Abstract
Introduction: Atrial fibrillation (AF) recurrence following pulmonary vein isolation (PVI) ablation has clinical significance. Identifying risk factors for AF recurrence is important. We investigated serum albumin (SA) levels (g/dL) as a prognostic factor for the recurrence of AF following cryoballoon PVI ablation. Methods: We included patients who underwent cryoballoon PVI ablation at our institution between the years 2013 and 2018. The primary outcome was recurrence of AF during follow up. Results: Our cohort consisted of 126 patients (67% males, mean age 61.8 ± 10.0 years). The pattern of AF amongst the cohort was paroxysmal in 62.5%, persistent in 25.4%, and longstanding persistent in 6.3%. Those with lower SA levels had a mean AF duration significantly less than those with higher SA levels (2.81 years, 7.34 years, and 6.37 years for SA levels of <3.8, 3.8–4.1, and ≥4.1, respectively; p = 0.003). Patients with lower SA levels were significantly more likely to have had more previous cardioversions and a larger left atrial area and volume. The mean follow-up was 380 days, in which the AF recurrence rate was 20.6%. Patients with lower SA level had significantly more AF recurrences (47.4%, 16.7%, and 2.2% for SA levels of <3.8, 3.8–4.1, and ≥4.1, respectively; p < 0.001). Upon multivariate analysis, an SA level < 3.8 was associated with a higher risk of AF recurrence (OR = 5.422 95% CI 1.134; 25.910; p < 0.001). Conclusion: SA levels were found to be a strong independent marker for AF recurrence following PVI ablation.
1. Introduction
Atrial fibrillation (AF) is the most common sustained arrhythmia worldwide, with a prevalence of up to 4% of the adult population [1]. It is associated with significant morbidity and mortality [2]. The maintenance of sinus rhythm is associated with improved symptom control and improvement of quality of life [2]. There is increasing evidence that this strategy is also associated with improved cardiovascular outcomes [3]. AF catheter ablation with pulmonary vein isolation has proven to be an effective and safe alternative to anti-arrhythmic drugs [4,5]. However, AF recurrence rates following pulmonary vein isolation (PVI) ablation are substantial. In contemporary randomized control trials, recurrence rates range up to 34–43% at one year following the procedure [6,7]. Various risk factor for AF recurrence have been described, and these should be taken into consideration in shared decision making with the patient [2]. Studies have shown that albumin levels are associated with the incidence of new onset AF [8,9]. Albumin as a prognostic marker of AF recurrence following PVI ablation has yet to be investigated. We investigated serum albumin (SA, g/dL) levels as a prognostic factor for the recurrence of AF following PVI ablation.
2. Materials and Methods
We included 126 consecutive patients with AF who had cryoballoon PVI ablation performed at our institution between the years 2013 and 2018. Patients with an inflammatory/rheumatic condition, active infection, acute illness, active malignancy, major surgery in the prior 6 months, and chronic liver disease/cirrhosis, and patients with a previous PVI ablation were excluded from this study (n = 9). Retrospectively, patient demographic data, echocardiographic parameters, and procedural data were collected from patient files. Medical conditions were obtained from patient clinic files and according to the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes, if available. Patients with AF were classified according to the Heart Rhythm Society expert consensus statement as follows: paroxysmal, persistent, and longstanding persistent [8,9]. Each patient file was reviewed, and the data of AF duration were taken from hospital records according to the dates recorded of initial diagnosis of atrial fibrillation on the electronic filing system. Symptoms alone without documentation of atrial fibrillation were not regarded as sufficient.
2.1. Ablation
The ablation procedure was performed under conscious sedation with midazolam and fentanyl or under general anesthesia. All PVI ablation procedures included in this cohort underwent cryoballoon PVI ablation. The PVI ablation was performed with a 15 F steerable trans-septal sheath (FlexCath), an Arctic Front Advance cryoballoon catheter, and an Achieve Mapping Catheter (Medtronic, Minneapolis, MN, USA). This procedure was done under fluoroscopic guidance and after trans-septal puncture, as previously described [10]. In brief, the cryoballoon was inserted into the pulmonary vein antrum and was then filled with a refrigerant to cool and occlude the tissue with the aim of creating electrical isolation of the tissue, as seen on electrophysiological recordings. The cooling was stopped if the temperature dropped below −55 degrees Celsius [11].
