Role of Glucose-Lowering Medications in Erectile Dysfunction
Abstract
1. Introduction
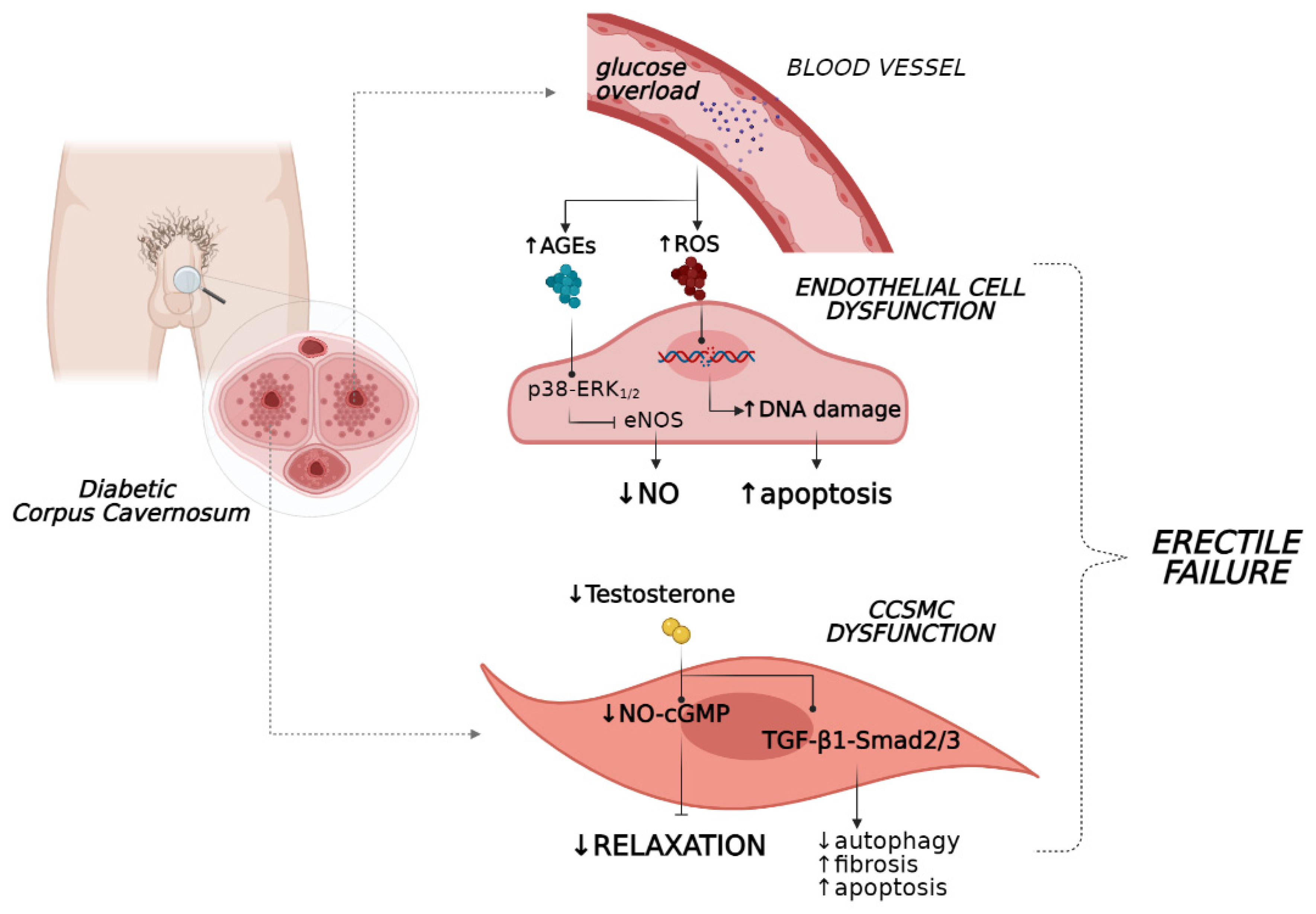
2. Pathophysiology of ED
2.1. Hyperglycaemia
2.2. Hypogonadism
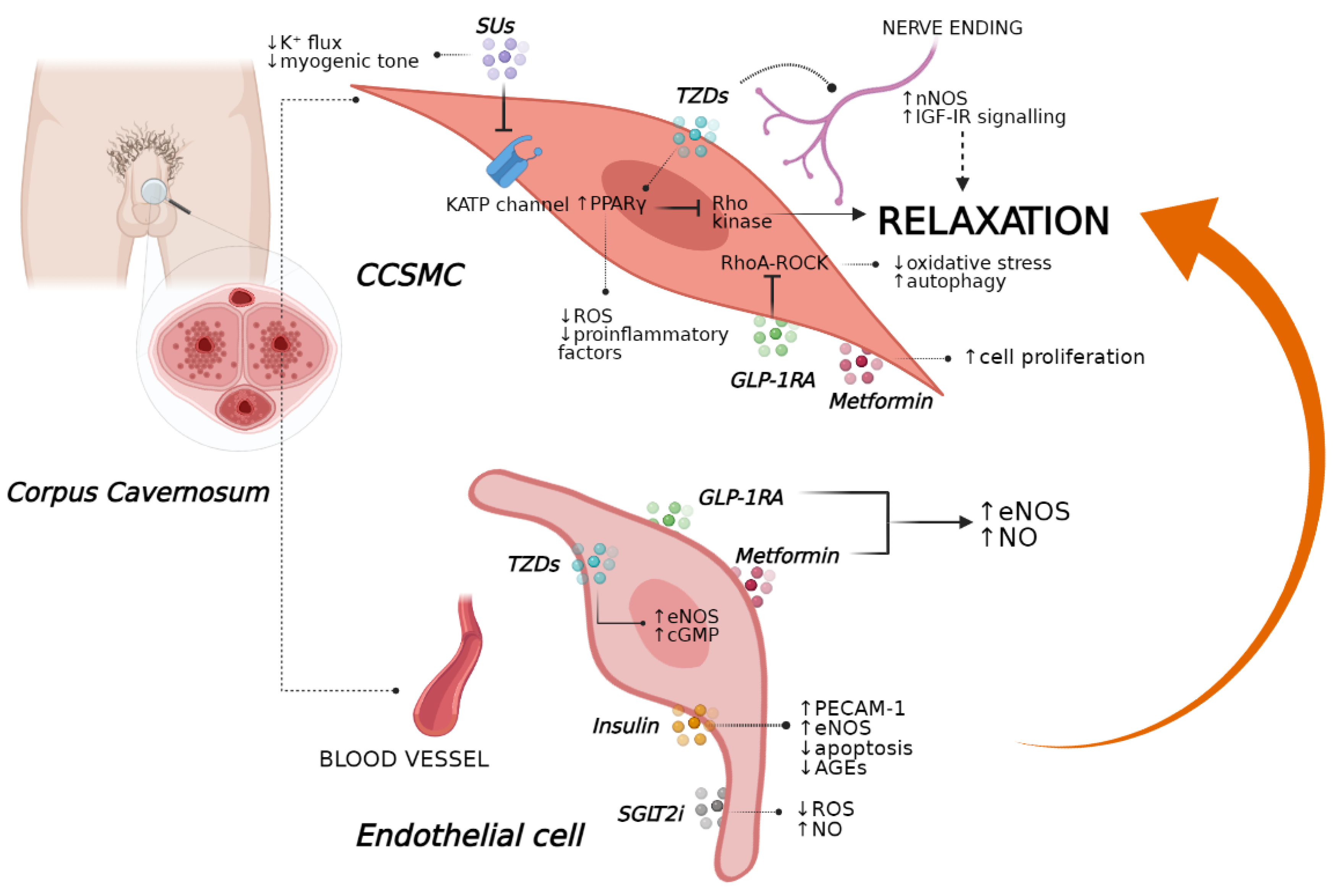
3. Glucose-Lowering Medications
3.1. Insulin
3.2. Sulfonylureas
3.3. Metformin
3.4. Acarbose (ACA)/α-Glucosidase Inhibitors
3.5. Thiazolidinediones
3.6. GLP-1RAs and DPP-4i
3.7. SGLT2is
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Corona, G.; Giorda, C.B.; Cucinotta, D.; Guida, P.; Nada, E.; Aglialoro, A.; Albanese, V.; Albano, S.; Antonangelo, C.; Baccetti, F.; et al. Sexual dysfunction at the onset of type 2 diabetes: The interplay of depression, hormonal and cardiovascular factors. J. Sex. Med. 2014, 11, 2065–2073. [Google Scholar] [CrossRef]
- Kouidrat, Y.; Pizzol, D.; Cosco, T.; Thompson, T.; Carnaghi, M.; Bertoldo, A.; Solmi, M.; Stubbs, B.; Veronese, N. High prevalence of erectile dysfunction in diabetes: A systematic review and meta-analysis of 145 studies. Diabet. Med. 2017, 34, 1185–1192. [Google Scholar] [CrossRef] [PubMed]
- Gandaglia, G.; Briganti, A.; Jackson, G.; Kloner, R.A.; Montorsi, F.; Montorsi, P.; Vlachopoulos, C. A systematic review of the association between erectile dysfunction and cardiovascular disease. Eur. Urol. 2014, 65, 968–978. [Google Scholar] [CrossRef] [PubMed]
- Baena-Díez, J.M.; Peñafiel, J.; Subirana, I.; Ramos, R.; Elosua, R.; Marín-Ibañez, A.; Guembe, M.J.; Rigo, F.; Tormo-Díaz, M.J.; Moreno-Iribas, C.; et al. Risk of Cause-Specific Death in Individuals With Diabetes: A Competing Risks Analysis. Diabetes Care 2016, 39, 1987–1995. [Google Scholar] [CrossRef]
- Corona, G.; Maseroli, E.; Rastrelli, G.; Francomano, D.; Aversa, A.; Hackett, G.I.; Ferri, S.; Sforza, A.; Maggi, M. Is late-onset hypogonadotropic hypogonadism a specific age-dependent disease, or merely an epiphenomenon caused by accumulating disease-burden? Minerva Endocrinol. 2016, 41, 196–210. [Google Scholar] [PubMed]
- Fried, M.; Yumuk, V.; Oppert, J.-M.; Scopinaro, N.; Torres, A.J.; Weiner, R.; Yashkov, Y.; Frühbeck, G. Interdisciplinary European Guidelines on Metabolic and Bariatric Surgery. Obes. Facts 2013, 6, 449–468. [Google Scholar] [CrossRef]
- Manicardi, V.; Adinolfi, V.; Aricò, N.; Botta, A.; Clemente, G.; Fava, D.; La Penna, G.; Lapice, E.; Miranda, C.; Nicolucci, A.; et al. Annali AMD 2020. 2020. Available online: https://aemmedi.it/nuovi-annali-amd-2020/ (accessed on 5 June 2021).
