Abstract
Background: Accurate knowledge of outcomes in Systemic Lupus Erythematosus (SLE) is crucial to understanding the true burden of the disease. The main objective of this systematic review was to gather all population-based studies on mortality, end-stage renal disease (ESRD) and cancer in SLE. Method: We performed a systematic literature search in two electronic databases (MEDLINE and Embase) to identify all population-based articles on SLE and survival, mortality, ESRD and cancer. The SLE diagnosis had to be verified. We used the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines (PRISMA). Results: We included 40/1041 articles on mortality (27), ESRD (11) and cancer (3), of which six were defined as inception studies. In the total SLE cohort, the standardized mortality ratio ranged from 1.9 to 4.6. Cardiovascular disease was the most frequent cause of death in studies with follow-up times over 15 years. SLE progressed to ESRD in 5–11% of all SLE patients. There are no data supporting increased cancer incidence from population-based inception cohorts. Conclusion: There is a need for more population-based studies on outcomes of SLE, especially inception studies, with the use of control groups and follow-up times over 15 years.
1. Introduction
Systemic Lupus Erythematosus (SLE) is a rare systemic and chronic disease often referred to as the prototype of autoimmune rheumatic diseases because of the varied spectrum of clinical manifestations and diversity of phenotypes. The etiology of SLE is believed to be multifactorial, and both genetic predisposition and environmental triggers are most likely involved [1]. The incidence, severity and phenotypic expression of the disease differ between ethnic groups, gender and age at disease onset. The annual incidence of SLE varies from 0.3 to 23.3/100,000, and the prevalence varies from 0 to 241/100,000 [1]. The variations are highly dependent on the method of retrieval and the definition of SLE diagnosis.
Several aspects of SLE make it one of the most challenging conditions to study at the population level. First, no diagnostic criteria for SLE exist and the diagnosis is based on the judgement of an experienced clinician. Diagnosing SLE can be challenging since SLE is a great imitator of other diseases. The symptoms of SLE overlap many other diseases that can easily be mistaken for SLE in as much as 40% [2,3,4,5] of cases.
Secondly, in many countries, SLE patients are not treated in the same hospital and/or specialization since different organs may be affected and the severity of the disease varies. Selected patient populations from tertiary hospitals tend to miss milder cases, and therefore underestimate the incidence and overestimate the severity of SLE. Thus, a closer estimate of the true frequency of clinical and laboratory SLE manifestations and outcomes is more likely from a geographically complete cohort of patients. All these aspects of the disease make it difficult and labour-intensive to collect epidemiological data. In Georgia, Lim et al. found 45,000 potential patients, screened 3142 records and found 1320 patients with a verified SLE diagnosis. In Sweden, Ingvarsson et al. screened 2461 cases and found 55 patients with a verified diagnosis, and Voss et al. in Denmark screened 980 cases to find 95 patients with a verified SLE diagnosis [4,5,6].
Earlier publications on SLE and epidemiology differ greatly in study-design. A good epidemiological study is highly dependent on valid data to obtain reliable results that are indicative of the total size of the problem and thus, a reliable assessment of outcome. Truly population-based research, with a verified and ascertained SLE diagnosis by chart review, is the best way to achieve the most accurate knowledge possible on this disease and its outcome measures. The use of standardized methods gives the best basis for comparison of epidemiological data across different studies and countries.
The objective of this study was to conduct a review of literature on population-based epidemiologic data on SLE and well-defined and hard outcomes; mortality, end-stage renal disease (ESRD) and cancer. The elected publications were thoroughly reviewed to ensure that they were from population-based cohorts and that the SLE diagnose was verified.
2. Materials and Methods
A senior medical librarian searched two electronic databases: MEDLINE (Ovid) and Embase (Ovid), from their inception to 25 June 2021, with language restricted to English. The systematic search used both controlled vocabulary (MeSH terms or EMTREE terms) and text word search in title, abstract or author keywords. The search consisted of two searches with different approaches. Search 1: Concepts for systemic lupus, SLE criteria, mortality or cancer, were combined with the Boolean operator AND. Search 2: Concepts for lupus nephritis, end stage renal disease or kidney transplantation were combined with the Boolean operator AND (Supplementary Materials S2). Both searches were restricted to population-based cohorts.
Two investigators (HH and KL or SRM and KL) independently evaluated all abstracts and titles to determine eligibility for inclusion. When necessary, the articles were reviewed in full, and, if in conflict, discussed in plenum (HH, SRM, KL). The authors also searched the reference list of included articles to find additional relevant studies.
For inclusion in this systematic review, the SLE diagnosis had to be verified by chart review. Studies on SLE were included on the relevant outcomes: mortality, overall and renal survival and risk of malignancy.
We excluded: (1) Studies that failed to validate the SLE diagnosis by chart review; (2) Studies based on administrative data; (3) Studies from tertiary centers only, if it was not specified that it was the only hospital serving the region; (4) Animal studies; (5) Meta-analysis; (6) Case reports; (7) Studies on unrelated outcomes; (8) Studies of selected SLE subsets (paediatric SLE, biopsy-proven lupus nephritis (LN), hospital inpatients); (9) Studies with fewer than 30 patients; (10) Studies on subset of relevant outcome (cardiovascular mortality).
