The Effect of Anti-Tumor Necrosis Factor-Alpha Therapy within 12 Weeks Prior to Surgery on Postoperative Complications in Inflammatory Bowel Disease: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Eligibility Criteria
2.2. Search Strategy
2.3. Selection Process
2.4. Data Collection Process
2.5. Data Items
- (1)
- general overall complication rates:
- overall postoperative complications
- overall infectious postoperative complications
- overall Clavien-Dindo minor and major complications
- readmission rates, reoperation rates, mortality rates
- (2)
- surgical-site complication rates:
- overall infectious surgical complications
- intra-abdominal septic complications, anastomotic leakages (AL), intra-abdominal abscesses (without drainage)
- surgical-site infections (SSI) (incisional- and deep or organ space)
- postoperative hemorrhages, ileus, small bowel obstructions, fistula formations
- (3)
- non-surgical-site complication rates:
- overall infectious and non-infectious non-surgical-site complications
- thrombosis, cardiovascular complications
- pneumonia, urinary tract infections and sepsis
2.6. Assessment of Risk of Bias and Quality of Included Studies
2.7. Synthesis Methods
3. Results
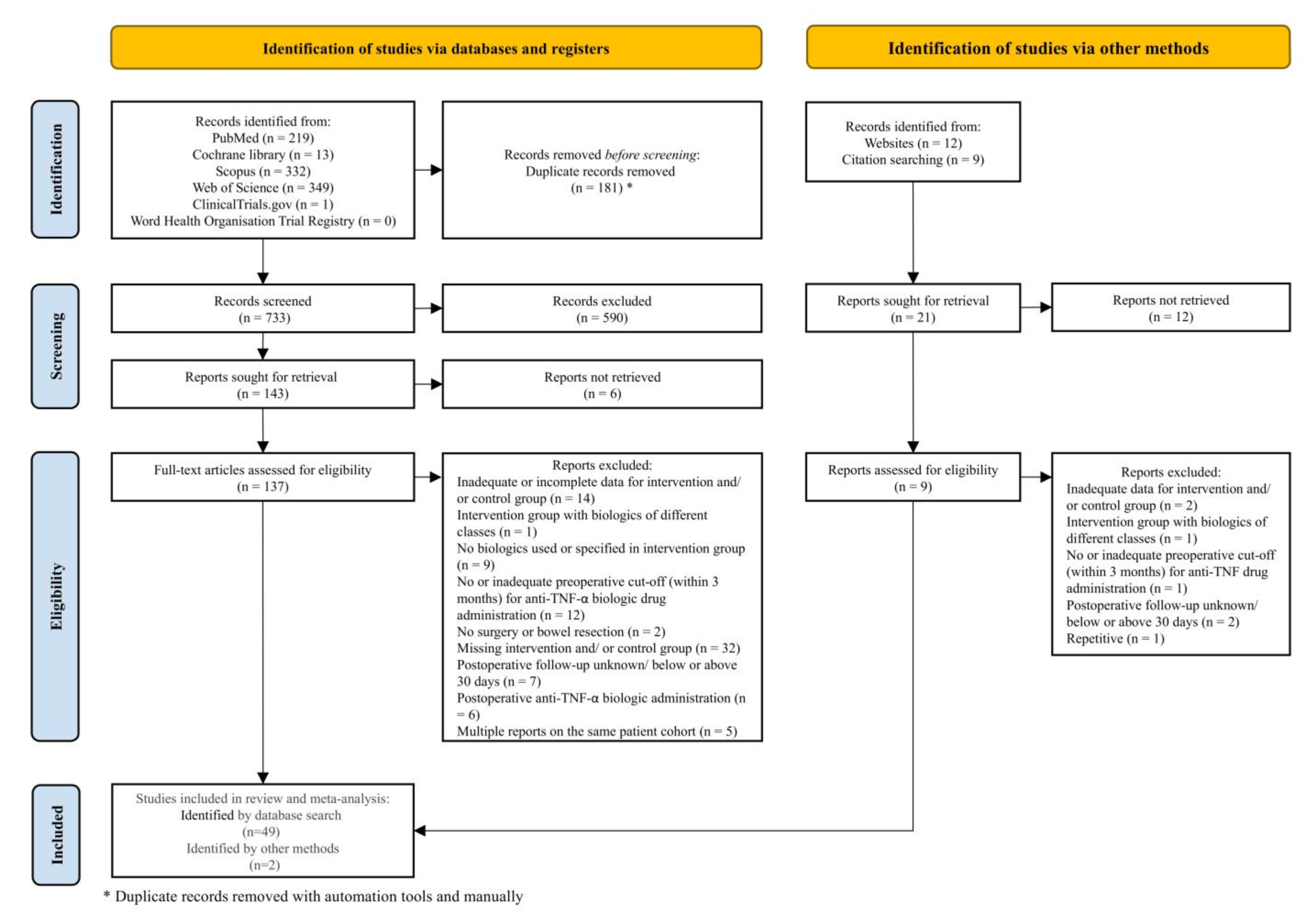
3.1. Study Selection
3.2. Study Characteristics
3.3. Results of Synthesis
3.3.1. Analysis of General Postoperative Complications
Overall Postoperative Complications
Overall Postoperative Infectious Complication
Overall Clavien-Dindo Major Complications
Readmission
Reoperation
Mortality
3.3.2. Analysis of Surgical Postoperative Complications
Overall Infectious Surgical-Site Complications
Intra-Abdominal Septic Complications
AL or Abscess
Intra-Adominal Abscesses
3.3.3. Analysis of Other Postoperative Complications
- (1)
- Overall non-infectious postoperative complications [48,54,71,89] (FE: OR, 0.85; 95% CI, 0.56–1.29; p = 0.45; I2 = 23%; p = 0.27) (Supplementary Figure S8).
- (2)
- (3)
- Superficial SSI [23,52,65,71,74,82,83,84,85,89] (RE: OR, 0.67; 95% CI, 0.32–1.40; p = 0.29; I2 = 69%; p = 0.0004–stable throughout sensitivity analysis; Egger’s Test: p = 0.643; no subgroup difference in subgroup analysis for subgroups stratified by IBD subtype: TSD: p = 0.93; sensitivity analysis: p = 0.74) (Supplementary Figure S10; Supplementary Table S12).
- (4)
- (5)
- Fistula formation [74,79,80] (FE: OR, 0.65; 95% CI, 0.17–2.47; p = 0.52; I2 = 0%; p = 0.83) (Supplementary Figure S12).
- (6)
- Ileus [23,48,55,59,62,66,70,74,75,89] (FE: OR, 1.11; 95% CI, 0.81–1.51; p = 0.52; I2 = 0%; p = 0.15; Egger’s Test: p = 0.115; no subgroup difference in subgroup analysis for subgroups stratified by IBD subtype: TSD: p = 0.76; sensitivity analysis: p = 0.5) (Supplementary Figure S13; Supplementary Table S13).
- (7)
- (8)
- (9)
- Overall infectious non-surgical-site complications [53,71,78] FE: OR, 1.14; 95% CI, 0.43–3.03; p = 0.79; I2 = 0%; p = 0.53) (Supplementary Figure S16).
- (10)
- (11)
- Cardiovascular complications [62,63,70,79] (FE: OR, 0.62; 95% CI, 0.23–1.73; p = 0.36; I2 = 0%; p = 0.96) (Supplementary Figure S18).
- (12)
- (13)
- Urinary tract infection [46,48,53,55,60,61,62,87,89] (FE: OR, 1.02; 95% CI, 0.54–1.91; p = 0.96; I2 = 0%; p = 0.65; no subgroup difference in subgroup analysis for subgroups stratified by IBD subtype: TSD: p = 0.68; sensitivity analysis: p = 0.76) (Supplementary Figure S20; Supplementary Table S18).
- (14)
4. Discussion
- (1)
- The overall postoperative complication rate was calculated individually, summation of all individual complications was not performed.
- (2)
- The correct number of patients was included for all studies evaluated in this analysis.
- (3)
- The study offers an insight on how authors assessed for risk of bias in accordance with the NOS scale—authors’ judgment and rationale, underlined by citations, were presented (Supplementary Tables S2.1.–S2.51.).