- Malavige, L.S.; Levy, J.C. Erectile dysfunction in diabetes mellitus. J. Sex. Med. 2009, 6, 1232–1247. [Google Scholar] [CrossRef] [PubMed]
- Nowotny, K.; Jung, T.; Höhn, A.; Weber, D.; Grune, T. Advanced glycation end products and oxidative stress in type 2 diabetes mellitus. Biomolecules 2015, 5, 194–222. [Google Scholar] [CrossRef] [PubMed]
- Afanas’Ev, I. Signaling of reactive oxygen and nitrogen species in diabetes mellitus. Oxid. Med. Cell. Longev. 2010, 3, 361–373. [Google Scholar] [CrossRef] [PubMed]
- D’Oria, R.; Schipani, R.; Leonardini, A.; Natalicchio, A.; Perrini, S.; Cignarelli, A.; Laviola, L.; Giorgino, F. The Role of Oxidative Stress in Cardiac Disease: From Physiological Response to Injury Factor. Oxid. Med. Cell. Longev. 2020, 2020, 5732956. [Google Scholar] [CrossRef] [PubMed]
- Incalza, M.A.; D’Oria, R.; Natalicchio, A.; Perrini, S.; Laviola, L.; Giorgino, F. Oxidative stress and reactive oxygen species in endothelial dysfunction associated with cardiovascular and metabolic diseases. Vasc. Pharm. 2018, 100, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Castela, Â.; Costa, C. Molecular mechanisms associated with diabetic endothelial-erectile dysfunction. Nat. Rev. Urol. 2016, 13, 266–274. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, F.S.; Giachini, F.R.C.; Carneiro, Z.N.; Lima, V.V.; Ergul, A.; Webb, R.C.; Tostes, R.C. Erectile dysfunction in young non-obese type II diabetic goto-kakizaki rats is associated with decreased eNOS phosphorylation at ser1177. J. Sex. Med. 2010, 7, 3620–3634. [Google Scholar] [CrossRef]
- Musicki, B.; Kramer, M.F.; Becker, R.E.; Burnett, A.L. Inactivation of phosphorylated endothelial nitric oxide synthase (Ser-1177) by O-GlcNAc in diabetes-associated erectile dysfunction. Proc. Natl. Acad. Sci. USA 2005, 102, 11870–11875. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Tian, W.; Uwais, Z.; Li, G.; Li, H.; Guan, R.; Gao, Z.; Xin, Z. AGE-Breaker ALT-711 Plus Insulin Could Restore Erectile Function in Streptozocin-Induced Type 1 Diabetic Rats. J. Sex. Med. 2014, 11, 1452–1462. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.; Ren, L.; Wei, Q.; Shao, H.; Chen, L.; Liu, N. Advanced glycation end-products decreases expression of endothelial nitric oxide synthase through oxidative stress in human coronary artery endothelial cells. Cardiovasc. Diabetol. 2017, 16, 52. [Google Scholar] [CrossRef] [PubMed]
- Yeh, C.H.; Sturgis, L.; Haidacher, J.; Zhang, X.N.; Sherwood, S.J.; Bjercke, R.J.; Juhasz, O.; Crow, M.T.; Tilton, R.G.; Denner, L. Requirement for p38 and p44/p42 mitogen-activated protein kinases in RAGE-mediated nuclear factor-κB transcriptional activation and cytokine secretion. Diabetes 2001, 50, 1495–1504. [Google Scholar] [CrossRef] [PubMed]
- Seftel, A.D.; Vaziri, N.D.; Zhemnin, N.I.; Razmjouei, K.; Fogarty, J.; Hampel, N.; Polak, J.; Wang, R.Z.; Ferguson, K.; Block, C.; et al. Advanced glycation end products in human penis: Elevation in diabetic tissue, site of deposition, and possible effect through iNos or eNos. Urology 1997, 50, 1016–1026. [Google Scholar] [CrossRef]
- Augustin, H.G.; Young Koh, G.; Thurston, G.; Alitalo, K. Control of vascular morphogenesis and homeostasis through the angiopoietin-Tie system. Nat. Rev. Mol. Cell Biol. 2009, 10, 165–177. [Google Scholar] [CrossRef] [PubMed]
- Fiedler, U.; Reiss, Y.; Scharpfenecker, M.; Grunow, V.; Koidl, S.; Thurston, G.; Gale, N.W.; Witzenrath, M.; Rosseau, S.; Suttorp, N.; et al. Angiopoietin-2 sensitizes endothelial cells to TNF-α and has a crucial role in the induction of inflammation. Nat. Med. 2006, 12, 235–239. [Google Scholar] [CrossRef] [PubMed]
- Singh, H.; Brindle, N.P.J.; Zammit, V.A. High glucose and elevated fatty acids suppress signaling by the endothelium protective ligand angiopoietin-1. Microvasc. Res. 2010, 79, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.X.; Stinnett, A. Disruption of Ang-1/Tie-2 signaling contributes to the impaired myocardial vascular maturation and angiogenesis in type II diabetic mice. Arter. Thromb. Vasc. Biol. 2008, 28, 1606–1613. [Google Scholar] [CrossRef]
- Puddu, A.; Sanguineti, R.; Maggi, D.; Nicolò, M.; Traverso, C.E.; Cordera, R.; Viviani, G.L. Advanced Glycation End-Products and Hyperglycemia Increase Angiopoietin-2 Production by Impairing Angiopoietin-1-Tie-2 System. J. Diabetes Res. 2019, 2019. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.W.; Lip, G.Y.H.; Blann, A.D. Plasma angiopoietin-1, angiopoietin-2, angiopoietin receptor Tie-2, and vascular endothelial growth factor levels in acute coronary syndromes. Circulation 2004, 110, 2355–2360. [Google Scholar] [CrossRef]
- Li, L.; Qian, L.; Yu, Z.Q. Serum angiopoietin-2 is associated with angiopathy in type 2 diabetes mellitus. J. Diabetes Complicat. 2015, 29, 568–571. [Google Scholar] [CrossRef] [PubMed]
- Deng, W.; Bivalacqua, T.J.; Champion, H.C.; Hellstrom, W.J.; Murthy, S.N.; Kadowitz, P.J. Superoxide dismutase—A target for gene therapeutic approach to reduce oxidative stress in erectile dysfunction. Methods Mol. Biol. 2010, 610, 213–227. [Google Scholar] [CrossRef]
- Angulo, J.; Peiró, C.; Cuevas, P.; Gabancho, S.; Fernández, A.; González-Corrochano, R.; La Fuente, J.M.; Baron, A.D.; Chen, K.S.; de Tejada, I.S. The novel antioxidant, AC3056 (2,6-di-t-butyl-4-((Dimethyl-4-Methoxyphenylsilyl)Methyloxy)Phenol), reverses erectile dysfunction in diabetic rats and improves NO-mediated responses in penile tissue from diabetic men. J. Sex. Med. 2009, 6, 373–387. [Google Scholar] [CrossRef] [PubMed]
- Goswami, S.K.; Gangadarappa, S.K.; Vishwanath, M.; Razdan, R.; Jamwal, R.; Bhadri, N.; Inamdar, M.N. Antioxidant Potential and Ability of Phloroglucinol to Decrease Formation of Advanced Glycation End Products Increase Efficacy of Sildenafil in Diabetes-Induced Sexual Dysfunction of Rats. Sex. Med. 2016, 4, e106–e114. [Google Scholar] [CrossRef] [PubMed]
- Castela, A.; Gomes, P.; Domingues, V.F.; Paíga, P.; Costa, R.; Vendeira, P.; Costa, C. Role of oxidative stress-induced systemic and cavernosal molecular alterations in the progression of diabetic erectile dysfunction. J. Diabetes 2015, 7, 393–401. [Google Scholar] [CrossRef] [PubMed]
- Costa, C.; Virag, R. The endothelial-erectile dysfunction connection: An essential update. J. Sex. Med. 2009, 6, 2390–2404. [Google Scholar] [CrossRef] [PubMed]
- Hui, J.; Chen, S.; Zhang, H.; Yang, C.; Wei, A.; He, S. Effects of “metabolic memory” on erectile function in diabetic men: A retrospective case-control study. Andrology 2021, 9, 288–296. [Google Scholar] [CrossRef] [PubMed]
- Camacho, E.M.; Huhtaniemi, I.T.; O’Neill, T.W.; Finn, J.D.; Pye, S.R.; Lee, D.M.; Tajar, A.; Bartfai, G.; Boonen, S.; Casanueva, F.F.; et al. Age-associated changes in hypothalamic-pituitary-testicular function in middle-aged and older men are modified by weight change and lifestyle factors: Longitudinal results from the European Male Ageing Study. Eur. J. Endocrinol. 2013, 168, 445–455. [Google Scholar] [CrossRef]
