Prognostic Value of Resistance Proteins in Plasma Cells from Multiple Myeloma Patients Treated with Bortezomib-Based Regimens
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Collection of MM Cells
2.3. Determination of Human Protein Level in Plasma Cells
2.4. Statistical Analysis
3. Results
3.1. Characteristics of the Patients Included in the Analysis
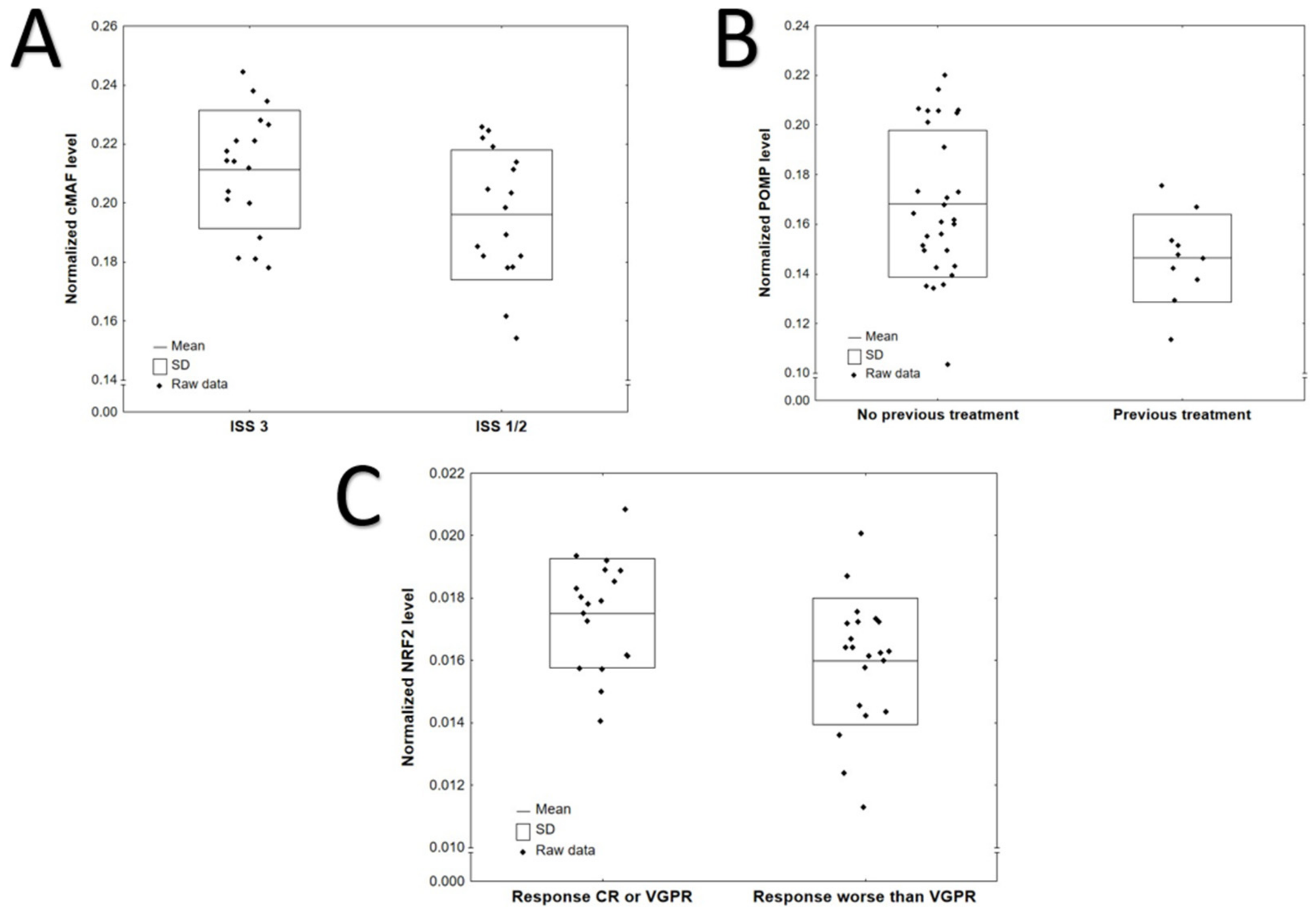
3.2. Protein Levels According to Clinical and Laboratory Characteristics
3.3. Influence of ASCT and ISS Protein Levels on Overall Survival and Progression-Free Survival
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ASCT | autologous stem cell transplantation |
| ACTB | Beta–actin protein |
| BM | bone marrow |
| ELISA | enzyme-linked immunosorbent assay |
| LCD | light chain disease |
| IMWG | International Myeloma Working Group |
| MAF | musculoaponeurotic fibrosarcoma |
| MM | Multiple myeloma |
| NRF2 | Nuclear factor erythroid 2-related factor 2 |
| NF-κB | nuclear factor kappa B |
| OS | overall survival |
| PC | plasma cells |
| PFS | progression-free survival |
| POMP | proteasome maturation protein |
| PI | Proteasome inhibitors |
| PSMB5 | proteasome subunit β5 |
| UPR | unfolded protein response |
| VCD | bortezomib, cyclophosphamide and dexamethasone; |
| VD | bortezomib and dexamethasone; |
| VMP | bortezomib, melphalan and prednisone; |
| VTD | bortezomib, thalidomide and dexamethasone |
| XBP1 | X–box binding protein 1 |
References
- International Myeloma Working Group. Criteria for the classification of monoclonal gammopathies, multiple myeloma and related disorders: A report of the International Myeloma Working Group. Br. J. Haematol. 2003, 121, 749–757. [Google Scholar] [CrossRef] [Green Version]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef]
- Gerecke, C.; Fuhrmann, S.; Strifler, S.; Schmidt-Hieber, M.; Einsele, H.; Knop, S. The diagnosis and treatment of multiple myeloma. Dtsch. Ärzteblatt Int. 2016, 113, 470–476. [Google Scholar] [CrossRef] [Green Version]
- Robak, P.; Robak, T. Novel drugs for multiple myeloma. Top. Anti-Cancer Res. 2019, 8, 1–43. [Google Scholar]
- Gandolfi, S.; Laubach, J.P.; Hideshima, T.; Chauhan, D.; Anderson, K.C.; Richardson, P.G. The proteasome and proteasome inhibitors in multiple myeloma. Cancer Metastasis Rev. 2017, 36, 561–584. [Google Scholar] [CrossRef]
- Davis, L.N.; Sherbenou, D.W. Emerging therapeutic strategies to overcome drug resistance in multiple myeloma. Cancers 2021, 13, 1686. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Frezza, M.; Schmitt, S.; Kanwar, J.; Dou, Q.P. Bortezomib as the first proteasome inhibitor anticancer drug: Current status and future perspectives. Curr. Cancer Drug Targets 2011, 11, 239–253. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robak, P.; Robak, T. Bortezomib for the treatment of hematologic malignancies: 15 years later. Drugs R&D 2019, 19, 73–92. [Google Scholar]