The procedure endpoint was electrical isolation between the pulmonary veins and left atrium, as confirmed with electrical entrance block. Patients were followed in our outpatient clinic.
2.2. Study Groups
Valid ambulatory SA levels within 24 h prior to the PVI ablation were obtained. This was included as part of the routine laboratory tests done prior to the procedure. SA levels were classified (as tertiles) as follows: T1, <3.8; T2, 3.8 to 4.19; and T3, ≥4.2 g/dL. SA levels were measured using a photometric color test for clinical chemistry analyzers.
2.3. Follow Up and Outcomes
Follow up duration was 1 year. Follow up was performed at 3, 6, 9, and 12 months. Prior to each follow up visit, patients underwent ambulatory Holter monitoring for 48 h. Antiarrhythmic drugs were ceased at the discretion of the treating physician at the 3-month follow up visit. Anticoagulation treatment was continued in all patients for at least 3 months, and then it was continued or ceased according to the stroke risk based on the CHA2DS2VASc score. Documentation of arrhythmia was taken from clinic notes and patient file documentation. The primary outcome was AF recurrence during follow up, which was defined as at least one episode of atrial arrhythmia that lasted more than 30s on Holter monitoring or symptomatic AF documented on electrocardiogram in patients’ files after a 3-month blanking period (therapy stabilization period) post ablation. If patients were symptomatic and complained of palpitations, they were invited for additional clinical review and Holter monitoring.
The study was a retrospective analysis and was conducted in compliance with the principles outlined in the declaration of Helsinki, and it was approved by the institutional Ethics Committee.
2.4. Statistical Analysis
Statistical analysis was performed using IBM SPSS 26 (SPSS Inc., Chicago, IL, USA) software. Patient characteristics were presented as mean ± standard deviation (SD) for continuous variables; range, median, and interquartile range (IQR) for count data; and numbers (n) and percent (%) for categorical data. Comparisons of baseline and follow up characteristics between the study groups were performed using Chi-square tests for categorical variables, non-parametric Kruskal–Wallis tests for count data, and analysis of variance (ANOVA) tests for continuous variables.
Associations between SA level (in the presented categories) and the risk recurrent AF in the univariate and multivariate levels were assessed using binomial logistic regression. The primary independent variable (SA) was used in the multivariable model using the Enter method. Other investigated independent variables were introduced to the model using the forward stepwise method. The results of the models were presented as the regression coefficients (B) and their standard errors (SE), odds ratios (OR)/adjusted odds ratios (adjOR), and 95% confidence intervals (CIs) for OR. For each test, p < 0.05 was considered statistically significant.
3. Results
Our cohort consisted of 126 patients, of whom 67% were male with mean age of 61.8 ± 10.0 years. Baseline characteristics of the total cohort and according to the SA tertiles are shown in Table 1, Table 2 and Table 3. Prominent comorbidities amongst the cohort included hypertension (59%), dyslipidemia (46.8%), and coronary artery disease (22.2%). The majority of the cohort had good left ventricular function (62.5%). The pattern of AF amongst the cohort was mostly paroxysmal in 62.5%, persistent in 25.4%, and long standing persistent in 6.3%. A quarter of the cohort had previous atrial flutter, and 20.6% had previously undergone cavotricuspid isthmus ablation for atrial flutter. The majority of the cohort (92.9%) had previously been treated with an antiarrhythmic drug, of which the most commonly used were propafenone (51.6%) followed by amiodarone (50.0%).

Table 1.
Baseline characteristics (demographics and comorbidities) according to serum albumin (SA) level group.

Table 2.
Atrial fibrillation (AF) characteristics (at the baseline) according to serum albumin (SA) level group.

Table 3.
Treatment and echocardiogram measurements (at the baseline) according to serum albumin (SA) level group.