- O’Connor, D.B.; Lee, D.M.; Corona, G.; Forti, G.; Tajar, A.; O’Neill, T.W.; Pendleton, N.; Bartfai, G.; Boonen, S.; Casanueva, F.F.; et al. The relationships between sex hormones and sexual function in middle-aged and older European men. J. Clin. Endocrinol. Metab. 2011, 96, E1577–E1587. [Google Scholar] [CrossRef] [PubMed]
- Traish, A.M.; Goldstein, I.; Kim, N.N. Testosterone and Erectile Function: From Basic Research to a New Clinical Paradigm for Managing Men with Androgen Insufficiency and Erectile Dysfunction. Eur. Urol. 2007, 52, 54–70. [Google Scholar] [CrossRef] [PubMed]
- Torres-Estay, V.; Carreño, V.D.; Fuenzalida, P.; Watts, A.; San Francisco, I.F.; Montecinos, V.P.; Sotomayor, P.C.; Ebos, J.; Smith, G.J.; Godoy, A.S. Androgens modulate male-derived endothelial cell homeostasis using androgen receptor-dependent and receptor-independent mechanisms. Angiogenesis 2017, 20, 25–38. [Google Scholar] [CrossRef] [PubMed]
- Torres-Estay, V.; Carreño, V.D.; San Francisco, I.F.; Sotomayor, P.; Godoy, A.S.; Smith, G.J. Androgen receptor in human endothelial cells. J. Endocrinol. 2015, 224, R131–R137. [Google Scholar] [CrossRef] [PubMed]
- Schultheiss, D.; Badalyan, R.; Pilatz, A.; Gabouev, A.I.; Schlote, N.; Wefer, J.; Von Wasielewski, R.; Mertsching, H.; Sohn, M.; Stief, C.G.; et al. Androgen and estrogen receptors in the human corpus cavernosum penis: Immunohistochemical and cell culture results. World J. Urol. 2003, 21, 320–324. [Google Scholar] [CrossRef]
- Aversa, A.; Isidori, A.M.; De Martino, M.U.; Caprio, M.; Fabbrini, E.; Rocchietti-March, M.; Frajese, G.; Fabbri, A. Androgens and penile erection: Evidence for a direct relationship between free testosterone and cavernous vasodilation in men with erectile dysfunction. Clin. Endocrinol. 2000, 53, 517–522. [Google Scholar] [CrossRef]
- Yamamoto, H.; Sasaki, S.; Tatsura, H.; Umemoto, Y.; Kubota, H.; Kamiya, H.; Kawai, T.; Kang, K.; Kohri, K. Penile apoptosis in association with p53 under lack of testosterone. Urol. Res. 2004, 32, 9–13. [Google Scholar] [CrossRef]
- Skogastierna, C.; Hotzen, M.; Rane, A.; Ekström, L. A Supraphysiological dose of testosterone induces nitric oxide production and oxidative stress. Eur. J. Prev. Cardiol. 2014, 21, 1049–1054. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Malhotra, N.; Jacob, J.J. Targeting testosterone measurements to patients with type 2 diabetes mellitus and moderate to severe symptomatic erectile dysfunction. Diabetes Res. Clin. Pract. 2018, 137, 221–223. [Google Scholar] [CrossRef]
- Makhsida, N.; Shah, J.; Yan, G.; Fisch, H.; Shabsigh, R. Hypogonadism and metabolic syndrome: Implications for testosterone therapy. J. Urol. 2005, 174, 827–834. [Google Scholar] [CrossRef] [PubMed]
- Saad, F.; Caliber, M.; Doros, G.; Haider, K.S.; Haider, A. Long-term treatment with testosterone undecanoate injections in men with hypogonadism alleviates erectile dysfunction and reduces risk of major adverse cardiovascular events, prostate cancer, and mortality. Aging Male 2020, 23, 81–92. [Google Scholar] [CrossRef] [PubMed]
- Efesoy, O.; Çayan, S.; Akbay, E. The Effect of Testosterone Replacement Therapy on Penile Hemodynamics in Hypogonadal Men With Erectile Dysfunction, Having Veno-Occlusive Dysfunction. Am. J. Mens. Health 2018, 12, 634–638. [Google Scholar] [CrossRef]
- Zhang, X.H.; Filippi, S.; Morelli, A.; Vignozzi, L.; Luconi, M.; Donati, S.; Forti, G.; Maggi, M. Testosterone restores diabetes-induced erectile dysfunction and sildenafil responsiveness in two distinct animal models of chemical diabetes. J. Sex. Med. 2006, 3, 253–266. [Google Scholar] [CrossRef]
- Aversa, A.; Francomano, D.; Lenzi, A. Does testosterone supplementation increase PDE5-inhibitor responses in difficult-to-treat erectile dysfunction patients? Expert Opin. Pharm. 2015, 16, 625–628. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Morelli, A.; Filippi, S.; Mancina, R.; Luconi, M.; Vignozzi, L.; Marini, M.; Orlando, C.; Vannelli, G.B.; Aversa, A.; Natali, A.; et al. Androgens regulate phosphodiesterase type 5 expression and functional activity in corpora cavernosa. Endocrinology 2004, 145, 2253–2263. [Google Scholar] [CrossRef]
- Zhu, J.; Zhang, W.; Ou, N.; Song, Y.; Kang, J.; Liang, Z.; Hu, R.; Yang, Y.; Liu, X. Do testosterone supplements enhance response to phosphodiesterase 5 inhibitors in men with erectile dysfunction and hypogonadism: A systematic review and meta-analysis. Transl. Urol. 2020, 9, 591–600. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.S.; Xin, Z.; Namiki, M.; Albersen, M.; Muller, D.; Lue, T.F. Direct androgen regulation of PDE5 gene or the lack thereof. Int. J. Impot. Res. 2013, 25, 81–85. [Google Scholar] [CrossRef] [PubMed]
- den Broeck, T.; Soebadi, M.A.; Falter, A.; Raets, L.; Duponselle, J.; Lootsma, J.; Heintz, A.; Philtjens, U.; Hofkens, L.; Gonzalez-Viedma, A.; et al. Testosterone Induces Relaxation of Human Corpus Cavernosum Tissue of Patients With Erectile Dysfunction. Sex. Med. 2020, 8, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.D.; English, K.M.; Hugh Jones, T.; Channer, K.S. Testosterone-induced coronary vasodilatation occurs via a non-genomic mechanism: Evidence of a direct calcium antagonism action. Clin. Sci. 2004, 107, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.J.; Xu, T.Y.; Xia, L.L.; Zhong, S.; Zhang, X.H.; Zhu, Z.W.; Chen, D.R.; Liu, Y.; Fan, Y.; Xu, C.; et al. Castration impairs erectile organ structure and function by inhibiting autophagy and promoting apoptosis of corpus cavernosum smooth muscle cells in rats. Int. Urol. Nephrol. 2015, 47, 1105–1115. [Google Scholar] [CrossRef] [PubMed]
- Cui, K.; Li, R.; Chen, R.; Li, M.; Wang, T.; Yang, J.; Chen, Z.; Wang, S.; Liu, J.; Rao, K. Androgen deficiency impairs erectile function in rats through promotion of corporal fibrosis. Andrologia 2018, 50, e12797. [Google Scholar] [CrossRef] [PubMed]
- Arakaki, R.F.; Blevins, T.C.; Wise, J.K.; Liljenquist, D.R.; Jiang, H.H.; Jacobson, J.G.; Martin, S.A.; Jackson, J.A. Comparison of insulin lispro protamine suspension versus insulin glargine once daily added to oral antihyperglycaemic medications and exenatide in type 2 diabetes: A prospective randomized open-label trial. Diabetes Obes. Metab. 2014, 16, 510–518. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.Y.; Chai, J.S.; Lee, S.H.; Park, K.; Paick, J.S.; Kim, S.W. Investigation of the Effects of the Level of Glycemic Control on Erectile Function and Pathophysiological Mechanisms in Diabetic Rats. J. Sex. Med. 2012, 9, 1550–1558. [Google Scholar] [CrossRef] [PubMed]
- Wessells, H.; Penson, D.F.; Cleary, P.; Rutledge, B.N.; Lachin, J.M.; McVary, K.T.; Schade, D.S.; Sarma, A.V. Effect of intensive glycemic therapy on erectile function in men with type 1 diabetes. J. Urol. 2011, 185, 1828–1834. [Google Scholar] [CrossRef]
- Park, J.; Kwon, O.; Cho, S.Y.; Cho, M.C.; Paick, J.S.; Kim, S.W. Comparison of improving effects for diabetic erectile dysfunction according to the anti-glycemic agents: Phlorizin and insulin. World J. Men Health 2019, 37, 210–218. [Google Scholar] [CrossRef]
- Shirai, M.; Yamanaka, M.; Shiina, H.; Igawa, M.; Ogishima, T.; Fujime, M.; Ishii, N.; Okuyama, A.; Lue, T.F.; Dahiya, R. Androgen, estrogen, and progesterone receptor gene regulation during diabetic erectile dysfunction and insulin treatment. Urology 2004, 64, 1244–1249. [Google Scholar] [CrossRef]
