Fludarabine–Cyclophosphamide-Based Conditioning with Antithymocyte Globulin Serotherapy Is Associated with Durable Engraftment and Manageable Infections in Children with Severe Aplastic Anemia
Abstract
:1. Introduction
2. Materials and Methods
Statistical Analysis
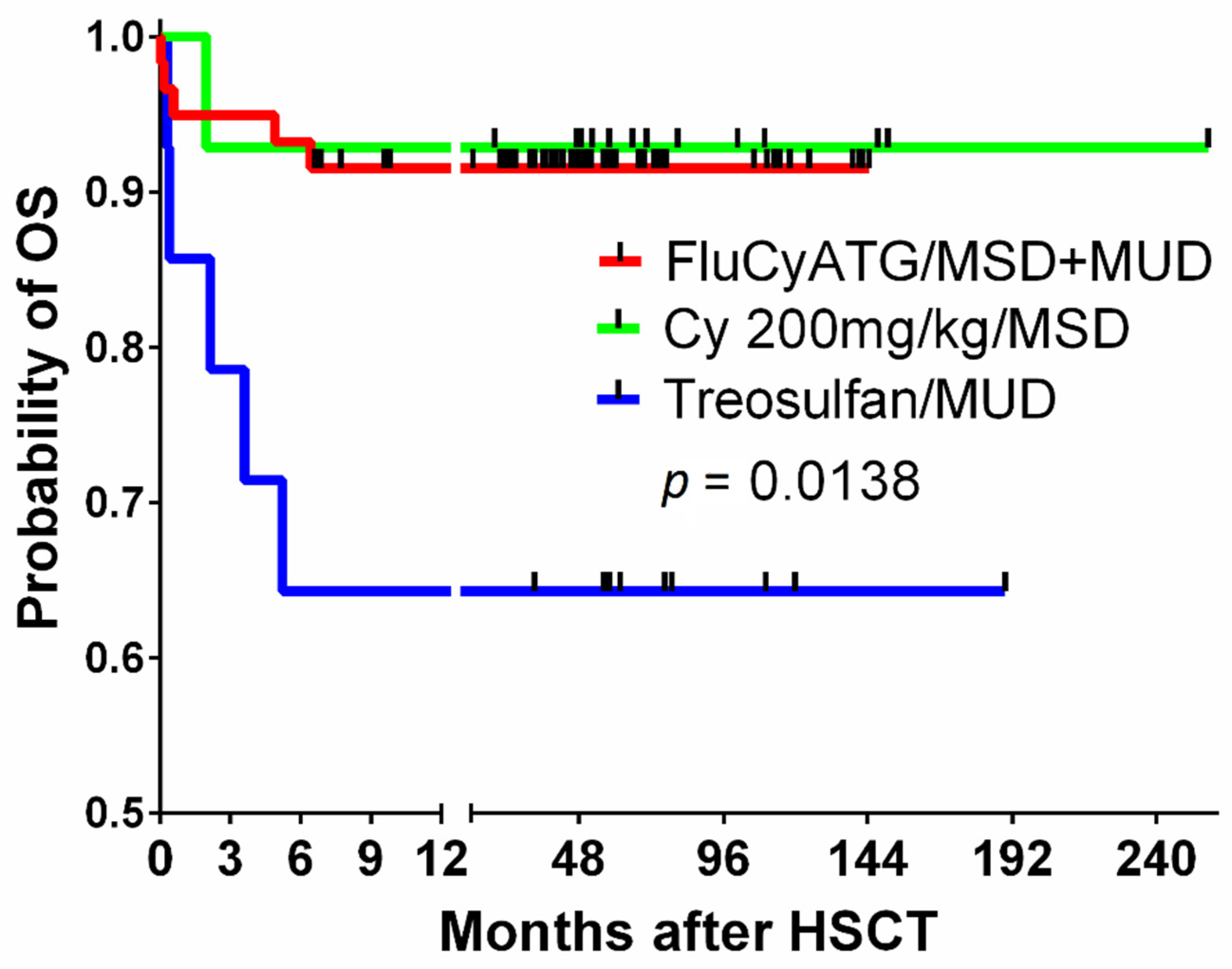
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A. Prophylaxis and Treatment of Infections
References
- Keel, S.B.; Scott, A.; Sanchez-Bonilla, M.; Ho, P.A.; Gulsuner, S.; Pritchard, C.C.; Abkowitz, J.L.; King, M.-C.; Walsh, T.; Shimamura, A. Genetic features of myelodysplastic syndrome and aplastic anemia in pediatric and young adult patients. Haematologica 2016, 101, 1343–1350. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Young, N.S.; Calado, R.T.; Scheinberg, P. Current concepts in the pathophysiology and treatment of aplastic anemia. Blood 2006, 108, 2509–2519. [Google Scholar] [CrossRef] [PubMed]
- Young, N.S.; Scheinberg, P.; Calado, R.T. Aplastic Anemia. N. Engl. J. Med. 2018, 379, 1643–1656. [Google Scholar] [CrossRef]
- Young, N.S.; Barrett, A.J. The treatment of severe acquired aplastic anemia. Blood 1995, 85, 3367–3377. [Google Scholar] [CrossRef]
- Marsh, J.C. Hematopoietic growth factors in the pathogenesis and for the treatment of aplastic anemia. Semin. Hematol. 2000, 37, 81–90. [Google Scholar] [CrossRef]
- EWOG-SAA-2010, DRKS-ID: DRKS00000610. Available online: https://www.kinderblutkrankheiten.de/content/fachinformationen/studienportal/studien_und_register/ewog_saa_2010/index_ger.html (accessed on 26 September 2021).
