Elastographic Evaluation of Thyroid Nodules in Children and Adolescents with Hashimoto’s Thyroiditis and Nodular Goiter with Reference to Cytological and/or Histopathological Diagnosis
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Serum Analysis
2.3. Thyroid Imaging—Ultrasonography and Elastography
2.4. Fine Needle Aspiration Biopsy
2.5. Data Analysis
3. Results
3.1. Cytological and Histopathological Results
3.2. Non-AIT Thyroid Nodules
3.3. AIT Thyroid Nodules
3.4. Benign Thyroid Nodules
3.5. Malignant Thyroid Nodules
3.6. Accuracy of Elastography
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Skarpa, V.; Kousta, E.; Tertipi, A.; Anyfandakis, K.; Vakaki, M.; Dolianiti, M.; Fotinou, A.; Papathanasiou, A. Epidemiological characteristics of children with autoimmune thyroid disease. Hormones 2011, 10, 207–214. [Google Scholar] [CrossRef]
- Zois, C.; Stavrou, I.; Kalogera, C.; Svarna, E.; Dimoliatis, I.; Seferiadis, K.; Tsatsoulis, A. High prevalence of autoimmune thyroiditis in schoolchildren after elimination of iodine deficiency in northwestern Greece. Thyroid 2003, 13, 485–489. [Google Scholar] [CrossRef]
- Miranda, D.M.; Massom, J.N.; Catarino, R.M.; Santos, R.T.; Toyoda, S.S.; Marone, M.M.; Tomimori, E.K.; Monte, O. Impact of nutritional iodine optimization on rates of thyroid hypoechogenicity and autoimmune thyroiditis: A cross-sectional, comparative study. Thyroid 2015, 25, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Radetti, G. Clinical aspects of Hashimoto’s thyroiditis. Endocr. Dev. 2014, 26, 158–170. [Google Scholar] [CrossRef]
- Penta, L.; Cofini, M.; Lanciotti, L.; Leonardi, A.; Principi, N.; Esposito, S. Hashimoto’s Disease and Thyroid Cancer in Children: Are They Associated? Front. Endocrinol. 2018, 9, 565. [Google Scholar] [CrossRef]
- Gan, T.; Randle, R.W. The Role of Surgery in Autoimmune Conditions of the Thyroid. Surg. Clin. N. Am. 2019, 99, 633–648. [Google Scholar] [CrossRef]
- Dermody, S.; Walls, A.; Harley, E.H., Jr. Pediatric thyroid cancer: An update from the SEER database 2007–2012. Int. J. Pediatr. Otorhinolaryngol. 2016, 89, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Zeng, R.; Zhao, M.; Niu, H.; Yang, K.X.; Shou, T.; Zhang, G.Q.; Yan, X.M. Relationship between Hashimoto’s thyroiditis and papillary thyroid carcinoma in children and adolescents. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 7778–7787. [Google Scholar] [CrossRef] [PubMed]