- Yamanaka, M.; Shirai, M.; Shiina, H.; Tanaka, Y.; Tsujimura, A.; Matsumiya, K.; Okuyama, A.; Dahiya, R. Diabetes induced erectile dysfunction and apoptosis in penile crura are recovered by insulin treatment in rats. J. Urol. 2003, 170, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Kesavadev, J.; Sadasivan Pillai, P.B.; Shankar, A.; Warrier, R.S.; Ramachandran, L.; Jothydev, S.; Krishnan, G. Exploratory CSII Randomized Controlled Trial on Erectile Dysfunction in T2DM Patients (ECSIITED). J. Diabetes Sci. Technol. 2018, 12, 1252–1253. [Google Scholar] [CrossRef]
- Zhou, F.; Hui, Y.; Xu, Y.; Lei, H.; Yang, B.; Guan, R.; Gao, Z.; Xin, Z.; Hou, J. Effects of adipose-derived stem cells plus insulin on erectile function in streptozotocin-induced diabetic rats. Int. Urol. Nephrol. 2016, 48, 657–669. [Google Scholar] [CrossRef]
- Wang, L.; Xu, Y.; Li, H.; Lei, H.; Guan, R.; Gao, Z.; Xin, Z. Antioxidant icariside II combined with insulin restores erectile function in streptozotocin-induced type 1 diabetic rats. J. Cell. Mol. Med. 2015, 19, 960–969. [Google Scholar] [CrossRef]
- Goldner, M.G.; Knatterud, G.L.; Prout, T.E. Effects of hypoglycemic agents on vascular complications in patients with adult-onset diabetes. 3. Clinical implications of UGDP results. Clin. Trial 1971, 218, 1400–1410. [Google Scholar] [CrossRef]
- Koren, S.; Shemesh-Bar, L.; Tirosh, A.; Peleg, R.K.; Berman, S.; Hamad, R.A.; Vinker, S.; Golik, A.; Efrati, S. The effect of sitagliptin versus glibenclamide on arterial stiffness, blood pressure, lipids, and inflammation in type 2 diabetes mellitus patients. Diabetes Technol. 2012, 14, 561–567. [Google Scholar] [CrossRef] [PubMed]
- Burke, M.A.; Mutharasan, R.K.; Ardehali, H. The sulfonylurea receptor, an atypical ATP-binding cassette protein, and its regulation of the KATP channel. Circ. Res. 2008, 102, 164–176. [Google Scholar] [CrossRef]
- Lee, S.W.; Wang, H.Z.; Christ, G.J. Characterization of ATP-sensitive potassium channels in human corporal smooth muscle cells. Int. J. Impot. Res. 1999, 11, 179–188. [Google Scholar] [CrossRef]
- Insuk, S.O.; Chae, M.R.; Choi, J.W.; Yang, D.K.; Sim, J.H.; Lee, S.W. Molecular basis and characteristics of KATP channel in human corporal smooth muscle cells. Int. J. Impot. Res. 2003, 15, 258–266. [Google Scholar] [CrossRef][Green Version]
- de Miranda Cará, A.; Fregonesi, A.; Antunes, E.; De Nucci, G.; Rodrigues Netto, N. Role of adenosine triphosphate-dependent potassium channels in canine penile erection. Urology 2004, 64, 603–607. [Google Scholar] [CrossRef]
- Sehra, D.; Sehra, S.; Sehra, S.T. Sulfonylureas: Do we need to introspect safety again? Expert Opin. Drug Saf. 2011, 10, 851–861. [Google Scholar] [CrossRef]
- Ruiz Rubio, J.L.; Hernández, M.; Rivera de los Arcos, L.; Benedito, S.; Recio, P.; García, P.; García-Sacristán, A.; Prieto, D. Role of ATP-sensitive K+ channels in relaxation of penile resistance arteries. Urology 2004, 63, 800–805. [Google Scholar] [CrossRef]
- Hsieh, G.; Kolasa, T.; Sullivan, J.; Brioni, J. Dual mechanism of action of nicorandil on rabbit corpus cavernosal smooth muscle tone. Int. J. Impot. Res. 2001, 13, 240–246. [Google Scholar] [CrossRef]
- Al-Kuraishy, H.M.; Al-Gareeb, A.I. Erectile Dysfunction and Low Sex Drive in Men with Type 2 DM: The Potential Role of Diabetic Pharmacotherapy. J. Clin. Diagn. Res. 2016, 10, FC21–FC26. [Google Scholar] [CrossRef]
- Wong, L.; Chen, H.; Lai, S.; Yang, H.; Kuang, J.; Pei, J. Effects of sulfonylurea as initial treatment on testosterone of middle-aged men with type 2 diabetes: A 16-week, pilot study. J. Diabetes Investig. 2015, 6, 454–459. [Google Scholar] [CrossRef] [PubMed]
- Brock, G.; Heiselman, D.; Maggi, M.; Kim, S.W.; Rodríguez Vallejo, J.M.; Behre, H.M.; McGettigan, J.; Dowsett, S.A.; Hayes, R.P.; Knorr, J.; et al. Effect of Testosterone Solution 2% on Testosterone Concentration, Sex Drive and Energy in Hypogonadal Men: Results of a Placebo Controlled Study. J. Urol. 2016, 195, 699–705. [Google Scholar] [CrossRef] [PubMed]
- Chan, T.Y.K. Outbreaks of severe hypoglycaemia due to illegal sexual enhancement products containing undeclared glibenclamide. Pharm. Drug Saf. 2009, 18, 1250–1251. [Google Scholar] [CrossRef] [PubMed]
- Kao, S.L.; Chan, C.L.; Tan, B.; Lim, C.C.T.; Dalan, R.; Gardner, D.; Pratt, E.; Lee, M.; Lee, K.O. An Unusual Outbreak of Hypoglycemia. N. Engl. J. Med. 2009, 360, 734–736. [Google Scholar] [CrossRef]
- Yosefy, C.; Magen, E.; Kiselevich, A.; Priluk, R.; London, D.; Volchek, L.; Viskoper, R.J. Rosiglitazone improves, while glibenclamide worsens blood pressure control in treated hypertensive diabetic and dyslipidemic subjects via modulation of insulin resistance and sympathetic activity. J. Cardiovasc. Pharm. 2004, 44, 215–222. [Google Scholar] [CrossRef]
- Cosenso-Martin, L.N.; Giollo-Júnior, L.T.; Fernandes, L.A.B.; Cesarino, C.B.; Nakazone, M.A.; de Nassau Machado, M.; Yugar-Toledo, J.C.; Vilela-Martin, J.F. Effect of vildagliptin versus glibenclamide on endothelial function and arterial stiffness in patients with type 2 diabetes and hypertension: A randomized controlled trial. Acta Diabetol. 2018, 55, 1237–1245. [Google Scholar] [CrossRef]
- Ma, L.; Lu, N.; Wu, G. Antiplatelet aggregation and endothelial protection of I4, a new synthetic anti-diabetes sulfonylurea compound. Platelets 2015, 26, 342–348. [Google Scholar] [CrossRef]
- Cignarelli, A.; Genchi, V.A.; Caruso, I.; Natalicchio, A.; Perrini, S.; Laviola, L.; Giorgino, F. Diabetes and cancer: Pathophysiological fundamentals of a ‘dangerous affair’. Diabetes Res. Clin. Pract. 2018, 143, 378–388. [Google Scholar] [CrossRef] [PubMed]
- Harden, K.A.; Cowan, P.A.; Velasquez-Mieyer, P.; Patton, S.B. Effects of lifestyle intervention and metformin on weight management and markers of metabolic syndrome in obese adolescents. J. Am. Acad. Nurse Pract. 2007, 19, 368–377. [Google Scholar] [CrossRef]
- Wilding, J.; Godec, T.; Khunti, K.; Pocock, S.; Fox, R.; Smeeth, L.; Clauson, P.; Fenici, P.; Hammar, N.; Medina, J. Changes in HbA1c and weight, and treatment persistence, over the 18 months following initiation of second-line therapy in patients with type 2 diabetes: Results from the United Kingdom Clinical Practice Research Datalink. BMC Med. 2018, 16, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Schernthaner, G.; Matthews, D.R.; Charbonnel, B.; Hanefeld, M.; Brunetti, P. Efficacy and safety of pioglitazone versus metformin in patients with type 2 diabetes mellitus: A double-blind, randomized trial. J. Clin. Endocrinol. Metab. 2004, 89, 6068–6076. [Google Scholar] [CrossRef] [PubMed]
- Mahrouf, M.; Ouslimani, N.; Peynet, J.; Djelidi, R.; Couturier, M.; Therond, P.; Legrand, A.; Beaudeux, J.L. Metformin reduces angiotensin-mediated intracellular production of reactive oxygen species in endothelial cells through the inhibition of protein kinase C. Biochem. Pharm. 2006, 72, 176–183. [Google Scholar] [CrossRef]