- Führer, M.; Rampf, U.; Baumann, I.; Faldum, A.; Niemeyer, C.; Janka-Schaub, G.; Friedrich, W.; Ebell, W.; Borkhardt, A.; Bender-Goetze, C.; et al. Immunosuppressive therapy for aplastic anemia in children: A more severe disease predicts better survival. Blood 2005, 106, 2102–2104. [Google Scholar] [CrossRef] [Green Version]
- Avery, S.; Shi, W.; Lubin, M.; Gonzales, A.M.; Heller, G.; Castro-Malaspina, H.; Giralt, S.; Kernan, N.A.; Scaradavou, A.; Barker, J.N. Influence of infused cell dose and HLA match on engraftment after double-unit cord blood allografts. Blood 2011, 117, 3277–3285; quiz 3478. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marsh, J.C.W. Treatment of acquired aplastic anemia. Haematologica 2007, 92, 2–5. [Google Scholar] [CrossRef] [PubMed]
- Szpecht, D.; Gorczyńska, E.; Kałwak, K.; Owoc-Lempach, J.; Choma, M.; Styczyński, J.; Goździk, J.; Dłużniewska, A.; Wysocki, M.; Kowalczyk, J.; et al. Matched sibling versus matched unrelated allogeneic hematopoietic stem cell transplantation in children with severe acquired aplastic anemia: Experience of the Polish pediatric group for hematopoietic stem cell transplantation. Arch. Immunol. Ther. Exp. 2012, 60, 225–233. [Google Scholar] [CrossRef]
- Holtan, S.G.; DeFor, T.E.; Lazaryan, A.; Bejanyan, N.; Arora, M.; Brunstein, C.G.; Blazar, B.R.; MacMillan, M.; Weisdorf, D.J. Composite end point of graft-versus-host disease-free, relapse-free survival after allogeneic hematopoietic cell transplantation. Blood 2015, 125, 1333–1338. [Google Scholar] [CrossRef] [Green Version]
- Dufour, C.; Veys, P.; Carraro, E.; Bhatnagar, N.; Pillon, M.; Wynn, R.F.; Gibson, B.; Vora, A.J.; Steward, C.G.; Ewins, A.M.; et al. Similar outcome of upfront-unrelated and matched sibling stem cell transplantation in idiopathic paediatric aplastic anaemia. A study on behalf of the UK Paediatric BMT Working Party, Paediatric Diseases Working Party and Severe Aplastic Anaemia Working P. Br. J. Haematol. 2015, 171, 585–594. [Google Scholar] [CrossRef]
- Perez-Albuerne, E.D.; Eapen, M.; Klein, J.; Gross, T.J.; Lipton, J.M.; Baker, K.S.; Woolfrey, A.; Kamani, N. Outcome of unrelated donor stem cell transplantation for children with severe aplastic anemia. Br. J. Haematol. 2008, 141, 216–223. [Google Scholar] [CrossRef]
- Deeg, H.J.; O’Donnell, M.; Tolar, J.; Agarwal, R.; Harris, R.E.; Feig, S.A.; Territo, M.C.; Collins, R.H.; McSweeney, P.A.; Copelan, E.A.; et al. Optimization of conditioning for marrow transplantation from unrelated donors for patients with aplastic anemia after failure of immunosuppressive therapy. Blood 2006, 108, 1485–1491. [Google Scholar] [CrossRef] [Green Version]
- Bacigalupo, A.; Socie’, G.; Lanino, E.; Prete, A.; Locatelli, F.; Locasciulli, A.; Cesaro, S.; Shimoni, A.; Marsh, J.; Brune, M.; et al. Fludarabine, cyclophosphamide, antithymocyte globulin, with or without low dose total body irradiation, for alternative donor transplants, in acquired severe aplastic anemia: A retrospective study from the EBMT-SAA Working Party. Haematologica 2010, 95, 976–982. [Google Scholar] [CrossRef] [Green Version]
- Samarasinghe, S.; Steward, C.; Hiwarkar, P.; Saif, M.A.; Hough, R.; Webb, D.; Norton, A.; Lawson, S.; Qureshi, A.; Connor, P.; et al. Excellent outcome of matched unrelated donor transplantation in paediatric aplastic anaemia following failure with immunosuppressive therapy: A United Kingdom multicentre retrospective experience. Br. J. Haematol. 2012, 157, 339–346. [Google Scholar] [CrossRef]