Causes of death analyses were excluded from this review if the study reported only multiple causes of death. We defined the study period as years from start of inclusion to end of follow-up. The total SLE population was defined as all SLE patients in the given study-period. Incident SLE were defined as patients diagnosed within the study-period. Inception SLE was defined as patients diagnosed within the study-period and captured within one year of the diagnosis.
This review was carried out in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [7].
3. Results
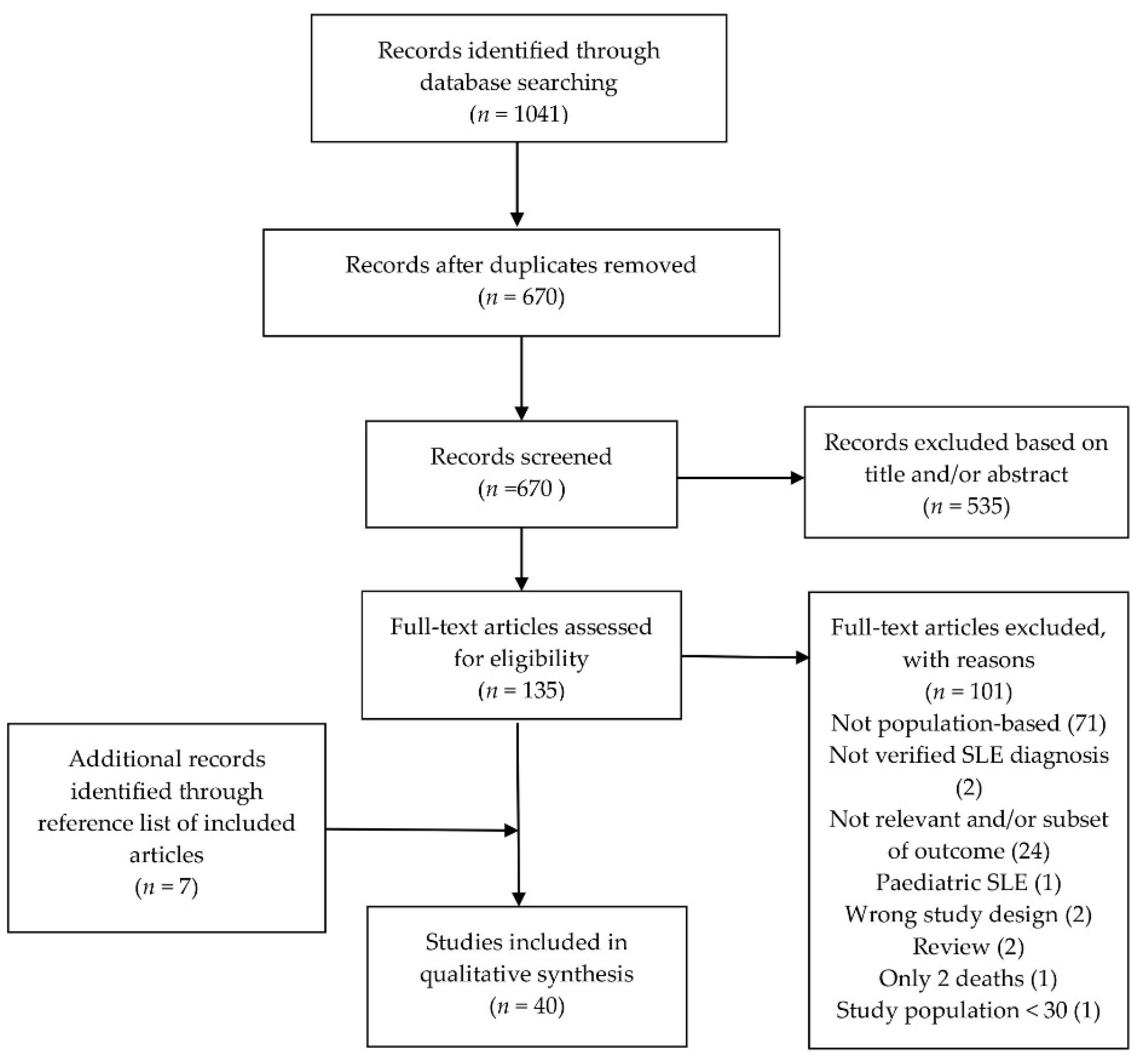
We screened 1041 titles/abstracts. Through the screening process, we identified 40 studies that met the criteria for inclusion, whereof 27 were for survival and mortality, 11 were for ESRD and three were for cancer (Figure 1). We found seven articles through manual search of the reference list of included articles. The case finding methodology and SLE ascertainment in all included cohorts is described in Supplementary Materials Table S1. All but three study locations included only SLE patients who fulfilled four or more of the American College of Rheumatology SLE classification criteria [8,9,10,11].

Figure 1.
