Abstract
Liver disease is often associated with dysfunctional potassium homeostasis but is not a well-established risk factor for hyperkalemia. This retrospective cohort study examined the potential relationship between liver disease and recurrent hyperkalemia. Patients with ≥1 serum potassium measurement between January 2004 and December 2018 who experienced hyperkalemia (serum potassium >5.0 mmol/L) were identified from the United States Veterans Affairs database. A competing risk regression model was used to analyze the relationship between patient characteristics and recurrent hyperkalemia. Of 1,493,539 patients with incident hyperkalemia, 71,790 (4.8%) had liver disease (one inpatient or two outpatient records) within 1 year before the index hyperkalemia event. Recurrent hyperkalemia within 1 year after the index event occurred in 234,807 patients (15.7%) overall, 19,518 (27.2%) with liver disease, and 215,289 (15.1%) without liver disease. The risk of recurrent hyperkalemia was significantly increased in patients with liver disease versus those without (subhazard ratio, 1.34; 95% confidence interval, 1.32–1.37; p < 0.0001). Aside from vasodilator therapy, the risk of recurrent hyperkalemia was not increased with concomitant medication. In this cohort study, liver disease was an independent risk factor strongly associated with recurrent hyperkalemia within 1 year, independent of concomitant renin–angiotensin–aldosterone system inhibitor or potassium-sparing diuretic use.
1. Introduction
Chronic liver disease is associated with a significant burden of disease, with >167,000 incident cases and >90,000 related deaths in the United States (US) in 2017 [1]. Among hospitalized US patients, the number of chronic liver disease-related admissions increased by 20.8% between 2012 and 2016 [2]. The incidence and mortality rates for chronic liver disease increased by 30% and 34%, respectively, between 2007 and 2017 [1].
Patients with advanced cirrhosis often experience dysfunctional potassium (K+) homeostasis, including hyperkalemia and hypokalemia [3]. In patients with advanced cirrhosis, the prevalence of hyperkalemia is 12–14%, and serum K+ levels correlate with markers of impaired kidney function (i.e., serum sodium, urea, and creatinine levels) [3]. Similarly, in hospitalized patients with acute-on-chronic liver failure, the reported incidence of hyperkalemia is 12%, with the presence of acute kidney injury and higher serum K+ levels at admission being independent risk factors for hyperkalemia [4]. In these two patient groups, the proportion of male patients was similar among those with or without hyperkalemia [3,4], indicating that the risk of hyperkalemia does not appear to be influenced by patient sex.
As with liver disease, hyperkalemia is also associated with a significant burden of disease, including an increased risk of severe cardiac arrhythmias and mortality [5,6]. Hyperkalemia has been shown to be an independent predictor of mortality in patients with acute decompensation or acute-on-chronic liver disease [7].
Despite the increasing prevalence of the incident chronic liver disease [1] and the significant prevalence and burden of hyperkalemia in these patients [3], liver disease is not a well-established risk factor for hyperkalemia. The aim of this study was to examine the potential relationship between liver disease and recurrent hyperkalemia in a large cohort of patients who had previously experienced hyperkalemia.
2. Materials and Methods
2.1. Study Design and Objectives
This study retrospectively identified adults (aged ≥ 18 years) from the US Veterans Affairs (VA) database with ≥1 serum K+ measurement of 0.5–8.0 mmol/L between January 2004 and December 2018 who had experienced a hyperkalemia event (defined as serum K+ > 5.0 mmol/L). Patients with missing demographic, comorbidity, index estimated glomerular filtration rate (eGFR), or medication data were excluded from the analysis.
The objective of this study was to examine the relationship between liver disease and hyperkalemia recurrence within 1 year after the index hyperkalemia event.
Liver disease was defined as the presence of mild-to-moderate or severe liver disease, according to the International Classification of Diseases, ninth and tenth revision, Clinical Modification codes (ICD-9 CM and ICD-10 CM), using one inpatient or two outpatient records within 1 year prior to the index hyperkalemia event. ICD-9 CM and ICD-10 CM codes were also used to identify comorbidities at baseline.
Hyperkalemia recurrence within 1 year after the index hyperkalemia event was defined as ≥1 subsequent serum K+ measurement >5.0 mmol/L (taken ≥7 days later), with ≥1 normal serum K+ measurement (≤5.0 mmol/L) between the index hyperkalemia event and the recurrence.