- Niedziela, M.; Handkiewicz-Junak, D.; Małecka-Tendera, E.; Czarniecka, A.; Dedecjus, M.; Lange, D.; Kucharska, A.; Gawlik, A.; Pomorski, L.; Włoch, J.; et al. Diagnostics and treatment of differentiated thyroid carcinoma in children—Guidelines of Polish National Societies. Endokrynol. Pol. 2016, 67, 628–642. [Google Scholar] [CrossRef][Green Version]
- Francis, G.L.; Waguespack, S.G.; Bauer, A.J.; Angelos, P.; Benvenga, S.; Cerutti, J.M.; Dinauer, C.A.; Hamilton, J.; Hay, I.D.; Luster, M.; et al. Management Guidelines for Children with Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 2015, 25, 716–759. [Google Scholar] [CrossRef]
- Perros, P.; Boelaert, K.; Colley, S.; Evans, C.; Evans, R.M.; Gerrard Ba, G.; Gilbert, J.; Harrison, B.; Johnson, S.J.; Giles, T.E.; et al. Guidelines for the management of thyroid cancer. Clin. Endocrinol. 2014, 81, 1–122. [Google Scholar] [CrossRef] [PubMed]
- Xie, C.; Cox, P.; Taylor, N.; LaPorte, S. Ultrasonography of thyroid nodules: A pictorial review. Insights Imaging 2016, 7, 77–86. [Google Scholar] [CrossRef]
- Tritou, I.; Vakaki, M.; Sfakiotaki, R.; Kalaitzaki, K.; Raissaki, M. Pediatric thyroid ultrasound: A radiologist’s checklist. Pediatr. Radiol. 2020, 50, 563–574. [Google Scholar] [CrossRef]
- Cozzolino, A.; Filardi, T.; Simonelli, I.; Grani, G.; Virili, C.; Stramazzo, I.; Santaguida, M.G.; Locantore, P.; Maurici, M.; Gianfrilli, D.; et al. Diagnostic accuracy of ultrasonographic features in detecting thyroid cancer in the transition age: A meta-analysis. Eur. Thyroid J. 2022, 11, e220039. [Google Scholar] [CrossRef]
- Cooper, D.S.; Doherty, G.M.; Haugen, B.R.; Kloos, R.T.; Lee, S.L.; Mandel, S.J.; Mazzaferri, E.L.; McIver, B.; Pacini, F.; Schlumberger, M.; et al. Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid 2009, 19, 1167–1214. [Google Scholar] [CrossRef] [PubMed]
- Cosgrove, D.; Barr, R.; Bojunga, J.; Cantisani, V.; Chammas, M.C.; Dighe, M.; Vinayak, S.; Xu, J.M.; Dietrich, C.F. WFUMB Guidelines and Recommendations on the Clinical Use of Ultrasound Elastography: Part 4. Thyroid. Ultrasound Med. Biol. 2017, 43, 4–26. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Chen, Y.; Ai, H.; Gong, W.; Zhou, B.; Xu, Y.; Chen, S.; Cheng, F. Combining real-time elastography with fine-needle aspiration biopsy to identify malignant thyroid nodules. J. Int. Med. Res. 2020, 48, 300060520976027. [Google Scholar] [CrossRef]
- Cantisani, V.; Lodise, P.; Grazhdani, H.; Mancuso, E.; Maggini, E.; Di Rocco, G.; D’Ambrosio, F.; Calliada, F.; Redler, A.; Ricci, P.; et al. Ultrasound elastography in the evaluation of thyroid pathology. Current status. Eur. J. Radiol. 2014, 83, 420–428. [Google Scholar] [CrossRef]