Flowchart of literature search and study inclusion. Studies identified through MEDLINE (Ovid) and Embase (Ovid) through 25 June 2021.
3.1. Standardized Mortality Rate and Survival
Twenty-three population-based studies reported survival with SLE, while a standardized mortality rate (SMR) was reported in 13 studies. Eighteen studies used incident patients for survival analysis, while five included all SLE patients (total). Six studies used only incident patients and seven used the total SLE population for SMR analysis (Table 1).

Table 1.
Survival and standardized mortality rate (SMR) in Systemic Lupus Erythematosus, from population-based cohorts.
The ten-year survival in incident cohorts ranges from 46% in Curacao to 92% in northern Norway, and from 90 to 92% in Europe and 76 to 89% in North America [10,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27]. Five and ten-year survival differed in incident cohorts with patient inclusion before and after 1990 (five-year survival 80% versus 92% and ten-year survival 63% versus 88%) [10,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28]. For all studies with patient inclusion starting after 1990, the five-year survival was 90% or more, except for Barbados and Wisconsin [9,11,14,20,23,27,29]. In studies on total SLE cohorts, the SMR ranges from 1.9 to 4.6 [9,19,20,26,27,30,31,32]. For female SLE patients, the SMR ranges from 1.8 to 4.7, while in male patients the SMR ranges from 1.5 to 4.6 [9,10,19,20,30,31,33]. There was no significant difference between the two groups. Among the incident SLE patients the SMR varied from 1.3 to 11.1, depending on follow-up time (one to 33 years) [10,16,17,33,34]. Only one incident study reported 25-year survival with SLE (60% survival versus 73% in the general population) [10].
3.2. The Main Causes of Death in Systemic Lupus Erythematosus
An average of 41% of patients in the studies from Asia died of infections, compared to an average of 12% in studies from Europe (Table 2) [9,10,14,16,17,20,21,23,30,33,35]. Renal failure was the underlying cause of death in about 17% (median) of SLE patients, except for a much higher frequency in Barbados (46%) [12,16,21,23,25,27,30,33,35]. From the article with the shortest follow-up time versus the longest, the causes of death varied from 60% infections and 6% cardio- and cerebrovascular disease (CVD) in Hong Kong [33] to 15% infections and 59% CVD in Sweden [10]. CVD was the most frequent cause of death in the two study locations with population-based cohorts over time [9,10].

Table 2.
The main causes of death in Systemic Lupus Erythematosus, from population-based studies.
3.3. End Stage Renal Disase
Within the primary studies reviewed, ESRD developed in 5–11% of the total SLE patients [35,36,37], of which 5–6% were in a Scandinavian population (Table 3) [36,38]. The incidence rate of ESRD varied from 2.3 to 11.1/1000 patient years in incident patient populations, depending on the population studied (Table 3) [38,39,40].

Table 3.
Risk of End Stage Renal Disease in Systemic Lupus Erythematosus, from population-based studies.
3.4. Cancer
We found only three studies on cancer in population-based cohorts, from three different countries. Only the study from Sweden was an inception study (Table 4) [41].

Table 4.
Cancer risk in Systemic Lupus Erythematosus relative to the general population, from population-based studies.
4. Discussion
The literature search on outcomes in SLE and mortality, ESRD and cancer revealed population-based studies from 22 different locations around the world. The main discovery is that from 1990 there is a higher survival rate during the first five to ten years of the disease. A cardiovascular cause of death is common later in the disease’s course, and improvement in survival is less clear. Death caused by infections differs between geographical area and the death rate due to infections is lowest in Europe. Development of ESRD occurs in 5–10% of SLE patients in cohorts of European and Asian ethnic population. ESRD is, however, more common in the African ethnic population. We only discovered one study on cancer from a population-based cohort with inclusion at the time of the SLE diagnosis.
It is well established that the change in treatment of SLE after the 1950s and 1960s caused the survival rate to improve tremendously, from less than a 50% survival rate over five years in the 1950s [47,48]. There are, however, some aspects of selected patient populations that may influence the reported outcome; a tertiary center may overestimate the severity of SLE by missing the diagnosis of milder SLE cases, for example.
Our search on survival with SLE revealed a ten-year survival rate varying with time and location, from 46% in Curacao in the 1980s to 93% in a more recent study from northern Norway [15,19]. The overall trend in survival indicates an improvement in five- and ten-year survival rates after 1990, with a five-year survival similar to the control population. This discovery is in accordance with the conclusion in a recent meta-analysis that survival with SLE improved up to the 1990s, but since appears to have stabilized [48].
A control group is necessary to enhance the quality of survival estimates in SLE. As survival from SLE improves, it may become similar to the survival rate in the general population. The reported survival rate from studies depends on the age composition of the SLE cohort and hence, the time since inception. In this systematic review, nine of the studies included made use of a control group in their survival analysis. They all included only incident cases and five studies were also defined as inception studies. From the inception studies with control groups conducted after 1990, the ten-year survival is only slightly lower in the SLE groups versus the control groups (91% vs. 96%) [19,20]. However, the gap seems to increase with time from diagnosis [10,21].