- Hattori, Y.; Suzuki, K.; Hattori, S.; Kasai, K. Metformin inhibits cytokine-induced nuclear factor κB activation via AMP-activated protein kinase activation in vascular endothelial cells. Hypertension 2006, 47, 1183–1188. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.W.; Park, S.Y.; Kim, J.Y.; Huh, J.Y.; Jeon, W.S.; Yoon, C.J.; Yun, S.S.; Moon, K.H. Metformin restores the penile expression of nitric oxide synthase in high-fat-fed obese rats. J. Androl. 2007, 28, 555–560. [Google Scholar] [CrossRef] [PubMed]
- Silva, F.H.; Alexandre, E.C.; Calmasini, F.B.; Calixto, M.C.; Antunes, E. Treatment with Metformin Improves Erectile Dysfunction in a Murine Model of Obesity Associated with Insulin Resistance. Urology 2015, 86, 423.e1–423.e6. [Google Scholar] [CrossRef]
- Zhang, C.; Luo, D.; Li, T.; Yang, Q.; Xie, Y.; Chen, H.; Lv, L.; Yao, J.; Deng, C.; Liang, X.; et al. Transplantation of Human Urine-Derived Stem Cells Ameliorates Erectile Function and Cavernosal Endothelial Function by Promoting Autophagy of Corpus Cavernosal Endothelial Cells in Diabetic Erectile Dysfunction Rats. Stem Cells Int. 2019, 2019. [Google Scholar] [CrossRef]
- Musicki, B.; Burnett, A.L. Constitutive NOS uncoupling and NADPH oxidase upregulation in the penis of type 2 diabetic men with erectile dysfunction. Andrology 2017, 5, 294–298. [Google Scholar] [CrossRef]
- Labazi, H.; Wynne, B.M.; Tostes, R.; Webb, R.C. Metformin treatment improves erectile function in an angiotensin II model of erectile dysfunction. J. Sex. Med. 2013, 10, 2154–2164. [Google Scholar] [CrossRef]
- Park, J.K.; Kim, S.Z.; Kim, J.U.; Kim, Y.G.; Kim, S.M.; Cho, K.W. Comparison of effects of angiotensin peptides in the regulation of clitoral cavernosum smooth muscle tone. Int. J. Impot. Res. 2002, 14, 72–80. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kilarkaje, N.; Yousif, M.H.M.; El-Hashim, A.Z.; Makki, B.; Akhtar, S.; Benter, I.F. Role of angiotensin II and angiotensin-(1-7) in diabetes-induced oxidative DNA damage in the corpus cavernosum. Fertil. Steril. 2013, 100, 226–233. [Google Scholar] [CrossRef]
- Zhang, J.; Li, S.; Zhang, S.; Wang, Y.; Jin, S.; Zhao, C.; Yang, W.; Liu, Y.; Kong, G. Effect of Icariside II and Metformin on Penile Erectile Function, Histological Structure, Mitochondrial Autophagy, Glucose-Lipid Metabolism, Angiotensin II and Sex Hormone in Type 2 Diabetic Rats With Erectile Dysfunction. Sex. Med. 2020, 8, 168–177. [Google Scholar] [CrossRef] [PubMed]
- Lanaspa, M.A.; Cicerchi, C.; Garcia, G.; Li, N.; Roncal-Jimenez, C.A.; Rivard, C.J.; Hunter, B.; Andrés-Hernando, A.; Ishimoto, T.; Sánchez-Lozada, L.G.; et al. Counteracting Roles of AMP Deaminase and AMP Kinase in the Development of Fatty Liver. PLoS ONE 2012, 7, e48801. [Google Scholar] [CrossRef]
- Ouyang, J.; Parakhia, R.A.; Ochs, R.S. Metformin activates AMP kinase through inhibition of AMP deaminase. J. Biol. Chem. 2011, 286, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Phatarpekar, V.P.; Wen, J.; Xia, Y. Role of adenosine signaling in penile erection and erectile disorders. J. Sex. Med. 2010, 7, 3553–3564. [Google Scholar] [CrossRef] [PubMed]
- Vignozzi, L.; Filippi, S.; Comeglio, P.; Cellai, I.; Morelli, A.; Rastrelli, G.; Maneschi, E.; Mannucci, E.; Maggi, M. Metformin in vitro and in vivo increases adenosine signaling in rabbit corpora cavernosa. J. Sex. Med. 2014, 11, 1694–1708. [Google Scholar] [CrossRef] [PubMed]
- Rey-Valzacchi, G.N.J.; Costanzo, P.R.; Finger, L.A.; Layus, A.O.; Gueglio, G.M.; Litwak, L.N.E.; Knoblovits, P. Addition of metformin to sildenafil treatment for erectile dysfunction in eugonadal nondiabetic men with insulin resistance. A prospective, randomized, double-blind pilot study. J. Androl. 2012, 33, 608–614. [Google Scholar] [CrossRef]
- Yan, W.J.; Mu, Y.; Yu, N.; Yi, T.L.; Zhang, Y.; Pang, X.L.; Cheng, D.; Yang, J. Protective effects of metformin on reproductive function in obese male rats induced by high-fat diet. J. Assist. Reprod. Genet. 2015, 32, 1097–1104. [Google Scholar] [CrossRef] [PubMed]
- Morgante, G.; Tosti, C.; Orvieto, R.; Musacchio, M.C.; Piomboni, P.; De Leo, V. Metformin improves semen characteristics of oligo-terato-asthenozoospermic men with metabolic syndrome. Fertil. Steril. 2011, 95, 2150–2152. [Google Scholar] [CrossRef]
- Furman, B.L. Acarbose. In xPharm: The Comprehensive Pharmacology Reference; Elsevier Inc.: Amsterdam, The Netherlands, 2007; pp. 1–3. ISBN 9780080552323. [Google Scholar]
- Sun, W.; Zeng, C.; Liao, L.; Chen, J.; Wang, Y. Comparison of acarbose and metformin therapy in newly diagnosed type 2 diabetic patients with overweight and/or obesity. Curr. Med. Res. Opin. 2016, 32, 1389–1396. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-S.; Lin, S.-D.; Lee, W.-J.; Su, S.-L.; Lee, I.-T.; Tu, S.-T.; Tseng, Y.-H.; Lin, S.-Y.; Sheu, W.H.-H. Effects of Acarbose Versus Glibenclamide on Glycemic Excursion and Oxidative Stress in Type 2 Diabetic Patients Inadequately Controlled by Metformin: A 24-Week, Randomized, Open-Label, Parallel-Group Comparison. Clin. Ther. 2011, 33, 1932–1942. [Google Scholar] [CrossRef]
- Chiasson, J.-L.; Josse, R.G.; Gomis, R.; Hanefeld, M.; Karasik, A.; Laakso, M. Acarbose for prevention of type 2 diabetes mellitus: The STOP-NIDDM randomised trial. Lancet 2002, 359, 2072–2077. [Google Scholar] [CrossRef]
- Chiasson, J.-L.; Josse, R.G.; Gomis, R.; Hanefeld, M.; Karasik, A.; Laakso, M.; STOP-NIDDM Trial Research Group. Acarbose treatment and the risk of cardiovascular disease and hypertension in patients with impaired glucose tolerance: The STOP-NIDDM trial. JAMA 2003, 290, 486–494. [Google Scholar] [CrossRef] [PubMed]
- Kato, T.; Inoue, T.; Node, K. Postprandial endothelial dysfunction in subjects with new-onset type 2 diabetes: An acarbose and nateglinide comparative study. Cardiovasc. Diabetol. 2010, 9. [Google Scholar] [CrossRef]
- Oboh, G.; Ogunbadejo, M.D.; Ogunsuyi, O.B.; Oyeleye, S.I. Can gallic acid potentiate the antihyperglycemic effect of acarbose and metformin? Evidence from streptozotocin-induced diabetic rat model. Arch. Physiol. Biochem. 2020, 1–9. [Google Scholar] [CrossRef]
- Oyeleye, S.I.; Ojo, O.R.; Oboh, G. Moringa oleifera leaf and seed inclusive diets influenced the restoration of biochemicals associated with erectile dysfunction in the penile tissue of STZ-induced diabetic male rats treated with/without Acarbose drug. J. Food Biochem. 2020, 45. [Google Scholar] [CrossRef] [PubMed]
- Derosa, G.; Mereu, R.; D’Angelo, A.; Salvadeo, S.A.; Ferrari, I.; Fogari, E.; Gravina, A.; Palumbo, I.; Maffioli, P.; Randazzo, S.; et al. Effect of pioglitazone and acarbose on endothelial inflammation biomarkers during oral glucose tolerance test in diabetic patients treated with sulphonylureas and metformin. J. Clin. Pharm. 2010, 35, 565–579. [Google Scholar] [CrossRef]
- Li, X.X.; Ling, S.K.; Hu, M.Y.; Ma, Y.; Li, Y.; Huang, P.L. Protective effects of acarbose against vascular endothelial dysfunction through inhibiting Nox4/NLRP3 inflammasome pathway in diabetic rats. Free Radic. Biol. Med. 2019, 145, 175–186. [Google Scholar] [CrossRef] [PubMed]