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Alatab, S.; Sepanlou, S.G.; Ikuta, K.; Vahedi, H.; Bisignano, C.; Safiri, S.; Sadeghi, A.; Nixon, M.R.; Abdoli, A.; Abolhassani, H.; et al. The global, regional, and national burden of inflammatory bowel disease in 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol. Hepatol. 2020, 5, 17–30. [Google Scholar] [CrossRef]
- Wong, D.J.; Roth, E.M.; Feuerstein, J.D.; Poylin, V.Y. Surgery in the age of biologics. Gastroenterol. Rep. 2019, 7, 77–90. [Google Scholar] [CrossRef] [PubMed]
- Ahmed Ali, U.; Martin, S.T.; Rao, A.D.; Kiran, R.P. Impact of preoperative immunosuppressive agents on postoperative outcomes in Crohn’s disease. Dis. Colon Rectum 2014, 57, 663–674. [Google Scholar] [CrossRef] [PubMed]
- El-Hussuna, A.; Krag, A.; Olaison, G.; Bendtsen, F.; Gluud, L.L. The effect of anti-tumor necrosis factor alpha agents on postoperative anastomotic complications in Crohn’s disease: A systematic review. Dis. Colon Rectum 2013, 56, 1423–1433. [Google Scholar] [CrossRef]
- Hanzel, J.; Almradi, A.; Istl, A.C.; Yang, M.L.; Fleshner, K.A.; Parker, C.E.; Guizzetti, L.; Ma, C.; Singh, S.; Jairath, V. Increased Risk of Infections with Anti-TNF Agents in Patients with Crohn’s Disease after Elective Surgery: Meta-Analysis. Dig. Dis. Sci. 2021, 67, 646–660. [Google Scholar] [CrossRef]
- Law, C.C.Y.; Koh, D.; Bao, Y.; Jairath, V.; Narula, N. Risk of Postoperative Infectious Complications from Medical Therapies in Inflammatory Bowel Disease: A Systematic Review and Meta-Analysis. Inflamm. Bowel Dis. 2020, 26, 1796–1807. [Google Scholar] [CrossRef]
- Lin, Y.S.; Cheng, S.W.; Wang, Y.H.; Chen, K.H.; Fang, C.J.; Chen, C. Systematic review with meta-analysis: Risk of post-operative complications associated with pre-operative exposure to anti-tumour necrosis factor agents for Crohn’s disease. Aliment. Pharmacol. Ther. 2019, 49, 966–977. [Google Scholar] [CrossRef]
- Moosvi, Z.; Duong, J.; Bechtold, M.L.; Nguyen, D.L. Systematic review and meta-analysis: Risks of postoperative complications with preoperative use of anti-tumor necrosis factor-alpha biologics in inflammatory bowel disease patients. Eur. J. Gastroenterol. Hepatol. 2021, 33, 799–816. [Google Scholar] [CrossRef]
- Narula, N.; Charleton, D.; Marshall, J.K. Meta-analysis: Peri-operative anti-TNFα treatment and post-operative complications in patients with inflammatory bowel disease. Aliment. Pharmacol. Ther. 2013, 37, 1057–1064. [Google Scholar] [CrossRef]
- Qiu, Y.; Zheng, Z.; Liu, G.; Zhao, X.; He, A. Effects of preoperative anti-tumour necrosis factor alpha infusion timing on postoperative surgical site infection in inflammatory bowel disease: A systematic review and meta-analysis. United Eur. Gastroenterol. J. 2019, 7, 1198–1214. [Google Scholar] [CrossRef]
- Rosenfeld, G.; Qian, H.; Bressler, B. The risks of post-operative complications following pre-operative infliximab therapy for Crohn’s disease in patients undergoing abdominal surgery: A systematic review and meta-analysis. J. Crohns Colitis 2013, 7, 868–877. [Google Scholar] [CrossRef] [PubMed]
- Selvaggi, F.; Pellino, G.; Canonico, S.; Sciaudone, G. Effect of preoperative biologic drugs on complications and function after restorative proctocolectomy with primary ileal pouch formation: Systematic review and meta-analysis. Inflamm. Bowel Dis. 2015, 21, 79–92. [Google Scholar] [CrossRef] [PubMed]
- Uchino, M.; Ikeuchi, H.; Shimizu, J.; Ohge, H.; Haji, S.; Mizuguchi, T.; Mohri, Y.; Yamashita, C.; Kitagawa, Y.; Suzuki, K.; et al. Association between preoperative tumor necrosis factor alpha inhibitor and surgical site infection after surgery for inflammatory bowel disease: A systematic review and meta-analysis. Surg. Today 2021, 51, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Xu, Y.; Zheng, T.; He, A.; Liu, G. Meta analysis on association between preoperative infliximab administration and morbidity of postoperative infectious complication in patients with ulcerative colitis. Chin. J. Dig. Dis. 2019, 3, 238–245. [Google Scholar] [CrossRef]
- Xu, Y.; Yang, L.; An, P.; Zhou, B.; Liu, G. Meta-Analysis: The Influence of Preoperative Infliximab Use on Postoperative Complications of Crohn’s Disease. Inflamm. Bowel Dis. 2019, 25, 261–269. [Google Scholar] [CrossRef]
- Yang, Z.P.; Hong, L.; Wu, Q.; Wu, K.C.; Fan, D.M. Preoperative infliximab use and postoperative complications in Crohn’s disease: A systematic review and meta-analysis. Int. J. Surg. 2014, 12, 224–230. [Google Scholar] [CrossRef]
- Zanelli, J.; Chandrapalan, S.; Patel, A.; Arasaradnam, R.P. The impact of pre-operative biologic therapy on post-operative surgical outcomes in ulcerative colitis: A systematic review and meta-analysis. Ther. Adv. Gastroenterol. 2020, 13, 1756284820937089. [Google Scholar] [CrossRef]
- Sandborn, W.J.; Hanauer, S.B. Antitumor necrosis factor therapy for inflammatory bowel disease: A review of agents, pharmacology, clinical results, and safety. Inflamm. Bowel Dis. 1999, 5, 119–133. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. (Eds.) Handbook for Systematic Reviews of Interventions Version 6.2 (Updated February 2021); Cochrane: London, UK, 2021; Available online: www.training.cochrane.org/handbook (accessed on 20 March 2022).
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Lau, C.; Dubinsky, M.; Melmed, G.; Vasiliauskas, E.; Berel, D.; McGovern, D.; Ippoliti, A.; Shih, D.; Targan, S.; Fleshner, P. The impact of preoperative serum anti-TNFα therapy levels on early postoperative outcomes in inflammatory bowel disease surgery. Ann. Surg. 2015, 261, 487–496. [Google Scholar] [CrossRef]
- Melo-Pinto, D.; Santos, J.V.; Barbosa, E. Risk factors for postoperative complications in Crohn disease: Analysis of 173 patients. J. Coloproctol. 2018, 38, 214–222. [Google Scholar] [CrossRef]
- Tang, S.; Dong, X.; Liu, W.; Qi, W.; Ye, L.; Yang, X.; Cao, Q.; Ge, X.; Zhou, W. Compare risk factors associated with postoperative infectious complication in Crohn’s disease with and without preoperative infliximab therapy: A cohort study. Int. J. Color. Dis. 2020, 35, 727–737. [Google Scholar] [CrossRef] [PubMed]