- Solomon, S.R.; Sizemore, C.A.; Zhang, X.; Brown, S.; Holland, H.K.; Morris, L.E.; Solh, M.; Bashey, A. Impact of Donor Type on Outcome after Allogeneic Hematopoietic Cell Transplantation for Acute Leukemia. Biol. Blood Marrow Transplant. 2016, 22, 1816–1822. [Google Scholar] [CrossRef] [Green Version]
- Ayuk, F.; Beelen, D.W.; Bornhäuser, M.; Stelljes, M.; Zabelina, T.; Finke, J.; Kobbe, G.; Wolff, D.; Wagner, E.-M.; Christopeit, M.; et al. Relative Impact of HLA Matching and Non-HLA Donor Characteristics on Outcomes of Allogeneic Stem Cell Transplantation for Acute Myeloid Leukemia and Myelodysplastic Syndrome. Biol. Blood Marrow Transplant. 2018, 24, 2558–2567. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gahrton, G. Risk assessment in haematopoietic stem cell transplantation: Impact of donor–recipient sex combination in allogeneic transplantation. Best Pract. Res. Clin. Haematol. 2007, 20, 219–229. [Google Scholar] [CrossRef]
- Viollier, R.; Socie, G.; Tichelli, A.; Bacigalupo, A.; Korthof, E.T.; Marsh, J.; Cornish, J.; Ljungman, P.; Oneto, R.; Békássy, A.N.; et al. Recent improvement in outcome of unrelated donor transplantation for aplastic anemia. Bone Marrow Transplant. 2008, 41, 45–50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, D.; Yang, J.; Hu, X.; Chen, J.; Gao, L.; Cheng, H.; Tang, G.; Luo, Y.; Zhang, W.; Wang, J. Aplastic Anemia Preconditioned with Fludarabine, Cyclophosphamide, and Anti-Thymocyte Globulin. Ann. Transplant. 2019, 24, 461–471. [Google Scholar] [CrossRef] [PubMed]
- Bacigalupo, A.; for the Severe Aplastic Anemia Working Party of the European Group for Blood and Marrow Transplantation (SAA WP-EBMT); Locatelli, F.; Lanino, E.; Marsh, J.C.W.; Socie, G.; Maury, S.; Prete, A.; Locasciulli, A.; Cesaro, S.; et al. Fludarabine, cyclophosphamide and anti-thymocyte globulin for alternative donor transplants in acquired severe aplastic anemia: A report from the EBMT-SAA Working Party. Bone Marrow Transplant. 2005, 36, 947–950. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Resnick, I.B.; Aker, M.; Shapira, M.Y.; Tsirigotis, P.D.; Bitan, M.; Abdul-Hai, A.; Samuel, S.; Ackerstein, A.; Gesundheit, B.; Zilberman, I.; et al. Allogeneic stem cell transplantation for severe acquired aplastic anaemia using a fludarabine-based preparative regimen. Br. J. Haematol. 2006, 133, 649–654. [Google Scholar] [CrossRef]
- Kang, H.J.; Hong, K.T.; Lee, J.W.; Kim, H.; Park, K.D.; Shin, H.Y.; Lee, S.H.; Yoo, K.H.; Sung, K.W.; Koo, H.H.; et al. Improved Outcome of a Reduced Toxicity-Fludarabine, Cyclophosphamide, plus Antithymocyte Globulin Conditioning Regimen for Unrelated Donor Transplantation in Severe Aplastic Anemia: Comparison of 2 Multicenter Prospective Studies. Biol. Blood Marrow Transplant. 2016, 22, 1455–1459. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Green, D.M.; Liu, W.; Kutteh, W.H.; Ke, R.W.; Shelton, K.C.; A Sklar, C.; Chemaitilly, W.; Pui, C.-H.; Klosky, J.L.; Spunt, S.L.; et al. Cumulative alkylating agent exposure and semen parameters in adult survivors of childhood cancer: A report from the St Jude Lifetime Cohort Study. Lancet Oncol. 2014, 15, 1215–1223. [Google Scholar] [CrossRef] [Green Version]
- Levine, J.M.; Whitton, J.A.; Ginsberg, J.P.; Green, D.M.; Leisenring, W.; Stovall, M.; Robison, L.L.; Armstrong, G.T.; Sklar, C.A. Nonsurgical premature menopause and reproductive implications in survivors of childhood cancer: A report from the Childhood Cancer Survivor Study. Cancer 2018, 124, 1044–1052. [Google Scholar] [CrossRef]
- Iftikhar, R.; Chaudhry, Q.U.N.; Anwer, F.; Neupane, K.; Rafae, A.; Mahmood, S.K.; Ghafoor, T.; Shahbaz, N.; Khan, M.A.; Khattak, T.A.; et al. Allogeneic hematopoietic stem cell transplantation in aplastic anemia: Current indications and transplant strategies. Blood Rev. 2021, 47, 100772. [Google Scholar] [CrossRef] [PubMed]