3.1. Comparison across SA Tertiles
Those with lower SA levels had mean AF durations significantly shorter than those with higher SA levels (2.81 years, 7.34 years, and 6.37 years in those with SA levels of T1, T2, and T3, respectively; p = 0.003). Patients with lower SA levels were significantly more likely to have had more previous cardioversions than those with higher SA levels (p = 0.002). Notably, left atrial areas were significantly larger with lower SA tertiles (24.97 ± 4.84 cm2, 22.79 ± 5.09 cm2, and 21.55 ± 4.46 cm2 in T1, T2, and T3, respectively; p = 0.024). There were also significant differences in mean left atrial volumes (mL/m2) in accordance with SA level (136.24 ± 42.56, 119.76 ± 34.96, and 105.54 ± 27.73 in T1, T2, and T3, respectively; p = 0.002).
3.2. Follow up and Outcome
Follow up data are shown in Table 4. There were follow up data for 123 participants, of which the mean follow up was 380 days. The overall AF recurrence rate was 20.6% (n = 26), as shown in Figure 1. Patients in the lower SA tertile had significantly more AF recurrences (47.4%, 16.7%, and 2.2% in T1, T2, and T3, respectively; p < 0.001). During the 3-month blanking period following the PVI ablation, those with higher SA had significantly fewer recurrences compared to those with lower SA levels (24.34%, 12.2%, and 4.8%, in T1, T2, and T3, respectively; p = 0.037).

Table 4.
Follow up characteristics by serum albumin (SA) level group.
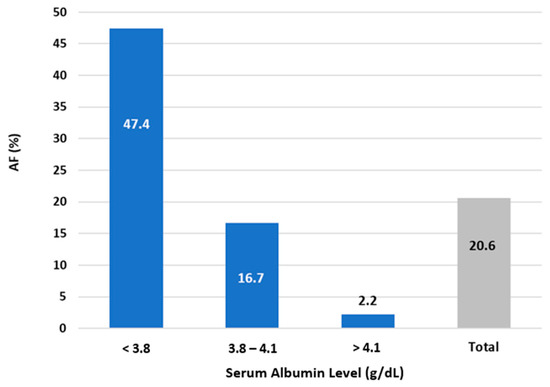
Figure 1.
Atrial fibrillation (AF) recurrence rate following pulmonary vein isolation (PVI) ablation according to serum albumin (SA) level group.
3.3. Association between SA Level and Recurrent AF
On univariate analysis SA levels < 3.8g/dL were nine times more likely to have a recurrence of AF following PVI ablation [OR = 9.00 (95% CI 3.424; 23.653), p < 0.001], compared to those with albumin levels ≥ 3.8g/dL. The results of the multivariate model (Table 5) showed a statistically significant association between SA and AF recurrence, with the lower categories of SA independently associated with a higher rate of AF recurrence following PVI ablation. Three parameters were found to be strong independent markers for AF recurrence upon multivariate analysis: SA levels > 3.8 with a adjOR = 5.422 (95% CI 1.134; 25.910), p < 0.001, followed by left atrial diameter with an adjOR = 1.206 (95% CI 1.026; 1.416), p = 0.023, for every 1 cm increase in diameter as well as direct current cardioversion with an adjOR = 2.596 (95% CI 1.411; 4.777), p = 0.002. The pattern of AF (paroxysmal vs. persistent/long persistent) was a significant marker for AF recurrence on univariate analysis with OR = 2.849 (95% CI 1.170; 6.944), p = 0.021, but this was not a significant factor on multivariable analysis.

Table 5.
Risk factors for atrial fibrillation (AF) recurrence following pulmonary vein isolation (PVI) ablation—univariate and multivariate analyses.
4. Discussion
Our study’s main finding is that albumin is a strong independent prognostic marker of AF recurrence following PVI ablation. Upon multivariate analysis, those with SA levels lower than 3.8g/dL were 5.4 times more likely to have AF recurrence following PVI ablation compared to patients with SA ≥ 3.8 g/dL. This was more influential than the risk associated with left atrial diameter or previous cardioversion. To our knowledge, this is the first study that has investigated the prognostic value of SA as a risk factor for AF recurrence.