- Ayoub, F.; Ewelukwa, O.; Brar, T.; Forde, J.; Mramba, L.; Glover, S.; Iqbal, A.; Tan, S. Evaluating the impact of vedolizumab on postoperative complications in inflammatory bowel disease patients. Dis. Colon Rectum 2018, 61, E141. [Google Scholar]
- de Buck van Overstraeten, A.; Eshuis, E.J.; Vermeire, S.; Van Assche, G.; Ferrante, M.; D’Haens, G.R.; Ponsioen, C.Y.; Belmans, A.; Buskens, C.J.; Wolthuis, A.M.; et al. Short- and medium-term outcomes following primary ileocaecal resection for Crohn’s disease in two specialist centres. Br. J. Surg. 2017, 104, 1713–1722. [Google Scholar] [CrossRef]
- Guasch, M.; Clos, A.; Manyosa, M.; Lobatón Ortega, T.; Gomez, J.; Pinol, M.; Cabre, E.; Troya, J.; Domenech, E. Prevalence and risk factors for postoperative septic complications in Crohn’s disease. J. Crohns Colitis 2016, 10, S350. [Google Scholar]
- Gudsoorkar, V.; Ibarra, S.; Gonzalez-Almada, A.; Oglat, A.; Koduru, P.; Abraham, B.; Haas, E. P133 an analysis of surgical outcomes in ibd patients treated with and without biologic therapy. Gastroenterology 2018, 154, S68–S69. [Google Scholar] [CrossRef]
- Guo, K.; Ren, J.; Li, G.; Hu, Q.; Wu, X.; Wang, Z.; Wang, G.; Gu, G.; Ren, H.; Hong, Z.; et al. Risk factors of surgical site infections in patients with Crohn’s disease complicated with gastrointestinal fistula. Int. J. Color. Dis. 2017, 32, 635–643. [Google Scholar] [CrossRef]
- Kim, J.; Zaghiyan, K.; Fleshner, P. P640 Risk of post-operative complications among Crohn’s disease patients treated pre-operatively with vedolizumab. A matched case-control study. J. Crohns Colitis 2018, 12, S433–S434. [Google Scholar] [CrossRef][Green Version]
- Oh, S.H.; Hong, S.N.; Kim, M.J.; Kim, E.R.; Chang, D.K.; Kim, Y.-H. P316 The risk of preoperative anti-TNF-α treatment on early postoperative complications in patients with Crohn’s disease. J. Crohns Colitis 2014, 8, S196. [Google Scholar] [CrossRef]
- Rizvi, A.; Kayal, M.; Plietz, M.; Zylberberg, H.; Radcliffe, M.; Yzet, C.; Khaitov, S.; Greenstein, A.; Sylla, P.; Dubinsky, M. 705—Pre-Operative Biologics Significantly Reduce Post-Operative Leaks After Staged Restorative Proctocolectomy. Gastroenterology 2019, 156, S-154. [Google Scholar] [CrossRef]
- Schils, N.; De Buck van Overstraeten, A.; Vermeire, S.; Van Assche, G.; Wolthuis, A.; D’Hoore, A.; Ferrante, M. P445 Perioperative use of vedolizumab seems not associated with short-term postoperative infectious complications in patients with Crohn’s disease undergoing right hemicolectomy with ileocolonic anastomosis. J. Crohns Colitis 2017, 11, S304. [Google Scholar] [CrossRef][Green Version]
- Aaron Brzezinski, L.A.; Del Real, G.A.; Parsi, M.; Lashner, B.; Achkar, J.-P. Infliximab Does Not Increase the Risk of Complications in the Perioperative Period in Patients with Crohn’s Disease. Gastroenterology 2002, 122, A616. [Google Scholar]
- Lau, C.C.; Dubinsky, M.; Melmed, G.Y.; Vasiliauskas, E.A.; McGovern, D.P.; Berel, D.; Ippoliti, A.; Murrell, Z.A.; Shih, D.Q.; Kaur, M.; et al. Higher Preoperative Serum Biologic Levels Are Associated with Postoperative Complications in Crohn’s Disease Patients. Gastroenterology 2013, 144, 190. [Google Scholar] [CrossRef]
- Lau, C.C.; Dubinsky, M.; Melmed, G.Y.; Vasiliauskas, E.A.; McGovern, D.P.; Berel, D.; Murrell, Z.A.; Ippoliti, A.; Shih, D.Q.; Kaur, M.; et al. Preoperative Serum Biologic Levels Do Not Impact Postoperative Outcomes in Ulcerative Colitis. Gastroenterology 2013, 144, 189–190. [Google Scholar] [CrossRef]
- de Silva, S.; Ma, C.; Proulx, M.C.; Crespin, M.; Kaplan, B.S.; Hubbard, J.; Prusinkiewicz, M.; Fong, A.; Panaccione, R.; Ghosh, S.; et al. Postoperative Complications and Mortality Following Colectomy for Ulcerative Colitis. Clin. Gastroenterol. Hepatol. 2011, 9, 972–980. [Google Scholar] [CrossRef]
- Desai, P.N.; Sharma, A.; Naik, A.S.; Otterson, M.F.; Zadvornova, Y.; Perera, L.P.; Venu, N.; Stein, D.J. Su1560 Timing of Pre-Operative Anti-Tumor Necrosis Factor Therapy Does Not Affect Early Post-Operative Complication Rates in Inflammatory Bowel Disease Patients Undergoing Intestinal Resection. Gastroenterology 2012, 142, S-1063. [Google Scholar] [CrossRef]
- García, M.J.; Rivero, M.; Miranda-Bautista, J.; Bastón-Rey, I.; Mesonero, F.; Leo-Carnerero, E.; Casas-Deza, D.; Cagigas Fernández, C.; Martin-Cardona, A.; El Hajra, I.; et al. Impact of Biological Agents on Postsurgical Complications in Inflammatory Bowel Disease: A Multicentre Study of Geteccu. J. Clin. Med. 2021, 10, 4402. [Google Scholar] [CrossRef]
- Karjalainen, E.K.; Renkonen-Sinisalo, L.; Mustonen, H.K.; Färkkilä, M.; Lepistö, A.H. Restorative Proctocolectomy in Ulcerative Colitis: Effect of Preoperative Immunomodulatory Therapy on Postoperative Complications and Pouch Failure. Scand. J. Surg. 2021, 110, 51–58. [Google Scholar] [CrossRef]
- Marchal, L.; D’Haens, G.; Van Assche, G.; Vermeire, S.; Noman, M.; Ferrante, M.; Hiele, M.; Bueno De Mesquita, M.; D’Hoore, A.; Penninckx, F.; et al. The risk of post-operative complications associated with infliximab therapy for Cron’s disease: A controlled cohort study. Aliment. Pharmacol. Ther. 2004, 19, 749–754. [Google Scholar] [CrossRef]
- Uchino, M.; Ikeuchi, H.; Matsuoka, H.; Tsuchida, T.; Tomita, N.; Takesue, Y. Risk Factors Associated with Surgical Site Infection After Ileal Pouch-Anal Anastomosis in Ulcerative Colitis. Dis. Colon. Rectum. 2010, 53, 143–149. [Google Scholar] [CrossRef]
- Weber, A.T.; Sack, J.; Ha, C.Y. Anti-TNF Exposure Is Not Associated with Increased Post-Operative Morbidity or Reoperation Among Crohn’s Disease Patients: 744. Am. J. Gastroenterol. 2017, 112, S411, S414–S415. [Google Scholar]
- Wells, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Non Randomised Studies in Meta-Analyses. Available online: www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 18 February 2022).
