1. Introduction
Twenty-four-hour ambulatory blood pressure monitoring (ABPM) is currently a more and more easily accessible tool to assess arterial blood pressure (BP) values. ABPM not only helps to detect white coat hypertension and is more useful to control the effectiveness of antihypertensive therapy than office measurements but also provides a valuable insight into the BP variability and fluctuations over the whole day sleep–wake cycle [
1]. Moreover, ABPM independence from the operator and available pediatric reference values makes it a useful non-invasive tool to measure cardiovascular risk in children. Up to now, there have been no better methods of BP assessment during the night. Blood pressure values differ during the day and the night. A physiological decline in BP at night is considered a normal circadian pattern—patients with such a nocturnal profile usually are called “dippers”. Patients with an insufficient night fall of BP values are referred to as “non-dippers”. However, more precisely, we can distinguish four different circadian BP rhythms: extreme dippers (over 20% decline in night BP values compared to daytime), dippers (between 20% and 10%), non-dippers (between 10% and 0%), and reverse dippers (nighttime surge in BP) (
Table 1) [
2].
There has been more than three decades of research on circadian BP patterns among hypertensive patients; and yet, there are still conflicting results as to whether dipping status is a valuable predictor of cardiovascular disease (CVD) morbidity and mortality and if it is associated with hypertension-mediated organ damage (HMOD). Studies in adults showed that both a lack of BP decline and excessive drop of BP at night might be indicators of CVD events [
3,
4]. For example, extreme dippers were found to be at the highest risk of stroke among all dipping patterns [
5]. On the other hand, a recent meta-analysis states that only reverse dippers are at high risk of complications [
6]. Similar discrepancies exist regarding whether normalizing blood pressure profile might improve cardiovascular risk. Of note, the results from a recently published yet strongly criticized Hygia study showed that prescribing one of the antihypertensive drugs at bedtime can improve dipping values and diminish the occurrence of CVD events [
7]. There are also some scarce and conflicted data in children [
8,
9]. Some papers suggest that non-dippers are associated with endothelial dysfunction and thicker intima-media [