- Bi, Y.; Zhang, B.; Xu, W.; Yang, H.; Feng, W.; Li, C.; Tong, G.; Li, M.; Wang, X.; Shen, S.; et al. Effects of exenatide, insulin, and pioglitazone on liver fat content and body fat distributions in drug-naive subjects with type 2 diabetes. Acta Diabetol. 2014, 51, 865–873. [Google Scholar] [CrossRef]
- Kaku, K.; Itayasu, T.; Hiroi, S.; Hirayama, M.; Seino, Y. Efficacy and safety of alogliptin added to pioglitazone in Japanese patients with type 2 diabetes: A randomized, double-blind, placebo-controlled trial with an open-label long-term extension study. Diabetes Obes. Metab. 2011, 13, 1028–1035. [Google Scholar] [CrossRef]
- Dormandy, J.A.; Charbonnel, B.; Eckland, D.J.; Erdmann, E.; Massi-Benedetti, M.; Moules, I.K.; Skene, A.M.; Tan, M.H.; Lefèbvre, P.J.; Murray, G.D.; et al. Secondary prevention of macrovascular events in patients with type 2 diabetes in the PROactive Study (PROspective pioglitAzone Clinical Trial In macroVascular Events): A randomised controlled trial. Lancet 2005, 366, 1279–1289. [Google Scholar] [CrossRef]
- Erdmann, E.; Dormandy, J.A.; Charbonnel, B.; Massi-Benedetti, M.; Moules, I.K.; Skene, A.M. The Effect of Pioglitazone on Recurrent Myocardial Infarction in 2,445 Patients With Type 2 Diabetes and Previous Myocardial Infarction. J. Am. Coll. Cardiol. 2007, 49, 1772–1780. [Google Scholar] [CrossRef]
- Wilcox, R.; Bousser, M.-G.; Betteridge, D.J.; Schernthaner, G.; Pirags, V.; Kupfer, S.; Dormandy, J. Effects of Pioglitazone in Patients With Type 2 Diabetes With or Without Previous Stroke. Stroke 2007, 38, 865–873. [Google Scholar] [CrossRef]
- Van Can, J.; Sloth, B.; Jensen, C.B.; Flint, A.; Blaak, E.E.; Saris, W.H.M. Effects of the once-daily GLP-1 analog liraglutide on gastric emptying, glycemic parameters, appetite and energy metabolism in obese, non-diabetic adults. Int. J. Obes. 2014, 38, 784–793. [Google Scholar] [CrossRef] [PubMed]
- Basolo, A.; Burkholder, J.; Osgood, K.; Graham, A.; Bundrick, S.; Frankl, J.; Piaggi, P.; Thearle, M.S.; Krakoff, J. Exenatide has a pronounced effect on energy intake but not energy expenditure in non-diabetic subjects with obesity: A randomized, double-blind, placebo-controlled trial. Metabolism 2018, 85, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Kovanecz, I.; Ferrini, M.G.; Vernet, D.; Nolazco, G.; Rajfer, J.; Gonzalez-Cadavid, N.F. Pioglitazone prevents corporal veno-occlusive dysfunction in a rat model of type 2 diabetes mellitus. BJU Int. 2006, 98, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Kovanecz, I.; Ferrini, M.G.; Vernet, D.; Nolazco, G.; Rajfer, J.; Gonzalez-Cadavid, N.F. Ageing-related corpora veno-occlusive dysfunction in the rat is ameliorated by pioglitazone. BJU Int. 2007, 100, 867–874. [Google Scholar] [CrossRef] [PubMed]
- Aliperti, L.A.; Lasker, G.F.; Hagan, S.S.; Hellstrom, J.A.; Gokce, A.; Trost, L.W.; Kadowitz, P.J.; Sikka, S.C.; Hellstrom, W.J.G. Efficacy of pioglitazone on erectile function recovery in a rat model of cavernous nerve injury. Urology 2014, 84, 1122–1127. [Google Scholar] [CrossRef]
- Heppner, T.J.; Bonev, A.D.; Eckman, D.M.; Gomez, M.F.; Petkov, G.V.; Nelson, M.T. Novel PPARγ agonists GI 262570, GW 7845, GW 1929, and pioglitazone decrease calcium channel function and myogenic tone in rat mesenteric arteries. Pharmacology 2005, 73, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Goya, K.; Sumitani, S.; Otsuki, M.; Xu, X.; Yamamoto, H.; Kurebayashi, S.; Saito, H.; Kouhara, H.; Kasayama, S. The thiazolidinedione drug troglitazone up-regulates nitric oxide synthase expression in vascular endothelial cells. J. Diabetes Complicat. 2006, 20, 336–342. [Google Scholar] [CrossRef]
- Katz, E.G.; Moustafa, A.A.; Heidenberg, D.; Haney, N.; Peak, T.; Lasker, G.F.; Knoedler, M.; Rittenberg, D.; Rezk, B.M.; Abd Elmageed, Z.Y.; et al. Pioglitazone Enhances Survival and Regeneration of Pelvic Ganglion Neurons after Cavernosal Nerve Injury. Urology 2016, 89, 76–82. [Google Scholar] [CrossRef]
- Heidenberg, D.J.; Haney, N.M.; Rezk, B.M.; Talwar, S.; Okpechi, S.C.; Srivastav, S.K.; Honda, M.; Song, B.; Swan, K.; Awadallah, S.; et al. Pioglitazone’s beneficial effects on erectile function preservation after cavernosal nerve injury in the rat are negated by inhibition of the insulin-like growth factor-1 receptor: A preclinical study. Int. J. Impot. Res. 2019, 31. [Google Scholar] [CrossRef] [PubMed]
- Gholamine, B.; Motevallian, M.; Shafiei, M.; Mahmoudian, M. Effects of pioglitazone on erectile dysfunction in sildenafil poor-responders: A randomized, controlled study. J. Pharm. Pharm. Sci. 2008, 11, 22. [Google Scholar] [CrossRef] [PubMed]
- Peuler, J.D.; Phelps, L.E. Sildenafil does not enhance but rather attenuates vasorelaxant effects of antidiabetic agents. J. Smooth Muscle Res. 2015, 51, 22–36. [Google Scholar] [CrossRef] [PubMed]
- Deng, H.; Lin, S.; Yang, X.; Lv, J.; Luo, S.; Zeng, L.; Weng, J.; Xu, W. Effect of baseline body mass index on glycemic control and weight change with exenatide monotherapy in Chinese drug-naïve type 2 diabetic patients. J. Diabetes 2019, 11, 509–518. [Google Scholar] [CrossRef]
- Nauck, M.A.; Heimesaat, M.M.; Behle, K.; Holst, J.J.; Nauck, M.S.; Ritzel, R.; Hüfner, M.; Schmiegel, W.H. Effects of glucagon-like peptide 1 on counterregulatory hormone responses, cognitive functions, and insulin secretion during hyperinsulinemic, stepped hypoglycemic clamp experiments in healthy volunteers. J. Clin. Endocrinol. Metab. 2002, 87, 1239–1246. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V. Pleiotropic effects of incretins. Indian J. Endocrinol. Metab. 2012, 16, 47. [Google Scholar] [CrossRef]
- Sinha, B.; Ghosal, S. Meta-analyses of the effects of DPP-4 inhibitors, SGLT2 inhibitors and GLP1 receptor analogues on cardiovascular death, myocardial infarction, stroke and hospitalization for heart failure. Diabetes Res. Clin. Pract. 2019, 150, 8–16. [Google Scholar] [CrossRef]
- Nauck, M.A.; Meier, J.J.; Cavender, M.A.; El Aziz, M.A.; Drucker, D.J. Cardiovascular actions and clinical outcomes with glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors. Circulation 2017, 136, 849–870. [Google Scholar] [CrossRef]
- Scheen, A.J. Cardiovascular effects of new oral glucose-lowering agents DPP-4 and SGLT-2 inhibitors. Circ. Res. 2018, 122, 1439–1459. [Google Scholar] [CrossRef]
- Muscogiuri, G.; Cignarelli, A.; Giorgino, F.; Prodram, F.; Santi, D.; Tirabassi, G.; Balercia, G.; Modica, R.; Faggiano, A.; Colao, A. GLP-1: Benefits beyond pancreas. J. Endocrinol. Investig. 2014, 37, 1143–1153. [Google Scholar] [CrossRef]
- Blackman, A.; Foster, G.D.; Zammit, G.; Rosenberg, R.; Aronne, L.; Wadden, T.; Claudius, B.; Jensen, C.B.; Mignot, E. Effect of liraglutide 3.0 mg in individuals with obesity and moderate or severe obstructive sleep apnea: The scale sleep apnea randomized clinical trial. Int. J. Obes. 2016, 40, 1310–1319. [Google Scholar] [CrossRef]
- Wadden, T.A.; Hollander, P.; Klein, S.; Niswender, K.; Woo, V.; Hale, P.M.; Aronne, L. Weight maintenance and additional weight loss with liraglutide after low-calorie-diet-induced weight loss: The SCALE Maintenance randomized study. Int. J. Obes. 2013, 37, 1443–1451. [Google Scholar] [CrossRef] [PubMed]