- Higgins, J.P.; Thompson, S.G. Quantifying heterogeneity in a meta-analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Smith, G.D.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Appau, K.A.; Fazio, V.W.; Shen, B.; Church, J.M.; Lashner, B.; Remzi, F.; Brzezinski, A.; Strong, S.A.; Hammel, J.; Kiran, R.P. Use of infliximab within 3 months of ileocolonic resection is associated with adverse postoperative outcomes in Crohn’s patients. J. Gastrointest. Surg. 2008, 12, 1738–1744. [Google Scholar] [CrossRef]
- Bafford, A.C.; Powers, S.; Ha, C.; Kruse, D.; Gorfine, S.R.; Chessin, D.B.; Bauer, J.J. Immunosuppressive Therapy Does Not Increase Operative Morbidity in Patients with Crohn’s Disease. J. Clin. Gastroenterol. 2013, 47, 491–495. [Google Scholar] [CrossRef]
- Bregnbak, D.; Mortensen, C.; Bendtsen, F. Infliximab and complications after colectomy in patients with ulcerative colitis. J. Crohns Colitis 2012, 6, 281–286. [Google Scholar] [CrossRef]
- Brouquet, A.; Maggiori, L.; Zerbib, P.; Lefevre, J.H.; Denost, Q.; Germain, A.; Cotte, E.; Beyer-Berjot, L.; Munoz-Bongrand, N.; Desfourneaux, V.; et al. Anti-TNF Therapy Is Associated with an Increased Risk of Postoperative Morbidity after Surgery for Ileocolonic Crohn Disease Results of a Prospective Nationwide Cohort. Ann. Surg. 2018, 267, 221–228. [Google Scholar] [CrossRef]
- Canedo, J.; Lee, S.H.; Pinto, R.; Murad-Regadas, S.; Rosen, L.; Wexner, S.D. Surgical resection in Crohn’s disease: Is immunosuppressive medication associated with higher postoperative infection rates? Color. Dis. 2011, 13, 1294–1298. [Google Scholar] [CrossRef]
- El-Hussuna, A.; Andersen, J.; Bisgaard, T.; Jess, P.; Henriksen, M.; Oehlenschlager, J.; Thorlacius-Ussing, O.; Olaison, G. Biologic treatment or immunomodulation is not associated with postoperative anastomotic complications in abdominal surgery for Crohn’s disease. Scand. J. Gastroenterol. 2012, 47, 662–668. [Google Scholar] [CrossRef]
- El-Hussuna, A.; Qvist, N.; Zangenberg, M.S.; Langkilde, A.; Siersma, V.; Hjort, S.; Gögenur, I. No effect of anti-TNF-α agents on the surgical stress response in patients with inflammatory bowel disease undergoing bowel resections: A prospective multi-center pilot study. BMC Surg. 2018, 18, 91. [Google Scholar] [CrossRef]
- Ferrante, M.; D’Hoore, A.; Vermeire, S.; Declerck, S.; Noman, M.; Van Assche, G.; Hoffman, I.; Rutgeerts, P.; Penninckx, F. Corticosteroids but not infliximab increase short-term postoperative infectious complications in patients with ulcerative colitis. Inflamm. Bowel Dis. 2009, 15, 1062–1070. [Google Scholar] [CrossRef] [PubMed]
- Gainsbury, M.L.; Chu, D.I.; Howard, L.A.; Coukos, J.A.; Farraye, F.A.; Stucchi, A.F.; Becker, J.M. Preoperative infliximab is not associated with an increased risk of short-term postoperative complications after restorative proctocolectomy and ileal pouch-anal anastomosis. J. Gastrointest. Surg. 2011, 15, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Remzi, F.H.; Shen, B.; Vogel, J.D.; Kiran, R.P. Operative strategy modifies risk of pouch-related outcomes in patients with ulcerative colitis on preoperative anti-tumor necrosis factor-α therapy. Dis. Colon Rectum 2013, 56, 1243–1252. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, A.; Rivero, M.; Martin-Arranz, M.D.; Sanchez, V.G.; Castro, M.; Barrio, J.; de Francisco, R.; Barreiro-de Acosta, M.; Julia, B.; Cea-Calvo, L.; et al. Perioperative management and early complications after intestinal resection with ileocolonic anastomosis in Crohn’s disease: Analysis from the PRACTICROHN study. Gastroenterol. Rep. 2019, 7, 168–175. [Google Scholar] [CrossRef]
- Indar, A.A.; Young-Fadok, T.M.; Heppell, J.; Efron, J.E. Effect of perioperative immunosuppressive medication on early outcome in Crohn’s disease patients. World J. Surg. 2009, 33, 1049–1052. [Google Scholar] [CrossRef]
- Jouvin, I.; Lefevre, J.H.; Creavin, B.; Pitel, S.; Chafai, N.; Tiret, E.; Beaugerie, L.; Parc, Y. Postoperative Morbidity Risks Following Ileocolic Resection for Crohn’s Disease Treated with Anti-TNF Alpha Therapy: A Retrospective Study of 360 Patients. Inflamm. Bowel Dis. 2018, 24, 422–432. [Google Scholar] [CrossRef]
- Kim, J.Y.; Zaghiyan, K.; Lightner, A.; Fleshner, P. Risk of postoperative complications among ulcerative colitis patients treated preoperatively with vedolizumab: A matched case-control study. BMC Surg. 2020, 20, 46. [Google Scholar] [CrossRef]
- Kotze, P.G.; Magro, D.O.; Martinez, C.A.R.; Saab, B.; Saab, M.P.; Pinheiro, L.V.; Olandoski, M.; Yamamoto, T.; Coy, C.S.R. Adalimumab and postoperative complications of elective intestinal resections in Crohn’s disease: A propensity score case-matched study. Color. Dis. 2018, 20, 211–218. [Google Scholar] [CrossRef]
- Kotze, P.G.; Saab, M.P.; Saab, B.; da Silva Kotze, L.M.; Olandoski, M.; Pinheiro, L.V.; Martinez, C.A.; Ayrizono, M.L.; Magro, D.O.; Coy, C.S. Tumor Necrosis Factor Alpha Inhibitors Did Not Influence Postoperative Morbidity after Elective Surgical Resections in Crohn’s Disease. Dig. Dis. Sci. 2017, 62, 456–464. [Google Scholar] [CrossRef]
- Krane, M.K.; Allaix, M.E.; Zoccali, M.; Umanskiy, K.; Rubin, M.A.; Villa, A.; Hurst, R.D.; Fichera, A. Preoperative infliximab therapy does not increase morbidity and mortality after laparoscopic resection for inflammatory bowel disease. Dis. Colon Rectum 2013, 56, 449–457. [Google Scholar] [CrossRef]
- Kunitake, H.; Hodin, R.; Shellito, P.C.; Sands, B.E.; Korzenik, J.; Bordeianou, L. Perioperative treatment with infliximab in patients with Crohn’s disease and ulcerative colitis is not associated with an increased rate of postoperative complications. J. Gastrointest. Surg. 2008, 12, 1730–1736. [Google Scholar] [CrossRef] [PubMed]
- Lightner, A.L.; McKenna, N.P.; Alsughayer, A.; Harmsen, W.S.; Taparra, K.; Parker, M.E.; Raffals, L.E.; Loftus, E.V., Jr. Biologics and 30-Day Postoperative Complications after Abdominal Operations for Crohn’s Disease: Are There Differences in the Safety Profiles? Dis. Colon Rectum 2019, 62, 1352–1362. [Google Scholar] [CrossRef] [PubMed]
- Maeda, K.; Nagahara, H.; Shibutani, M.; Otani, H.; Sakurai, K.; Toyokawa, T.; Tanaka, H.; Kubo, N.; Muguruma, K.; Kamata, N.; et al. A preoperative low nutritional prognostic index correlates with the incidence of incisional surgical site infections after bowel resection in patients with Crohn’s disease. Surg. Today 2015, 45, 1366–1372. [Google Scholar] [CrossRef] [PubMed]
- Mascarenhas, C.; Nunoo, R.; Asgeirsson, T.; Rivera, R.; Kim, D.; Hoedema, R.; Dujovny, N.; Luchtefeld, M.; Davis, A.T.; Figg, R. Outcomes of ileocolic resection and right hemicolectomies for Crohn’s patients in comparison with non-Crohn’s patients and the impact of perioperative immunosuppressive therapy with biologics and steroids on inpatient complications. Am. J. Surg. 2012, 203, 375–378, discussion 378. [Google Scholar] [CrossRef] [PubMed]