10]. On the other hand, Seeman et al. found no difference in HMOD assessed as a left ventricular mass index between dippers and non-dippers [
11].
Therefore, our study aimed to assess dipping status, its determinants, and its relationship with precisely evaluated HMOD: left ventricular mass, urinary albumin excretion, and indicators of arterial damage: common carotid artery intima-media thickness (cIMT), arterial stiffness (augmentation index—AIx75HR, and aortic pulse wave velocity—aPWV) in a cohort of pediatric patients with untreated primary hypertension (PH).
4. Discussion
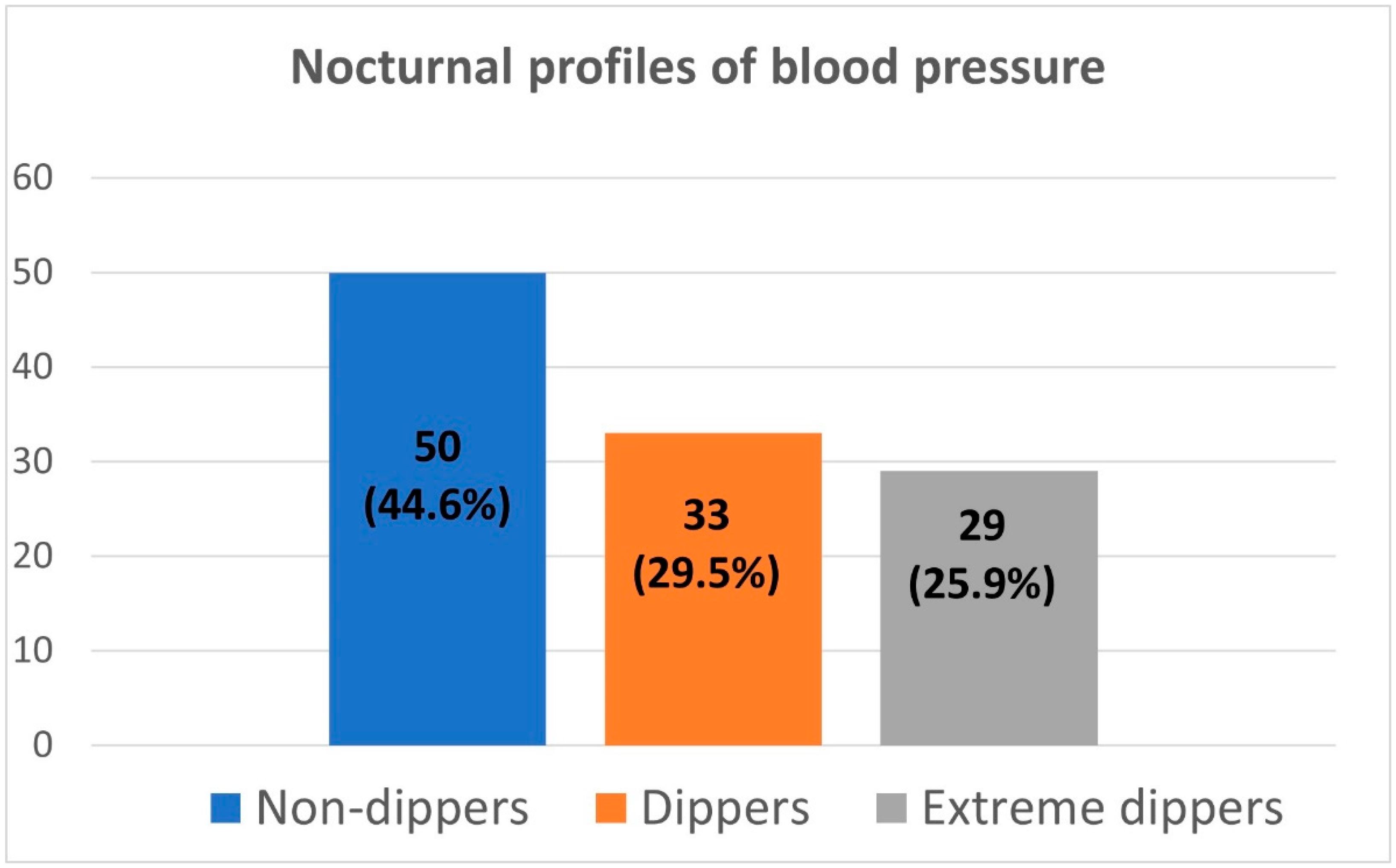
Our single-center cross-sectional study aimed to assess the dipping status in pediatric patients with untreated primary (essential) hypertension. First, we revealed that the non-dipping phenomenon is present in as many as half of the patients, and almost a quarter of them can be classified as extreme dippers leaving only a minority of patients with normal dipping status. Secondly, body mass and body mass index were significant determinants of dipping status. As for the analysis of the relation between dipping status and HMOD, systolic blood pressure dipping correlated negatively with left ventricular mass index, and non-dippers were found to have significantly higher left ventricular mass compared to dippers. Conversely, the augmentation index correlated positively with blood pressure dipping, and extreme dippers were characterized by the highest augmentation index. The latter association was confirmed by logistic regression and ROC analysis. Interestingly, no significant associations were revealed between central blood pressure, aortic pulse wave velocity, common carotid artery intima-media thickness, and dipping status.
We analyzed patients at one tertiary nephrology center with confirmed primary hypertension. We managed to collect a group of more than 100 treatment-naive patients. Our cohort can be considered representative of adolescent patients with primary hypertension. The mean age in our group was between 14.5 and 15 years, and the male gender was predominant. Additionally, more than 2/3 of the patients were overweight and obese—similar percentages are found in studies by other authors [
28,
29,
30,
31]. We found hyperuricemia in a large portion of patients, a hallmark of primary hypertension, and uric acid may be involved in its pathogenesis [
16]. Noteworthy, the predominant vascular phenotype in our cohort was isolated systolic hypertension, which also coincides with the results of other authors’ works [
32].