2.2. Statistical Analysis
Descriptive statistics were used to assess patient characteristics; no formal statistical hypotheses were tested. Mean ± standard deviation (SD) or median (interquartile range [IQR]) were used to describe continuous variables, and counts and percentages were used for categorical variables.
A multivariate Fine and Gray competing risk regression model was used to analyze the relationship between patient characteristics, including demographics (age, sex, race, and ethnicity), eGFR, comorbidities (diabetes, chronic obstructive pulmonary disease [COPD], cancer, congestive heart failure, peripheral vascular disease, cerebrovascular disease, myocardial infarction, liver disease, dementia, rheumatologic disease, peptic ulcer disease, hemiplegia/paraplegia, or acquired immunodeficiency syndrome [AIDS]/human immunodeficiency virus [HIV] infection), and concomitant medications (renin–angiotensin–aldosterone system inhibitor [RAASi], beta-blocker, calcium channel blocker, thiazide, loop diuretic, alpha-blocker, K+-sparing diuretic, or vasodilator use), and hyperkalemia recurrence within 1 year after the index hyperkalemia event, in which the outcome was hyperkalemia recurrence and the competing event was all-cause mortality within 1 year after the index hyperkalemia event. Subhazard ratios (sHRs) and 95% confidence intervals (CIs) were used for regression analysis outcomes. Variables in the multivariate model were evaluated jointly and with adjustments for the other variables included. The variables were selected a priori based on clinically relevant risk factors identified in the literature that were captured in the Veterans Affairs database.
2.3. Ethical Considerations
This study was approved by the Institutional Review Board of the Tibor Rubin VA Medical Center of Long Beach, CA, USA. Written consent was waived given the research’s nonintrusive nature, large sample size, and patient anonymity.
3. Results
3.1. Patients
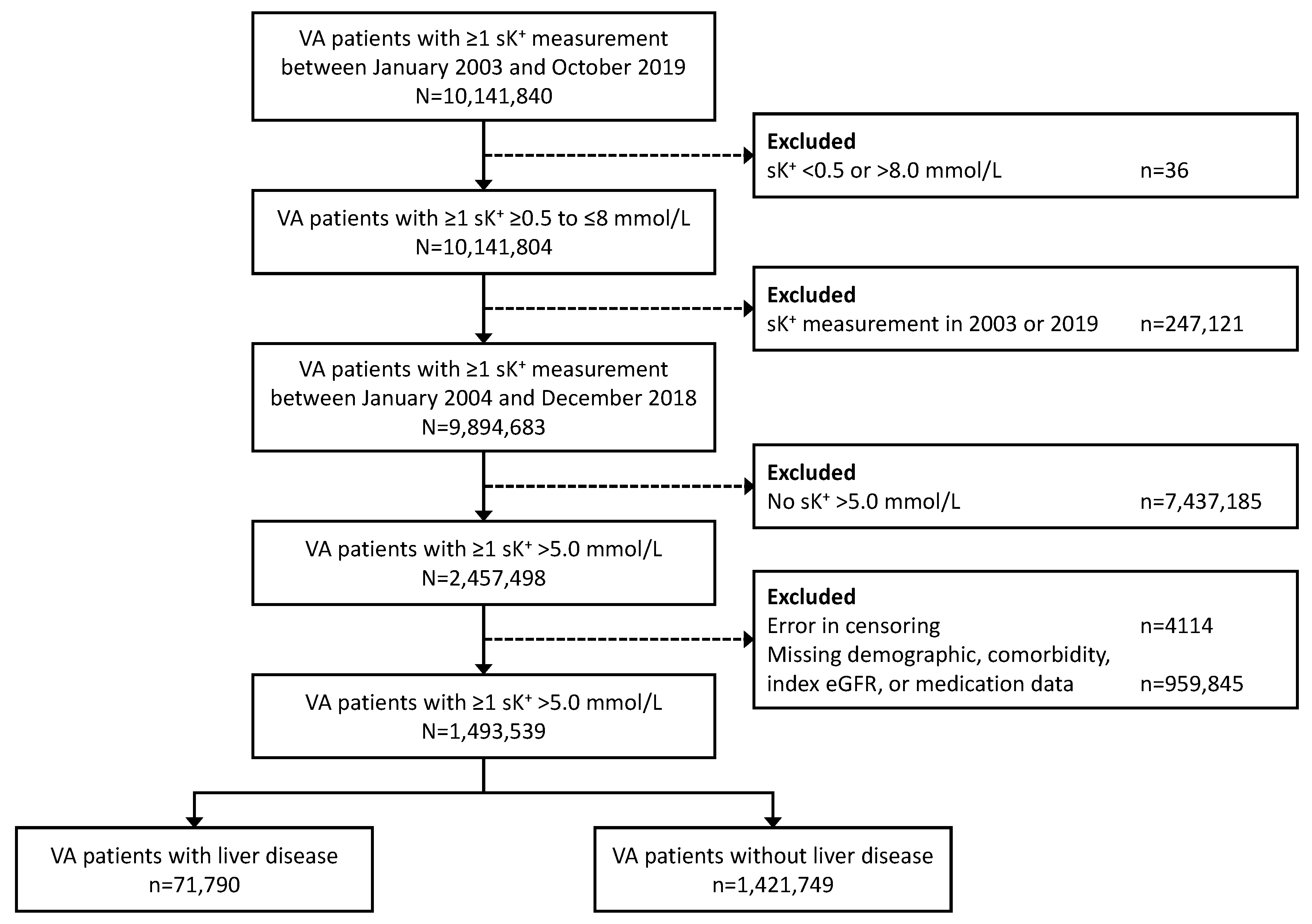