- Davies, M.J.; Bergenstal, R.; Bode, B.; Kushner, R.F.; Lewin, A.; Skjøth, T.V.; Andreasen, A.H.; Jensen, C.B.; DeFronzo, R.A.; Valensi, P.; et al. Efficacy of liraglutide for weight loss among patients with type 2 diabetes: The SCALE diabetes randomized clinical trial. JAMA J. Am. Med. Assoc. 2015, 314, 687–699. [Google Scholar] [CrossRef] [PubMed]
- Kanoski, S.E.; Hayes, M.R.; Skibicka, K.P. GLP-1 and weight loss: Unraveling the diverse neural circuitry. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2016, 310, R885–R895. [Google Scholar] [CrossRef] [PubMed]
- Inoue, T.; Inoguchi, T.; Sonoda, N.; Hendarto, H.; Makimura, H.; Sasaki, S.; Yokomizo, H.; Fujimura, Y.; Miura, D.; Takayanagi, R. GLP-1 analog liraglutide protects against cardiac steatosis, oxidative stress and apoptosis in streptozotocin-induced diabetic rats. Atherosclerosis 2015, 240, 250–259. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.T.; Tang, H.Q.; Su, H.; Wang, Y.; Zhou, Q.; Zhang, Q.; Wang, Y.; Zhu, H.Q. Glucagon-like peptide-1 attenuates endothelial barrier injury in diabetes via cAMP/PKA mediated down-regulation of MLC phosphorylation. Biomed. Pharm. 2019, 113. [Google Scholar] [CrossRef]
- Ishibashi, Y.; Matsui, T.; Takeuchi, M.; Yamagishi, S.I. Glucagon-like peptide-1 (GLP-1) inhibits advanced glycation end product (AGE)-induced up-regulation of VCAM-1 mRNA levels in endothelial cells by suppressing AGE receptor (RAGE) expression. Biochem. Biophys. Res. Commun. 2010, 391, 1405–1408. [Google Scholar] [CrossRef]
- Nyström, T.; Gutniak, M.K.; Zhang, Q.; Zhang, F.; Holst, J.J.; Ahrén, B.; Sjöholm, Å. Effects of glucagon-like peptide-1 on endothelial function in type 2 diabetes patients with stable coronary artery disease. Am. J. Physiol. Endocrinol. Metab. 2004, 287. [Google Scholar] [CrossRef]
- Erdogdu, Ö.; Nathanson, D.; Sjöholm, Å.; Nyström, T.; Zhang, Q. Exendin-4 stimulates proliferation of human coronary artery endothelial cells through eNOS-, PKA- and PI3K/Akt-dependent pathways and requires GLP-1 receptor. Mol. Cell. Endocrinol. 2010, 325, 26–35. [Google Scholar] [CrossRef]
- Gaspari, T.; Liu, B.H.; Welungoda, I.; Hu, Y.; Widdop, R.E.; Knudsen, L.B.; Simpson, R.W.; Dear, A.E. A GLP-1 receptor agonist liraglutide inhibits endothelial cell dysfunction and vascular adhesion molecule expression in an ApoE-/- mouse model. Diabetes Vasc. Dis. Res. 2011, 8, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.K.; Luo, J.Y.; Lau, C.W.; Cho, W.C.; Ng, S.C.F.; Ma, R.C.W.; Tian, X.Y.; Huang, Y. A GLP-1 analog lowers ER stress and enhances protein folding to ameliorate homocysteine-induced endothelial dysfunction. Acta Pharm. Sin. 2021, 1–12. [Google Scholar] [CrossRef]
- Koska, J.; Sands, M.; Burciu, C.; D’Souza, K.M.; Raravikar, K.; Liu, J.; Truran, S.; Franco, D.A.; Schwartz, E.A.; Schwenke, D.C.; et al. Exenatide protects against glucoseand lipid-induced endothelial dysfunction: Evidence for direct vasodilation effect of glp-1 receptor agonists in humans. Diabetes 2015, 64, 2624–2635. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.; She, M.; Xu, M.; Chen, H.; Li, J.; Chen, X.; Zheng, D.; Liu, J.; Chen, S.; Zhu, J.; et al. GLP-1 treatment protects endothelial cells from oxidative stress-induced autophagy and endothelial dysfunction. Int. J. Biol. Sci. 2018, 14, 1696–1708. [Google Scholar] [CrossRef]
- Bjørnholm, K.D.; Skovsted, G.F.; Mitgaard-Thomsen, A.; Rakipovski, G.; Tveden-Nyborg, P.; Lykkesfeldt, J.; Povlsen, G.K. Liraglutide treatment improves endothelial function in the Ldlr-/- mouse model of atherosclerosis and affects genes involved in vascular remodelling and inflammation. Basic Clin. Pharm. Toxicol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Han, F.; Hou, N.; Liu, Y.; Huang, N.; Pan, R.; Zhang, X.; Mao, E.; Sun, X. Liraglutide improves vascular dysfunction by regulating a cAMP-independent PKA-AMPK pathway in perivascular adipose tissue in obese mice. Biomed. Pharm. 2019, 120, 109537. [Google Scholar] [CrossRef] [PubMed]
- Altabas, V.; Altabas, K. DPP-4 inhibition improves a sexual condition? Med. Hypotheses 2015, 85, 124–126. [Google Scholar] [CrossRef] [PubMed]
- Yuan, P.; Ma, D.; Gao, X.; Wang, J.; Li, R.; Liu, Z.; Wang, T.; Wang, S.; Liu, J.; Liu, X. Liraglutide Ameliorates Erectile Dysfunction via Regulating Oxidative Stress, the RhoA/ROCK Pathway and Autophagy in Diabetes Mellitus. Front. Pharm. 2020, 11. [Google Scholar] [CrossRef]
- Maclusky, N.J.; Cook, S.; Scrocchi, L.; Shin, J.; Kim, J.; Vaccarino, F.; Asa, S.L.; Drucker, D.J. Neuroendocrine function and response to stress in mice with complete disruption of glucagon-like peptide-1 receptor signaling. Endocrinology 2000, 141, 752–762. [Google Scholar] [CrossRef]
- Zhang, E.; Xu, F.; Liang, H.; Yan, J.; Xu, H.; Li, Z.; Wen, X.; Weng, J. GLP-1 Receptor Agonist Exenatide Attenuates the Detrimental Effects of Obesity on Inflammatory Profile in Testis and Sperm Quality in Mice. Am. J. Reprod. Immunol. 2015, 74, 457–466. [Google Scholar] [CrossRef]
- Giagulli, V.A.; Carbone, M.D.; Ramunni, M.I.; Licchelli, B.; De Pergola, G.; Sabbà, C.; Guastamacchia, E.; Triggiani, V. Adding liraglutide to lifestyle changes, metformin and testosterone therapy boosts erectile function in diabetic obese men with overt hypogonadism. Andrology 2015, 3, 1094–1103. [Google Scholar] [CrossRef] [PubMed]
- Shao, N.; Yu, X.Y.; Yu, Y.M.; Li, B.W.; Pan, J.; Wu, W.H.; Zhang, H.J.; Ma, X.F.; Hao, M.; Kuang, H.Y. Short-term combined treatment with exenatide and metformin is superior to glimepiride combined metformin in improvement of serum testosterone levels in type 2 diabetic patients with obesity. Andrologia 2018, 50. [Google Scholar] [CrossRef] [PubMed]
- Fontoura, P.; Cardoso, M.C.D.A.; Erthal-Martins, M.C.; Werneck, C.; Sartorio, C.; Ramos, C.F. The effects of liraglutide on male fertility: A case report. Reprod. Biomed. Online 2014, 29, 644–646. [Google Scholar] [CrossRef]
- Sopko, N.A.; Hannan, J.L.; Bivalacqua, T.J. Understanding and targeting the Rho kinase pathway in erectile dysfunction. Nat. Rev. Urol. 2014, 11, 622–628. [Google Scholar] [CrossRef] [PubMed]
- Dalaklioglu, S.; Tasatargil, A.; Kuscu, N.; Celik, S.; Celik-Ozenci, C.; Ozdem, S.; Barutcigil, A.; Kucukcetin, I. Protective effect of exendin-4 treatment on erectile dysfunction induced by chronic methylglyoxal administration in rats. Peptides 2018, 106, 1–8. [Google Scholar] [CrossRef]
- Takeda, H.; Sasai, N.; Ito, S.; Obana, M.; Takuma, T.; Takai, M.; Kaneshige, H.; Machimura, H.; Kanamori, A.; Nakajima, K.; et al. Efficacy and Safety of Alogliptin in Patients With Type 2 Diabetes: Analysis of the ATTAK-J Study. J. Clin. Med. Res. 2016, 8, 130–140. [Google Scholar] [CrossRef] [PubMed]
- Mechanism of Action of Inhibitors of Dipeptidyl-Peptidase-4 (DPP-4) | Elsevier Enhanced Reader. Available online: https://reader.elsevier.com/reader/sd/pii/S1521690X09000232?token=CD1B2CAC3076F481A8BBC18456AF1EF7A483BCC4BD49BB908C4B2A0F620E9D0218ADAEFBC0CDE9B573BAEAE798A9DB99&originRegion=eu-west-1&originCreation=20210414141938 (accessed on 14 April 2021).