Arterial hypertension is the most important independent risk factor for cardiovascular morbidity and mortality worldwide. A particularly strong relationship is found between systolic blood pressure and the risk of stroke [
2]. Hard end-points are fortunately virtually non-existent in pediatrics, and incident cardiovascular events are observed primarily in patients with vasculopathies and additional risk factors such as chronic kidney disease, vasculitis, familial hypercholesterolemia, or recently as a consequence of COVID-19 infection [
33,
34,
35]. In contrast, subclinical hypertension-mediated organ damage (HMOD) is common in adolescents with PH. Evaluation of HMOD is of particular importance because it indicates the need for antihypertensive treatment, on the one hand, and, on the other, it identifies patients who should be subjected to special surveillance. According to ESH recommendations, assessment of left ventricular mass by ECHO is the most important, objective way to assess HMOD [
12]. Additional methods include assessment of urinary albumin excretion and evaluation of arterial lesions: especially cIMT and central arterial stiffness assessed as aortic PWV and additionally, AIx75HR. In our cohort, any form of HMOD was observed in about half of the patients, which also coincides with the findings of other authors [
36,
37,
38].
For many years, the importance of assessing not only the BP value itself but also other derivatives such as pulse pressure or BP variability has been raised. Assessment of pressure variability can be done in the long-term (visit-to-visit) range but also in the shorter term. Since the introduction of the ABPM study into widespread use, circadian variability has become the most commonly assessed variability. Arbitrarily years ago, it was accepted that the normal nocturnal drop in blood pressure was 10% (dippers vs. non-dippers). In subsequent years, data emerged indicating that it was also useful to separate patients with a high (>20%) nocturnal drop (extreme dippers) and those whose nighttime pressure increases (reverse dippers) [
1,
2]. Risk factors for impaired nocturnal BP drop have been identified. It has been repeatedly shown that secondary forms of AH (renal; endocrine e.g., primary hyperaldosteronism, pheochromocytoma/paraganglioma; autonomic dysfunction) are characterized by a higher prevalence of an impaired blood pressure profile [
39]. As for pediatric data, Flynn’s classic paper found that patients with secondary AH were characterized by higher nighttime diastolic pressure and an impaired diurnal profile of diastolic pressure. It is worth noting that Flynn based his conclusions on a small group of patients [
40]. In our study published in the local press, we compared 31 patients with PH and 33 patients with renal hypertension. Patients with PH were characterized by a tendency toward a lower diastolic pressure load at night and a greater decrease in systolic and diastolic blood pressure [
41]. Interestingly, in our cohort of pediatric patients with PH, as many as about half of the patients with confirmed PH were characterized by non-dipping and almost a quarter by extreme dipping. This indicates that assessment of the diurnal profile alone should not be an indication to look for or exclude secondary forms of hypertension.
The nocturnal drop in systolic blood pressure correlated negatively with body weight and BMI (both values normalized to pediatric values). These results indicate that overweight and obesity are risk factors for impaired nocturnal pressure drop. Other studies have found similar results in adults and children [
42,
43]. Obesity and overweight are associated with overactivation of the sympathetic nervous system and hyperinsulinism, which may be responsible for elevated nighttime blood pressure. In addition, patients with excess body weight may suffer from obstructive sleep apnea (OSA), a well-known risk factor for nocturnal hypertension [
44].
A sub-analysis of 26 patients showed a strong positive relationship between urinary potassium excretion and a nocturnal blood pressure drop. Evaluation of urinary excretion of the component is a simple marker of its dietary supply. Population studies have shown that increasing dietary potassium is associated with a reduction in blood pressure and cardiovascular risk. Bankir et al. found a positive association between daytime urinary potassium excretion and blood pressure dipping [
45]. Conversely, in a study by Libianto et al., no relation between urinary potassium excretion and blood pressure dipping was found in adults with diabetes mellitus [
46]. Further studies on larger numbers of pediatric patients evaluating the relationship between dietary sodium and potassium supply and the diurnal pressure profile are needed.