Of 9,894,683 US veterans with ≥1 serum K+ measurement of 0.5–8.0 mmol/L between January 2004 and December 2018, we identified 1,493,539 individuals who experienced a hyperkalemia event; of these patients, 71,790 (4.8%) had liver disease prior to the index hyperkalemia event (Figure 1).
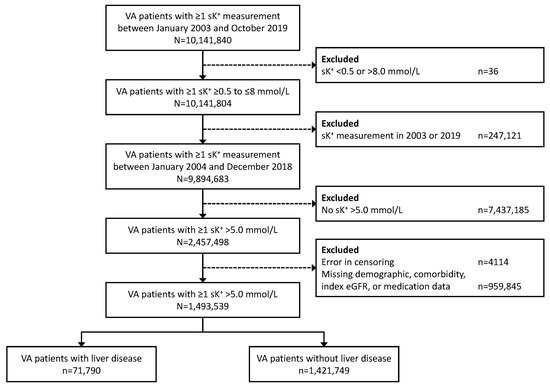
Figure 1.
Patient cohort disposition. sK+, serum potassium level; VA, Veterans Affairs.
The mean ± SD age was 61.5 ± 13.0 years in the total population, 56.4 ± 9.4 years among patients with liver disease, and 61.7 ± 13.1 years in those without liver disease (Table 1). Overall, 96.0% of the patients were male, and the mean ± SD index eGFR was 67.5 ± 25.1 mL/min/1.73 m2. Patients with liver disease had a numerically higher median (IQR) Charlson Comorbidity Index than that of those without liver disease (4 (3, 6) vs. 1 (0, 2)). Numerically greater proportions of patients with liver disease also had comorbid diabetes, COPD, cancer, congestive heart failure, myocardial infarction, and AIDS/HIV infection. The use of concomitant medications was similar in patients with and without liver disease.

Table 1.
Patient demographics and clinical characteristics.
Patients with liver disease had numerically higher mean ± SD levels of serum alkaline phosphatase (129.8 ± 127.2 vs. 83.5 ± 60.9 U/L), blood urea nitrogen (27.0 ± 21.1 vs. 22.7 ± 15.0 mg/dL), and serum ferritin (587.4 ± 2988.8 vs. 286.1 ± 867.9 ng/mL) than those without liver disease (Table 2). Numerically lower mean ± SD levels of serum bicarbonate (24.9 ± 5.5 vs. 26.9 ± 4.6 mmol/L), total cholesterol (158.5 ± 52.2 vs. 173.8 ± 44.6 mg/dL), low-density lipoprotein cholesterol (89.4 ± 41.9 vs. 99.6 ± 37.9 mg/dL), and triglycerides (143.7 ± 197.8 vs. 150.5 ± 158.8 mg/dL) were observed in patients with versus without liver disease. Other serum electrolyte levels, including serum K+, sodium, and calcium, showed non-significant numerical differences between patients with liver disease and those without liver disease.