- Monsinjon, M.; Mege, D.; Maggiori, L.; Treton, X.; Bouhnik, Y.; Panis, Y. Postoperative course of laparoscopic subtotal colectomy is affected by prolonged preoperative anti-TNF therapy in patients with acute colitis complicating inflammatory bowel disease. Int. J. Color. Dis. 2017, 32, 1499–1502. [Google Scholar] [CrossRef]
- Morar, P.S.; Hodgkinson, J.D.; Thalayasingam, S.; Koysombat, K.; Purcell, M.; Hart, A.L.; Tarne, J.W.; Faiz, O. Determining Predictors for Intra-abdominal Septic Complications Following Ileocolonic Resection for Crohn’s Disease-Considerations in Pre-operative and Peri-operative Optimisation Techniques to Improve Outcome. J. Crohns Colitis 2015, 9, 483–491. [Google Scholar] [CrossRef]
- Myrelid, P.; Marti-Gallostra, M.; Ashraf, S.; Sunde, M.L.; Tholin, M.; Oresland, T.; Lovegrove, R.E.; Tøttrup, A.; Kjaer, D.W.; George, B.D. Complications in surgery for Crohn’s disease after preoperative antitumour necrosis factor therapy. Br. J. Surg. 2014, 101, 539–545. [Google Scholar] [CrossRef]
- Nasir, B.S.; Dozois, E.J.; Cima, R.R.; Pemberton, J.H.; Wolff, B.G.; Sandborn, W.J.; Loftus, E.V.; Larson, D.W. Perioperative Anti-Tumor Necrosis Factor Therapy Does Not Increase the Rate of Early Postoperative Complications in Crohn’s Disease. J. Gastrointest. Surg. 2010, 14, 1859–1866. [Google Scholar] [CrossRef]
- Nelson, R.; Liao, C.; Fichera, A.; Rubin, D.T.; Pekow, J. Rescue therapy with cyclosporine or infliximab is not associated with an increased risk for postoperative complications in patients hospitalized for severe steroid-refractory ulcerative colitis. Inflamm. Bowel Dis. 2014, 20, 14–20. [Google Scholar] [CrossRef]
- Nørgård, B.M.; Nielsen, J.; Qvist, N.; Gradel, K.O.; de Muckadell, O.B.; Kjeldsen, J. Pre-operative use of anti-TNF-α agents and the risk of post-operative complications in patients with ulcerative colitis—A nationwide cohort study. Aliment. Pharmacol. Ther. 2012, 35, 1301–1309. [Google Scholar] [CrossRef]
- Nørgård, B.M.; Nielsen, J.; Qvist, N.; Gradel, K.O.; de Muckadell, O.B.; Kjeldsen, J. Pre-operative use of anti-TNF-α agents and the risk of post-operative complications in patients with Crohn’s disease—A nationwide cohort study. Aliment. Pharmacol. Ther. 2013, 37, 214–224. [Google Scholar] [CrossRef]
- Regadas, F.S.; Pinto, R.A.; Murad-Regadas, S.M.; Canedo, J.A.; Leal, M.; Nogueras, J.J.; Wexner, S.D. Short-term outcome of infliximab and other medications on patients with inflammatory bowel disease undergoing ileostomy reversal. Color. Dis. 2011, 13, 555–560. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, G.; Armuzzi, A.; Pugliese, D.; Verbo, A.; Papa, A.; Mattana, C.; Rapaccini, G.L.; Guidi, L.; Coco, C. Anti-TNF-alpha therapies do not increase early postoperative complications in patients with inflammatory bowel disease. An Italian single-center experience. Int. J. Color. Dis. 2011, 26, 1435–1444. [Google Scholar] [CrossRef] [PubMed]
- Selvasekar, C.R.; Cima, R.R.; Larson, D.W.; Dozois, E.J.; Harrington, J.R.; Harmsen, W.S.; Loftus, E.V., Jr.; Sandborn, W.J.; Wolff, B.G.; Pemberton, J.H. Effect of infliximab on short-term complications in patients undergoing operation for chronic ulcerative colitis. J. Am. Coll. Surg. 2007, 204, 956–962, discussion 962–963. [Google Scholar] [CrossRef] [PubMed]
- Serradori, T.; Germain, A.; Scherrer, M.L.; Ayav, C.; Perez, M.; Romain, B.; Palot, J.P.; Rohr, S.; Peyrin-Biroulet, L.; Bresler, L. The effect of immune therapy on surgical site infection following Crohn’s Disease resection. Br. J. Surg. 2013, 100, 1089–1093. [Google Scholar] [CrossRef] [PubMed]
- Shwaartz, C.; Fields, A.C.; Sobrero, M.; Cohen, B.D.; Divino, C.M. Effect of Anti-TNF Agents on Postoperative Outcomes in Inflammatory Bowel Disease Patients: A Single Institution Experience. J. Gastrointest. Surg. 2016, 20, 1636–1642. [Google Scholar] [CrossRef]
- Syed, A.; Cross, R.K.; Flasar, M.H. Anti-tumor necrosis factor therapy is associated with infections after abdominal surgery in Crohn’s disease patients. Am. J. Gastroenterol. 2013, 108, 583–593. [Google Scholar] [CrossRef]
- Tay, G.S.; Binion, D.G.; Eastwood, D.; Otterson, M.F. Multivariate analysis suggests improved perioperative outcome in Crohn’s disease patients receiving immunomodulator therapy after segmental resection and/or strictureplasty. Surgery 2003, 134, 565–572. [Google Scholar] [CrossRef]
- Tiberi, A.; Pesi, B.; Giudici, F.; Zambonin, D.; Nelli, T.; Cupellini, C.; Ficari, F.; Cianchi, F.; Scaringi, S. Laparoscopic ileo-colic resection and right hemicolectomy for Crohn’s disease and colon cancer: A preliminary comparative study on post-operative outcome. Updates Surg. 2020, 72, 821–826. [Google Scholar] [CrossRef]
- Uchino, M.; Ikeuchi, H.; Bando, T.; Chohno, T.; Sasaki, H.; Horio, Y.; Kuwahara, R.; Minagawa, T.; Goto, Y.; Ichiki, K.; et al. Associations between multiple immunosuppressive treatments before surgery and surgical morbidity in patients with ulcerative colitis during the era of biologics. Int. J. Color. Dis. 2019, 34, 699–710. [Google Scholar] [CrossRef]
- Uchino, M.; Ikeuchi, H.; Bando, T.; Hirose, K.; Hirata, A.; Chohno, T.; Sasaki, H.; Takahashi, Y.; Takesue, Y.; Hida, N.; et al. Does Pre-Operative Multiple Irnmunosuppressive Therapy Associate with Surgical Site Infection in Surgery for Ulcerative Colitis? Digestion 2015, 92, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Uchino, M.; Ikeuchi, H.; Matsuoka, H.; Bando, T.; Ichiki, K.; Nakajima, K.; Tomita, N.; Takesue, Y. Risk factors for surgical site infection and association with infliximab administration during surgery for Crohn’s disease. Dis. Colon Rectum 2013, 56, 1156–1165. [Google Scholar] [CrossRef] [PubMed]
- Uchino, M.; Ikeuchi, H.; Matsuoka, H.; Bando, T.; Ichiki, K.; Nakajima, K.; Tomita, N.; Takesue, Y. Infliximab administration prior to surgery does not increase surgical site infections in patients with ulcerative colitis. Int. J. Color. Dis. 2013, 28, 1295–1306. [Google Scholar] [CrossRef] [PubMed]
- Ward, S.T.; Mytton, J.; Henderson, L.; Amin, V.; Tanner, J.R.; Evison, F.; Radley, S. Anti-TNF therapy is not associated with an increased risk of post-colectomy complications, a population-based study. Color. Dis. 2018, 20, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Waterman, M.; Xu, W.; Dinani, A.; Steinhart, A.H.; Croitoru, K.; Nguyen, G.C.; McLeod, R.S.; Greenberg, G.R.; Cohen, Z.; Silverberg, M.S. Preoperative biological therapy and short-term outcomes of abdominal surgery in patients with inflammatory bowel disease. Gut 2013, 62, 387–394. [Google Scholar] [CrossRef]