Epidemiological data are growing, linking low SA levels to the incidence of a variety of cardiovascular diseases [12]. Recent data are accumulating linking albumin as a risk factor in the incidence of AF [13,14]. On an epidemiological level, albumin was shown by Liao et al. to be inversely associated to the future risk of AF in a population-based prospective cohort without pre-existing AF (hazard ratio (HR) = 0.90 (95% CI 0.86; 0.94) [14]. Van Beek et al. [13] showed a similar association amongst patients in intensive care. Lower SA levels were associated with the early development of AF during admission as well as with an increased number of AF episodes. This association was linear, with a lowered risk of AF by 14% (95% CI 3%; 23%) for every gram per deciliter increase in SA. We focused our study on albumin as a biomarker that has been researched in the cardiovascular sphere and is known to have many important biochemical properties. We did not use total serum protein for risk stratification, as this measure includes many proteins, including albumin and globulins. The globulin fraction is made up of different proteins, including immunoglobulins, enzymes, and carrier proteins, which all have important physiological roles, including those in the immune system. We focused our risk stratification on albumin in an aim to be more specific and to not be confounded by these other protein fractions.
The postulation is that albumin is a marker of the nutritional status of the patients and is an indicator of underlying health status. However, there are further physiological reasons that albumin could be an important disease risk factor. Firstly, the physiological properties of SA are multiple and are related to inflammatory properties, the coagulation pathway, and anti-oxidant activity [12,15,16]. Albumin is a negative acute phase protein, and hypoalbuminemia is a maker of a heightened inflammatory state of the patient. Indeed, several inflammatory biomarkers have been shown to be associated with the incidence and perpetuation of AF. Both c-reactive protein and interleukin-6 have been associated with AF recurrence following catheter ablation [17,18]. It is hypothesized that cardiac fibroblasts are stimulated by inflammation mediators, which contribute to atrial fibrosis and the ensuing atriopathy, which increases the risk and persistence of AF [19].
We found that patients with lower SA levels had significantly shorter durations of AF. We hypothesize that this could be due to lower SA levels being a marker of the chronicity of comorbid condition systemic diseases, and AF may be a later manifestation of various comorbid conditions such as advancing age, hypertension, etc. [2].
Albumin also binds both endogenous and exogenous substances. It is an important factor in drug bioavailability affecting drug pharmacokinetics, duration of drug activity, and metabolism of drugs [20]. It also has a fundamental role in maintaining plasma colloid osmotic pressure, preventing extravascular fluid extravasation [21]. Furthermore, SA levels have been associated with increased extracellular volume, a noninvasive marker of myocardial fibrosis on magnetic resolution imaging, in patients with various cardiovascular diseases such as aortic stenosis and heart failure [19,22]. Lower albumin could be a marker of myocardial fibrosis and could be a marker of the atrial fibrosis in patients with AF. Indeed, we found that lower SA levels were significantly associated with more previous cardioversions for AF as well as larger atrial areas and volumes. This could be a surrogate marker for the severity of the atriopathy of AF and thus could be associated with an increased risk of recurrence following PVI ablation.
Upon multivariate analysis, we found that AF recurrence following PVI ablation was significant, with a cut-off of SA levels lower than 3.8g/dL. SA levels less than 3.5g/dL are usually regarded as the cut-off for hypoalbuminemia. However, it has been shown that albumin levels within the lower normal range of 3.5–4.0g/dL have an association with poorer outcomes in other cardiovascular conditions [23]. A decreased admission serum albumin level has been found to be an independent predictor of long-term mortality in hospital survivors of acute myocardial infarction [24]. It is important to note that patients with low SA levels, as well as lower normal range SA, are at increased risk of AF recurrence following PVI ablation.
Our study is the first to show a significant association between SA levels and recurrence of AF following PVI ablation. Recurrence of AF has clinical significance, not only in terms of patients’ symptomatology, but also in terms of cardiovascular outcomes. The EAST-AFNET 4 Trial found that early rhythm control was significantly associated with a lower risk of adverse cardiovascular outcomes. This trial was ended prematurely due to a significantly lower composite of death from cardiovascular causes, stroke, or hospitalization with worsening of heart failure or acute coronary syndrome amongst those with early rhythm control [3]. PVI ablation has been shown to be an effective form of rhythm control. However, contemporary randomized control trials have shown that the documentation of AF recurrence following the procedure remains significant [6,10]. Identification of risk factors can assist in selecting patients who will have a higher chance of maintaining sinus rhythm following the procedure.
