarray Analysis of Long Noncoding RNAs in Female Diabetic Peripheral Neuropathy Patients. Cell. Physiol. Biochem. 2018, 46, 1209–1217. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Song, C.; Chen, K.; Zhang, X. Inhibition of long non-coding RNA IGF2AS protects apoptosis and neuronal loss in anesthetic-damaged mouse neural stem cell derived neurons. Biomed. Pharm. 2017, 85, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Gong, H.-Y.; Xu, L. PVT1 protects diabetic peripheral neuropathy via PI3K/AKT pathway. Eur. Rev. Med. Pharm. Sci. 2018, 22, 6905–6911. [Google Scholar] [CrossRef]
- Liu, C.; Li, C.-P.; Wang, J.-J.; Shan, K.; Liu, X.; Yan, B. RNCR3 knockdown inhibits diabetes mellitus-induced retinal reactive gliosis. Biochem. Biophys. Res. Commun. 2016, 479, 198–203. [Google Scholar] [CrossRef]
- Yu, J.-L.; Li, C.; Che, L.-H.; Zhao, Y.-H.; Guo, Y.-B. Downregulation of long noncoding RNA H19 rescues hippocampal neurons from apoptosis and oxidative stress by inhibiting IGF2 methylation in mice with streptozotocin-induced diabetes mellitus. J. Cell. Physiol. 2019, 234, 10655–10670. [Google Scholar] [CrossRef]
- Li, C.-P.; Wang, S.-H.; Wang, W.-Q.; Song, S.-G.; Liu, X.-M. Long Noncoding RNA-Sox2OT Knockdown Alleviates Diabetes Mellitus-Induced Retinal Ganglion Cell (RGC) injury. Cell. Mol. Neurobiol. 2017, 37, 361–369. [Google Scholar] [CrossRef]
- Zhang, Y.-L.; Hu, H.-Y.; You, Z.-P.; Li, B.-Y.; Shi, K. Targeting long non-coding RNA MALAT1 alleviates retinal neurodegeneration in diabetic mice. Int. J. Ophthalmol. 2020, 13, 213–219. [Google Scholar] [CrossRef]
- Jiang, L.; Wang, C.; Shen, X. LncRNA GAS5 suppresses ER stress‑induced apoptosis and inflammation by regulating SERCA2b in HG‑treated retinal epithelial cell. Mol. Med. Rep. 2020, 22, 1072–1080. [Google Scholar] [CrossRef]
- Tu, W.; Wang, H.; Li, S.; Liu, Q.; Sha, H. The Anti-Inflammatory and Anti-Oxidant Mechanisms of the Keap1/Nrf2/ARE Signaling Pathway in Chronic Diseases. Aging Dis. 2019, 10, 637–651. [Google Scholar] [CrossRef]
- Li, H.; Liu, Y.; Huang, J.; Liu, Y.; Zhu, Y. Association of genetic variants in lncRNA GAS5/miR-21/mTOR axis with risk and prognosis of coronary artery disease among a Chinese population. J. Clin. Lab. Anal. 2020, 34, 521. [Google Scholar] [CrossRef]
- Chen, L.; Ren, P.; Zhang, Y.; Gong, B.; Yu, D.; Sun, X. Long non‑coding RNA GAS5 increases the radiosensitivity of A549 cells through interaction with the miR‑21/PTEN/Akt axis. Oncol. Rep. 2020, 43, 897–907. [Google Scholar] [CrossRef]
- Zhou, Y.; Chen, B. GAS5‑mediated regulation of cell signaling (Review). Mol. Med. Rep. 2020, 22, 3049–3056. [Google Scholar] [CrossRef]
- Khoshnoodi, M.; Truelove, S.; Polydefkis, M. Effect of diabetes type on long-term outcome of epidermal axon regeneration. Ann. Clin. Transl. Neurol. 2019, 6, 2088–2096. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).