- White, E.C.; Melmed, G.Y.; Vasiliauskas, E.; Dubinsky, M.; Ippoliti, A.; McGovern, D.; Targan, S.; Fleshner, P. Does Preoperative Immunosuppression Influence Unplanned Hospital Readmission after Surgery in Patients with Crohn’s Disease? Dis. Colon Rectum 2012, 55, 563–568. [Google Scholar] [CrossRef]
- Yamada, A.; Komaki, Y.; Patel, N.; Komaki, F.; Aelvoet, A.S.; Tran, A.L.; Pekow, J.; Dalal, S.; Cohen, R.D.; Cannon, L.; et al. Risk of Postoperative Complications among Inflammatory Bowel Disease Patients Treated Preoperatively with Vedolizumab. Am. J. Gastroenterol. 2017, 112, 1423–1429. [Google Scholar] [CrossRef]
- Yamamoto, T.; Spinelli, A.; Suzuki, Y.; Saad-Hossne, R.; Teixeira, F.V.; de Albuquerque, I.C.; da Silva, R.N.; de Barcelos, I.F.; Takeuchi, K.; Yamada, A.; et al. Risk factors for complications after ileocolonic resection for Crohn’s disease with a major focus on the impact of preoperative immunosuppressive and biologic therapy: A retrospective international multicentre study. United Eur. Gastroenterol. J. 2016, 4, 784–793. [Google Scholar] [CrossRef]
- Yu, C.S.; Jung, S.W.; Lee, J.L.; Lim, S.B.; Park, I.J.; Yoon, Y.S.; Kim, C.W.; Yang, S.K.; Ye, B.D.; Park, S.H.; et al. The Influence of Preoperative Medications on Postoperative Complications in Patients after Intestinal Surgery for Crohn’s Disease. Inflamm. Bowel Dis. 2019, 25, 1559–1568. [Google Scholar] [CrossRef]
- Zhu, F.; Li, Y.; Guo, Z.; Cao, L.; Feng, D.; Zhang, T.; Zhu, W.; Gong, J. Nomogram to Predict Postoperative Intra-abdominal Septic Complications after Bowel Resection and Primary Anastomosis for Crohn’s Disease. Dis. Colon Rectum 2020, 63, 629–638. [Google Scholar] [CrossRef]
- Zittan, E.; Milgrom, R.; Ma, G.W.; Wong-Chong, N.; O’Connor, B.; McLeod, R.S.; MacRae, H.M.; Greenberg, G.R.; Nguyen, G.C.; Croitoru, K.; et al. Preoperative Anti-tumor Necrosis Factor Therapy in Patients with Ulcerative Colitis Is Not Associated with an Increased Risk of Infectious and Noninfectious Complications after Ileal Pouch-anal Anastomosis. Inflamm. Bowel Dis. 2016, 22, 2442–2447. [Google Scholar] [CrossRef] [PubMed]
- Myrelid, P.; Söderholm, J.D.; Olaison, G.; Sjödahl, R.; Andersson, P. Split stoma in resectional surgery of high-risk patients with ileocolonic Crohn’s disease. Color. Dis. 2012, 14, 188–193. [Google Scholar] [CrossRef] [PubMed]



| Author and Year | Country | Years of Study | IBD Subtype † | Type of Anti-TNF-α Agent ‡ | Last Preoperative Anti-TNF-α Agent Exposure in Weeks | Duration of Postoperative Follow-Up in Days | Number of Patients | Number of Patients with Concomitant Preoperative Medication Use: Corticosteroids/Immunomodulators | NOS Score ¶ | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| I § | I § | I §/C § | I § | C § | I § | C § | |||||
| Appau et al., 2008 [46] | USA | 1998–2007 | CD | IFX | <12 | 30 | 60 | 329 | 39/n.a. | 253/n.a. | 7 |
| Bafford et al., 2013 [47] | USA | 1999–2010 | CD | U | <12 | 30 | 35 | 161 | 16/19 | 56/50 | 5 |
| Bregnbak et al., 2012 [48] | Denmark | 2005–2010 | UC | IFX | <12 | 30 | 20 | 51 | 15/n.a. | 33/n.a. | 5 |
| Brouquet et al., 2018 [49] | France | 2013–2015 | CD | ADA, IFX, U | <12 | 30 | 143 | 449 | 1/n.a. | 46/n.a. | 7 |
| Canedo et al., 2010 [50] | USA | 2001–2008 | CD | IFX | <12 | 30 | 65 | 160 | n.a. | n.a. | 6 |
| El-Hussuna et al., 2012 [51] | Denmark | 2000–2007 | CD | CZP, IFX | <12 | 30 | 32 | 385 | 28/11 | 135/135 | 6 |
| El-Hussuna et al., 2018 [52] | Denmark | 2014–2016 | IBD (CD/UC) | ADA, GO, IFX, U | <12 | 30 | 18 | 28 | 7/8 | 9/7 | 6 |
| Ferrante et al., 2009 [53] | Belgium | 1998–2008 | UC and IC | IFX | <12 | 30 | 22 | 119 | 14/13 | 82/65 | 5 |
| Gainsbury et al., 2011 [54] | USA | 2005–2009 | UC | IFX | <12 | 30 | 29 | 52 | 27/n.a. | 36/n.a. | 7 |
| Gu et al., 2013 (1) * [55] | USA | 2006–2010 | UC and IC | ADA, IFX, | 4–12 | 30 | 25 | 156 | 10/7 | 80/41 | 5 |
| Gu et al., 2013 (2) * [55] | ADA, CZP, IFX | 142 | 265 | 100/34 | 212/74 | ||||||
| Gutierrez et al., 2019 [56] | Spain | 2007–2010 | CD | U | 0 | 30 | 64 | 300 | n.a. | n.a. | 7 |
| Indar et al., 2009 [57] | USA | 1999–2007 | CD | IFX | <6 | 30 | 17 | 95 | 10/8 | 37/31 | 5 |
| Jouvin et al., 2018 [58] | France | 2002–2013 | CD | ADA, CZP, IFX | <8 | 30 | 55 | 305 | 15/29 | 139/118 | 4 |
| Kim et al., 2020 [59] | USA | 2007–2017 | UC | ADA, IFX | <12 | 30 | 74 | 54 | 48/62 | 34/24 | 7 |
| Kotze et al., 2017 [61] | Brazil | 2007–2014 | CD | ADA, IFX | <8 | 30 | 71 | 52 | 22/58 | 20/33 | 7 |
| Kotze et al., 2018 [60] | Brazil | 7-year period | CD | ADA | <8 | 30 | 25 | 25 | 8/22 | 11/17 | 6 |
| Krane et al., 2013 [62] | USA | 2004–2011 | IBD (CD/IC/UC) | IFX | <12 | 30 | 142 | 376 | 105/46 | 221/77 | 8 |
| Kunitake et al., 2008 [63] | USA | 1993–2007 | IBD (CD/IC/UC) | IFX | <12 | 30 | 101 | 312 | 76/37 | 240/81 | 5 |
| Lau et al., 2015 (1) * [21] | USA | 1999–2012 | CD | ADA, CZP, IFX | 0 | 30 | 50 | 73 | 43/n.a. | 56/n.a. | 6 |
| Lau et al., 2015 (2) * [21] | UC | 30 | 17 | 77 | 4/n.a. | 13/n.a. | |||||
| Lightner et al., 2019 [64] | USA | 2014–2017 | CD | ADA, CZP, IFX | <12 | 30 | 197 | 105 | 37/18 | 22/38 | 6 |
| Maeda et al., 2015 [65] | Japan | 2005–2013 | CD | ADA, IFX | <4 | 30 | 65 | 112 | n.a. | n.a. | 4 |
| Mascarenhas et al., 2012 [66] | USA | 2003–2010 | CD | IFX | <12 | 30 | 19 | 74 | n.a. | n.a. | 5 |
| Melo-Pinto et al., 2018 [22] | Portugal | 2010–2015 | CD | U | <12 | 30 | 62 | 106 | 13/n.a. | 33/n.a. | 7 |
| Monsinjon et al., 2017 [67] | France | 2006–2015 | IBD (CD/UC) | U | 0 | 30 | 25 | 40 | n.a. | n.a. | 5 |
| Morar et al., 2015 [68] | UK | 2005–2010 | CD | ADA, IFX | <4 | 30 | 4 | 126 | n.a. | n.a. | 6 |
| Myrelid et al., 2014 [69] | Sweden | 2005–2011 | CD | ADA, IFX | <8 | 30 | 111 | 187 | n.a. | n.a. | 5 |
| Nasir et al., 2010 [70] | USA | 2005–2009 | CD | ADA, CZP, IFX | <8 | 30 | 119 | 251 | 16/63 | 25/110 | 5 |
| Nelson et al., 2014 [71] | USA | 2006–2012 | UC | IFX | <4 | 30 | 24 | 50 | 37/32 | 114/83 | 5 |
| Nørgård et al., 2012 [72] | Denmark | 2003–2010 | UC | ADA, IFX, U | <12 | 30 | 199 | 1027 (D/M)/997 (C) | 24/0 | 50/19 | 6 |
| Nørgård et al., 2013 [73] | Denmark | 2000–2010 | CD | ADA, IFX, U | <12 | 30 | 214 (D/M)/213 (C) | 2079 (D/M)/2025 (C) | 49/n.a. | 300/n.a. | 6 |
| Regadas et al., 2011 [74] | USA | 2001–2008 | IBD (CD/IC/UC) | IFX | <8 | 30 | 28 | 221 | 19/n.a. | 294/n.a. | 5 |
| Rizzo et al., 2011 [75] | Italy | 2004–2010 | IBD (CD/UC) | ADA, CZP, IFX | <12 | 30 | 54 | 60 | 18/15 | 107/35 | 7 |
| Selvasekar et al., 2007 [76] | USA | 2002–2005 | UC | IFX | <8 | 30 | 23 | 254 | 19/21 | 29/6 | 7 |
| Serradori et al., 2013 [77] | France | 2000–2010 | CD | ADA, IFX | <12 | 30 | 42 | 175 | n.a. | n.a. | 5 |
| Shwaartz et al., 2016 [78] | USA | 2013–2015 | IBD (CD/UC) | ADA, CZP, IFX | <8 | 30 | 73 | 209 | 16/n.a. | 71/n.a. | 6 |
| Syed et al., 2013 [79] | USA | 2004–2011 | CD | ADA, CZP, IFX | <8 | 30 | 150 | 175 | 26/29 | 50/55 | 5 |