Findings from this review also indicate that the main causes of death from SLE differ with the length of follow-up time of the studies; CVD is more frequent in studies with the longest follow-up time [10,21]. It is well known from earlier studies that death due to CVD is more frequent later in the course of the disease [49,50]. Urowitch et al. identified this bimodal pattern of mortality in 1976 [51]. In the included studies, European SLE patients died less often of infections compared to Asians. It appears we still do not manage to prevent CVD over time, as up to 59% of SLE patients die of CVD. This might indicate better treatment for the acute phase of SLE, but not for damage accrual due to SLE. However, death from infections remains prominent in certain parts of the world.
In this review, SMR in total SLE cohorts ranges from 1.9 to 4.6, similar, but with a slightly lower range of variation, compared to two previous meta-analyses [52,53]. Studying SMR in incident populations makes comparison difficult as the inclusion periods differ, the highest SMR being from Taiwan within the first year after diagnosis [34]. Several studies have identified ethnicity as a modifier of outcome in SLE, with lower survival in patients of African descent [47,54]. This corresponds with our findings of the lowest SMR in a predominantly white Scandinavian population. The discrepancy in prognosis might be due to both genetic and socioeconomic factors. A possible gender disparity in SLE prognosis has been proposed; however, the results have been inconsistent and contradictory [55]. In this review, we found no significant sex differences in SMR.
Many studies have reported the risk of ESRD development in SLE, and, as registries of biopsy-proven LN are quite common, outcomes in this particular patient subset have been widely investigated. However, as many as 44% of all LN patients are not biopsy-proven [56]. Thus, we excluded studies of this selected SLE patient subset, as they might differ from other LN patients. In this review, we found that only 11 population-based studies estimated the frequency of ESRD in SLE populations. An estimated 5–11% of SLE patients progressed to ESRD, fewer than in a recent meta-analysis [57]. A lower frequency of ESRD in the white population is in line with previous reports [54,57]. The trend in ESRD development seems to be stable over time, despite improvements in therapy. This corresponds to findings from a recent meta-analysis where the risk of ESRD development remained unchanged during the last decade [57].
We identified only three studies on cancer development in SLE patients. Only one was an inception study [41]. In these studies, the cancer risk was increased by 1.2–1.8 times. By comparison, a prior review, which also included non-population-based studies, found an increased risk of cancer ranging from 1.1 to 3.6 times in the SLE population [58]. The lowest cancer risk (SMR 1.2) found in our review was from an old Swedish study with 116 SLE patients. The study from the National Health Insurance Research Database from Taiwan is on the other end of the scale, with a SIR of 1.8 [46].
Earlier studies, mostly non-population-based or without a verified diagnosis, have found that hematological cancers appear more often in the SLE population compared to the general population [58]. All three studies in this review found significantly higher numbers of lymphomas, and especially non-Hodgkin lymphomas, with a reported SMR of 11.6 from Sweden and SIR of 7.3 from Taiwan [41,45,46]. In addition, all three studies found an increased incidence of lung cancer [41,45,46]. Taiwan reports a significant increase for lung/mediastinum (SIR 1.2) [46], yet data from Sweden (SMR 5.6) and Iceland (O/E ratio 1.7) are not significant [41,45].
Cancer development in SLE patients is particularly difficult to study for two reasons. First, cancer sometimes leads to death; subsequently, patients who get cancer early in the course of the disease may not be captured. Secondly, some people with cancer might have paraneoplastic symptoms that may mimic SLE and then be mistakenly diagnosed with SLE. This emphasizes both the importance of a verified SLE diagnosis in studies on cancer and SLE, and the need for further population-based, and preferably inception-based (early capture), studies on cancer.
Considerable differences in the methods for case finding, verification of diagnosis, and study design can make comparing the results of the SLE outcomes difficult. To overcome some of these problems, all studies in this systematic review have employed comprehensive case-finding and case ascertainment methods, or it has been indicated in the article that all patients in a defined geographic region were included. However, the geographic area and its location for care of SLE patients is not always described in detail, and it is likely that we have missed some population-based studies.
The composition of the cohorts used for analysis of outcomes differs as some studies include all patients and some include only incident patients, making comparisons more difficult. Only seven studies of incident SLE patients had a follow-up period over 15 years [10,13,14,21,25,27,41]. The reason for this may be that hospital data registries going back before the year 2000 are rare and not so easily accessible. They may also not contain the entire volume of ICD-codes on outpatients [59].
Most of the population-based studies, except for Taiwan, are small due to the work effort necessary to identify all patients and verify their diagnoses. Taiwan has a good health system, and 96%–99% of its population is included in the National Health Insurance Database. All SLE patients must fulfill the ACR criteria to receive their benefit claim checks as in- and outpatients [34,39,42,43,46]. However, this may also give the patients and their doctors an additional motive towards approving the SLE diagnosis. In addition, verification of the SLE diagnosis is processed earlier on in the course of the disease in Taiwan compared to the other studies. Hence, an early misdiagnosis of SLE would not be reclassified retrospectively.