- Radu, T.G.; Ciurea, M.E.; Mogoantă, S.Ş.; Busuioc, C.J.; Grosu, F.; Ţenovici, M.; Petrescu, I.O.; Vladu, I.M. Papillary thyroid cancer stroma—Histological and immunohistochemical study. Rom. J. Morphol. Embryol. 2016, 57, 801–809. [Google Scholar]
- Yi, L.; Qiong, W.; Yan, W.; Youben, F.; Bing, H. Correlation between Ultrasound Elastography and Histologic Characteristics of Papillary Thyroid Carcinoma. Sci. Rep. 2017, 22, 45042. [Google Scholar] [CrossRef]
- Ruchala, M.; Szczepanek-Parulska, E.; Zybek, A.; Moczko, J.; Czarnywojtek, A.; Kaminski, G.; Sowinski, J. The role of sonoelastography in acute, subacute and chronic thyroiditis: A novel application of the method. Eur. J. Endocrinol. 2012, 166, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Sigrist, R.M.S.; Liau, J.; Kaffas, A.E.; Chammas, M.C.; Willmann, J.K. Ultrasound Elastography: Review of Techniques and Clinical Applications. Theranostics 2017, 7, 1303–1329. [Google Scholar] [CrossRef]
- Borysewicz-Sanczyk, H.; Dzieciol, J.; Sawicka, B.; Bossowski, A. Practical Application of Elastography in the Diagnosis of Thyroid Nodules in Children and Adolescents. Horm. Res. Paediatr. 2016, 86, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Chen, J.; Sun, Y.; Xu, F.; Wu, L.; Huang, P. A retrospective study of reducing unnecessary thyroid biopsy for American College of Radiology Thyroid Imaging Reporting and Data Systems 4 assessment through applying shear wave elastography. Arch. Endocrinol. Metab. 2020, 64, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Russ, G.; Royer, B.; Bigorgne, C.; Rouxel, A.; Bienvenu-Perrard, M.; Leenhardt, L. Prospective evaluation of thyroid imaging reporting and data system on 4550 nodules with and without elastography. Eur. J. Endocrinol. 2013, 168, 649–655. [Google Scholar] [CrossRef]
- Pani, F.; Yasuda, Y.; Di Dalmazi, G.; Chalan, P.; Gabrielson, K.; Adamo, L.; Sabini, E.; Mariotti, S.; Caturegli, P. Pre-existing Thyroiditis Ameliorates Papillary Thyroid Cancer: Insights From a New Mouse Model. Endocrinology 2021, 162, bqab144. [Google Scholar] [CrossRef] [PubMed]
- Pani, F.; Caria, P.; Yasuda, Y.; Makoto, M.; Mariotti, S.; Leenhardt, L.; Roshanmehr, S.; Caturegli, P.; Buffet, C. The Immune Landscape of Papillary Thyroid Cancer in the Context of Autoimmune Thyroiditis. Cancers 2022, 14, 4287. [Google Scholar] [CrossRef]