- Okazuka, K.; Ishida, T. Proteasome inhibitors for multiple myeloma. Jpn. J. Clin. Oncol. 2018, 48, 785–793. [Google Scholar] [CrossRef] [Green Version]
- San Miguel, J.F.; Schlag, R.; Khuageva, N.K.; Dimopoulos, M.A.; Shpilberg, O.; Kropff, M.; Spicka, I.; Petrucci, M.T.; Palumbo, A.; Samoilova, O.S.; et al. Bortezomib plus melphalan and prednisone for initial treatment of multiple myeloma. N. Engl. J. Med. 2008, 359, 906–917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robak, P.; Drozdz, I.; Szemraj, J.; Robak, T. Drug resistance in multiple myeloma. Cancer Treat. Rev. 2018, 70, 199–208. [Google Scholar] [CrossRef]
- Nikesitch, N.; Ling, S.C. Molecular mechanisms in multiple myeloma drug resistance. J. Clin. Pathol. 2016, 69, 97–101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, N.; Sharma, A.; Sharma, A. Emerging biomarkers in multiple myeloma: A review. Clin. Chim. Acta 2020, 503, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Dytfeld, D.; Rosebeck, S.; Kandarpa, M.; Mayampurath, A.; Mellacheruvu, D.; Alonge, M.M.; Ngoka, L.; Jasielec, J.; Richardson, P.G.; Volchenboum, S.; et al. Proteomic profiling of naïve multiple myeloma patient plasma cells identifies pathways associated with favourable response to bortezomib-based treatment regimens. Br. J. Haematol. 2015, 170, 66–79. [Google Scholar] [CrossRef] [PubMed]
- Dytfeld, D.; Luczak, M.; Wrobel, T.; Usnarska-Zubkiewicz, L.; Brzezniakiewicz, K.; Jamroziak, K.; Giannopoulos, K.; Przybylowicz-Chalecka, A.; Ratajczak, B.; Czerwinska-Rybak, J.; et al. Comparative proteomic profiling of refractory/relapsed multiple myeloma reveals biomarkers involved in resistance to bortezomib-based therapy. Oncotarget 2016, 7, 56726–56736. [Google Scholar] [CrossRef] [Green Version]
- Rajpal, R.; Dowling, P.; Meiller, J.; Clarke, C.; Murphy, W.G.; O’Connor, R.; Kell, M.; Mitsiades, C.; Richardson, P.; Anderson, K.C.; et al. A novel panel of protein biomarkers for predicting response to thalidomide-based therapy in newly diagnosed multiple myeloma patients. Proteomics 2011, 11, 1391–1402. [Google Scholar] [CrossRef]
- Li, B.; Fu, J.; Chen, P.; Ge, X.; Li, Y.; Kuiatse, I.; Wang, H.; Wang, H.; Zhang, X.; Orlowski, R.Z. The Nuclear factor (Erythroid-derived 2)-like 2 and proteasome maturation protein axis mediate bortezomib resistance in multiple myeloma. J. Biol. Chem. 2015, 290, 29854–29868. [Google Scholar] [CrossRef] [Green Version]
- Ri, M.; Iida, S.; Nakashima, T.; Miyazaki, H.; Mori, F.; Ito, A.; Inagaki, A.; Kusumoto, S.; Ishida, T.; Komatsu, H.; et al. Bortezomib-resistant myeloma cell lines: A role for mutated PSMB5 in preventing the accumulation of unfolded proteins and fatal ER stress. Leukemia 2010, 24, 1506–1512. [Google Scholar] [CrossRef]
- Oerlemans, R.; Franke, N.E.; Assaraf, Y.G.; Cloos, J.; van Zantwijk, I.; Berkers, C.R.; Scheffer, G.L.; Debipersad, K.; Vojtekova, K.; Lemos, C.; et al. Molecular basis of bortezomib resistance: Proteasome subunit beta5 (PSMB5) gene mutation and overexpression of PSMB5 protein. Blood 2008, 112, 2489–2499. [Google Scholar] [CrossRef] [Green Version]