- Markiewicz, M.; Drozd-Sokolowska, J.; Biecek, P.; Dzierzak-Mietla, M.; Boguradzki, P.; Staniak, M.; Piatkowska-Jakubas, B.; Piekarska, A.; Tormanowska, M.; Halaburda, K.; et al. Allogeneic hematopoietic stem cell transplantation for paroxysmal nocturnal hemoglobinuria: Multicenter analysis by Polish Adult Leukemia Group. Biol. Blood Marrow Transplant. 2020, 26, 1833–1839. [Google Scholar] [CrossRef] [PubMed]
- Drexler, B.; Zurbriggen, F.; Diesch, T.; Viollier, R.; Halter, J.P.; Heim, D.; Holbro, A.; Infanti, L.; Buser, A.; Gerull, S.; et al. Very long-term follow-up of aplastic anemia treated with immunosuppressive therapy or allogeneic hematopoietic cell transplantation. Ann. Hematol. 2020, 99, 2529–2538. [Google Scholar] [CrossRef]
- Admiraal, R.; van Kesteren, C.; Jol-van der Zijde, C.M.; van Tol, M.J.D.; Bartelink, I.H.; Bredius, R.G.M.; Boelens, J.J.; Knibbe, C.A.J. Population Pharmacokinetic Modeling of Thymoglobulin® in Children Receiving Allogeneic-Hematopoietic Cell Transplantation: Towards Improved Survival Through Individualized Dosing. Clin. Pharmacokinet. 2015, 54, 435–446. [Google Scholar] [CrossRef]
- El Amrani, M.; Admiraal, R.; Willaert, L.; Raaij, L.J.C.E.-V.; Lacna, A.M.; Hack, C.E.; Huitema, A.D.R.; Nierkens, S.; Van Maarseveen, E.M. Quantification of T Cell Binding Polyclonal Rabbit Anti-thymocyte Globulin in Human Plasma with Liquid Chromatography Tandem-Mass Spectrometry. AAPS J. 2020, 22, 43. [Google Scholar] [CrossRef]
- Admiraal, R.; van Kesteren, C.; Jol-van der Zijde, C.M.; Lankester, A.C.; Bierings, M.B.; Egberts, T.; van Tol, M.J.D.; Knibbe, C.A.J.; Bredius, R.G.M.; Boelens, J.J. Association between anti-thymocyte globulin exposure and CD4+ immune reconstitution in paediatric haemopoietic cell transplantation: A multicentre, retrospective pharmacodynamic cohort analysis. Lancet Haematol. 2015, 2, e194–e203. [Google Scholar] [CrossRef]
- Bejanyan, N.; Kim, S.; Hebert, K.M.; Kekre, N.; Abdel-Azim, H.; Ahmed, I.; Aljurf, M.; Badawy, S.M.; Beitinjaneh, A.; Boelens, J.J.; et al. Choice of conditioning regimens for bone marrow transplantation in severe aplastic anemia. Blood Adv. 2019, 3, 3123–3131. [Google Scholar] [CrossRef] [Green Version]
- Bacigalupo, A.; Socié, G.; Hamladji, R.M.; Aljurf, M.; Maschan, A.; Kyrcz-Krzemien, S.; Cybicka, A.; Sengelov, H.; Unal, A.; Beelen, D.; et al. Current outcome of HLA identical sibling versus unrelated donor transplants in severe aplastic anemia: An EBMT analysis. Haematologica 2015, 100, 696–702. [Google Scholar] [CrossRef]
- Champlin, R.E.; Perez, W.S.; Passweg, J.R.; Klein, J.P.; Camitta, B.M.; Gluckman, E.; Bredeson, C.N.; Eapen, M.; Horowitz, M.M. Bone marrow transplantation for severe aplastic anemia: A randomized controlled study of conditioning regimens. Blood 2007, 109, 4582–4585. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marsh, J.C.; Gupta, V.; Lim, Z.; Ho, A.Y.; Ireland, R.M.; Hayden, J.; Potter, V.; Koh, M.B.; Islam, M.S.; Russell, N.; et al. Alemtuzumab with fludarabine and cyclophosphamide reduces chronic graft-versus-host disease after allogeneic stem cell transplantation for acquired aplastic anemia. Blood 2011, 118, 2351–2357. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Viale, M.; Merli, A.; Bacigalupo, A. Analysis at the clonal level of T-cell phenotype and functions in severe aplastic anemia patients. Blood 1991, 78, 1268–1274. Available online: http://www.ncbi.nlm.nih.gov/pubmed/1715221 (accessed on 26 September 2021).