- Corrias, A.; Cassio, A.; Weber, G.; Mussa, A.; Wasniewska, M.; Rapa, A.; Gastaldi, R.; Einaudi, S.; Baronio, F.; Vigone, M.C.; et al. Study Group for Thyroid Diseases of Italian Society for Pediatric Endocrinology and Diabetology (SIEDP/ISPED). Thyroid nodules and cancer in children and adolescents affected by autoimmune thyroiditis. Arch. Pediatr. Adolesc. Med. 2008, 162, 526–531. [Google Scholar] [CrossRef]
- Radetti, G.; Loche, S.; D’Antonio, V.; Salerno, M.; Guzzetti, C.; Aversa, T.; Cassio, A.; Cappa, M.; Gastaldi, R.; Deluca, F.; et al. Influence of Hashimoto Thyroiditis on the Development of Thyroid Nodules and Cancer in Children and Adolescents. J. Endocr. Soc. 2019, 3, 607–616. [Google Scholar] [CrossRef]
- Keskin, M.; Savas-Erdeve, S.; Aycan, Z. Co-Existence of thyroid nodule and thyroid cancer in children and adolescents with Hashimoto thyroiditis: A single-center study. Horm. Res. Paediatr. 2016, 85, 181–187. [Google Scholar] [CrossRef]
- Won, J.H.; Lee, J.Y.; Hong, H.S.; Jeong, S.H. Thyroid nodules and cancer in children and adolescents affected by Hashimoto’s thyroiditis. Br. J. Radiol. 2018, 91, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Januś, D.; Wójcik, M.; Taczanowska, A.; Sołtysiak, P.; Wędrychowicz, A.; Roztoczyńska, D.; Drabik, G.; Wyrobek, Ł.; Starzyk, J.B. Follow-up of parenchymal changes in the thyroid gland with diffuse autoimmune thyroiditis in children prior to the development of papillary thyroid carcinoma. J. Endocrinol. Investig. 2019, 42, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Consorti, F.; Loponte, M.; Milazzo, F.; Potasso, L.; Antonaci, A. Risk of malignancy from thyroid nodular disease as an element of clinical management of patients with Hashimoto’s thyroiditis. Eur. Surg. Res. 2010, 45, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Kim, Y.; Choi, J.W.; Kim, Y.S. The association between papillary thyroid carcinoma and histologically proven Hashimoto’s thyroiditis: A meta-analysis. Eur. J. Endocrinol. 2013, 168, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Moon, S.; Chung, H.S.; Yu, J.M.; Yoo, H.J.; Park, J.H.; Kim, D.S.; Park, Y.J. Associations between Hashimoto thyroiditis and clinical outcomes of papillary thyroid cancer: A meta-analysis of observational studies. Endocrinol. Metab. 2018, 33, 473–484. [Google Scholar] [CrossRef] [PubMed]
- Resende de Paiva, C.; Grønhøj, C.; Feldt-Rasmussen, U.; von Buchwald, C. Association between Hashimoto’s Thyroiditis and Thyroid Cancer in 64,628 Patients. Front. Oncol. 2017, 10, 53. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Zeng, W.; Fang, F.; Yu, T.; Zhao, Y.; Fan, X.; Guo, N.; Gao, X. Clinical analysis of Hashimoto thyroiditis coexistent with papillary thyroid cancer in 1392 patients. Acta Otorhinolaryngol. Ital. 2017, 37, 393–400. [Google Scholar] [CrossRef]