Table 2.
Index laboratory measurements.
3.2. Hyperkalemia Recurrence
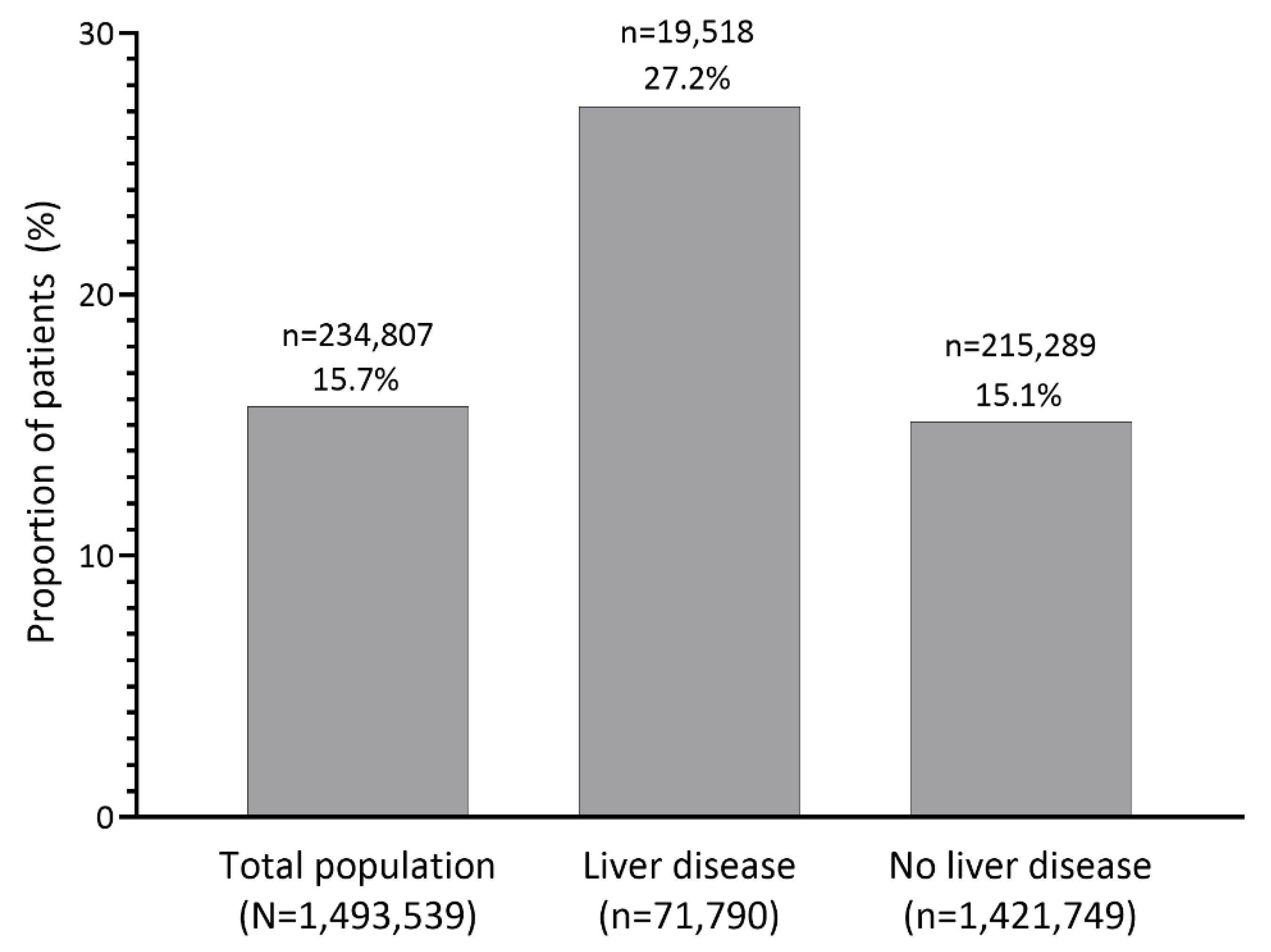
In the total population, recurrent hyperkalemia within 1 year after the index hyperkalemia event occurred in 234,807 (15.7%) patients (Figure 2). Recurrent hyperkalemia occurred in 19,518 (27.2%) patients with liver disease and 215,289 (15.1%) patients without liver disease.
