The Role of Iron in Calciphylaxis—A Current Review
Abstract
1. Introduction
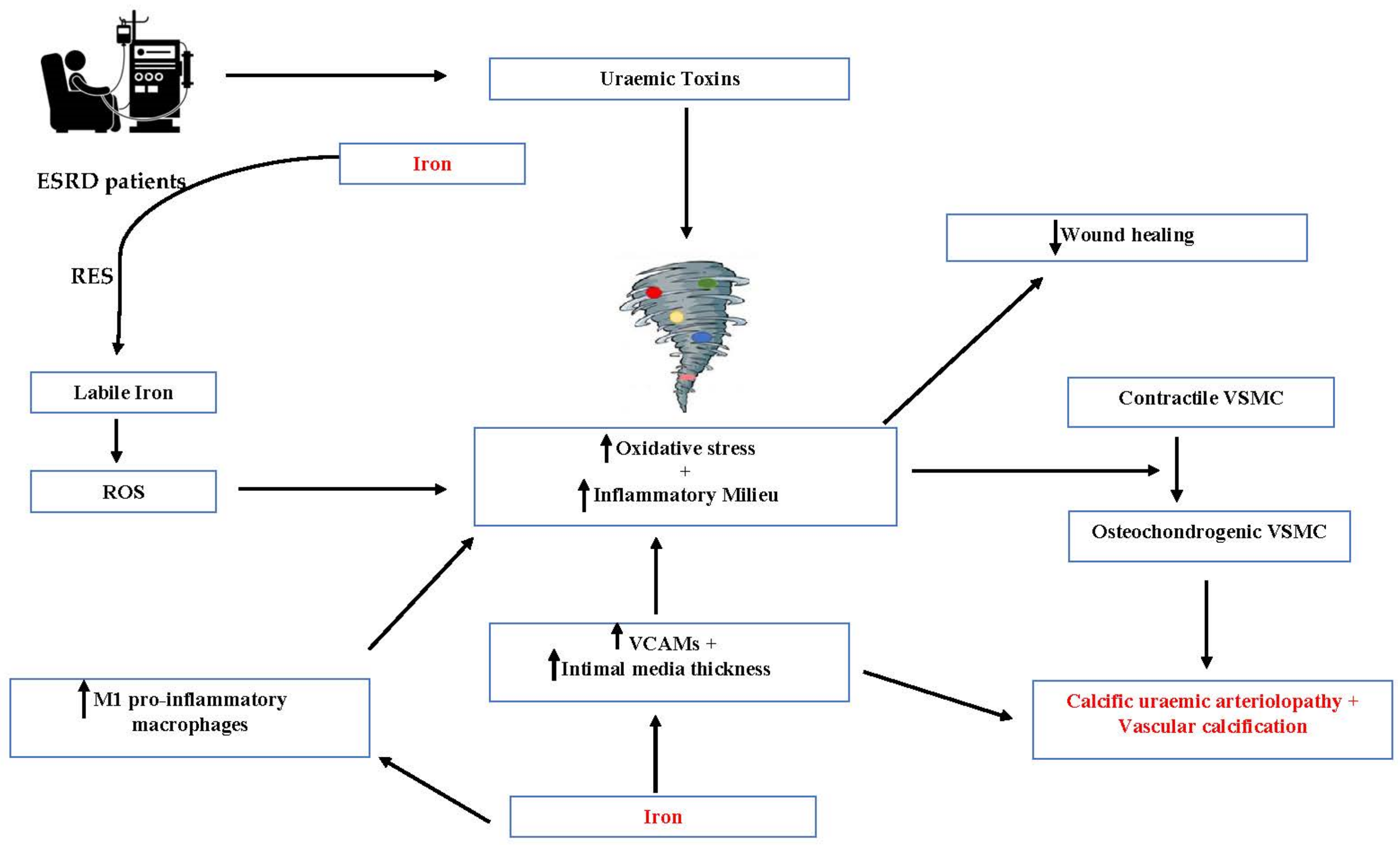
2. Role of Iron in Calciphylaxis
3. Mechanistic Insights on the Role of Iron in the Pathogenesis of Calciphylaxis
4. Pivotal Randomised Controlled Trial Data and Its Implications
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bhambri, A.; Del Rosso, J.Q. Calciphylaxis: A Review. J Clin. Aesthet. Dermatol. 2008, 1, 38. [Google Scholar] [PubMed]
- Nigwekar, S.U.; Thadhani, R.; Brandenburg, V.M. Calciphylaxis. N. Engl. J. Med. 2018, 378, 1704–1714. [Google Scholar] [CrossRef] [PubMed]
- Chinnadurai, R.; Huckle, A.; Hegarty, J.; Kalra, P.A.; Sinha, S. Calciphylaxis in end-stage kidney disease: Outcome data from the UK Calciphylaxis Study. J. Nephrol. 2021, 34, 1537–1545. [Google Scholar] [CrossRef] [PubMed]
- Peña Esparragoza, J.K.; Pérez Fernández, M.; Martínez Miguel, P.; Bouarich, H. La asociación de la sobrecarga de hierro y el desarrollo de calcifilaxis. Nefrología 2020, 40, 209–210. [Google Scholar] [CrossRef] [PubMed]
- Panchal, S.; Holtermann, K.; Trivedi, N.; Regunath, H.; Yerram, P. Calciphylaxis: An Analysis of Concomitant Factors, Treatment Effectiveness and Prognosis in 30 Patients. Int. J. Nephrol. Renov. Dis. 2020, 13, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Amuluru, L.; High, W.; Hiatt, K.M.; Ranville, J.; Shah, S.V.; Malik, B.; Swaminathan, S. Metal deposition in calcific uremic arteriolopathy. J. Am. Acad. Dermatol. 2009, 61, 73–79. [Google Scholar] [CrossRef]
- Higgins, S. Yale-Waterbury Internal Medicine Residency Program Calciphylaxis and Hypotension: Report of a Case and Review of the Literature. Clin. Dermatol. Ther. 2016, 3, 1–4. [Google Scholar] [CrossRef]
- Farah, M.; Crawford, R.I.; Levin, A.; Chan Yan, C. Calciphylaxis in the current era: Emerging “ironic” features? Nephrol. Dial. Transplant. 2011, 26, 191–195. [Google Scholar] [CrossRef]