We found that six locations (Iceland, Lund in Sweden, Funen in Denmark, northern Norway, Rochester in the USA and New Territories in Hong Kong) have repeated the retrieval of patients at several time points [10,19,27,31,33]. Scandinavia is highly represented in publishing from population-based studies, probably due to the health care system being mostly public, making it easier to identify the patients. Despite small study populations, these are valuable contributions to population-based knowledge of outcomes for SLE. Lund in Sweden already published the very first data on survival from a population-based cohort in 1989 and has, to date, the longest follow-up time on an inception cohort reporting on 25-year survival (60%) [10]. However, four locations from the USA have made a tremendous effort collecting larger population-based cohorts that were published in the last decade [25,27,32,37,40].
5. Conclusions
Population-based studies on SLE patients with a verified diagnosis is considered the gold standard in the pursuit of finding the true outcomes of suffering from SLE. Studies using the 1997 ACR criteria are easier to compare over time, as most studies included only SLE patients with four or more ACR criteria. There is a special need for cancer studies and studies with longer follow-up time on survival in population-based inception cohorts.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/jcm10194306/s1, Table S1: Case finding methodology and SLE ascertainment in population-based Systemic Lupus Erythematosus cohorts; Supplementary Materials S2: Search strings.
Author Contributions
K.L. received the invitation to write the article. Ø.M. and K.L. concepted and designed the article. H.H. and S.R.M. performed the literature search. H.H., S.R.M. and K.L. performed the screening and eligibility phase and study identification. H.H. and S.R.M. wrote the first draft of the article; they contributed equally to this paper and share first authorship. All authors contributed to the writing of subsequent drafts. All authors have read and agreed to the published version of the manuscript.
Funding
This research received funding from The Dam Foundation (Grant number 296618), The Norwegian Women’s Public Health Association (Grant number 40108) and Vivi Irene Hansens fond (Grant number 190613).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The authors would like to thank Liberian Hilde Iren Flaatten, University of Oslo, for performing the literature search, and Torild Garen for valuable technical assistance.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Rees, F.; Doherty, M.; Grainge, M.; Lanyon, P.; Zhang, W. The worldwide incidence and prevalence of systemic lupus erythematosus: A systematic review of epidemiological studies. Rheumatology 2017, 56, 1945–1961. [Google Scholar] [CrossRef]
- Lerang, K.; Gilboe, I.-M.; Gran, J.T. Differences between rheumatologists and other internists regarding diagnosis and treatment of systemic lupus erythematosus. Rheumatology 2011, 51, 663–669. [Google Scholar] [CrossRef]
- Gergianaki, I.; Fanouriakis, A.; Repa, A.; Tzanakakis, M.; Adamichou, C.; Pompieri, A.; Spirou, G.; Bertsias, A.; Kabouraki, E.; Tzanakis, I.; et al. Epidemiology and burden of systemic lupus erythematosus in a Southern European population: Data from the community-based lupus registry of Crete, Greece. Ann. Rheum. Dis. 2017, 76, 1992–2000. [Google Scholar] [CrossRef]
- Voss, A.; Green, A.; Junker, P. Systemic lupus erythematosus in Denmark: Clinical and epidemiological characterization of a county-based cohort. Scand. J. Rheumatol. 1998, 27, 98–105. [Google Scholar]
- Ingvarsson, R.F.; Bengtsson, A.A.; Jönsen, A. Variations in the epidemiology of systemic lupus erythematosus in southern Sweden. Lupus 2016, 25, 772–780. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.S.; Bayakly, A.R.; Helmick, C.G.; Gordon, C.; Easley, K.; Drenkard, C. The Incidence and Prevalence of Systemic Lupus Erythematosus, 2002–2004: The Georgia Lupus Registry. Arthritis Rheumatol. 2013, 66, 357–368. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; The PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Hochberg, M.C. Updating the American college of rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum. 1997, 40, 1725. [Google Scholar] [CrossRef] [PubMed]