- De Vries, A.C.H.; Langerak, A.W.; Verhaaf, B.; Niemeyer, C.M.; Stary, J.; Schmiegelow, K.; Van Wering, E.R.; Zwaan, C.M.; Beishuizen, A.; Pieters, R.; et al. T-cell receptor Vβ CDR3 oligoclonality frequently occurs in childhood refractory cytopenia (MDS-RC) and severe aplastic anemia. Leukemia 2008, 22, 1170–1174. [Google Scholar] [CrossRef] [Green Version]
- Shah, R.M.; Truong, T.H.; Leaker, M.T.; Wright, N.A.; Le, D.; Steele, M.; Bruce, A.A.; Desai, S.; Guilcher, G.M.; Lewis, V. Fludarabine, Campath, and Low-Dose Cyclophosphamide (FCClow) with or without TBI Conditioning Results in Excellent Transplant Outcomes in Children with Severe Aplastic Anemia. Biol. Blood Marrow Transplant. 2020, 26, 1900–1905. [Google Scholar] [CrossRef]
- Schönberger, S.; Meisel, R.; Adams, O.; Pufal, Y.; Laws, H.; Enczmann, J.; Dilloo, D. Prospective, Comprehensive, and Effective Viral Monitoring in Children Undergoing Allogeneic Hematopoietic Stem Cell Transplantation. Biol. Blood Marrow Transplant. 2010, 16, 1428–1435. [Google Scholar] [CrossRef] [Green Version]
- Im, S.H.; Kim, B.R.; Park, S.M.; Yoon, B.A.; Hwang, T.J.; Baek, H.J.; Kook, H. Better Failure-Free Survival and Graft-versus-Host Disease-Free/Failure Free Survival with Fludarabine-Based Conditioning in Stem Cell Transplantation for Aplastic Anemia in Children. J. Korean Med. Sci. 2020, 35, e46. [Google Scholar] [CrossRef]
- Chaudhry, Q.U.N.; Iftikhar, R.; Satti, T.M.; Mahmood, S.K.; Ghafoor, T.; Shamshad, G.U.; Farhan, M.; Shahbaz, N.; Khan, M.A.; Khattak, T.A.; et al. Outcome of Fludarabine-Based Conditioning in High-Risk Aplastic Anemia Patients Undergoing Matched Related Donor Transplantation: A Single-Center Study from Pakistan. Biol. Blood Marrow Transplant. 2019, 25, 2375–2382. [Google Scholar] [CrossRef]
- Kang, H.J.; Shin, H.Y.; Park, J.E.; Chung, N.G.; Cho, B.; Kim, H.K.; Kim, S.Y.; Lee, Y.H.; Lim, Y.T.; Yoo, K.H.; et al. Successful Engraftment with Fludarabine, Cyclophosphamide, and Thymoglobulin Conditioning Regimen in Unrelated Transplantation for Severe Aplastic Anemia: A Phase II Prospective Multicenter Study. Biol. Blood Marrow Transplant. 2010, 16, 1582–1588. [Google Scholar] [CrossRef] [Green Version]
- Giraud, G.; Bogdanovic, G.; Priftakis, P.; Remberger, M.; Svahn, B.-M.; Barkholt, L.; Ringden, O.; Winiarski, J.; Ljungman, P.; Dalianis, T. The incidence of hemorrhagic cystitis and BK-viruria in allogeneic hematopoietic stem cell recipients according to intensity of the conditioning regimen. Haematologica 2006, 91, 401–404. Available online: http://www.ncbi.nlm.nih.gov/pubmed/16531266 (accessed on 26 September 2021).