- Balsas, P.; Galán-Malo, P.; Marzo, I.; Naval, J. Bortezomib resistance in a myeloma cell line is associated to PSMB5 overexpression and polyploidy. Leuk. Res. 2012, 36, 212–218. [Google Scholar] [CrossRef]
- Shi, C.X.; Zhu, Y.; Bruins, L.A.; Bonolo de Campos, C.; Stewart, W.; Braggio, E.; Stewart, A.K. proteasome subunits differentially control myeloma cell viability and proteasome inhibitor sensitivity. Mol. Cancer Res. 2020, 18, 1453–1464. [Google Scholar] [CrossRef]
- Sun, Y.; Abdul Aziz, A.; Bowles, K.; Rushworth, S. High NRF2 expression controls endoplasmic reticulum stress induced apoptosis in multiple myeloma. Cancer Lett. 2018, 412, 37–45. [Google Scholar] [CrossRef] [Green Version]
- Moi, P.; Chan, K.; Asunis, I.; Cao, A.; Kan, Y.W. Isolation of NF-E2-related factor 2 (Nrf2), a NF-E2-like basic leucine zipper transcriptional activator that binds to the tandem NF-E2/AP1 repeat of the β-globin locus control region. Proc. Natl. Acad. Sci. USA 1994, 91, 9926–9930. [Google Scholar] [CrossRef] [Green Version]
- Borjan, B.; Kern, J.; Steiner, N.; Gunsilius, E.; Wolf, D.; Untergasser, G. Spliced XBP1 levels determine sensitivity of multiple myeloma cells to proteasome inhibitor bortezomib independent of the unfolded protein response mediator GRP78. Front. Oncol. 2020, 9, 1530. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Chen, J.; Hua, X.; Sun, Y.; Cui, R.; Sha, J.; Zhu, X. The emerging role of XBP1 in cancer. Biomed. Pharmacother. 2020, 127, 110069. [Google Scholar] [CrossRef]
- Eychene, A.; Rocques, N.; Pouponnot, C. A new MAFia in cancer. Nat. Rev. Cancer 2008, 8, 683–693. [Google Scholar] [CrossRef]
- Chen, X.H.; Xu, Y.J.; Wang, X.G.; Lin, P.; Cao, B.Y.; Zeng, Y.Y.; Wang, Q.; Zhang, Z.B.; Mao, X.L.; Zhang, T. Mebendazole elicits potent antimyeloma activity by inhibiting the USP5/c-Maf axis. Acta Pharmacol. Sin. 2019, 40, 1568–1577. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Juan, J.; Zhang, Z.; Du, Y.; Xu, Y.; Tong, J.; Cao, B.; Moran, M.F.; Zeng, Y.; Mao, X. Inhibition of the deubiquitinase USP5 leads to c-Maf protein degradation and myeloma cell apoptosis. Cell Death Dis. 2017, 8, e3058. [Google Scholar] [CrossRef] [Green Version]
- Kannan, M.B.; Solovieva, V.; Blank, V. The small MAF transcription factors MAFF, MAFG and MAFK: Current knowledge and perspectives. Biochim. Biophys. Acta 2012, 1823, 1841–1846. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiang, Y.W.; Ye, S.; Huang, Y.; Chen, Y.; Van Rhee, F.; Epstein, J.; Walker, B.A.; Morgan, G.J.; Davies, F.E. MAFb protein confers intrinsic resistance to proteasome inhibitors in multiple myeloma. BMC Cancer 2018, 18, 724. [Google Scholar] [CrossRef] [Green Version]