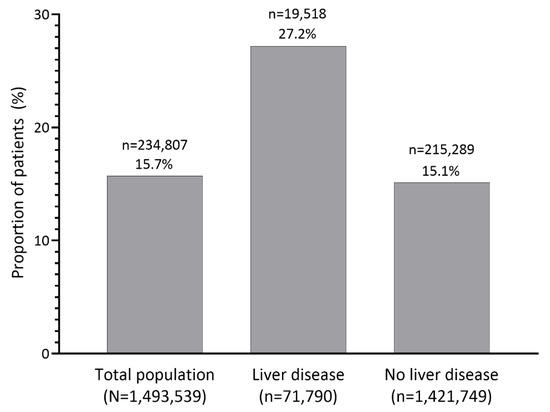
Figure 2.
Proportion of patients with recurrent hyperkalemia within 1 year after the index hyperkalemia event in the overall population and in patients with and without liver disease. The number of patients with recurrent hyperkalemia is indicated above the percentage.
3.3. Characteristics Associated with Hyperkalemia Recurrence
In the fully adjusted, multivariate, Fine and Gray competing regression analysis, several factors showed a higher risk of recurrent hyperkalemia within 1 year after the index hyperkalemia event compared with their respective referents. The presence of liver disease was associated with a 34% higher risk of the outcome versus the absence of liver disease (sHR, 1.34; 95% CI, 1.32–1.37; p < 0.0001) (Figure 3). In this analysis, liver disease had the second strongest association (based on effect estimates) between comorbidity and recurrent hyperkalemia after diabetes (sHR, 1.37; 95% CI, 1.35–1.38; p < 0.0001). Other comorbidities were also associated with a higher risk of hyperkalemia recurrence, including hemiplegia/paraplegia, congestive heart failure, cancer, COPD, rheumatologic disease, peptic ulcer disease, and peripheral vascular disease. A lower eGFR category was also associated with recurrent hyperkalemia, with patients in the eGFR 0 to <15, ≥15 to <30, and ≥30 to <45 mL/min/1.73 m2 groups having a significantly higher risk of hyperkalemia recurrence than those in the eGFR ≥90 mL/min/1.73 m2 group. A higher Charlson Comorbidity Index was also associated with a significantly higher risk of hyperkalemia recurrence (sHR, 1.06; 95% CI, 1.06–1.07; p < 0.0001) (Figure 3).
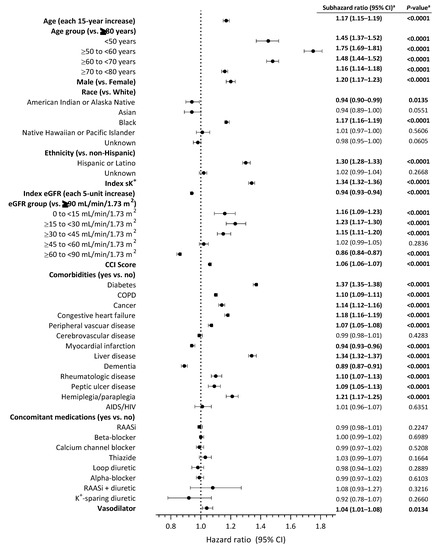
Figure 3.
Regression analysis of patient demographic and clinical characteristics and concomitant medications and risk of recurrent hyperkalemia within 1 year after the index hyperkalemia event. a Statistically significant values are shown in bold text. AIDS, acquired immunodeficiency syndrome; CCI, Charlson Comorbidity Index; CI, confidence interval; COPD, chronic obstructive pulmonary disease; eGFR, estimated glomerular filtration rate; HIV, human immunodeficiency virus; K+, potassium; RAASi, renin-angiotensin-aldosterone system inhibitor; sK+, serum potassium level.
The risk of recurrent hyperkalemia within 1 year of the index hyperkalemia event was significantly increased with concomitant vasodilator therapy (sHR, 1.04; 95% CI, 1.01–1.08; p = 0.0134). The risk of recurrent hyperkalemia was not significantly increased with any other concomitant medication in this analysis, including RAASis or K+-sparing diuretics. Other significant risk factors for recurrent hyperkalemia were advancing age, younger age group (vs. ≥80 years), male sex (vs. female), black race (vs. white), Hispanic or Latino ethnicity (vs. non-Hispanic), and index serum K+ level.