- Salamonowicz-Bodzioch, M.; Frączkiewicz, J.; Czyżewski, K.; Zając-Spychała, O.; Gorczyńska, E.; Panasiuk, A.; Ussowicz, M.; Kałwak, K.; Szmit, Z.; Wróbel, G.; et al. Prospective analysis of BKV hemorrhagic cystitis in children and adolescents undergoing hematopoietic cell transplantation. Ann. Hematol. 2021, 100, 1283–1293. [Google Scholar] [CrossRef]
- Riachy, E.; Krauel, L.; Rich, B.S.; McEvoy, M.P.; Honeyman, J.N.; Boulad, F.; Wolden, S.L.; Herr, H.W.; La Quaglia, M.P. Risk Factors and Predictors of Severity Score and Complications of Pediatric Hemorrhagic Cystitis. J. Urol. 2014, 191, 186–192. [Google Scholar] [CrossRef]
- Mori, Y.; Miyamoto, T.; Kato, K.; Kamezaki, K.; Kuriyama, T.; Oku, S.; Takenaka, K.; Iwasaki, H.; Harada, N.; Shiratsuchi, M.; et al. Different Risk Factors Related to Adenovirus- or BK Virus-Associated Hemorrhagic Cystitis following Allogeneic Stem Cell Transplantation. Biol. Blood Marrow Transplant. 2012, 18, 458–465. [Google Scholar] [CrossRef] [Green Version]
- Matthes-Martin, S.; Feuchtinger, T.; Shaw, P.J.; Engelhard, D.; Hirsch, H.H.; Cordonnier, C.; Ljungman, P.; Fourth European Conference on Infections in Leukemia. European guidelines for diagnosis and treatment of adenovirus infection in leukemia and stem cell transplantation: Summary of ECIL-4 (2011). Transpl. Infect. Dis. 2012, 14, 555–563. [Google Scholar] [CrossRef] [PubMed]
- Feghoul, L.; Chevret, S.; Cuinet, A.; Dalle, J.-H.; Ouachée, M.; Yacouben, K.; Fahd, M.; Khourouj, V.G.-E.; Roupret-Serzec, J.; Sterkers, G.; et al. Adenovirus infection and disease in paediatric haematopoietic stem cell transplant patients: Clues for antiviral pre-emptive treatment. Clin. Microbiol. Infect. 2015, 21, 701–709. [Google Scholar] [CrossRef] [Green Version]
- Mynarek, M.; Ganzenmueller, T.; Mueller-Heine, A.; Mielke, C.; Gonnermann, A.; Beier, R.; Sauer, M.; Eiz-Vesper, B.; Kohstall, U.; Sykora, K.-W.; et al. Patient, Virus, and Treatment-Related Risk Factors in Pediatric Adenovirus Infection after Stem Cell Transplantation: Results of a Routine Monitoring Program. Biol. Blood Marrow Transplant. 2014, 20, 250–256. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schrezenmeier, H.; Passweg, J.R.; Marsh, J.C.W.; Bacigalupo, A.; Bredeson, C.N.; Bullorsky, E.; Camitta, B.M.; Champlin, R.E.; Gale, R.P.; Fuhrer, M.; et al. Worse outcome and more chronic GVHD with peripheral blood progenitor cells than bone marrow in HLA-matched sibling donor transplants for young patients with severe acquired aplastic anemia. Blood 2007, 110, 1397–1400. [Google Scholar] [CrossRef] [PubMed]
- Eapen, M.; Le Rademacher, J.; Antin, J.H.; Champlin, R.E.; Carreras, J.; Fay, J.; Passweg, J.R.; Tolar, J.; Horowitz, M.M.; Marsh, J.C.W.; et al. Effect of stem cell source on outcomes after unrelated donor transplantation in severe aplastic anemia. Blood 2011, 118, 2618–2621. [Google Scholar] [CrossRef] [PubMed]
- Platzbecker, U.; Binder, M.; Schmid, C.; Rutt, C.; Ehninger, G.; Bornhäuser, M. Second donation of hematopoietic stem cells from unrelated donors for patients with relapse or graft failure after allogeneic transplantation. Haematologica 2008, 93, 1276–1278. [Google Scholar] [CrossRef] [PubMed]
- Horan, J.T.; Carreras, J.; Tarima, S.; Camitta, B.M.; Gale, R.P.; Hale, G.A.; Hinterberger, W.; Marsh, J.; Passweg, J.R.; Walters, M.C.; et al. Risk Factors Affecting Outcome of Second HLA-Matched Sibling Donor Transplantations for Graft Failure in Severe Acquired Aplastic Anemia. Biol. Blood Marrow Transplant. 2009, 15, 626–631. [Google Scholar] [CrossRef] [PubMed]