- Voss, A.; Laustrup, H.; Hjelmborg, J.; Junker, P. Survival in systemic lupus erythematosus, 1995–2010. A prospective study in a Danish community. Lupus 2013, 22, 1185–1191. [Google Scholar] [CrossRef]
- Ingvarsson, R.F.; Landgren, A.J.; Bengtsson, A.A.; Jönsen, A. Good survival rates in systemic lupus erythematosus in southern Sweden, while the mortality rate remains increased compared with the population. Lupus 2019, 28, 1488–1494. [Google Scholar] [CrossRef]
- Al-Adhoubi, N.K.; Al-Balushi, F.; Al Salmi, I.; Ali, M.; Al Lawati, T.; Al Lawati, B.S.H.; Abdwani, R.; Al Shamsi, A.; Al Kaabi, J.; Al Mashaani, M.; et al. A multicenter longitudinal study of the prevalence and mortality rate of systemic lupus erythematosus patients in Oman: Oman Lupus Study. Int. J. Rheum. Dis. 2021, 24, 847–854. [Google Scholar] [CrossRef]
- Flower, C.; Hennis, A.J.M.; Hambleton, I.R.; Nicholson, G.D.; Liang, M.H.; the The Barbados National Lupus Registry Group. Systemic lupus erythematosus in an Afro-Caribbean population: Incidence, clinical manifestations and survival in the Barbados national lupus registry. Arthritis Rheum. 2012, 64, 1151–1158. [Google Scholar] [CrossRef]
- Peschken, C.A.; Esdaile, J.M. Systemic lupus erythematosus in North American Indians: A population based study. J. Rheumatol. 2000, 27. [Google Scholar]
- Mok, C.C.; Mak, A.; Chu, W.P.; To, C.H.; Nin Wong, S. Long-term Survival of Southern Chinese Patients With Systemic Lupus Erythematosus. Medicine 2005, 84, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Nossent, J. Systemic lupus erythematosus on the Caribbean island of Curacao: An epidemiological investigation. Ann. Rheum. Dis. 1992, 51, 1197–1201. [Google Scholar] [CrossRef] [PubMed]
- Alamanos, Y.; Voulgari, P.V.; Siozos, C.; Katsimpri, P.; Tsintzos, S.; Dimou, G.; Politi, E.N.; Rapti, A.; Laina, G.; Drosos, A.A. Epidemiology of systemic lupus erythematosus in northwest Greece 1982–2001. J. Rheumatol. 2003, 30, 731–735. [Google Scholar]
- Gudmundsson, S.; Steinsson, K. Systemic lupus erythematosus in Iceland 1975 through 1984. A nationwide epidemiological study in an unselected population. J. Rheumatol. 1990, 17, 1162–1167. [Google Scholar]
- Nossent, H.C. Systemic lupus erythematosus in the Arctic region of Norway. J. Rheumatol. 2001, 28, 539–546. [Google Scholar]
- Eilertsen, G.; Becker-Merok, A.; Nossent, J.C. The Influence of the 1997 Updated Classification Criteria for Systemic Lupus Erythematosus: Epidemiology, Disease Presentation, and Patient Management. J. Rheumatol. 2009, 36, 552–559. [Google Scholar] [CrossRef][Green Version]
- Lerang, K.; Gilboe, I.-M.; Thelle, D.S.; Gran, J.T. Mortality and years of potential life loss in systemic lupus erythematosus: A population-based cohort study. Lupus 2014, 23, 1546–1552. [Google Scholar] [CrossRef]
- Alonso, M.D.; Llorca, J.; Martinez-Vazquez, F.; Miranda-Filloy, J.A.; de Teran, T.D.; Dierssen, T.; Rodriguez, T.R.V.; Gomez-Acebo, I.; Blanco, R.; Gonzalez-Gay, M.A. Systemic Lupus Erythematosus in Northwestern Spain. Medicine 2011, 90, 350–358. [Google Scholar] [CrossRef]
- Ståhl-Hallengren, C.; Jönsen, A.; Nived, O.; Sturfelt, G. Incidence studies of systemic lupus erythematosus in Southern Sweden: Increasing age, decreasing frequency of renal manifestations and good prognosis. J. Rheumatol. 2000, 27, 685–691. [Google Scholar] [PubMed]
- Pamuk, O.N.; Balci, M.A.; Donmez, S.; Tsokos, G.C. The incidence and prevalence of systemic lupus erythematosus in Thrace, 2003–2014: A 12-year epidemiological study. Lupus 2015, 25, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Naleway, A.L.; Davis, M.E.; Greenlee, R.T.; Wilson, D.A.; Mccarty, D.J. Epidemiology of systemic lupus erythematosus in rural Wisconsin. Lupus 2005, 14, 862–866. [Google Scholar] [CrossRef] [PubMed]
- Bartels, C.M.; Buhr, K.A.; Goldberg, J.W.; Bell, C.L.; Visekruna, M.; Nekkanti, S.; Greenlee, R.T. Mortality and Cardiovascular Burden of Systemic Lupus Erythematosus in a US Population-based Cohort. J. Rheumatol. 2014, 41, 680–687. [Google Scholar] [CrossRef] [PubMed]