4. Discussion
In this large retrospective cohort study of approximately 1.5 million individuals with incident hyperkalemia, the prevalence of recurrent hyperkalemia within 1 year among patients with liver disease was 27.2%, and liver disease was strongly associated with hyperkalemia recurrence, independent of concomitant RAASi and K+-sparing diuretic use. Other significant clinical risk factors for hyperkalemia recurrence included diabetes, increased Charlson Comorbidity Index score, lower eGFR, concomitant vasodilator therapy, index serum K+ level, and several patient demographics, including advancing age, male sex, black race, and Hispanic or Latino ethnicity.
The prevalence of recurrent hyperkalemia among patients with liver disease in this study was higher than the prevalence of incident hyperkalemia reported in previous studies of patients with advanced cirrhosis (12–14%) [3] or acute-on-chronic liver failure (12%) [4]. This is likely because many patients who develop hyperkalemia are at risk for a recurrent hyperkalemia event, especially those with chronic kidney disease (CKD), chronic heart failure, or diabetes [8].
A previous study showed that the Model for End-Stage Liver Disease (MELD) score, which is considered to be a marker of the severity of hepatic dysfunction, is a stronger predictor for angiotensin receptor blocker-related hyperkalemia than serum creatinine or eGFR alone, with MELD scores of ≥10 being associated with an increased risk of hyperkalemia [9].
In a study of patients at high risk for hyperkalemia (i.e., patients with incident CKD or chronic heart failure or those initiating RAASi therapy), independent predictors of hyperkalemia recurrence were the severity of the index hyperkalemia event, low eGFR levels, diabetes, and spironolactone use [8]. Similarly, our study showed that comorbid diabetes and reduced eGFR were independently associated with recurrent hyperkalemia; however, K+-sparing diuretic use was not associated with an increased risk of hyperkalemia recurrence.
The mechanisms underlying hyperkalemia development in patients with liver disease are not fully understood, but reduced liver uptake of potassium, the presence of sarcopenia, use of diuretics or aldosterone antagonists, and a progressive decline in eGFR are thought to be closely related to the increase in hyperkalemia risk in patients with advanced cirrhosis [7]. The development of hyperkalemia is hypothesized to occur via renin–angiotensin–aldosterone system (RAAS) activation, which typically occurs in conjunction with elevated plasma antidiuretic hormone levels, resulting in water and sodium retention, ascites, and hyponatremia [3]. In patients with acute-on-chronic liver disease, hyperkalemia often develops secondary to reduced kidney function, resulting in impaired urinary K+ excretion and increased renal sodium and water absorption [3,7]. Hyperkalemia may also be caused by the use of K+-sparing diuretics, such as spironolactone or eplerenone [10], which are recommended as first-line treatment in patients with cirrhosis and grade 2 ascites, either alone or in conjunction with a loop diuretic [11]. However, in this analysis, patients with chronic liver disease were at increased risk of hyperkalemia recurrence regardless of concomitant K+-sparing diuretic use. The prevalence of hyperkalemia recurrence in our study was higher among patients with liver disease than that in those without liver disease (27.2% vs. 15.1%) independent of RAASi or K+-sparing diuretic use and despite the absence of a major between-group difference in serum sodium levels. This suggests that other mechanisms that are independent of the RAAS may play a role in recurrent hyperkalemia in patients with liver disease.
The limitations of this study include its retrospective observational design, which may have led to additional confounding factors that were not included in the regression analysis. Patients treated with potassium binders and those on dialysis accounted for less than 1% each of the study population and were not excluded from this study; due to the small proportions of these patients, we do not believe these to be strong confounders of the presented results. The use of ICD-9 CM and ICD-10 CM diagnosis codes to identify patients with liver disease is another potential limitation, although this would usually be biased toward the null hypothesis [12]. The study population comprised patients treated through the VA health system. As such, the results are not necessarily generalizable to the general US population because of differences in demographics and clinical characteristics between veterans and non-veterans and differences in practice patterns between VA and non-VA health systems.
5. Conclusions
In our large retrospective cohort study of US veterans with hyperkalemia, liver disease was an independent risk factor strongly associated with recurrent hyperkalemia within 1 year, independent of concomitant RAASi or K+-sparing diuretic use. Further studies are needed to understand the potential mechanisms underlying the association between liver disease and hyperkalemia recurrence.