- Sowers, K.M.; Hayden, M.R. Calcific Uremic Arteriolopathy: Pathophysiology, Reactive Oxygen Species and Therapeutic Approaches. Oxidative Med. Cell. Longev. 2010, 3, 109–121. [Google Scholar] [CrossRef]
- Portales-Castillo, I.; Kroshinsky, D.; Malhotra, C.K.; Culber-Costley, R.; Cozzolino, M.G.; Karparis, S.; Halasz, C.L.; Goverman, J.; Manley, H.J.; Malhotra, R.; et al. Calciphylaxis-as a drug induced adverse event. Expert Opin. Drug Saf. 2018, 18, 29–35. [Google Scholar] [CrossRef]
- Budisavljevic, M.N.; Cheek, D.; Ploth, D.W. Calciphylaxis in chronic renal failure. J. Am. Soc. Nephrol. 1996, 7, 978–982. [Google Scholar] [CrossRef]
- Floege, J.; Kubo, Y.; Floege, A.; Chertow, G.M.; Parfrey, P.S. The effect of cinacalcet on calcific uremic arteriolopathy events in patients receiving hemodialysis: The EVOLVE trial. Clin. J. Am. Soc. Nephrol. 2015, 10, 800–807. [Google Scholar] [CrossRef]
- Zacharias, J.; Fontaine, B.; Fine, A. Calcium Use Increases Risk of Calciphylaxis: A Case-Control Study. Perit. Dial. Int. J. Int. Soc. Perit. Dial. 1999, 19, 248–252. [Google Scholar] [CrossRef]
- Gaisne, R.; Péré, M.; Menoyo, V.; Hourmant, M.; Larmet-Burgeot, D. Calciphylaxis epidemiology, risk factors, treatment and survival among French chronic kidney disease patients: A case-control study. BMC Nephrol. 2020, 21, 63. [Google Scholar] [CrossRef]
- Papanikolaou, G.; Pantopoulos, K. Iron metabolism and toxicity. Toxicol. Appl. Pharmacol. 2005, 202, 199–211. [Google Scholar] [CrossRef]
- Kohgo, Y.; Ikuta, K.; Ohtake, T.; Torimoto, Y.; Kato, J. Body iron metabolism and pathophysiology of iron overload. Int. J. Hematol. 2008, 88, 7–15. [Google Scholar] [CrossRef]
- Wessling-Resnick, M. Iron Homeostasis and the Inflammatory Response. Annu. Rev. Nutr. 2010, 30, 105–122. [Google Scholar] [CrossRef]
- Selye, H. The Dermatologic Implications of Stress and Calciphylaxis. J. Investig. Dermatol. 1962, 39, 259–275. [Google Scholar] [CrossRef]
- Jovanovich, A.; Chonchol, M. Calcific Uremic Arteriolopathy Revisited. J. Am. Soc. Nephrol. 2016, 27, 3233–3235. [Google Scholar] [CrossRef]