- Uramoto, K.M.; Michet, C.J., Jr.; Thumboo, J.; Sunku, J.; O’Fallon, W.M.; Gabriel, S.E. Trends in the incidence and mortality of systemic lupus erythematosus, 1950-1992. Arthritis Rheum. 1999, 42, 46–50. [Google Scholar] [CrossRef]
- Jarukitsopa, S.; Hoganson, D.D.; Crowson, C.S.; Sokumbi, O.; Davis, M.D.; Michet, C.J.; Matteson, E.L.; Kremers, H.M.; Chowdhary, V.R. Epidemiology of Systemic Lupus Erythematosus and Cutaneous Lupus Erythematosus in a Predominantly White Population in the United States. Arthritis Rheum. 2014, 67, 817–828. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, H.; Nived, O.; Sturfelt, G. Outcome in systemic lupus erythematosus: A prospective study of patients from a defined population. Medicine 1989, 68, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Lucero, L.G.; Barbaglia, A.L.; Bellomio, V.I.; Bertolaccini, M.C.; Escobar, M.A.M.; Sueldo, H.R.; Yacuzzi, M.S.; Carrizo, G.A.; Robles, N.; Rengel, S.; et al. Prevalence and incidence of systemic lupus erythematosus in Tucumán, Argentina. Lupus 2020, 29, 1815–1820. [Google Scholar] [CrossRef]
- Jacobsen, S. Mortality and Causes of Death of 513 Danish Patients with Systemic Lupus Erythematosus. Scand. J. Rheumatol. 1999, 28, 75–80. [Google Scholar] [CrossRef]
- Laustrup, H.; Voss, A.; Green, A.; Junker, P. Occurrence of systemic lupus erythematosus in a Danish community: An 8-year prospective study. Scand. J. Rheumatol. 2009, 38, 128–132. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.S.; Helmick, C.G.; Bao, G.; Hootman, J.; Bayakly, R.; Gordon, C.; Drenkard, C. Racial Disparities in Mortality Associated with Systemic Lupus Erythematosus—Fulton and DeKalb Counties, Georgia, 2002–2016. MMWR. Morb. Mortal. Wkly. Rep. 2019, 68, 419–422. [Google Scholar] [CrossRef] [PubMed]
- Mok, C.C.; To, C.H.; Ho, L.Y.; Yu, K.L. Incidence and mortality of systemic lupus erythematosus in a southern Chinese population, 2000-2006. J. Rheumatol. 2008, 35, 1978–1982. [Google Scholar]
- Yeh, K.-W.; Yu, C.-H.; Chan, P.-C.; Horng, J.-T.; Huang, J.-L. Burden of systemic lupus erythematosus in Taiwan: A population-based survey. Rheumatol. Int. 2013, 33, 1805–1811. [Google Scholar] [CrossRef]
- Iseki, K.; Miyasato, F.; Oura, T.; Uehara, H.; Nishime, K.; Fukiyama, K. An Epidemiologic Analysis of End-stage Lupus Nephritis. Am. J. Kidney Dis. 1994, 23, 547–554. [Google Scholar] [CrossRef]
- Jacobsen, S.; Petersen, J.; Ullman, S.; Junker, P.; Voss, A.; Rasmussen, J.M.; Tarp, U.; Poulsen, L.H.; Hansen, G.V.O.; Skaarup, B.; et al. A multicentre study of 513 Danish patients with systemic lupus erythematosus. I. Disease manifestations and analyses of clinical subsets. Clin. Rheumatol. 1998, 17, 468–477. [Google Scholar] [CrossRef]
- Somers, E.C.; Marder, W.; Cagnoli, P.; Lewis, E.E.; Deguire, P.; Gordon, C.; Helmick, C.G.; Wang, L.; Wing, J.J.; Dhar, J.P.; et al. Population-Based Incidence and Prevalence of Systemic Lupus Erythematosus: The Michigan Lupus Epidemiology and Surveillance Program. Arthritis Rheumatol. 2013, 66, 369–378. [Google Scholar] [CrossRef]
- Moe, S.E.R.; Molberg, Ø.; Strøm, E.H.; Lerang, K. Assessing the relative impact of lupus nephritis on mortality in a population-based systemic lupus erythematosus cohort. Lupus 2019, 28, 818–825. [Google Scholar] [CrossRef]
- Yu, K.-H.; Kuo, C.-F.; Chou, I.-J.; Chiou, M.-J.; See, L.-C. Risk of end-stage renal disease in systemic lupus erythematosus patients: A nationwide population-based study. Int. J. Rheum. Dis. 2016, 19, 1175–1182. [Google Scholar] [CrossRef] [PubMed]
- Plantinga, L.C.; Lim, S.S.; Patzer, R.E.; McClellan, W.M.; Kramer, M.; Klein, M.; Pastan, S.; Gordon, C.; Helmick, C.G.; Drenkard, C. Incidence of End-Stage Renal Disease Among Newly Diagnosed Systemic Lupus Erythematosus Patients: The Georgia Lupus Registry. Arthritis Rheum. 2015, 68, 357–365. [Google Scholar] [CrossRef]
- Nived, O.; Bengtsson, A.; Jönsen, A.; Sturfelt, G.; Olsson, H. Malignancies during follow-up in an epidemiologically defined systemic lupus erythematosus inception cohort in southern Sweden. Lupus 2001, 10, 500–504. [Google Scholar] [CrossRef]
- Lin, C.-H.; Hung, P.-H.; Hu, H.-Y.; Chen, Y.-J.; Guo, H.-R.; Hung, K.-Y. Infection-related hospitalization and risk of end-stage renal disease in patients with systemic lupus erythematosus: A nationwide population-based study. Nephrol. Dial. Transplant. 2016, 32, 1683–1690. [Google Scholar] [CrossRef]