- Sur, M.L.; Gaga, R.; Lazăr, C.; Lazea, C.; Aldea, C.; Sur, D. Papillary thyroid carcinoma in children with Hashimoto’s thyroiditis—A review of the literature between 2000 and 2020. J. Pediatr. Endocrinol. Metab. 2020, 33, 1511–1517. [Google Scholar] [CrossRef]
- Niedziela, M.; Flader, M.; Harasymczuk, J.; Trejster, E.; Rabska-Pietrzak, B.; Breborowicz, D.; Kurzawa, P.; Bręborowicz, J. The increased coexistence of thyroid carcinoma (TC) and autoimmune thyroiditis (AIT) in children and adolescents of Greater Poland in years 2001–2015 compared to years 1996–2000. Endokrynol. Pol. 2015, 66A, A76. [Google Scholar]
- Ben-Skowronek, I.; Sieniawska, J.; Pac, E.; Wrobel, W.; Skowronek, A.; Tomczyk, Z.; Mlodawska, A.; Makuch, M.; Malka, M.; Cielecki, C.; et al. Thyroid Cancer Risk Factors in Children with Thyroid Nodules: A One-Center Study. J. Clin. Med. 2021, 10, 4455. [Google Scholar] [CrossRef]
- Gupta, A.; Ly, S.; Castroneves, L.A.; Frates, M.C.; Benson, C.B.; Feldman, H.A.; Wassner, A.J.; Smith, J.R.; Marqusee, E.; Alexander, E.K.; et al. A standardized assessment of thyroid nodules in children confirms higher cancer prevalence than in adults. J. Clin. Endocrinol. Metab. 2013, 98, 3238–3245. [Google Scholar] [CrossRef] [PubMed]
- Celletti, I.; Fresilli, D.; De Vito, C.; Bononi, M.; Cardaccio, S.; Cozzolino, A.; Durante, C.; Grani, G.; Grimaldi, G.; Isidori, A.M.; et al. TIRADS, SRE and SWE in INDETERMINATE thyroid nodule characterization: Which has better diagnostic performance? Radiol. Med. 2021, 126, 1189–1200. [Google Scholar] [CrossRef] [PubMed]
- Cantisani, V.; De Silvestri, A.; Scotti, V.; Fresilli, D.; Tarsitano, M.G.; Polti, G.; Guiban, O.; Polito, E.; Pacini, P.; Durante, C.; et al. US-Elastography With Different Techniques for Thyroid Nodule Characterization: Systematic Review and Meta-analysis. Front. Oncol. 2022, 16, 845549. [Google Scholar] [CrossRef]
- Borysewicz-Sańczyk, H.; Sawicka, B.; Karny, A.; Bossowski, F.; Marcinkiewicz, K.; Rusak, A.; Dzięcioł, J.; Bossowski, A. Suspected Malignant Thyroid Nodules in Children and Adolescents According to Ultrasound Elastography and Ultrasound-Based Risk Stratification Systems—Experience from One Center. J. Clin. Med. 2022, 11, 1768. [Google Scholar] [CrossRef]