| Tang et al., 2020 [23] | China | 2014–2018 | CD | IFX | <8 | 30 | 85 | 305 | 23/53 | 30/63 | 6 |
| Tay et al., 2003 [80] | USA | 1998–2002 | CD | IFX | <8 | 30 | 22 | 78 | 35/n.a. | 85/n.a. | 7 |
| Tiberi et al., 2020 [81] | Japan | 2008–2019 | CD | U | <12 | 30 | 32 | 101 | 0/n.a. | 14/n.a. | 5 |
| Uchino et al., 2013 [84] | Japan | 2008–2011 | CD | IFX | <12 | 30 | 79 | 326 | n.a. | n.a. | 6 |
| Uchino et al., 2013 [85] | Japan | 2010–2012 | UC | IFX | <12 | 30 | 22 | 174 | 29/1 | 110/5 | 7 |
| Uchino et al., 2015 [83] | Japan | 2012–2014 | UC | U | <12 | 30 | 44 | 137 | 17/14 | 104/80 | 7 |
| Uchino et al., 2019 [82] | UK | 2015–2018 | UC | ADA, GO, IFX | <12 | 30 | 146 | 155 | n.a. | n.a. | 7 |
| Ward et al., 2018 [86] | Canada | 2006–2015 | UC | ADA, GO, IFX | <12 | 30 | 753 | 5472 | n.a. | n.a. | 6 |
| Waterman et al., 2012 [87] | USA | 2000–2010 | IBD (CD/IC/UC) | AD, IFX | <12 | 30 | 104 | 278 | n.a. | n.a. | 8 |
| White et al., 2012 [88] | USA | 1999–2009 | CD | ADA, CZP, IFX | <12 | 30 | 59 | 279 | 24/35 | 47/56 | 5 |
| Yamada et al., 2017 (1) * [89] | USA | 2014–2016 | CD | ADA, IFX, U | <4 | 30 | 96 | 121 | n.a. | n.a. | 8 |
| Yamada et al., 2017 (2) * [89] | UC | 33 | 129 | ||||||||
| Yamamoto et al., 2016 [90] | Japan | 2008–2013 | CD | ADA, IFX | <8 | 30 | 79 | 152 | n.a. | n.a. | 8 |
| Yu et al., 2019 [91] | Korea | 2006–2015 | CD | ADA, IFX | <8 | 30 | 122 | 339 | 26/31 | 42/34 | 5 |
| Zhu et al., 2020 [92] | China | 2011–2017 | CD | IFX | <8 | 30 | 98 | 614 | 20/66 | 85/296 | 6 |
| Zittan et al., 2016 [93] | Canada | 2002–2013 | UC | U | <4 | 30 | 27 | 562 | n.a. | n.a. | 7 |
| Author and Year | General Postoperative Complications † | Surgical-Site Postoperative Complications ‡ | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OPC, n | OIC, n | OC-DMC, n | RA, n | RO, n | M, n | OISSC, n | AL, n | IAA, n | IASC, n | |||||||||||
| I § | C § | I § | C § | I § | C § | I § | C § | I § | C § | I § | C § | I § | C § | I § | C § | I § | C § | I § | C § | |
| Appau et al., 2008 [46] | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | ↑12 | ↓31 | 5 | 10 | 1 | 0 | n.a. | n.a. | 6 | 14 | 6 | 14 | n.a. | n.a. |
| Bafford et al., 2013 [47] | 7 | 38 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 0 | 0 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Bregnbak et al., 2012 [48] | 10 | 25 | 4 | 21 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 0 | 0 | n.a. | n.a. | n.a. | n.a. | 1 | 2 | n.a. | n.a. |
| Brouquet et al., 2018 [49] | ↑64 | ↓112 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 0 | 0 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | ↑18 | ↓32 |
| Canedo et al., 2010 [50] | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 2 | 9 | n.a. | n.a. | n.a. | n.a. | 2 | 5 | 2 | 8 | n.a. | n.a. |
| El-Hussuna et al., 2012 [51] | 3 | 49 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| El-Hussuna et al., 2018 [52] | 5 | 8 | n.a. | n.a. | n.a. | n.a. | 4 | 7 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 1 | 2 |
| Ferrante et al., 2009 [53] | n.a. | n.a. | 2 | 29 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 0 | 0 | 1 | 23 | 0 | 15 | 0 | 9 | n.a. | n.a. |
| Gainsbury et al., 2011 [54] | 13 | 23 | 5 | 14 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 0 | 0 | n.a. | n.a. | n.a. | n.a. | 4 | 7 | n.a. | n.a. |
| Gu et al., 2013 (1) * [55] | 11 | 84 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Gu et al., 2013 (2) * [55] | 67 | 135 | n.a. | n.a. | n.a. | n.a. | 28 | 50 | 8 | 20 | 2 | 1 | n.a. | n.a. | 16 | 24 | 3 | 12 | n.a. | n.a. |
| Gutierrez et al., 2019 [56] | 22 | 42 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Indar et al., 2009 [57] | 6 | 27 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 0 | 0 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Jouvin et al., 2018 [58] | ↑20 | ↓67 | ↑14 | ↓44 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 1 | 2 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Kim et al., 2020 [59] | 33 | 20 | 8 | 11 | n.a. | n.a. | 11 | 7 | 0 | 0 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 2 | 0 | n.a. | n.a. |
| Kotze et al., 2017 [61] | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 8 | 1 | 12 | 7 | 2 | 2 | 26 | 11 | 6 | 6 | 10 | 2 | n.a. | n.a. |
| Kotze et al., 2018 [60] | 17 | 12 | n.a. | n.a. | 7 | 9 | 2 | 1 | 4 | 4 | 0 | 1 | 9 | 6 | 2 | 4 | 3 | 2 | n.a. | n.a. |
| Krane et al., 2013 [62] | 36 | 92 | 17 | 42 | 12 | 32 | 4 | 19 | 5 | 9 | 0 | 1 | n.a. | n.a. | 3 | 5 | 8 | 20 | n.a. | n.a. |
| Kunitake et al., 2008 [63] | 17 | 49 | 6 | 32 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 2 | 1 | n.a. | n.a. | 3 | 9 | n.a. | n.a. | n.a. | n.a. |
| Lau et al., 2015 (1) * [21] | 16 | 13 | 11 | 7 | n.a. | n.a. | 8 | 5 | n.a. | n.a. | 1 | 0 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Lau et al., 2015 (2) * [21] | 8 | 31 | 2 | 10 | n.a. | n.a. | 4 | 15 | n.a. | n.a. | 0 | 0 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Lightner et al., 2019 [64] | n.a. | n.a. | 33 | 30 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 16 | 6 |
| Maeda et al., 2015 [65] | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 8 | 27 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Mascarenhas et al., 2012 [66] | n.a. | n.a. | n.a. | n.a. | 2 | 3 | n.a. | n.a. | n.a. | n.a. | 0 | 0 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Melo-Pinto et al., 2018 [22] | 2 | 17 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Monsinjon et al., 2017 [67] | 7 | 12 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 0 | 0 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Morar et al., 2015 [68] | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | ↑2 | ↓10 |
| Myrelid et al., 2014 [69] | 38 | 54 | 18 | 26 | 12 | 20 | n.a. | n.a. | 9 | 13 | n.a. | n.a. | n.a. | n.a. | 8 | 15 | n.a. | n.a. | n.a. | n.a. |
| Nasir et al., 2010 [70] | 36 | 70 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 0 | 1 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 4 | 5 |
| Nelson et al., 2014 [71] | n.a. | n.a. | 6 | 12 | n.a. | n.a. | 3 | 12 | n.a. | n.a. | 0 | 0 | 4 | 8 | n.a. | n.a. | 2 | 2 | n.a. | n.a. |
| Nørgård et al., 2012 [72] | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 43 | 230 | 0 | 30 | n.a. | n.a. | 1 | 16 | n.a. | n.a. | n.a. | n.a. |
| Nørgård et al., 2013 [73] | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 16 | 175 | 1 | 54 | n.a. | n.a. | 8 | 56 | n.a. | n.a. | n.a. | n.a. |
| Regadas et al., 2011 [74] | 1 | 21 | 0 | 22 | n.a. | n.a. | n.a. | n.a. | 0 | 9 | 0 | 0 | n.a. | n.a. | 0 | 3 | 0 | 5 | n.a. | n.a. |
| Rizzo et al., 2011 [75] | 14 | 10 | 9 | 8 | n.a. | n.a. | 6 | 3 | 3 | 1 | 0 | 0 | n.a. | n.a. | 4 | 3 | n.a. | n.a. | n.a. | n.a. |