- Lin, W.-H.; Guo, C.-Y.; Wang, W.-M.; Yang, D.-C.; Kuo, T.-H.; Liu, M.-F.; Wang, M.-C. Incidence of progression from newly diagnosed systemic lupus erythematosus to end stage renal disease and all-cause mortality: A nationwide cohort study in Taiwan. Int. J. Rheum. Dis. 2013, 16, 747–753. [Google Scholar] [CrossRef]
- Eilertsen, G.O.; Fismen, S.; Hanssen, T.-A.; Nossent, J.C. Decreased incidence of lupus nephritis in northern Norway is linked to increased use of antihypertensive and anticoagulant therapy. Nephrol. Dial. Transplant. 2010, 26, 620–627. [Google Scholar] [CrossRef][Green Version]
- Ragnarsson, O.; Gröndal, G.; Steinsson, K. Risk of malignancy in an unselected cohort of Icelandic patients with systemic lupus erythematosus. Lupus 2003, 12, 687–691. [Google Scholar] [CrossRef]
- Chen, Y.-J.; Chang, Y.-T.; Wang, C.-B.; Wu, C.-Y. Malignancy in Systemic Lupus Erythematosus: A Nationwide Cohort Study in Taiwan. Am. J. Med. 2010, 123, 1150.e1–1150.e6. [Google Scholar] [CrossRef]
- Borchers, A.T.; Keen, C.L.; Shoenfeld, Y.; Gershwin, M. Surviving the butterfly and the wolf: Mortality trends in systemic lupus erythematosus. Autoimmun. Rev. 2004, 3, 423–453. [Google Scholar] [CrossRef]
- Tektonidou, M.; Lewandowski, L.B.; Hu, J.; Dasgupta, A.; Ward, M.M. Survival in adults and children with systemic lupus erythematosus: A systematic review and Bayesian meta-analysis of studies from 1950 to 2016. Ann. Rheum. Dis. 2017, 76, 2009–2016. [Google Scholar] [CrossRef]
- Schoenfeld, S.R.; Kasturi, S.; Costenbader, K.H. The epidemiology of atherosclerotic cardiovascular disease among patients with SLE: A systematic review. Semin. Arthritis Rheum. 2013, 43, 77–95. [Google Scholar] [CrossRef]
- Lu, X.; Wang, Y.; Zhang, J.; Pu, D.; Hu, N.; Luo, J.; An, Q.; He, L. Patients with systemic lupus erythematosus face a high risk of cardiovascular disease: A systematic review and Meta-analysis. Int. Immunopharmacol. 2021, 94, 107466. [Google Scholar] [CrossRef]
- Urowitz, M.B.; Bookman, A.A.; Koehler, B.E.; Gordon, D.A.; Smythe, H.A.; Ogryzlo, M.A. The bimodal mortality pattern of systemic lupus erythematosus. Am. J. Med. 1976, 60, 221–225. [Google Scholar] [CrossRef]
- Yurkovich, M.; Vostretsova, K.; Chen, W.; Aviña-Zubieta, J.A. Overall and Cause-Specific Mortality in Patients With Systemic Lupus Erythematosus: A Meta-Analysis of Observational Studies. Arthritis Rheum. 2013, 66, 608–616. [Google Scholar] [CrossRef]
- Lee, Y.H.; Choi, S.J.; Ji, J.D.; Song, G.G. Overall and cause-specific mortality in systemic lupus erythematosus: An updated meta-analysis. Lupus 2016, 25, 727–734. [Google Scholar] [CrossRef] [PubMed]
- Lewis, M.J.; Jawad, A.S. The effect of ethnicity and genetic ancestry on the epidemiology, clinical features and outcome of systemic lupus erythematosus. Rheumatology 2016, 56, i67–i77. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.-J.; Wallace, D.; Ishimori, M.; Scofield, R.; Weisman, M. Review: Male systemic lupus erythematosus: A review of sex disparities in this disease. Lupus 2009, 19, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Hanly, J.G.; O’Keeffe, A.G.; Su, L.; Urowitz, M.B.; Romero-Diaz, J.; Gordon, C.; Bae, S.-C.; Bernatsky, S.; Clarke, A.E.; Wallace, D.J.; et al. The frequency and outcome of lupus nephritis: Results from an international inception cohort study. Rheumatology 2015, 55, 252–262. [Google Scholar] [CrossRef]
- Tektonidou, M.; Dasgupta, A.; Ward, M.M. Risk of End-Stage Renal Disease in Patients With Lupus Nephritis, 1971-2015: A Systematic Review and Bayesian Meta-Analysis. Arthritis Rheumatol. 2016, 68, 1432–1441. [Google Scholar] [CrossRef]
- Choi, M.Y.; Flood, K.; Bernatsky, S.; Ramsey-Goldman, R.; Clarke, A.E. A review on SLE and malignancy. Best Pr. Res. Clin. Rheumatol. 2017, 31, 373–396. [Google Scholar] [CrossRef]
- Jørgensen, K.T.; Pedersen, B.V.; Nielsen, N.M.; Jacobsen, S.; Frisch, M. Childbirths and risk of female predominant and other autoimmune diseases in a population-based Danish cohort. J. Autoimmun. 2012, 38, J81–J87. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).