- Menzilcioglu, M.S.; Duymus, M.; Gungor, G.; Citil, S.; Sahin, T.; Boysan, S.N.; Sarica, A. The value of real-time ultrasound elastography in chronic autoimmune thyroiditis. Br. J. Radiol. 2014, 87, 20140604. [Google Scholar] [CrossRef] [PubMed]
- Kara, T.; Ateş, F.; Durmaz, M.S.; Akyürek, N.; Durmaz, F.G.; Özbakır, B.; Öztürk, M. Assessment of thyroid gland elasticity with shear-wave elastography in Hashimoto’s thyroiditis patients. J. Ultrasound 2020, 23, 543–551. [Google Scholar] [CrossRef] [PubMed]
- Bakırtaş Palabıyık, F.; İnci, E.; Papatya Çakır, E.D.; Hocaoğlu, E. Evaluation of Normal Thyroid Tissue and Autoimmune Thyroiditis in Children Using Shear Wave Elastography. J. Clin. Res. Pediatr. Endocrinol. 2019, 11, 132–139. [Google Scholar] [CrossRef]
- Kandemirli, S.G.; Bayramoglu, Z.; Caliskan, E.; Sari, Z.N.A.; Adaletli, I. Quantitative assessment of thyroid gland elasticity with shear-wave elastography in pediatric patients with Hashimoto’s thyroiditis. J. Med. Ultrason 2018, 45, 417–423. [Google Scholar] [CrossRef]
- Magri, F.; Chytiris, S.; Capelli, V.; Gaiti, M.; Zerbini, F.; Carrara, R.; Malovini, A.; Rotondi, M.; Bellazzi, R.; Chiovato, L. Comparison of elastographic strain index and thyroid fine-needle aspiration cytology in 631 thyroid nodules. J. Clin. Endocrinol. Metab. 2013, 98, 4790–4797. [Google Scholar] [CrossRef]
- Liu, B.J.; Xu, H.X.; Zhang, Y.F.; Xu, J.M.; Li, D.D.; Bo, X.W.; Li, X.L.; Guo, L.H.; Xu, X.H.; Qu, S. Acoustic radiation force impulse elastography for differentiation of benign and malignant thyroid nodules with concurrent Hashimoto’s thyroiditis. Med. Oncol. 2015, 32, 50. [Google Scholar] [CrossRef]
- Magri, F.; Chytiris, S.; Capelli, V.; Alessi, S.; Nalon, E.; Rotondi, M.; Cassibba, S.; Calliada, F.; Chiovato, L. Shear wave elastography in the diagnosis of thyroid nodules: Feasibility in the case of coexistent chronic autoimmune Hashimoto’s thyroiditis. Clin. Endocrinol. 2012, 76, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Brylka, D.; Sun, L.N.; Lin, Y.Q.; Sui, G.Q.; Gao, J. Comparison of strain ratio with elastography score system in differentiating malignant from benign thyroid nodules. Clin. Imaging 2013, 37, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Ning, C.P.; Jiang, S.Q.; Zhang, T.; Sun, L.T.; Liu, Y.J.; Tian, J.W. The value of strain ratio in differential diagnosis of thyroid solid nodules. Eur. J. Radiol. 2012, 81, 286–291. [Google Scholar] [CrossRef] [PubMed]


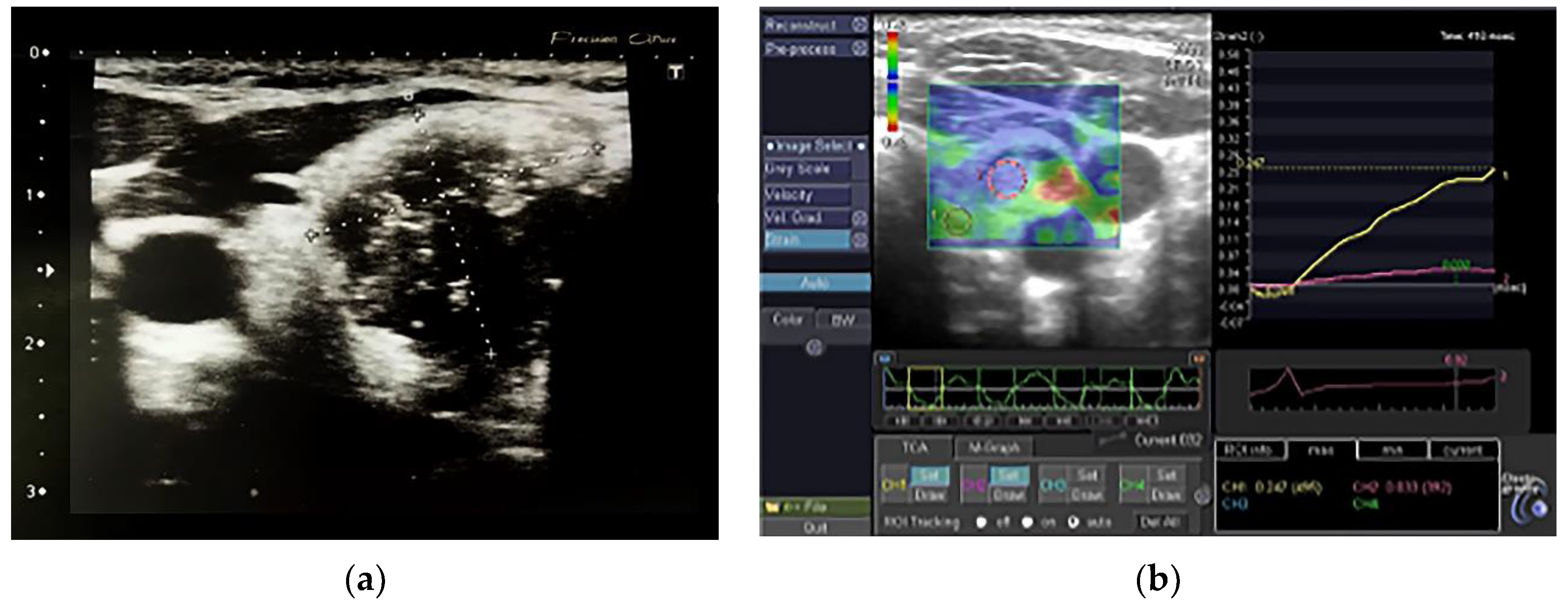
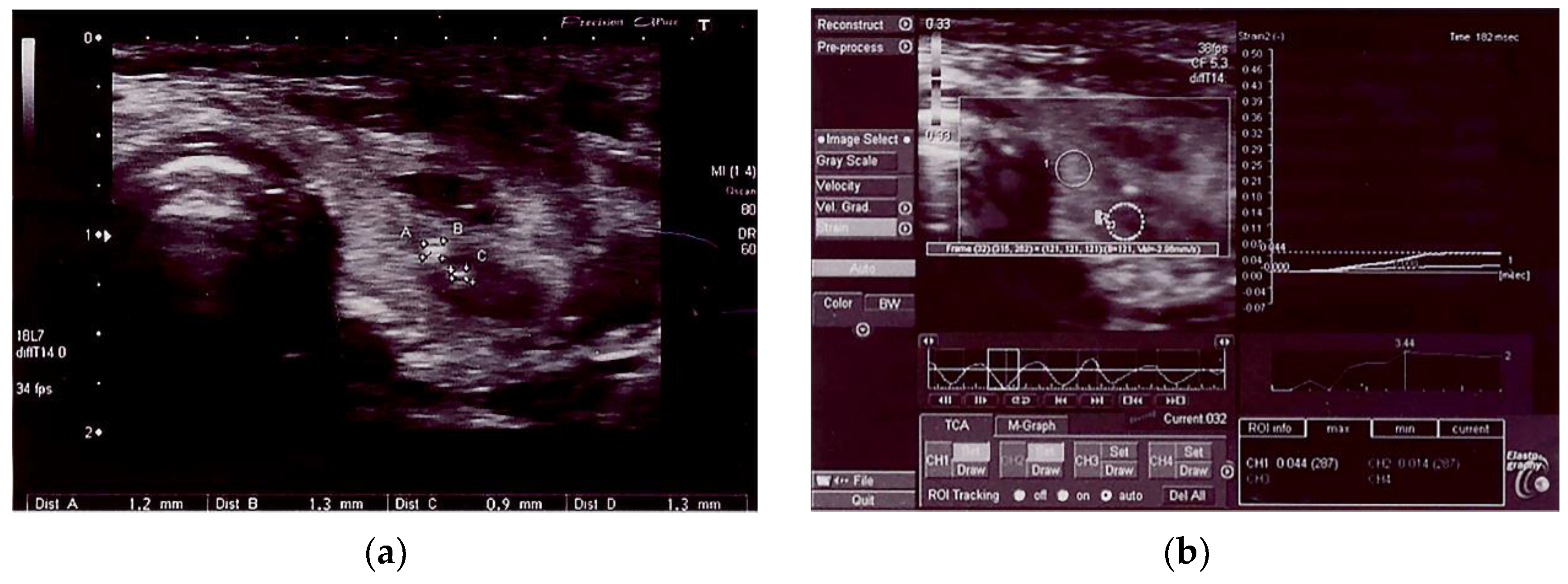
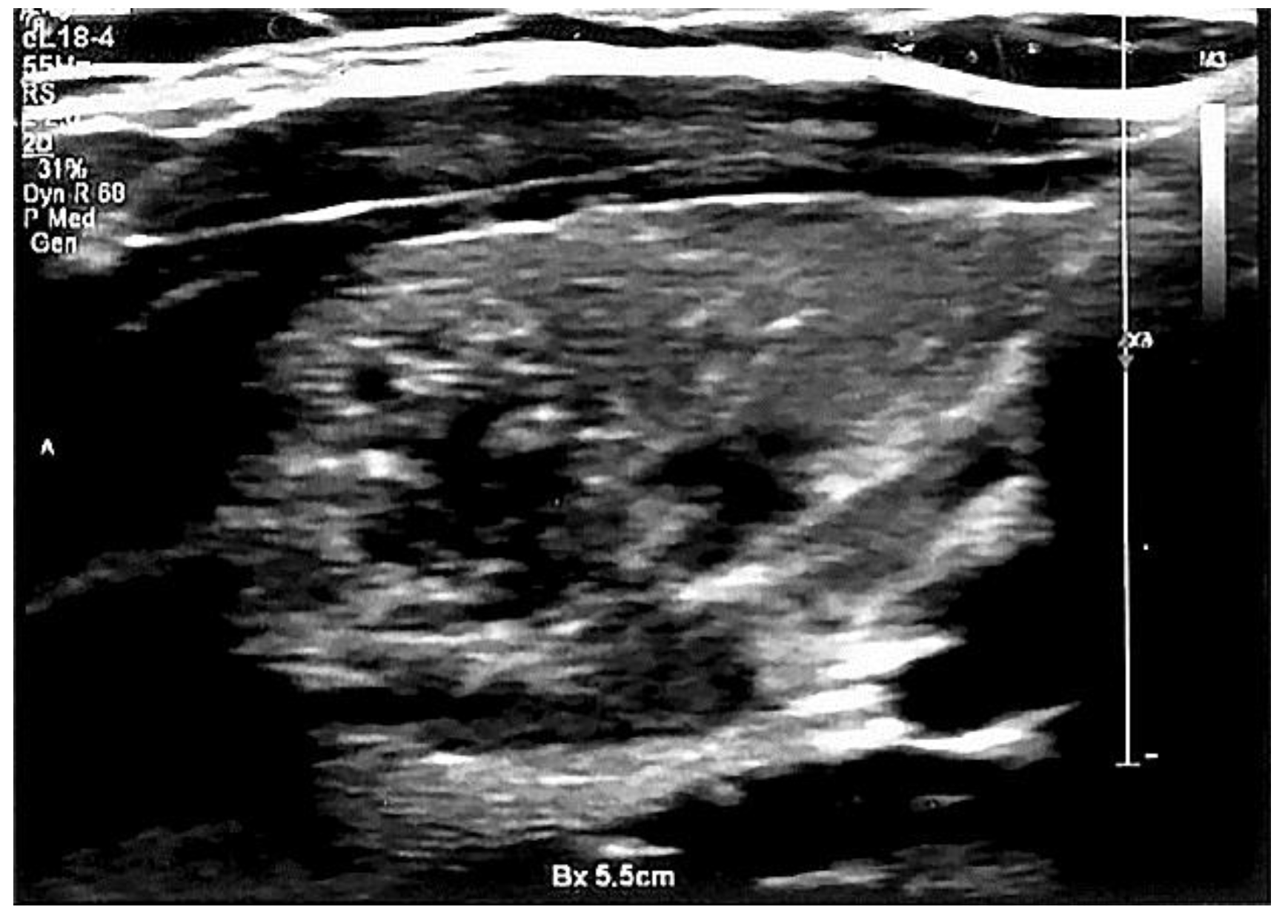
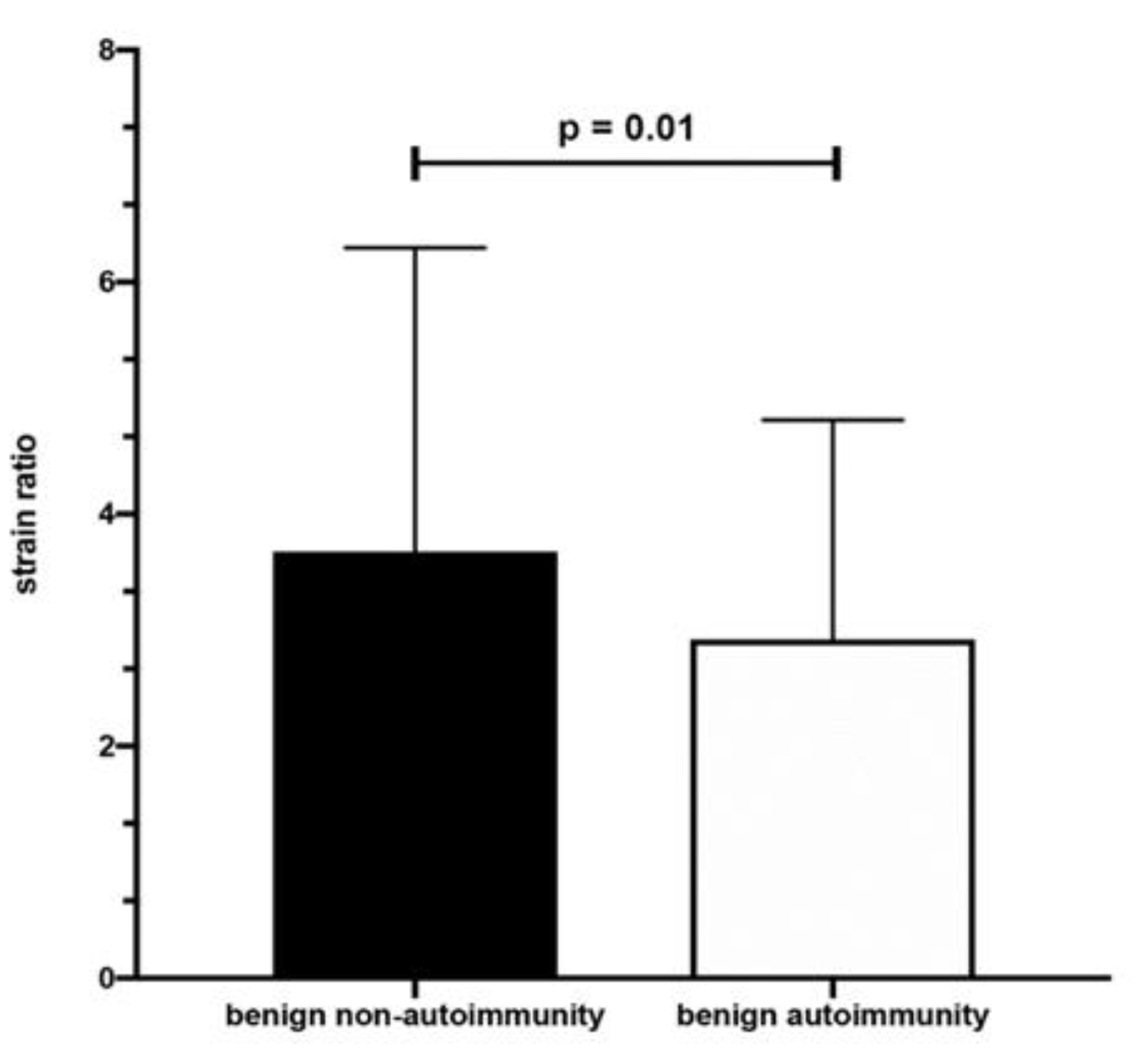