- Katsuoka, F.; Yamamoto, M. Small Maf proteins (MafF, MafG, MafK): History, structure and function. Gene 2016, 586, 197–205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robak, P.; Węgłowska, E.; Dróżdż, I.; Mikulski, D.; Jarych, D.; Ferlińska, M.; Wawrzyniak, E.; Misiewicz, M.; Smolewski, P.; Fendler, W.; et al. Cytokine and chemokine profile in patients with multiple myeloma treated with bortezomib. Mediat. Inflamm. 2020, 2020, 1835836. [Google Scholar] [CrossRef]
- Robak, P.; Dróżdż, I.; Jarych, D.; Mikulski, D.; Węgłowska, E.; Siemieniuk-Ryś, M.; Misiewicz, M.; Stawiski, K.; Fendler, W.; Szemraj, J.; et al. The value of serum microRNA expression signature in predicting refractoriness to bortezomib-based therapy in multiple myeloma patients. Cancers 2020, 12, 2569. [Google Scholar] [CrossRef] [PubMed]
- Durie, B.G.; Harousseau, J.L.; Miguel, J.S.; Bladé, J.; Barlogie, B.; Anderson, K.; Gertz, M.; Dimopoulos, M.; Westin, J.; Sonneveld, P.; et al. International uniform response criteria for multiple myeloma. Leukemia 2006, 20, 1467–1473. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kyle, R.A.; Rajkumar, S.V. Criteria for diagnosis, staging, risk stratification and response assessment of multiple myeloma. Leukemia 2009, 23, 3–9. [Google Scholar] [CrossRef]
- Anderson, K.C.; Kyle, R.A.; Rajkumar, S.V.; Stewart, A.K.; Weber, D.; Richardson, P. ASH/FDA Panel on Clinical Endpoints in Multiple Myeloma. Leukemia 2008, 22, 231–239. [Google Scholar] [CrossRef] [Green Version]
- Pellett, S.; Tepp, W.H.; Johnson, E.A.; Sesardic, D. Assessment of ELISA as endpoint in neuronal cell-based assay for BoNT detection using hiPSC derived neurons. J. Pharmacol. Toxicol. Methods 2017, 88 Pt 1, 1–6. [Google Scholar] [CrossRef]
- Yadirgi, G.; Stickings, P.; Rajagopal, S.; Liu, Y.; Sesardic, D. Immuno-detection of cleaved SNAP-25 from differentiated mouse embryonic stem cells provides a sensitive assay for determination of botulinum A toxin and antitoxin potency. J. Immunol. Methods 2017, 451, 90–99. [Google Scholar] [CrossRef]
- Budczies, J.; Klauschen, F.; Sinn, B.V.; Győrffy, B.; Schmitt, W.D.; Darb-Esfahani, S.; Denkert, C. Cutoff Finder: A comprehensive and straightforward Web application enabling rapid biomarker cutoff optimization. PLoS ONE 2012, 7, e51862. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robak, P.; Jarych, D.; Mikulski, D.; Dróżdż, I.; Węgłowska, E.; Kotkowska, A.; Misiewicz, M.; Smolewski, P.; Stawiski, K.; Fendler, W.; et al. The prognostic value of whole-blood PSMB5, CXCR4, POMP, and RPL5 mRNA expression in patients with multiple myeloma treated with bortezomib. Cancers 2021, 13, 951. [Google Scholar] [CrossRef]
- Yang, Y.; Chen, Y.; Saha, M.N.; Chen, J.; Evans, K.; Qiu, L.; Reece, D.; Chen, G.A.; Chang, H. Targeting phospho-MARCKS overcomes drug resistance and induces antitumor activity in preclinical models of multiple myeloma. Leukemia 2015, 29, 715–726. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.; Yang, Y.; Wang, J.; Zhang, L.; Wang, F.; He, A. Variability of serum novel serum peptide biomarkers correlates with the disease states of multiple myeloma. Clin. Proteomics 2019, 16, 17. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Protein | Characteristics | Significance in MM | Reference |