- Huang, C.-Y.; Shih, C.-M.; Tsao, N.-W.; Lin, Y.-W.; Huang, P.-H.; Wu, S.-C.; Lee, A.-W.; Kao, Y.-T.; Chang, N.-C.; Nakagami, H.; et al. Dipeptidyl peptidase-4 inhibitor improves neovascularization by increasing circulating endothelial progenitor cells. Br. J. Pharm. 2012, 167, 1506–1519. [Google Scholar] [CrossRef]
- Fadini, G.P.; Avogaro, A. Dipeptidyl peptidase-4 inhibition and vascular repair by mobilization of endogenous stem cells in diabetes and beyond. Atherosclerosis 2013, 229, 23–29. [Google Scholar] [CrossRef]
- Amadesi, S.; Reni, C.; Katare, R.; Meloni, M.; Oikawa, A.; Beltrami, A.P.; Avolio, E.; Cesselli, D.; Fortunato, O.; Spinetti, G.; et al. Role for substance P-based nociceptive signaling in progenitor cell activation and angiogenesis during ischemia in mice and in human subjects. Circulation 2012, 125, 1774–1786. [Google Scholar] [CrossRef] [PubMed]
- Castellani, M.L.; Galzio, R.J.; Felaco, P.; Tripodi, D.; Toniato, E.; De Lutiis, M.A.; Conti, F.; Fulcheri, M.; Conti, C.; Theoharides, T.C.; et al. VEGF, substance P and stress, new aspects: A revisited study. J. Biol. Regul. Homeost. Agents 2010, 24, 229–237. [Google Scholar] [PubMed]
- Harmar, A.J.; Fahrenkrug, J.; Gozes, I.; Laburthe, M.; May, V.; Pisegna, J.R.; Vaudry, D.; Vaudry, H.; Waschek, J.A.; Said, S.I. Pharmacology and functions of receptors for vasoactive intestinal peptide and pituitary adenylate cyclase-activating polypeptide: IUPHAR Review 1. Br. J. Pharm. 2012, 166, 4–17. [Google Scholar] [CrossRef]
- Traish, A.M.; Galoosian, A. Androgens Modulate Endothelial Function and Endothelial Progenitor Cells in Erectile Physiology. Korean J. Urol. 2013, 54, 721. [Google Scholar] [CrossRef] [PubMed]
- Aini, K.; Fukuda, D.; Tanaka, K.; Higashikuni, Y.; Hirata, Y.; Yagi, S.; Kusunose, K.; Yamada, H.; Soeki, T.; Sata, M. Vildagliptin, a DPP-4 Inhibitor, Attenuates Endothelial Dysfunction and Atherogenesis in Nondiabetic Apolipoprotein E-Deficient Mice. Int. Heart J. 2019, 60, 1421–1429. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Bai, Z.; Wu, H.; Wang, W. The DPP-4 inhibitor saxagliptin ameliorates ox-LDL-induced endothelial dysfunction by regulating AP-1 and NF-κB. Eur. J. Pharm. 2019, 851, 186–193. [Google Scholar] [CrossRef]
- Hibi, H.; Ohori, T.; Yamada, Y. DPP-IV inhibitor may affect spermatogenesis. Diabetes Res. Clin. Pract. 2011, 93. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.; McMurray, J.J.V. SGLT2 inhibitors and mechanisms of cardiovascular benefit: A state-of-the-art review. Diabetologia 2018, 61, 2108–2117. [Google Scholar] [CrossRef]
- Lee, M.M.Y.; Petrie, M.C.; McMurray, J.J.V.; Sattar, N. How Do SGLT2 (Sodium-Glucose Cotransporter 2) inhibitors and GLP-1 (glucagon-like peptide-1) receptor agonists reduce cardiovascular outcomes?: Completed and ongoing mechanistic trials. Arter. Thromb. Vasc. Biol. 2020, 40, 506–522. [Google Scholar] [CrossRef]
- Morieri, M.L.; Consoli, A.; Sesti, G.; Purrello, F.; Avogaro, A.; Fadini, G.P. Comparative effectiveness of dapagliflozin vs DPP-4 inhibitors on a composite endpoint of HbA1c, body weight and blood pressure reduction in the real world. Diabetes. Metab. Res. Rev. 2021, 37. [Google Scholar] [CrossRef]
- Cowie, M.R.; Fisher, M. SGLT2 inhibitors: Mechanisms of cardiovascular benefit beyond glycaemic control. Nat. Rev. Cardiol. 2020, 17, 761–772. [Google Scholar] [CrossRef]
- Jayawardene, D.; Ward, G.M.; O’Neal, D.N.; Theverkalam, G.; MacIsaac, A.I.; MacIsaac, R.J. New treatments for type 2 diabetes: Cardiovascular protection beyond glucose lowering? Hear. Lung Circ. 2014, 23, 997–1008. [Google Scholar] [CrossRef] [PubMed]
- Kluger, A.Y.; Tecson, K.M.; Lee, A.Y.; Lerma, E.V.; Rangaswami, J.; Lepor, N.E.; Cobble, M.E.; McCullough, P.A. Class effects of SGLT2 inhibitors on cardiorenal outcomes. Cardiovasc. Diabetol. 2019, 18, 99. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Farooq, M.A.; Gaertner, S.; Bruckert, C.; Qureshi, A.W.; Lee, H.H.; Benrahla, D.; Pollet, B.; Stephan, D.; Ohlmann, P.; et al. Empagliflozin improved systolic blood pressure, endothelial dysfunction and heart remodeling in the metabolic syndrome ZSF1 rat. Cardiovasc. Diabetol. 2020, 19. [Google Scholar] [CrossRef] [PubMed]
- Ganbaatar, B.; Fukuda, D.; Shinohara, M.; Yagi, S.; Kusunose, K.; Yamada, H.; Soeki, T.; Hirata, K.I.; Sata, M. Empagliflozin ameliorates endothelial dysfunction and suppresses atherogenesis in diabetic apolipoprotein E-deficient mice. Eur. J. Pharm. 2020, 875. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.M.; Battson, M.L.; Jarrell, D.K.; Hou, S.; Ecton, K.E.; Weir, T.L.; Gentile, C.L. SGLT2 inhibition via dapagliflozin improves generalized vascular dysfunction and alters the gut microbiota in type 2 diabetic mice. Cardiovasc. Diabetol. 2018, 17. [Google Scholar] [CrossRef]
- Solini, A.; Giannini, L.; Seghieri, M.; Vitolo, E.; Taddei, S.; Ghiadoni, L.; Bruno, R.M. Dapagliflozin acutely improves endothelial dysfunction, reduces aortic stiffness and renal resistive index in type 2 diabetic patients: A pilot study. Cardiovasc. Diabetol. 2017, 16. [Google Scholar] [CrossRef]
- Packer, M.; Anker, S.D.; Butler, J.; Filippatos, G.; Zannad, F. Effects of sodium-glucose cotransporter 2 inhibitors for the treatment of patients with heart failure—Proposal of a novel mechanism of action. JAMA Cardiol. 2017, 2, 1025–1029. [Google Scholar] [CrossRef] [PubMed]
- Kimura, G. Diuretic action of sodium-glucose cotransporter 2 inhibitors and its importance in the management of heart failure. Circ. J. 2016, 80, 2277–2281. [Google Scholar] [CrossRef]
- Martens, P.; Mathieu, C.; Verbrugge, F.H. Promise of SGLT2 Inhibitors in Heart Failure: Diabetes and Beyond. Curr. Treat. Options Cardiovasc. Med. 2017, 19, 23. [Google Scholar] [CrossRef]
- Uthman, L.; Homayr, A.; Juni, R.P.; Spin, E.L.; Kerindongo, R.; Boomsma, M.; Hollmanna, M.W.; Preckel, B.; Koolwijk, P.; Van Hinsbergh, V.W.M.; et al. Empagliflozin and dapagliflozin reduce ROS generation and restore no bioavailability in tumor necrosis factor α-stimulated human coronary arterial endothelial cells. Cell. Physiol. Biochem. 2019, 53, 865–886. [Google Scholar] [CrossRef]
- Assaly, R.; Gorny, D.; Compagnie, S.; Mayoux, E.; Bernabe, J.; Alexandre, L.; Giuliano, F.; Behr-Roussel, D. The Favorable Effect of Empagliflozin on Erectile Function in an Experimental Model of Type 2 Diabetes. J. Sex. Med. 2018, 15, 1224–1234. [Google Scholar] [CrossRef] [PubMed]
| Drug | Effect on HbA1c | Effect on Body Weight | Effect on Erectile Function (Demonstrated or Potential) |
|---|---|---|---|
| Insulin | ↓↓ | ↑↑ | ? |
| SUs | ↓ | ↑ | ↑/↓ |
| Metformin | ↓ | ↓ | ↑ |
| Acarbose | ↓ | ↓ | ? |
| TZDs | ↓ | ↑ | ↑ |
| GLP1-RA | ↓↓ | ↓↓ | ↑ |
| DPP4i | ↓ | = | ? |
| SGLT2i | ↓↓ | ↓ | ↑ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cignarelli, A.; Genchi, V.A.; D’Oria, R.; Giordano, F.; Caruso, I.; Perrini, S.; Natalicchio, A.; Laviola, L.; Giorgino, F. Role of Glucose-Lowering Medications in Erectile Dysfunction. J. Clin. Med. 2021, 10, 2501. https://doi.org/10.3390/jcm10112501
Cignarelli A, Genchi VA, D’Oria R, Giordano F, Caruso I, Perrini S, Natalicchio A, Laviola L, Giorgino F. Role of Glucose-Lowering Medications in Erectile Dysfunction. Journal of Clinical Medicine. 2021; 10(11):2501. https://doi.org/10.3390/jcm10112501
Chicago/Turabian StyleCignarelli, Angelo, Valentina Annamaria Genchi, Rossella D’Oria, Fiorella Giordano, Irene Caruso, Sebastio Perrini, Annalisa Natalicchio, Luigi Laviola, and Francesco Giorgino. 2021. "Role of Glucose-Lowering Medications in Erectile Dysfunction" Journal of Clinical Medicine 10, no. 11: 2501. https://doi.org/10.3390/jcm10112501
APA StyleCignarelli, A., Genchi, V. A., D’Oria, R., Giordano, F., Caruso, I., Perrini, S., Natalicchio, A., Laviola, L., & Giorgino, F. (2021). Role of Glucose-Lowering Medications in Erectile Dysfunction. Journal of Clinical Medicine, 10(11), 2501. https://doi.org/10.3390/jcm10112501