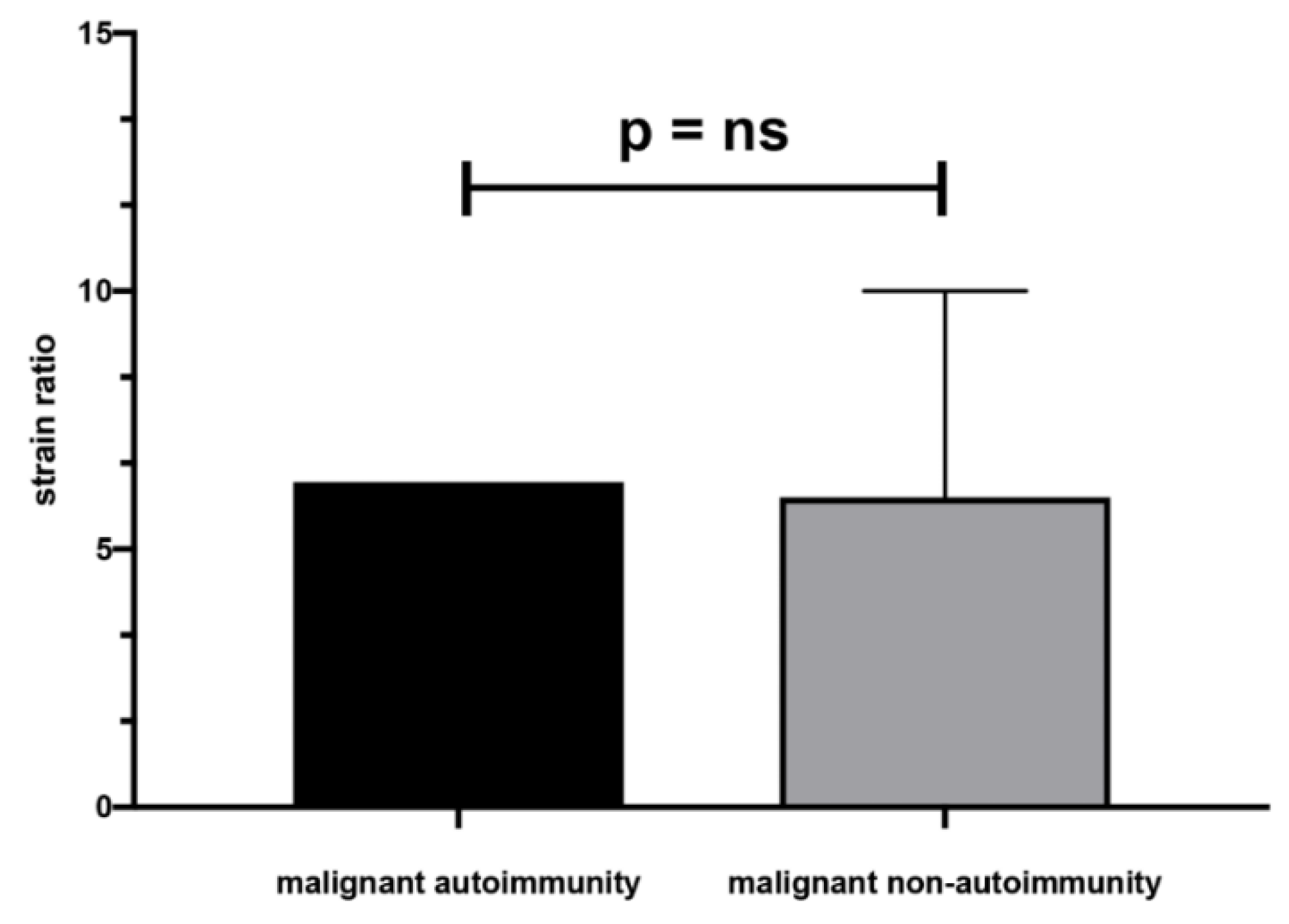

| All, Range (Mean ± SD) | AIT, Range (Mean ± SD) | Non-AIT, Range (Mean ± SD) | p | |
|---|---|---|---|---|
| number of thyroid nodules | 261 | 118 | 143 | |
| sex (boys/girls) | 73/188 | 37/81 | 36/107 | |
| left/right | 116/145 | 55/63 | 60/83 | |
| malignant/benign | 5/256 | 2/116 | 3/140 | |
| age (years) | 5–18 (14.60 ± 3.12) | 5–18 (14.02 ± 3.34) | 5–18 (15.08 ± 2.85) | <0.01 |
| TSH (μIU/mL) | 0.13–14.55 (2.90 ± 2.04) | 0.13–14.55 (3.55 ± 2.47) | 0.34–6.72 (2.29 ± 1.27) | <0.0001 |
| fT4 (ng/dL) | 0.84–5.81 (1.4 ± 0.54) | 0.92–1.8 (1.3 ± 0.23) | 0.84–5.81 (1.4 ± 0.74) | ns |
| TPO antibodies (IU/mL) | 1.08–1300 (108.6 ± 192.0) | 1.08–1300 (183.7 ± 230.4) | 5–34 (12.87 ± 7.5) | <0.0001 |
| Tg antibodies (IU/mL) | 10–1617 (161.1–304.1) | 10–1617 (229.67 ± 347.14) | 10–17.2 (11.38 ± 2.32) | <0.01 |
| length (mm) | 3.0–28.0 (7.9 ± 4.2) | 3.5–21.0 (7.3 ± 3.5) | 3.0–28.0 (8.3 ± 4.8) | ns |
| width (mm) | 1.7–24.0 (6.2 ± 3.6) | 1.7–22.0 (5.8 ± 3.3) | 1.8–24.0 (6.5 ± 3.9) | ns |
| Bethesda System | hp | Echogenicity | Margin | Compositoin | Taller-than-Wide Shape | Suspected Lymph Nodes | Echogenic Foci |
|---|---|---|---|---|---|---|---|
| VI | ptc | hypoechogenic | irregular | solid | yes | no | microcalcifications |
| VI | ptc | hypoechogenic | irregular | solid | yes | no | micro- and macrocalcifications |
| V | ptc | hyperechogenic | irregular | solid | yes | no | microcalcifications |
| V | ptc | hypoechogenic | irregular | solid | yes | no | microcalcifications |
| V | ptc | hypoechogenic | irregular | solid | yes | no | microcalcifications |
| IV | benign | hypo- and isoechogenic | regular | solid and cystic | yes | no | no |
| IV | benign | hypoechogenic | irregular | solid | no | no | microcalcifications |
| III | benign | hypoechogenic | regular | solid | yes | no | no |
| III | benign | hypoechogenic | irregular | solid | no | no | microcalcifications |
| III | benign | hypoechogenic | regular | solid | no | no | no |
| III | benign | hypoechogenic | regular | solid | no | no | no |
| Malignant Mean ± SD | Benign | p | |
|---|---|---|---|
| Bethesda system | 5.33 ± 0.58 | 2.31 ± 0.61 | <0.001 |
| length | 12.83 ± 7.00 | 8.21 ± 4.69 | 0.097 |
| width | 10.50 ± 6.26 | 6.37 ± 3.90 | 0.075 |
| TSH | 1.66 ± 0.50 | 2.31 ± 1.29 | ns |
| fT4 | 1.19 ± 0.15 | 1.42 ± 0.75 | ns |
| TPO antibodies | 9.40 ± 0.01 | 12.93 ± 7.60 | ns |
| Malignant Mean ± SD | Benign | p | |
|---|---|---|---|
| Bethesda system | 5.50 ± 0.70 | 2.12 ± 0.33 | <0.001 |