Author Contributions
Conceptualization: E.S., K.C., Y.G.B. and K.K.-Z.; Methodology and Design: E.S., J.-T.H., A.A., Y.G.B., S.F. and L.W.; Formal Analysis: E.S., J.-T.H. and Y.G.B.; Investigation: E.S., J.-T.H. and Y.G.B.; Resources: E.S., S.F. and L.W.; Data Curation: E.S. and J.-T.H.; Writing—Original Draft: E.S., J.-T.H., R.S.A., K.K.-Z. and Y.G.B.; Writing—Review and Editing: E.S., J.-T.H., R.S.A., K.K.-Z., A.A., S.F., L.W., K.C. and Y.G.B.; Visualization: E.S., J.-T.H. and Y.G.B.; Supervision: E.S. and Y.G.B.; Project Administration: E.S. and S.F. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by AstraZeneca. Article processing charges were funded by AstraZeneca.
Institutional Review Board Statement
This study was approved by the Institutional Review Board of the Tibor Rubin VA Medical Center of Long Beach, CA, USA. All methods were carried out in accordance with the guidelines of the Institutional Review Board. Written consent was not required given the research’s nonintrusive nature, large sample size, patient anonymity, and use of only de-identified data; therefore, the requirement for informed consent was waived by the Institutional Review Board.
Informed Consent Statement
Patient written consent was waived given the research’s nonintrusive nature, large sample size, and patient anonymity.
Data Availability Statement
Restrictions apply to the availability of data generated or analyzed during this study. The United States Department of Veterans Affairs places legal restrictions on veterans’ healthcare data, which includes both identifying data and sensitive patient information. The corresponding author will, on request, detail the restrictions and any conditions under which access to some data may be provided. Requests for access to data from this study can be directed to the corresponding author, Elani Streja (estreja@hs.uci.edu).
Acknowledgments
The authors thank Rubeen Israni, Jill Davis, and Nipun Atreja for their contributions to study methodology and design and to the investigation; all were employees of AstraZeneca (Wilmington, DE, USA) at the time of their contributions. Sarah Greig and Raewyn M. Poole of inScience Communications provided medical writing support funded by AstraZeneca. The data reported in this manuscript have been supplied by the US Veterans Administration. Support for Veterans Administration/Centers for Medicare and Medicaid Services (VA/CMS) data is provided by the Department of Veterans Affairs, Veterans Health Administration, Office of Research and Development, Health Services Research and Development, VA Information Resource Center (project numbers SDR 02-237 and 98-004). Opinions expressed in this publication are those of the authors and do not represent the official opinion of the US Department of Veterans Affairs or the US Government. Results from this study were presented at the American Society of Nephrology Kidney Week 2021 (virtual meeting, 27 October–7 November 2021).
Conflicts of Interest
R.S.A. has served as a consultant for Otsuka. J.-T.H. and E.S. are employees of the Department of Veteran Affairs. A.A., S.F. and L.W. are employees and stockholders of AstraZeneca. K.C. and Y.G.B. were employees and stockholders of AstraZeneca at the time of this study. K.K.-Z. is an employee of the Department of Veteran Affairs and has received honoraria and/or support from Abbott, AbbVie, Alexion, Amgen, American Society of Nephrology, AstraZeneca, AVEO Oncology, Chugai, DaVita, Fresenius, Genentech, Haymarket Media, Hofstra Medical School, International Federation of Kidney Foundations, International Society for Hemodialysis, International Society of Renal Nutrition and Metabolism, Japanese Society for Dialysis Therapy, Hospira, Kabi, Keryx, Novartis, National Institutes of Health, National Kidney Foundation, OPKO, Pfizer, Relypsa, Resverlogix, Sandoz, Sanofi, Shire, Vifor, UpToDate, and ZS Pharma. This study was supported by AstraZeneca (Wilmington, DE, USA). AstraZeneca funded the study and manuscript development (medical writing and publication fees) and was not involved in data collection or analysis. AstraZeneca employees participated in the study conceptualization and design and the interpretation of data (authors and the former AstraZeneca employees listed in the acknowledgments). AstraZeneca authors were involved in drafting the manuscript, and the manuscript underwent AstraZeneca medical/legal review prior to submission. The non-AstraZeneca authors take sole accountability for data collection and analysis. The final decision to submit the manuscript for publication was the shared responsibility of all authors, and the content of the manuscript does not reflect the position of the Department of Veterans Affairs or AstraZeneca.