|---|---|---|---|
| POMP | A short-lived maturation factor essential for 20S proteasome subunit biogenesis. | POMP over-expression contributes to proteasome inhibitor resistance, while suppression enhances bortezomib and carfilzomib activity. | [12,17] |
| PSMB5 | A component of the 20S core proteasome complex involved in the proteolytic degradation of most intracellular proteins. | Overexpression observed in bortezomib-resistant cell lines, PSMB5 contributes to bortezomib resistance in MM patients. | [18,19,20,21] |
| NRF2 | A transcription activator that binds to antioxidant response elements in the promoter regions of target genes. | A key regulator of MM survival in treatment naive and PI-treated cells, PI increases expression of NRF2 in MM cells. | [22,23] |
| XBP1 | A transcription factor found during endoplasmic reticulum stress; a regulator of the UPR; needed for differentiation of B cells into PCs. | XBP1 levels correlate with bortezomib resistance in MM; XBP1 levels are low in bortezomib-refractory MM patients | [24,25] |
| cMAF | A bZIP zipper transcription factors, belonging to the AP-1 family. | Overexpressed in MM, enhancing tumor-stroma interactions. | [26,27,28,29] |
| MAFB | bZIP transcription factor that plays an important role in the regulation of lineage-specific hematopoiesis. | High expression is associated with resistance to proteasome inhibitors, frequent event in the progression of MM. | [26,27,28,30,31] |
| Characteristics | Overall (N = 39) |
|---|---|
| Sex | M: 23 (59.0) |
| F: 16 (41.0) | |
| Age mean ± SD | 66.8 ± 8.9 |
| (range) | (39–81) |
| ISS at diagnosis | I: 12 (30.8) |
| II: 7 (17.9) | |
| III: 17 (43.6) | |
| Data missing: 3 (7.7) | |
| Paraprotein | |
| IgG | 23 (59.0) |
| LCD | 8 (20.5) |
| IgA | 8 (20.5) |
| HB < 10 g/dL at diagnosis | 14 (35.9) |
| Creatinine > 2 mg/dL at diagnosis | 4 (10.3) |
| Calcium > 2.5 mmol/L at diagnosis | 11 (28.2) |
| Bone disease | 20 (51.3) |
| CRP > 5 mg/L | 16 (41) |
| LDH > 240 U/L | 10 (25.6) |
| Cytogenetics * | |
| amp(1q) | 11 (28.2) |
| t(4;14) | 4 (10.3) |
| del(13q) | 2 (5.1) |
| del(17p) | 2 (5.1) |
| t(11;14) | 1 (2.6) |
| del(1p) | 1 (2.6) |
| t(14;16) | 0 (0) |
| t(14;20) | 0 (0) |
| IGH rearrangements | 7 (17.9) |
| Prior treatment | 10 (25.6) |
| Bortezomib regimen: | |
| VCD | 30 (76.9) |
| VMP | 4 (10.3) |
| VD | 4 (10.3) |
| VTD | 1 (2.6) |
| ASCT | 18 (46.2) |
| RTx | 10 (25.6) |
| Response to treatment | |
| CR | 10 (25.6) |
| VGPR | 10 (25.6) |
| PR | 8 (20.5) |
| SD | 5 (12.8) |
| PD | 6 (15.4) |
| Refractoriness to bortezomib | 12 (30.8) |
| Variables | PFS | OS | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Coefficient | p-Value | HR | 95% CI | Coefficient | p-Value | HR | 95% CI | |||