| length | 19.00 ± 2.83 | 7.10 ± 3.08 | <0.001 |
| width | 17.00 ± 2.83 | 5.53 ± 2.87 | <0.001 |
| TSH | 3.00 ± 0.01 | 3.55 ± 2.48 | ns |
| fT4 | - | 1.32 ± 0.23 | - |
| TPO antibodies | 167.5 ± 106.77 | 184.16 ± 233.11 | ns |
| Tg antibodies | 1617.0 ± 0.001 | 200.15 ± 283.54 | <0.001 |
| Non-AIT Mean ± SD | AIT Mean ± SD | p | |
|---|---|---|---|
| Bethesda system | 2.31 ± 0.61 | 2.12 ± 0.33 | ns |
| length | 8.21 ± 4.69 | 7.10 ± 3.08 | 0.044 |
| width | 6.37 ± 3.90 | 5.53 ± 2.87 | 0.075 |
| TSH | 2.31 ± 1.28 | 3.55 ± 2.48 | <0.001 |
| fT4 | 1.42 ± 0.75 | 1.32 ± 0.23 | ns |
| TPO antibodies | 12.93 ± 7.60 | 184.16 ± 233.11 | <0.001 |
| Tg antibodies | 11.39 ± 2.32 | 200.15 ± 283.54 | 0.003 |
| Non-AIT Mean ± SD | AIT Mean ± SD | p | |
|---|---|---|---|
| Bethesda system | 5.33 ± 0.58 | 5.50 ± 0.70 | ns |
| length | 12.83 ± 7.00 | 19.00 ± 2.83 | ns |
| width | 10.50 ± 6.26 | 17.00 ± 2.83 | ns |
| TSH | 1.66 ± 0.50 | 3.00 ± 0.01 | ns |
| fT4 | 1.19 ± 0.15 | - | - |
| TPO antibodies | 9.40 ± 0.01 | 167.5 ± 106.77 | ns |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borysewicz-Sańczyk, H.; Sawicka, B.; Bossowski, F.; Dzięcioł, J.; Bossowski, A. Elastographic Evaluation of Thyroid Nodules in Children and Adolescents with Hashimoto’s Thyroiditis and Nodular Goiter with Reference to Cytological and/or Histopathological Diagnosis. J. Clin. Med. 2022, 11, 6339. https://doi.org/10.3390/jcm11216339
Borysewicz-Sańczyk H, Sawicka B, Bossowski F, Dzięcioł J, Bossowski A. Elastographic Evaluation of Thyroid Nodules in Children and Adolescents with Hashimoto’s Thyroiditis and Nodular Goiter with Reference to Cytological and/or Histopathological Diagnosis. Journal of Clinical Medicine. 2022; 11(21):6339. https://doi.org/10.3390/jcm11216339
Chicago/Turabian StyleBorysewicz-Sańczyk, Hanna, Beata Sawicka, Filip Bossowski, Janusz Dzięcioł, and Artur Bossowski. 2022. "Elastographic Evaluation of Thyroid Nodules in Children and Adolescents with Hashimoto’s Thyroiditis and Nodular Goiter with Reference to Cytological and/or Histopathological Diagnosis" Journal of Clinical Medicine 11, no. 21: 6339. https://doi.org/10.3390/jcm11216339
APA StyleBorysewicz-Sańczyk, H., Sawicka, B., Bossowski, F., Dzięcioł, J., & Bossowski, A. (2022). Elastographic Evaluation of Thyroid Nodules in Children and Adolescents with Hashimoto’s Thyroiditis and Nodular Goiter with Reference to Cytological and/or Histopathological Diagnosis. Journal of Clinical Medicine, 11(21), 6339. https://doi.org/10.3390/jcm11216339