- Cesaro, S.; De Latour, R.P.; Tridello, G.; Pillon, M.; Carlson, K.; Fagioli, F.; Jouet, J.-P.; Koh, M.B.C.; Panizzolo, I.S.; Kyrcz-Krzemien, S.; et al. Second allogeneic stem cell transplant for aplastic anaemia: A retrospective study by the severe aplastic anaemia working party of the European society for blood and marrow transplantation. Br. J. Haematol. 2015, 171, 606–614. [Google Scholar] [CrossRef] [PubMed]
- Simonin, M.; Pdwp-Ebmt, O.B.O.; Dalissier, A.; Labopin, M.; Willasch, A.; Zecca, M.; Mouhab, A.; Chybicka, A.; Balduzzi, A.; Volin, L.; et al. More chronic GvHD and non-relapse mortality after peripheral blood stem cell compared with bone marrow in hematopoietic transplantation for paediatric acute lymphoblastic leukemia: A retrospective study on behalf of the EBMT Paediatric Diseases Working Pa. Bone Marrow Transplant. 2017, 52, 1071–1073. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferrara, J.L.; Levine, J.E.; Reddy, P.; Holler, E. Graft-versus-host disease. Lancet 2009, 373, 1550–1561. [Google Scholar] [CrossRef]
- Groarke, E.M.; Patel, B.A.; Gutierrez-Rodrigues, F.; Rios, O.; Lotter, J.; Baldoni, D.; Pierre, A.S.; Shalhoub, R.; Wu, C.O.; Townsley, D.M.; et al. Eltrombopag added to immunosuppression for children with treatment-naïve severe aplastic anaemia. Br. J. Haematol. 2021, 192, 605–614. [Google Scholar] [CrossRef]
- Townsley, D.M.; Scheinberg, P.; Winkler, T.; Desmond, R.; Dumitriu, B.; Rios, O.; Weinstein, B.; Valdez, J.; Lotter, J.; Feng, X.; et al. Eltrombopag Added to Standard Immunosuppression for Aplastic Anemia. N. Engl. J. Med. 2017, 376, 1540–1550. [Google Scholar] [CrossRef] [Green Version]



| Category | Value | |
|---|---|---|
| Number of Patients | 56 | |
| Sex | male | 33 |
| female | 23 | |
| Age at HCT in years | median | 9.4 |
| range | 0.8–17.8 | |
| Time from diagnosis to HCT in months | median | 4.57 |
| range | 1.23–66.6 | |
| Previous IS protocol | yes | 14 |
| no | 32 | |
| PNH clone | detectable | 8 |
| undetectable | 48 | |
| Donor | matched sibling donor | 18 |
| matched unrelated donor | 38 | |
| Degree of HLA match | matched sibling donor, 6/6 | 15 |
| matched sibling donor, 10/10 | 2 | |
| matched unrelated donor, 10/10 | 33 | |
| matched sibling/unrelated donor, 9/10 | 6 | |
| Stem cell source | bone marrow | 17 |
| peripheral blood stem cells | 39 | |
| Bone marrow CD34 + cells/kg in millions | median (range) | 3.54 (0.34–12.14) |
| Peripheral blood CD34 + cells/kg in millions | median (range) | 9.31 (2.42–29.78) |
| Antithymocyte globulin | Grafalon | 15 |
| Thymoglobulin | 41 | |
| Posttransplantation methotrexate | yes | 54 |
| no | 2 | |
| Posttransplantation follow-up in months | median | 44 |
| range | 0–144 | |
| Time to neutrophil count > 500/µL in days | median | 14 |
| range | 10–22 | |
| Time to platelet count > 20,000/µL in days | median | 16 |
| range | 5–212 | |
| Acute graft versus host disease | any grade | 22 |
| grade III-IV | 6 | |
| Chronic graft versus host disease | any grade | 7 |
| moderate–severe grade | 5 | |
| Category | Number of Patients | 5-Year OS | Log-Rank p | 2-Year GFS | Log-Rank p | |
|---|---|---|---|---|---|---|
| Sex | male | 33 | 93.9 | ns | 84.5 | ns |
| female | 23 | 91.3 | 63.4 | |||
| Previous IS protocol | yes | 14 | 83.3 | p = 0.017 | 66.4 | ns |