Our study showed that an impaired circadian blood pressure profile is a risk factor for left ventricular hypertrophy. These findings are consistent with the results of numerous adult studies [
47]. Nevertheless, there are doubts about the real significance of non-dipping for developing left ventricular hypertrophy. It is known that the importance of diurnal blood pressure profile is significantly lower in treated patients and, in addition, some studies indicate that the value of nighttime and weighted 24-h average BP is the real predictor of left ventricular hypertrophy [
48]. Seeman showed no relationship between blood pressure dipping and left ventricular mass or left ventricular hypertrophy in pediatric patients. However, what is noteworthy is that the authors analyzed both patients on antihypertensive treatment (87/114) and patients with various causes of hypertension (80/114 children with renoparenchymal hypertension) [
11]. Other factors, such as renal function, may have played a role in the failure to show a relationship between left ventricular mass and dipping status in the cited study. Similarly, a pediatric Chinese study revealed no relation between BP dipping and left ventricular hypertrophy [
9]. In this case, the difference in results could be derivative of younger age (13 years), different ethnicity, and different thresholds for LVH.
In addition, we showed that the phenomenon of extreme dipping is associated with an increase in the augmentation index. Patients defined as extreme dippers are characterized by a significant (>20%) decrease in nighttime blood pressure. Palatini, in a study involving more than 10,000 adults, showed that extreme dipping is a risk factor for cardiovascular complications, but only in patients over the age of 70 [
4]. Interestingly, recently extreme dippers were found to have an increased risk for left ventricular hypertrophy [
49,
50]. Our results indicate that excessive nocturnal blood pressure drop might also be associated with adverse arterial changes. Noteworthy, unfortunately, we did not show such a relationship for a parameter that is the gold standard for assessing vascular stiffness—aortic pulse wave velocity. It should also be stressed that some data indicate an opposite association between dipping status and arterial stiffness. Two adult Asian studies [
51,
52] and one small Portuguese pediatric study [
8] showed that augmentation index was positively associated with rather non-dipping not extreme dipping phenomenon.
Elevated urinary albumin excretion indicates subclinical endothelial damage not only in the kidneys and is an independent risk factor for cardiovascular morbidity and mortality [
2]. Evaluation of albuminuria is recommended in both adults and children with hypertension [
2,
12]. We did not demonstrate a statistically significant association of albuminuria with BP dipping in our cohort. Otherwise, some of the data in adults suggest an association of impaired diurnal profile with albuminuria [
53], e.g., in patients with uncontrolled nocturnal hypertension [
54]. One pediatric study also revealed that a higher urine protein-to-creatinine ratio was associated with significantly higher odds of non-dipping [
55]. Further studies, including prospective analyses on the significance of this phenomenon for the development of HMOD and hard-end points, are needed.
To date, two questions remain unresolved: first, whether we can effectively influence the circadian blood pressure profile and whether restoring a normal blood pressure profile reduces cardiovascular risk. Current European guidelines recommend prescribing treatment with long-acting drugs in a single morning dose (in adults, preferably a single pill composition) [
2]. Most studies did not show any additional benefit from supplying an antihypertensive drug in the evening. The only study that unequivocally showed a benefit of drug delivery in the evening is the HYGIA study [
7]. Nevertheless, after its publication, a debate swept through the literature questioning its credibility. Even more so, there are no such data in children. Here, too, there is a need for interventional studies that could demonstrate the benefit of drug delivery in the evening in pediatric patients with nocturnal hypertension.
The strength of our study is the relatively large group of pediatric patients with untreated (including non-pharmacological treatment) hypertension and the in-depth analysis of HMOD. Limitations are certainly the cross-sectional nature of the study, and the lack of validation of the OSCAR 2 SUNTECH device in the pediatric population (although the device has been validated in adults and has shown utility in pediatric patients in numerous of our publications [
18,
19,
25,
41]). The incomplete analysis of urinary potassium excretion is also a limitation. Patients were also not screened for obstructive sleep apnea and sleep quality; the ABPM could have affected the latter test itself.