| Lower | Higher | Lower | Higher | |||||||
| ISS III | 0.38 | 0.0308 | 2.12 | 1.07 | 4.19 | 0.34 | 0.1284 | 1.95 | 0.82 | 4.64 |
| Previous treatment | 0.49 | 0.0178 | 2.65 | 1.18 | 5.91 | 0.16 | 0.5265 | 1.36 | 0.52 | 3.56 |
| ≥VGPR | −0.38 | 0.0292 | 0.47 | 0.24 | 0.93 | −0.18 | 0.4102 | 0.69 | 0.29 | 1.66 |
| ASCT | −0.53 | 0.0035 | 0.35 | 0.17 | 0.71 | −0.65 | 0.0074 | 0.27 | 0.10 | 0.70 |
| Sex (M) | 0.12 | 0.4781 | 1.28 | 0.65 | 2.53 | 0.05 | 0.8298 | 1.10 | 0.45 | 2.69 |
| HB < 10 g/dL | 0.12 | 0.4967 | 1.27 | 0.64 | 2.54 | 0.07 | 0.7431 | 1.16 | 0.48 | 2.81 |
| Calcium > 2.5 mmol/L | −0.13 | 0.4838 | 0.77 | 0.38 | 1.59 | 0.01 | 0.9747 | 1.02 | 0.40 | 2.55 |
| Creatinine > 2 mg/dL | −0.14 | 0.6125 | 0.76 | 0.27 | 2.17 | 0.22 | 0.482 | 1.57 | 0.45 | 5.46 |
| Bone disease | 0.03 | 0.8753 | 1.06 | 0.54 | 2.06 | −0.06 | 0.7691 | 0.88 | 0.37 | 2.08 |
| High POMP | −0.064 | 0.7434 | 0.880 | 0.410 | 1.889 | 0.515 | 0.0277 | 2.802 | 1.120 | 7.010 |
| High PSMB5 | 0.059 | 0.7343 | 1.125 | 0.570 | 2.219 | 0.271 | 0.2319 | 1.720 | 0.707 | 4.188 |
| High NRF2 | −0.205 | 0.2784 | 0.663 | 0.315 | 1.394 | −0.264 | 0.3104 | 0.590 | 0.213 | 1.635 |
| High XBP1 | −0.047 | 0.7954 | 0.911 | 0.450 | 1.845 | −0.301 | 0.2419 | 0.548 | 0.200 | 1.501 |
| High cMAF | 0.237 | 0.1972 | 1.608 | 0.781 | 3.310 | 0.280 | 0.0988 | 2.522 | 0.841 | 7.565 |
| High MAFB | −0.270 | 0.1582 | 0.583 | 0.276 | 1.233 | −0.572 | 0.0147 | 0.319 | 0.127 | 0.798 |
| Variables | PFS | ||||
|---|---|---|---|---|---|
| Coefficient | p-Value | HR | 95% CI | ||
| Lower | Higher | ||||
| ≥VGPR | −0.43 | 0.0170 | 0.43 | 0.21 | 0.86 |
| ASCT | −0.56 | 0.0021 | 0.32 | 0.16 | 0.66 |
| OS | |||||
| ASCT | −0.79 | 0.0025 | 0.20 | 0.07 | 0.57 |
| High MAFB | −0.92 | 0.0005 | 0.16 | 0.06 | 0.45 |
| High POMP | 0.60 | 0.0189 | 3.30 | 1.22 | 8.94 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Robak, P.; Szemraj, J.; Mikulski, D.; Drozdz, I.; Juszczak, K.; Jarych, D.; Misiewicz, M.; Kościelny, K.; Fendler, W.; Robak, T. Prognostic Value of Resistance Proteins in Plasma Cells from Multiple Myeloma Patients Treated with Bortezomib-Based Regimens. J. Clin. Med. 2021, 10, 5028. https://doi.org/10.3390/jcm10215028
Robak P, Szemraj J, Mikulski D, Drozdz I, Juszczak K, Jarych D, Misiewicz M, Kościelny K, Fendler W, Robak T. Prognostic Value of Resistance Proteins in Plasma Cells from Multiple Myeloma Patients Treated with Bortezomib-Based Regimens. Journal of Clinical Medicine. 2021; 10(21):5028. https://doi.org/10.3390/jcm10215028
Chicago/Turabian StyleRobak, Paweł, Janusz Szemraj, Damian Mikulski, Izabela Drozdz, Karolina Juszczak, Dariusz Jarych, Małgorzata Misiewicz, Kacper Kościelny, Wojciech Fendler, and Tadeusz Robak. 2021. "Prognostic Value of Resistance Proteins in Plasma Cells from Multiple Myeloma Patients Treated with Bortezomib-Based Regimens" Journal of Clinical Medicine 10, no. 21: 5028. https://doi.org/10.3390/jcm10215028