| no | 32 | 100 | 83.5 | |||
| Time from diagnosis to HCT | <3 months | 14 | 100 | ns | 91.7 | ns |
| >3 months | 41 | 90.2 | 70.4 | |||
| PNH clone | detectable | 8 | 100 | ns | 100 | ns |
| undetectable | 48 | 91.7 | 72 | |||
| Donor | matched sibling donor | 18 | 94.1 | ns | 87.8 | ns |
| matched unrelated donor | 38 | 92.3 | 71 | |||
| Recipient–donor gender | Male donor–male recipient | 24 | 100 | p = 0.04 | 86.8 | ns |
| Female donor–female recipient | 11 | 81.8 | 53 | |||
| Male donor–female recipient | 9 | 77.8 | 77.8 | |||
| Female donor–male recipient | 12 | 100 | 72.9 | |||
| Donor gender | Male donor | 36 | 100 | p = 0.005 | 82.5 | ns |
| Female donor | 20 | 80 | 64.6 | |||
| CMV IgG status | Donor positive–recipient positive | 28 | 90.3 | ns | 73.1 | ns |
| Donor positive–recipient negative | 4 | 100 | 100 | |||
| Donor negative–recipient positive | 15 | 93.8 | 75 | |||
| Donor negative–recipient negative | 4 | 100 | 75 | |||
| Stem cell source | bone marrow | 17 | 88.2 | ns | 69.7 | ns |
| peripheral blood stem cells | 39 | 94.9 | 79 | |||
| Conditioning | Flu 160 mg/m2 + Cy 200 mg/kg BW | 12 | 85 | p = 0.0063 | 58.3 | ns |
| Flu 120 mg/m2 + Cy 100 mg/kg BW | 44 | 97.7 | 81 | |||
| Antithymocyte globulin | Grafalon | 15 | 100 | ns | 100 | ns |
| Thymoglobulin | 41 | 90.2 | 70.7 | |||
| Acute graft versus host disease | no | 34 | 91.2 | ns | n/a | |
| grade I-II | 16 | 100 | ||||
| grade III-IV | 6 | 83.3 | ||||
| Chronic graft versus host disease | no | 49 | 97.8 | ns | n/a | |
| any grade | 7 | 100 | ||||
| Posttransplantation ADV replication | yes | 7 | 100 | ns | 76.9 | ns |
| no | 49 | 91.8 | 71.4 | |||
| Posttransplantation BKV replication | yes | 16 | 100 | ns | 87.5 | ns |
| no | 40 | 90 | 71.9 | |||
| Posttransplantation CMV replication | yes | 16 | 100 | ns | 93.8 | ns |
| no | 40 | 90 | 69.1 | |||
| Posttransplantation EBV replication | yes | 12 | 91.7 | ns | 74.1 | ns |
| no | 44 | 93.2 | 76.7 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salamonowicz-Bodzioch, M.; Rosa, M.; Frączkiewicz, J.; Gorczyńska, E.; Gul, K.; Janeczko-Czarnecka, M.; Jarmoliński, T.; Kałwak, K.; Mielcarek-Siedziuk, M.; Olejnik, I.; et al. Fludarabine–Cyclophosphamide-Based Conditioning with Antithymocyte Globulin Serotherapy Is Associated with Durable Engraftment and Manageable Infections in Children with Severe Aplastic Anemia. J. Clin. Med. 2021, 10, 4416. https://doi.org/10.3390/jcm10194416
Salamonowicz-Bodzioch M, Rosa M, Frączkiewicz J, Gorczyńska E, Gul K, Janeczko-Czarnecka M, Jarmoliński T, Kałwak K, Mielcarek-Siedziuk M, Olejnik I, et al. Fludarabine–Cyclophosphamide-Based Conditioning with Antithymocyte Globulin Serotherapy Is Associated with Durable Engraftment and Manageable Infections in Children with Severe Aplastic Anemia. Journal of Clinical Medicine. 2021; 10(19):4416. https://doi.org/10.3390/jcm10194416
Chicago/Turabian StyleSalamonowicz-Bodzioch, Małgorzata, Monika Rosa, Jowita Frączkiewicz, Ewa Gorczyńska, Katarzyna Gul, Małgorzata Janeczko-Czarnecka, Tomasz Jarmoliński, Krzysztof Kałwak, Monika Mielcarek-Siedziuk, Igor Olejnik, and et al. 2021. "Fludarabine–Cyclophosphamide-Based Conditioning with Antithymocyte Globulin Serotherapy Is Associated with Durable Engraftment and Manageable Infections in Children with Severe Aplastic Anemia" Journal of Clinical Medicine 10, no. 19: 4416. https://doi.org/10.3390/jcm10194416