Multiple risk factors for AF recurrence have been identified, with many being linked to atrial size and function, atriopathy associated with AF, and the nature of AF [25,26,27]. Our study is the first to suggest that albumin could be a modifiable risk factor for AF recurrence.
Our study has limitations that include the retrospective design of the study and a relatively small sample size, which also limited our ability to add additional AF risk factors into the modelling of outcomes. It is important to note that our study found an association between SA and AF recurrence following PVI ablation. However, our study was not a mechanistic study, and our findings are hypothesis generating and not mechanistic in nature. Renal function was not assessed as correlated to AF recurrence following PVI ablation, and this is a further limitation. Endocardial mapping was not done in our cohort, and the relationship between quantity of low voltage areas and SA levels was not assessed. The outcome was collected from 24–48-h Holter monitoring done during 3-month periods or documentation in patient file data. In many cases it was not possible to report the exact date of recurrence. Accordingly, a logistic regression model was used and not a Cox regression model.
5. Conclusions
In our single-center cohort, SA levels were found to be a strong independent marker for AF recurrence following PVI ablation. This relationship may be multifactorial in nature, and further research is needed to clarify this association.
Author Contributions
Conceptualization, N.S.P. and A.S.; Methodology, N.S.P., G.G. (Gregory Golovchiner), Y.P., K.S., T.G., Y.A. and A.S.; Formal analysis, G.G. (Gregory Golovchiner), Y.P., K.S. and Y.A.; Investigation, G.G. (Gustavo Goldenberg), S.T., A.B., E.K., A.E. and A.S.; Resources, M.W., S.T., A.B., E.K., I.N.A., R.K. and A.H.; Data curation, G.G. (Gustavo Goldenberg), Y.P., A.E., T.G. and I.N.A.; Writing—original draft, N.S.P., M.W., A.S., and A.H.; Writing—review and editing, G.G. (Gregory Golovchiner), G.G. (Gustavo Goldenberg), M.W., S.T., A.B., E.K., A.E., K.S., T.G., I.N.A., Y.A. and R.K.; Supervision, R.K. and A.H. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by Rabin Medical Center Helsinki Committee (Approval Code: 0594-21 RMC, Approval Date: 7 December 2021).
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to ethical restrictions and patient data that was taken from Rabin Medical Center.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Benjamin, E.J.; Muntner, P.; Alonso, A.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Das, S.R.; et al. Heart disease and stroke statistics—2019 update: A report from the American heart association. Circulation 2019, 139, e56–e528. [Google Scholar] [CrossRef]
- Hindricks, G.; Potpara, T.; Dagres, N.; Arbelo, E.; Bax, J.J.; Blomström-Lundqvist, C.; Boriani, G.; Castella, M.; Dan, G.-A.; Dilaveris, P.E.; et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS): The Task Force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) Developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. Eur. Heart J. 2021, 42, 373–498. [Google Scholar] [CrossRef] [PubMed]
- Kirchhof, P.; Camm, A.J.; Goette, A.; Brandes, A.; Eckardt, L.; Elvan, A.; Fetsch, T.; van Gelder, I.C.; Haase, D.; Haegeli, L.M.; et al. Early Rhythm-Control Therapy in Patients with Atrial Fibrillation. N. Engl. J. Med. 2020, 383, 1305–1316. [Google Scholar] [CrossRef] [PubMed]