| Selvasekar et al., 2007 [76] | n.a. | n.a. | 6 | 25 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 0 | 0 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Serradori et al., 2013 [77] | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 0 | 0 | 9 | 15 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Shwaartz et al., 2016 [78] | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 6 | 11 | n.a. | n.a. | 0 | 0 | n.a. | n.a. | 4 | 11 | 4 | 4 | n.a. | n.a. |
| Syed et al., 2013 [79] | n.a. | n.a. | ↑54 | ↓44 | 47 | 47 | 28 | 23 | 24 | 23 | 2 | 1 | n.a. | n.a. | 9 | 9 | 21 | 18 | 28 | 27 |
| Tang et al., 2020 [23] | 35 | 94 | ↑22 | ↓43 | 19 | 61 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 24 | 50 | 5 | 18 | 2 | 6 | n.a. | n.a. |
| Tay et al., 2003 [80] | n.a. | n.a. | 3 | 8 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 1 | 3 | 2 | 4 | 3 | 8 |
| Tiberi et al., 2020 [81] | 2 | 4 | n.a. | n.a. | 2 | 4 | n.a. | n.a. | n.a. | n.a. | 0 | 0 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Uchino et al., 2013 [84] | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | ↓9 | ↑99 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Uchino et al., 2013 [85] | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | ↓1 | ↑46 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Uchino et al., 2015 [83] | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 5 | 32 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Uchino et al., 2019 [82] | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 16 | 25 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Ward et al., 2018 [86] | 94 | 656 | 35 | 270 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Waterman et al., 2012 [87] | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 25 | 41 | 3 | 12 | 1 | 1 | n.a. | n.a. | 2 | 15 | n.a. | n.a. | n.a. | n.a. |
| White et al., 2012 [88] | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 7 | 21 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Yamada et al., 2017 (1) * [89] | 25 | 41 | 10 | 16 | n.a. | n.a. | 4 | 8 | 6 | 4 | 0 | 0 | n.a. | n.a. | 2 | 2 | 6 | 8 | n.a. | n.a. |
| Yamada et al., 2017 (2) * [89] | 15 | 48 | 9 | 18 | n.a. | n.a. | 2 | 4 | 2 | 8 | 0 | 0 | n.a. | n.a. | 2 | 2 | 2 | 10 | n.a. | n.a. |
| Yamamoto et al., 2016 [90] | 17 | 38 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 7 | 12 | n.a. | n.a. | 9 | 18 |
| Yu et al., 2019 [91] | 37 | 167 | 28 | 126 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Zhu et al., 2020 [92] | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | ↑27 | ↓56 |
| Zittan et al., 2016 [93] | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | 2 | 66 | 3 | 96 | n.a. | n.a. |
| Readmission | Intra-Abdominal Abscesses | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OR with 95% CI–FE | Heterogeneity | Egger’s Test [45] | OR with 95% CI–FE | Heterogeneity | Egger’s Test [45] | ||||||||
| OR, 1.39; 95% CI, 1.11–1.73; p = 0.004 [↑ (I); ↓(C)] | I2 = 0%; p = 0.45 | p = 0.994 | OR, 1.19; 95% CI, 0.89–1.60; p = 0.24 | I2 = 0%; p = 0.76 | p = 0.850 | ||||||||
| Subgroup analysis | |||||||||||||
| Subgroups | OR with 95% CI–FE | TSD | SA | TSD | Subgroups | OR with 95% CI–FE | TSD | SA | TSD | ||||
| IBD | CD | OR, 1.77; 95% CI, 1.23–2.52; p = 0.002 [↑ (I); ↓(C)] | p = 0.15 | CD | p = 0.05 | IBD | CD | OR, 1.50; 95% CI, 1.00–2.25; p = 0.05 [↑ (I); ↓(C)] | p = 0.15 | CD | p = 0.05 | ||
| UC/IC | OR, 1.04; 95% CI, 0.70–1.54; p = 0.86 | UC/IC | UC/IC | OR, 0.76; 95% CI, 0.43–1.32; p = 0.33 | UC/IC | ||||||||
| IBD | OR, 1.40; 95% CI, 0.94–2.08; p = 0.10 | x | IBD | OR, 1.30; 95% CI, 0.66–1.60; p = 0.45 | x | ||||||||
| Potential risk factors | |||||||||||||
| OA | >50% | OR, 2.01; 95% CI, 1.32–3.08; p = 0.001 [↑ (I); ↓(C)] | p = 0.06 | >50% | p = 0.02 | OA | >50% | OR, 1.37; 95% CI, 0.91–2.06; p = 0.13 | p = 0.62 | >50% | p = 0.35 | ||
| <50% | OR, 1.01; 95% CI, 0.70–1.47; p = 0.94 | <50% | <50% | OR, 1.00; 95% CI, 0.58–1.70; p = 0.99 | <50% | ||||||||
| U | OR, 1.43; 95% CI, 1.00–2.07; p = 0.05 [↑ (I); ↓(C)] | x | U | OR, 1.09; 95% CI, 0.55–2.16; p = 0.80 | x | ||||||||
| ELEMS | EL | OR, 1.21; 95% CI, 0.69–2.13; p = 0.50 | p = 0.35 | EL | p = 0.98 | ELEMS | EL | OR, 1.65; 95% CI, 0.95–2.87; p = 0.07 | p = 0.38 | EL | p = 0.16 | ||
| ELEMS | OR, 1.22; 95% CI, 0.88–1.70; p = 0.23 | ELEMS | ELEMS | OR, 1.01; 95% CI, 0.66–1.54; p = 0.96 | ELEMS | ||||||||
| U | OR, 1.70; 95% CI, 1.20–2.40; p = 0.003 [↑ (I); ↓(C)] | x | U | OR, 1.16; 95% CI, 0.63–2.12; p = 0.64 | x | ||||||||
| PRI | <50% | OR, 1.79; 95% CI, 1.32–2.42; p = 0.0002 [↑ (I); ↓(C)] | p = 0.06 | <50% | p = 0.03 | PRI | <50% | OR, 1.58; 95% CI, 1.06–2.37; p = 0.02 [↑ (I); ↓(C)] | p = 0.13 | <50% | p = 0.05 | ||
| >50% | OR, 1.07; 95% CI, 0.76–1.51; p = 0.69 | >50% | >50% | OR, 0.80; 95% CI, 0.47–1.37; p = 0.42 | >50% | ||||||||
| U | OR, 0.87; 95% CI, 0.32–2.38; p = 0.79 | x | U | OR, 1.03; 95% CI, 0.48–2.20; p = 0.94 | x | ||||||||
| CSIM | >50% | OR, 1.27; 95% CI, 0.92–1.74; p = 0.15 | p = 0.73 | >50% | p = 0.56 | CSIM | >50% | OR, 1.28; 95% CI, 0.86–1.91; p = 0.22 | p = 0.53 | >50% | p = 1.0 | ||
| <50% | OR, 1.48; 95% CI, 0.96–2.28; p = 0.07 | <50% | <50% | OR, 1.28; 95% CI, 0.76–2.16; p = 0.35 | <50% | ||||||||
| U | OR, 1.54; 95% CI, 1.01–2.37; p = 0.05 | x | U | OR, 0.80; 95% CI, 0.37–1.72; p = 0.56 | x | ||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cira, K.; Weber, M.-C.; Wilhelm, D.; Friess, H.; Reischl, S.; Neumann, P.-A. The Effect of Anti-Tumor Necrosis Factor-Alpha Therapy within 12 Weeks Prior to Surgery on Postoperative Complications in Inflammatory Bowel Disease: A Systematic Review and Meta-Analysis. J. Clin. Med. 2022, 11, 6884. https://doi.org/10.3390/jcm11236884
Cira K, Weber M-C, Wilhelm D, Friess H, Reischl S, Neumann P-A. The Effect of Anti-Tumor Necrosis Factor-Alpha Therapy within 12 Weeks Prior to Surgery on Postoperative Complications in Inflammatory Bowel Disease: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine. 2022; 11(23):6884. https://doi.org/10.3390/jcm11236884
Chicago/Turabian StyleCira, Kamacay, Marie-Christin Weber, Dirk Wilhelm, Helmut Friess, Stefan Reischl, and Philipp-Alexander Neumann. 2022. "The Effect of Anti-Tumor Necrosis Factor-Alpha Therapy within 12 Weeks Prior to Surgery on Postoperative Complications in Inflammatory Bowel Disease: A Systematic Review and Meta-Analysis" Journal of Clinical Medicine 11, no. 23: 6884. https://doi.org/10.3390/jcm11236884
APA StyleCira, K., Weber, M.-C., Wilhelm, D., Friess, H., Reischl, S., & Neumann, P.-A. (2022). The Effect of Anti-Tumor Necrosis Factor-Alpha Therapy within 12 Weeks Prior to Surgery on Postoperative Complications in Inflammatory Bowel Disease: A Systematic Review and Meta-Analysis. Journal of Clinical Medicine, 11(23), 6884. https://doi.org/10.3390/jcm11236884