- Rees, J.K.H.; Coles, G.A. Calciphylaxis in Man. Br. Med. J. 1969, 2, 670–672. [Google Scholar] [CrossRef][Green Version]
- Rubinger, D.; Friedlaender, M.M.; Silver, J.; Kopolovic, Y.; Czaczkes, W.J.; Popovtzer, M.M. Progressive Vascular Calcification with Necrosis of Extremities in Hemodialysis Patients: A Possible Role of Iron Overload. Am. J. Kidney Dis. 1986, 7, 125–129. [Google Scholar] [CrossRef]
- Rostoker, G.; Senet, P.; Lepeytre, F.; Griuncelli, M.; Loridon, C.; Rabaté, C.; Cohen, Y. Analysis of liver iron concentration in an elderly female undergoing hemodialysis with calcific uremic arteriolopathy does not support the role of iron overload in calciphylaxis: Lesson for the clinical nephrologist. J. Nephrol. 2021, 34, 1547–1551. [Google Scholar] [CrossRef] [PubMed]
- Macdougall, I.C.; White, C.; Anker, S.D.; Bhandari, S.; Farrington, K.; Kalra, P.A.; McMurray, J.J.; Murray, H.; Tomson, C.R.; Wheeler, D.C.; et al. Intravenous Iron in Patients Undergoing Maintenance Hemodialysis. N. Engl. J. Med. 2019, 380, 447–458. [Google Scholar] [CrossRef] [PubMed]
- Del Vecchio, L.; Ekart, R.; Ferro, C.J.; Malyszko, J.; Mark, P.B.; Ortiz, A.; Sarafidis, P.; Valdivielso, J.M.; Mallamaci, F. Intravenous iron therapy and the cardiovascular system: Risks and benefits. Clin. Kidney J. 2021, 14, 1067–1076. [Google Scholar] [CrossRef]
- Zickler, D.; Willy, K.; Girndt, M.; Fiedler, R.; Martus, P.; Storr, M.; Schindler, R. High cut-off dialysis in chronic haemodialysis patients reduces serum procalcific activity. Nephrol. Dial. Transplant. 2016, 31, 1706–1712. [Google Scholar] [CrossRef]
- Neven, E.; De Schutter, T.M.; Behets, G.J.; Gupta, A.; D’Haese, P.C. Iron and vascular calcification. Is there a link? Nephrol. Dial. Transplant. 2011, 26, 1137–1145. [Google Scholar] [CrossRef]
- National Kidney Foundation. Clinical Update: Issues in Iron Management in the Hemodialysis Patient. 2014. Available online: https://www.kidney.org/sites/default/files/02-10-6796_IBE_IronManagement_Bulletin_Print_1.pdf (accessed on 8 June 2022).