- Pappone, C.; Augello, G.; Sala, S.; Gugliotta, F.; Vicedomini, G.; Gulletta, S.; Paglino, G.; Mazzone, P.; Sora, N.; Greiss, I.; et al. A Randomized Trial of Circumferential Pulmonary Vein Ablation Versus Antiarrhythmic Drug Therapy in Paroxysmal Atrial Fibrillation: The APAF Study. J. Am. Coll. Cardiol. 2006, 48, 2340–2347. [Google Scholar] [CrossRef] [PubMed]
- Nyong, J.; Amit, G.; Adler, A.J.; Owolabi, O.O.; Perel, P.; Prieto-Merino, D.; Lambiase, P.; Casas, J.P.; Morillo, C.A. Efficacy and safety of ablation for people with non-paroxysmal atrial fibrillation. Cochrane Database Syst. Rev. 2011, 2016, CD012088. [Google Scholar] [CrossRef] [PubMed]
- Andrade, J.G.; Wells, G.A.; Deyell, M.W.; Bennett, M.; Essebag, V.; Champagne, J.; Roux, J.-F.; Yung, D.; Skanes, A.; Khaykin, Y.; et al. Cryoablation or Drug Therapy for Initial Treatment of Atrial Fibrillation. N. Engl. J. Med. 2021, 384, 305–315. [Google Scholar] [CrossRef] [PubMed]
- Kuck, K.-H.; Brugada, J.; Fürnkranz, A.; Metzner, A.; Ouyang, F.; Chun, K.J.; Elvan, A.; Arentz, T.; Bestehorn, K.; Pocock, S.J.; et al. Cryoballoon or Radiofrequency Ablation for Paroxysmal Atrial Fibrillation. N. Engl. J. Med. 2016, 374, 2235–2245. [Google Scholar] [CrossRef] [PubMed]
- January, C.T.; Wann, L.S.; Alpert, J.S.; Calkins, F.H.; Cigarroa, J.E.; Cleveland, J.C.; Conti, J.B.; Ellinor, P.; Ezekowitz, M.D.; Field, M.E.; et al. 2014 AHA/ACC/HRS Guideline for the Management of Patients With Atrial Fibrillation: A report of the American College of Cardiology/American Heart Association Task Force on practice guidelines and the Heart Rhythm Society. Circulation 2014, 130, e199–e267. [Google Scholar] [CrossRef]
- Calkins, H.; Hindricks, G.; Cappato, R.; Kim, Y.-H.; Saad, E.B.; Aguinaga, L.; Akar, J.G.; Badhwar, V.; Brugada, J.; Camm, J.; et al. 2017 HRS/EHRA/ECAS/APHRS/SOLAECE expert consensus statement on catheter and surgical ablation of atrial fibrillation. Heart Rhythm. 2017, 14, e275–e444. [Google Scholar] [CrossRef]
- Wazni, O.M.; Dandamudi, G.; Sood, N.; Hoyt, R.; Tyler, J.; Durrani, S.; Niebauer, M.; Makati, K.; Halperin, B.; Gauri, A.; et al. Cryoballoon Ablation as Initial Therapy for Atrial Fibrillation. N. Engl. J. Med. 2021, 384, 316–324. [Google Scholar] [CrossRef]
- Su, W.; Aryana, A.; Passman, R.; Singh, G.; Hokanson, R.; Kowalski, M.; Andrade, J.; Wang, P. Cryoballoon Best Practices II: Practical guide to procedural monitoring and dosing during atrial fibrillation ablation from the perspective of experienced users. Heart Rhythm 2018, 15, 1348–1355. [Google Scholar] [CrossRef]
- Arques, S. Human serum albumin in cardiovascular diseases. Eur. J. Intern. Med. 2018, 52, 8–12. [Google Scholar] [CrossRef]
- van Beek, D.E.; Kuijpers, Y.A.; Königs, M.H.; van der Horst, I.C.; Scheeren, T.W. Low serum albumin levels and new-onset atrial fibrillation in the ICU: A prospective cohort study. J. Crit. Care 2020, 56, 26–30. [Google Scholar] [CrossRef]
- Liao, L.-Z.; Zhang, S.-Z.; Li, W.-D.; Liu, Y.; Li, J.-P.; Zhuang, X.-D.; Liao, X.-X. Serum albumin and atrial fibrillation: Insights from epidemiological and mendelian randomization studies. Eur. J. Epidemiol. 2020, 35, 113–122. [Google Scholar] [CrossRef]