References
- Paik, J.M.; Golabi, P.; Younossi, Y.; Saleh, N.; Nhyira, A.; Younossi, Z.M. The growing burden of disability related to chronic liver disease in the United States: Data from the Global Burden of Disease Study 2007–2017. Hepatol. Commun. 2021, 5, 749–759. [Google Scholar] [CrossRef] [PubMed]
- Hirode, G.; Saab, S.; Wong, R.J. Trends in the burden of chronic liver disease among hospitalized US adults. JAMA Netw. Open 2020, 3, e201997. [Google Scholar] [CrossRef] [PubMed]
- Maiwall, R.; Kumar, S.; Sharma, M.K.; Wani, Z.; Ozukum, M.; Sarin, S.K. Prevalence and prognostic significance of hyperkalemia in hospitalized patients with cirrhosis. J. Gastroenterol. Hepatol. 2016, 31, 988–994. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.J.; Wang, K.; Jiang, H.Q.; Han, T. Characteristics, risk factors, and adverse outcomes of hyperkalemia in acute-on-chronic liver failure patients. Biomed. Res. Int. 2019, 2019, 6025726. [Google Scholar] [CrossRef] [PubMed]
- Hunter, R.W.; Bailey, M.A. Hyperkalemia: Pathophysiology, risk factors and consequences. Nephrol. Dial. Transpl. 2019, 34, iii2–iii11. [Google Scholar] [CrossRef] [PubMed]
- Collins, A.J.; Pitt, B.; Reaven, N.; Funk, S.; McGaughey, K.; Wilson, D.; Bushinsky, D.A. Association of serum potassium with all-cause mortality in patients with and without heart failure, chronic kidney disease, and/or diabetes. Am. J. Nephrol. 2017, 46, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Mezzano, G.; Cárdenas, A.; Aguilar, F.; Pavesi, M.; Sole, C.; Napoleone, L.; Graupera, I.; Juanola, A.; Carol, M.; Pose, E.; et al. Hyperkalemia influences the outcome of patients with cirrhosis with acute decompensation (AD) and acute-on-chronic liver failure (ACLF). Dig. Liver Dis. 2021, 53, 738–745. [Google Scholar] [CrossRef] [PubMed]
- Adelborg, K.; Nicolaisen, S.K.; Hasvold, P.; Palaka, E.; Pedersen, L.; Thomsen, R.W. Predictors for repeated hyperkalemia and potassium trajectories in high-risk patients—A population-based cohort study. PLoS ONE 2019, 14, e0218739. [Google Scholar] [CrossRef] [PubMed]
- Sheen, S.S.; Park, R.W.; Yoon, D.; Shin, G.T.; Kim, H.; Park, I.W. The Model for End-stage Liver Disease score is potentially a useful predictor of hyperkalemia occurrence among hospitalized angiotensin receptor blocker users. J. Clin. Pharm. Ther. 2015, 40, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Gurnani, V.; Kumar, N.; Khan, S.I.; Nawaz, M.U.; Ahmed, H.; Naz, S.; Masood Shah, A.; Jahangir, M. Biochemical risk factors associated with hyperkalemia in cirrhotic patients. Cureus 2021, 13, e18356. [Google Scholar] [CrossRef]
- Biggins, S.W.; Angeli, P.; Garcia-Tsao, G.; Gines, P.; Ling, S.C.; Nadim, M.K.; Wong, F.; Kim, W.R. Diagnosis, evaluation, and management of ascites, spontaneous bacterial peritonitis and hepatorenal syndrome: 2021 practice guidance by the American Association for the Study of Liver Diseases. Hepatology 2021, 74, 1014–1048. [Google Scholar] [CrossRef]
- Burkholder, D.A.; Moran, I.J.; DiBattista, J.V.; Lok, A.S.; Parikh, N.D.; Chen, V.L. Accuracy of International Classification of Diseases-10 Codes for cirrhosis and portal hypertensive complications. Dig. Dis. Sci. 2021, 67, 3623–3631. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).