- Rick, J.; Rrapi, R.; Chand, S.; Shinohara, M.M.; Strowd, L.; Pasieka, H.B.; Micheletti, R.; Kroshinsky, D.; Ortega-Loayza, A.G. Calciphylaxis: Treatment and outlook—CME part II. J. Am. Acad. Dermatol. 2022, 86, 985–992. [Google Scholar] [CrossRef]
- Peng, T.; Zhuo, L.; Wang, Y.; Jun, M.; Li, G.; Wang, L.; Hong, D. Systematic review of sodium thiosulfate in treating calciphylaxis in chronic kidney disease patients. Nephrology 2018, 23, 669–675. [Google Scholar] [CrossRef]
- Nigwekar, S.U.; Brunelli, S.M.; Meade, D.; Wang, W.; Hymes, J.; Lacson, L., Jr. Sodium Thiosulfate Therapy for Calcific Uremic Arteriolopathy. Clin. J. Am. Soc. Nephrol. 2013, 8, 1162–1170. [Google Scholar] [CrossRef]
- Wright, J.A.; Richards, T.; Srai, S.K.S. The role of iron in the skin and cutaneous wound healing. Front. Pharmacol. 2014, 5, 156. [Google Scholar] [CrossRef]
- Sindrilaru, A.; Peters, T.; Wieschalka, S.; Baican, C.; Baican, A.; Peter, H.; Hainzl, A.; Schatz, S.; Qi, Y.; Schlecht, A.; et al. An unrestrained proinflammatory M1 macrophage population induced by iron impairs wound healing in humans and mice. J. Clin. Investig. 2011, 121, 985–997. [Google Scholar] [CrossRef]
- Papatriantafyllou, M. Macrophages: Iron macrophages. Nat. Rev. Immunol. 2011, 11, 158. [Google Scholar]
- Hénaut, L.; Candellier, A.; Boudot, C.; Grissi, M.; Mentaverri, R.; Choukroun, G.; Brazier, M.; Kamel, S.; Massy, Z.A. New Insights into the Roles of Monocytes/Macrophages in Cardiovascular Calcification Associated with Chronic Kidney Disease. Toxins 2019, 11, 529. [Google Scholar] [CrossRef]
- Zarjou, A.; Jeney, V.; Arosio, P.; Poli, M.; Antal-Szalmás, P.; Agarwal, A.; Balla, G.; Balla, J. Ferritin Prevents Calcification and Osteoblastic Differentiation of Vascular Smooth Muscle Cells. J. Am. Soc. Nephrol. 2009, 20, 1254–1263. [Google Scholar] [CrossRef]
- Balogh, E.; Tolnai, E.; Nagy, B., Jr.; Nagy, B.; Balla, G.; Balla, J.; Jeney, V. Iron overload inhibits osteogenic commitment and differentiation of mesenchymal stem cells via the induction of ferritin. Biochim. Biophys. Acta 2016, 1862, 1640–1649. [Google Scholar] [CrossRef]
- Sikura, K.; Potor, L.; Szerafin, T.; Zarjou, A.; Agarwal, A.; Arosio, P.; Poli, M.; Hendrik, Z.; Méhes, G.; Oros, M.; et al. Potential Role of H-Ferritin in Mitigating Valvular Mineralization. Arter. Thromb. Vasc. Biol. 2019, 39, 413–431. [Google Scholar] [CrossRef]
- Fine, A.; Zacharias, J. Calciphylaxis is usually non-ulcerating: Risk factors, outcome and therapy. Kidney Int. 2002, 61, 2210–2217. [Google Scholar] [CrossRef]
- Hayashi, M. Calciphylaxis: Diagnosis and clinical features. Clin. Exp. Nephrol. 2013, 17, 498–503. [Google Scholar] [CrossRef]
- Jeong, H.; Dominguez, A.R. Calciphylaxis: Controversies in Pathogenesis, Diagnosis and Treatment. Am. J. Med Sci. 2016, 351, 217–227. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wickens, O.; Rengarajan, S.; Chinnadurai, R.; Ford, I.; Macdougall, I.C.; Kalra, P.A.; Sinha, S. The Role of Iron in Calciphylaxis—A Current Review. J. Clin. Med. 2022, 11, 5779. https://doi.org/10.3390/jcm11195779
Wickens O, Rengarajan S, Chinnadurai R, Ford I, Macdougall IC, Kalra PA, Sinha S. The Role of Iron in Calciphylaxis—A Current Review. Journal of Clinical Medicine. 2022; 11(19):5779. https://doi.org/10.3390/jcm11195779
Chicago/Turabian StyleWickens, Olivia, Sharmilee Rengarajan, Rajkumar Chinnadurai, Ian Ford, Iain C. Macdougall, Philip A. Kalra, and Smeeta Sinha. 2022. "The Role of Iron in Calciphylaxis—A Current Review" Journal of Clinical Medicine 11, no. 19: 5779. https://doi.org/10.3390/jcm11195779
APA StyleWickens, O., Rengarajan, S., Chinnadurai, R., Ford, I., Macdougall, I. C., Kalra, P. A., & Sinha, S. (2022). The Role of Iron in Calciphylaxis—A Current Review. Journal of Clinical Medicine, 11(19), 5779. https://doi.org/10.3390/jcm11195779