- Roche, M.; Rondeau, P.; Singh, N.R.; Tarnus, E.; Bourdon, E. The antioxidant properties of serum albumin. FEBS Lett. 2008, 582, 1783–1787. [Google Scholar] [CrossRef]
- Paar, M.; Rossmann, C.; Nusshold, C.; Wagner, T.; Schlagenhauf, A.; Leschnik, B.; Oettl, K.; Koestenberger, M.; Cvirn, G.; Hallström, S. Anticoagulant action of low, physiologic, and high albumin levels in whole blood. PLoS ONE 2017, 12, e0182997. [Google Scholar] [CrossRef]
- Jiang, Z.; Dai, L.; Song, Z.; Li, H.; Shu, M. Association Between C-Reactive Protein and Atrial Fibrillation Recurrence After Catheter Ablation: A Meta-analysis. Clin. Cardiol. 2013, 36, 548–554. [Google Scholar] [CrossRef]
- Wu, N.; Xu, B.; Xiang, Y.; Wu, L.; Zhang, Y.; Ma, X.; Tong, S.; Shu, M.; Song, Z.; Li, Y.; et al. Association of inflammatory factors with occurrence and recurrence of atrial fibrillation: A meta-analysis. Int. J. Cardiol. 2013, 169, 62–72. [Google Scholar] [CrossRef]
- Harada, M.; Nattel, S. Implications of Inflammation and Fibrosis in Atrial Fibrillation Pathophysiology. Card. Electrophysiol. Clin. 2021, 13, 25–35. [Google Scholar] [CrossRef]
- Nicholson, J.; Wolmarans, M.; Park, G. The role of albumin in critical illness. Br. J. Anaesth. 2000, 85, 599–610. [Google Scholar] [CrossRef]
- Arques, S.; Ambrosi, P. Human Serum Albumin in the Clinical Syndrome of Heart Failure. J. Card. Fail. 2011, 17, 451–458. [Google Scholar] [CrossRef]
- Prenner, S.B.; Kumar, A.; Zhao, L.; Cvijic, M.E.; Basso, M.; Spires, T.; Li, Z.; Yarde, M.; Bhattacharya, P.; Zamani, P.; et al. Effect of Serum Albumin Levels in Patients with Heart Failure With Preserved Ejection Fraction (from the TOPCAT Trial). Am. J. Cardiol. 2020, 125, 575–582. [Google Scholar] [CrossRef]
- Kurtul, A.; Murat, S.N.; Yarlioglues, M.; Duran, M.; Ocek, A.H.; Koseoglu, C.; Celık, I.E.; Kilic, A.; Aksoy, O. Usefulness of Serum Albumin Concentration to Predict High Coronary SYNTAX Score and In-Hospital Mortality in Patients With Acute Coronary Syndrome. Angiology 2016, 67, 34–40. [Google Scholar] [CrossRef]
- Plakht, Y.; Gilutz, H.; Shiyovich, A. Decreased admission serum albumin level is an independent predictor of long-term mortality in hospital survivors of acute myocardial infarction. Soroka Acute Myocardial Infarction II (SAMI-II) project. Int. J. Cardiol. 2016, 219, 20–24. [Google Scholar] [CrossRef]
- Chen, L.Y.; Chung, M.K.; Allen, L.A.; Ezekowitz, M.; Furie, K.L.; McCabe, P.; Noseworthy, P.A.; Perez, M.V.; Turakhia, M.P.; American Heart Association Council on Clinical Cardiology; et al. Atrial Fibrillation Burden: Moving Beyond Atrial Fibrillation as a Binary Entity: A Scientific Statement From the American Heart Association. Circulation 2018, 137, e623–e644. [Google Scholar] [CrossRef]
- Njoku, A.; Kannabhiran, M.; Arora, R.; Reddy, P.; Gopinathannair, R.; Lakkireddy, D.; Dominic, P. Left atrial volume predicts atrial fibrillation recurrence after radiofrequency ablation: A meta-analysis. Europace 2018, 20, 33–42. [Google Scholar] [CrossRef]
- Marrouche, N.F.; Wilber, D.; Hindricks, G.; Jais, P.; Akoum, N.; Marchlinski, F.; Kholmovski, E.; Burgon, N.; Hu, N.; Mont, L.; et al. Association of Atrial Tissue Fibrosis Identified by Delayed Enhancement MRI and Atrial Fibrillation Catheter Ablation. JAMA 2014, 311, 498–506. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).